Coeliac Disease in Adults
Coeliac disease is a chronic, immune-mediated systemic disorder triggered by dietary gluten in genetically predisposed i... MRCP exam preparation.
What matters first
Coeliac disease is a chronic, immune-mediated systemic disorder triggered by dietary gluten in genetically predisposed i... MRCP exam preparation.
Severe malabsorption with multiple nutrient deficiencies
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Severe malabsorption with multiple nutrient deficiencies
- Refractory coeliac disease (Type I or Type II)
- Suspected enteropathy-associated T-cell lymphoma (EATL)
- Dermatitis herpetiformis with systemic involvement
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Irritable Bowel Syndrome
- Non-Coeliac Gluten Sensitivity
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Coeliac Disease in Adults
1. Clinical Overview
Summary
Coeliac disease is a chronic, immune-mediated systemic disorder triggered by dietary gluten in genetically predisposed individuals carrying HLA-DQ2 or HLA-DQ8 haplotypes. [1] Ingestion of gluten (specifically gliadin peptides from wheat, barley, and rye) initiates an adaptive immune response in the small intestinal mucosa, mediated by tissue transglutaminase (tTG)-modified gliadin peptides presented to CD4+ T cells via HLA-DQ2/DQ8 molecules. [2] This results in characteristic histological changes including villous atrophy, crypt hyperplasia, and intraepithelial lymphocytosis (Marsh classification), leading to malabsorption and a spectrum of gastrointestinal and extraintestinal manifestations. [3]
The clinical phenotype ranges from classic malabsorptive disease (diarrhoea, steatorrhoea, weight loss) to non-classic presentations (iron deficiency anaemia, osteoporosis, fatigue, infertility) or entirely silent disease detected through screening of at-risk groups. [4] Diagnosis requires a combination of positive serological markers (IgA anti-tissue transglutaminase antibodies) obtained while the patient is consuming gluten, confirmed by duodenal biopsy demonstrating villous atrophy. [5] Total IgA level must be checked concurrently, as 2-3% of coeliac patients have selective IgA deficiency requiring IgG-based testing. [6]
The only established treatment is a strict, lifelong gluten-free diet (GFD), which results in clinical improvement, serological normalisation, and mucosal healing in the majority of patients. [7] Expert dietitian input is essential for successful dietary management. Untreated or poorly controlled coeliac disease carries increased risks of osteoporosis, anaemia, infertility, and malignancy, most notably enteropathy-associated T-cell lymphoma (EATL). [8]
Key Facts
| Parameter | Detail |
|---|---|
| Definition | Immune-mediated enteropathy triggered by dietary gluten in genetically susceptible individuals |
| Prevalence | 1% worldwide (0.5-1.5%); 3-5 undiagnosed for every diagnosed case [9] |
| Genetic Basis | HLA-DQ2 (90-95%) or HLA-DQ8 (5-10%); necessary but not sufficient [10] |
| Key Enzyme | Tissue transglutaminase (tTG/TG2) - deamidates gliadin peptides |
| Pathognomonic | Villous atrophy + positive serology + clinical/histological response to GFD |
| Gold Standard Diagnosis | IgA-tTG + duodenal biopsy (while on gluten-containing diet) |
| First-line Treatment | Strict lifelong gluten-free diet with expert dietitian support |
| Prognosis | Excellent with strict GFD adherence; normalised mortality after 3-5 years [8] |
Clinical Pearls
IgA Deficiency Pearl: Selective IgA deficiency occurs in 2-3% of coeliac patients (10-15x higher than general population). ALWAYS check total IgA level with serology. If IgA deficient, use IgG-tTG or IgG-DGP antibodies. [6]
Gluten Challenge Pearl: Serological and histological findings require gluten exposure. Patients must consume gluten-containing diet for at least 6 weeks (ideally 3+ months with 10g gluten/day, equivalent to 4 slices bread) before testing. [5]
Dermatitis Herpetiformis Pearl: This is the pathognomonic cutaneous manifestation of coeliac disease. 100% of DH patients have coeliac enteropathy on biopsy (often subclinical). Skin biopsy shows granular IgA deposits at dermal papillae tips on direct immunofluorescence. [11]
Family Screening Pearl: First-degree relatives have 10% prevalence of coeliac disease; second-degree relatives 5%. HLA-DQ2/DQ8 testing can exclude coeliac in family members (negative predictive value > 99%). [12]
Bone Health Pearl: Osteoporosis/osteopenia affects 30-40% of newly diagnosed adults. All adults should have DEXA scan at diagnosis. Bone density improves with strict GFD and calcium/vitamin D supplementation. [13]
Oats Pearl: Pure, uncontaminated oats (certified less than 20ppm gluten) are tolerated by most (95%) coeliac patients and can be introduced after initial GFD stabilisation. Small minority develop avenin sensitivity. [14]
Why This Matters Clinically
Coeliac disease is one of the most common lifelong conditions, affecting approximately 1% of the global population, yet remains substantially underdiagnosed with an estimated 5:1 ratio of undiagnosed to diagnosed cases. [9] This "coeliac iceberg" phenomenon means that many patients experience years of preventable morbidity before diagnosis. A high index of suspicion and appropriate serological testing in at-risk groups transforms outcomes. Early diagnosis and strict dietary adherence prevent long-term complications including osteoporosis, anaemia, infertility, and the significantly increased risk of enteropathy-associated T-cell lymphoma. [8,15]
2. Epidemiology
Prevalence and Incidence
The global seroprevalence of coeliac disease is approximately 1.4%, with biopsy-confirmed prevalence of 0.7%. [9] Prevalence varies geographically and has increased over time, attributed to genuine increases in incidence, improved diagnostic awareness, and better serological testing. [16]
| Population | Prevalence | Source |
|---|---|---|
| General population (global) | 1.0% (0.5-1.5%) | [9] |
| Europe | 1.0% | [16] |
| North America | 0.7-1.0% | [17] |
| Middle East/North Africa | 0.5-1.0% | [9] |
| Asia (increasing recognition) | 0.5-1.0% | [9] |
| First-degree relatives | 10% (7.5-12.5%) | [12] |
| Second-degree relatives | 5% | [12] |
| Type 1 diabetes mellitus | 3-8% | [18] |
| Autoimmune thyroid disease | 2-5% | [18] |
| Down syndrome | 5-12% | [18] |
| Turner syndrome | 4-8% | [18] |
| Williams syndrome | 9% | [18] |
| Selective IgA deficiency | 10-15% | [6] |
Demographics
| Factor | Detail |
|---|---|
| Age at diagnosis | Bimodal: childhood (8-12 months after gluten introduction) and adulthood (40-60 years peak) |
| Sex | Female predominance (1.5-2:1 in diagnosed cases; equalises when screened) |
| Ethnicity | All ethnicities affected; historically underdiagnosed in non-European populations |
| Iceberg phenomenon | For every diagnosed case, 3-5 remain undiagnosed [9] |
| Diagnostic delay | Average 10-13 years from symptom onset to diagnosis in adults [4] |
Risk Factors for Coeliac Disease
| Risk Factor | Relative Risk/Prevalence | Evidence |
|---|---|---|
| HLA-DQ2 (DQ2.5: DQA105/DQB102) | Present in 90-95% of coeliacs | [10] |
| HLA-DQ8 (DQA103/DQB103:02) | Present in 5-10% (often DQ2-negative) | [10] |
| First-degree relative with coeliac | 10% prevalence | [12] |
| Type 1 diabetes mellitus | 3-8% prevalence | [18] |
| Autoimmune thyroid disease | 2-5% prevalence | [18] |
| Selective IgA deficiency | 10-15% prevalence | [6] |
| Down syndrome (Trisomy 21) | 5-12% prevalence | [18] |
| Turner syndrome | 4-8% prevalence | [18] |
| Autoimmune hepatitis | 3-6% prevalence | [18] |
| Primary biliary cholangitis | 3% prevalence | [18] |
Environmental Factors
While HLA-DQ2/DQ8 is necessary for coeliac disease development, only 2-3% of HLA-DQ2/DQ8-positive individuals develop disease, indicating that environmental and non-HLA genetic factors are important. [10]
| Environmental Factor | Evidence |
|---|---|
| Timing of gluten introduction | Earlier studies suggested early (less than 4 months) or late (> 7 months) introduction increased risk; recent evidence suggests timing less important than thought [19] |
| Breastfeeding | Previously thought protective; recent RCTs show no protective effect [19] |
| Gastrointestinal infections | Rotavirus, adenovirus infections may trigger onset [2] |
| Gut microbiome dysbiosis | Altered microbiome composition observed; causality uncertain [2] |
| Quantity of gluten consumed | Higher gluten intake in infancy may increase risk [19] |
3. Pathophysiology
Molecular Pathogenesis
The pathogenesis of coeliac disease involves a complex interplay between environmental triggers (gluten), genetic susceptibility (HLA-DQ2/DQ8), and immune dysregulation. [2,20]
Exam Detail: #### Step 1: Genetic Susceptibility (HLA-DQ2/DQ8)
The HLA class II molecules DQ2 and DQ8 are the strongest genetic risk factors for coeliac disease:
| HLA Haplotype | Alleles | Frequency in Coeliacs | Population Frequency |
|---|---|---|---|
| HLA-DQ2.5 (highest risk) | DQA105:01/DQB102:01 | 90% | 20-30% |
| HLA-DQ2.2 | DQA102:01/DQB102:02 | 5% | 10-15% |
| HLA-DQ8 | DQA103:01/DQB103:02 | 5-10% | 10-15% |
| DQ2.5 homozygous | Double dose DQB1*02 | 25% of coeliacs | 2% |
Key concept: DQ2.5 homozygotes have 5x higher risk than heterozygotes due to enhanced gluten peptide presentation. [10]
The HLA-DQ2/DQ8 molecules have peptide-binding grooves with preference for negatively charged amino acids at specific anchor positions, explaining their affinity for deamidated gliadin peptides. [20]
Step 2: Gluten Structure and Digestion
Gluten composition:
- Gliadin (monomeric, soluble) - primary immunogenic component
- Glutenin (polymeric, insoluble)
Gluten proteins are uniquely rich in proline and glutamine residues, making them resistant to complete proteolytic digestion by gastric, pancreatic, and brush border enzymes. [2] The resulting peptides (particularly the immunodominant 33-mer from alpha-gliadin) reach the lamina propria intact and are highly immunogenic.
Immunodominant peptides:
- α-gliadin 33-mer (residues 56-88): Contains multiple overlapping T-cell epitopes
- α-gliadin p31-43: Innate immune activator
- γ-gliadin and ω-gliadin peptides: Additional T-cell epitopes
Step 3: Intestinal Permeability and Peptide Entry
Gliadin peptides cross the intestinal epithelium via:
- Transcellular transport: Via CD71 (transferrin receptor)-mediated transcytosis of IgA-gliadin complexes [20]
- Paracellular transport: Via zonulin-mediated tight junction opening
- Epithelial damage: During active inflammation
Zonulin pathway: Gliadin peptides bind to CXCR3 on enterocytes, triggering zonulin release and tight junction disassembly, increasing intestinal permeability. [2]
Step 4: Tissue Transglutaminase Modification
Tissue transglutaminase (tTG/TG2) is the key enzyme in coeliac pathogenesis:
| Function | Mechanism | Consequence |
|---|---|---|
| Deamidation | Converts glutamine to glutamic acid (negative charge) | Enhanced binding to HLA-DQ2/DQ8 |
| Cross-linking | Creates gliadin-tTG complexes | tTG becomes autoantigen target |
| Location | Subepithelial, released during tissue damage | Available in lamina propria |
The deamidation reaction converts neutral glutamine (Q) residues to negatively charged glutamic acid (E) residues at specific positions, creating peptides that bind with high affinity to the positively charged pockets of HLA-DQ2/DQ8. [2,20]
Key deamidation sites on α-gliadin 33-mer:
- Q65→E65
- Q72→E72
- Q80→E80
Step 5: Adaptive Immune Response
Antigen Presentation:
- Dendritic cells in lamina propria capture deamidated gliadin peptides
- Peptides loaded onto HLA-DQ2/DQ8 molecules
- Presented to gliadin-specific CD4+ T cells
CD4+ T-Cell Response (Th1 dominant):
- Secretion of IFN-γ (primary effector cytokine)
- IL-21, IL-17 production
- Activation of cytotoxic pathways
- Help for B cells → antibody production
B-Cell Response:
- Production of anti-gliadin antibodies (AGA)
- Production of anti-tTG antibodies (tTG-IgA, tTG-IgG)
- Production of anti-endomysial antibodies (EMA)
- Anti-deamidated gliadin peptide antibodies (DGP)
Autoantibody generation: Anti-tTG antibodies arise because tTG becomes covalently linked to gliadin during deamidation, and B cells recognising gliadin-tTG complexes receive T-cell help from gliadin-specific T cells (hapten-carrier mechanism). [2]
Step 6: Innate Immune Response
In parallel with adaptive immunity, gliadin peptides (particularly p31-43) trigger innate immune responses:
| Component | Effect |
|---|---|
| IL-15 overexpression | Upregulated in epithelium and lamina propria |
| NK receptor upregulation | NKG2D, CD94/NKG2C on IELs |
| MICA expression | Stress ligand on enterocytes |
| IEL activation | Cytotoxic T lymphocytes kill enterocytes |
IL-15 is the key innate cytokine, driving:
- Intraepithelial lymphocyte (IEL) expansion
- Upregulation of NKG2D on CD8+ IELs
- MICA/MICB stress molecule expression on enterocytes
- NK-like cytotoxicity independent of TCR recognition
This creates a "perfect storm" where adaptive immune recognition of gliadin combines with innate immune-mediated epithelial destruction. [20]
Step 7: Epithelial Damage and Villous Atrophy
The final common pathway involves:
- Cytotoxic IELs: CD8+ αβ and γδ T cells kill enterocytes via perforin/granzyme
- Apoptosis induction: Fas/FasL and MICA/NKG2D pathways
- Matrix remodelling: MMPs degrade basement membrane
- Crypt hyperplasia: Compensatory proliferation (increased crypt mitotic index)
- Villous atrophy: Net loss of absorptive surface area
Histological Changes: Marsh-Oberhuber Classification
The Marsh classification, modified by Oberhuber, grades the severity of small intestinal damage: [3]
| Stage | IEL Count | Crypt Architecture | Villi | Clinical Significance |
|---|---|---|---|---|
| Marsh 0 | Normal (less than 25/100 enterocytes) | Normal | Normal | Normal mucosa; pre-infiltrative |
| Marsh 1 | Increased (> 25/100 enterocytes) | Normal | Normal | Infiltrative; seen in latent coeliac, NCGS, infections |
| Marsh 2 | Increased | Hyperplastic (elongated crypts) | Normal | Hyperplastic; rare in isolation |
| Marsh 3a | Increased | Hyperplastic | Partial atrophy (mild blunting) | Coeliac disease |
| Marsh 3b | Increased | Hyperplastic | Subtotal atrophy (marked blunting) | Coeliac disease |
| Marsh 3c | Increased | Hyperplastic | Total atrophy (flat mucosa) | Severe coeliac disease |
Marsh 3 (any subtype) = diagnostic of coeliac disease when combined with positive serology and clinical context.
Exam Detail: Histological features to identify:
- Intraepithelial lymphocytes (IELs): > 25 per 100 enterocytes; predominantly CD3+CD8+ T cells; also γδ T cells
- Crypt hyperplasia: Increased crypt depth, increased mitotic figures, crypt branching
- Villous atrophy: Reduced villous height:crypt depth ratio (normal > 3:1)
- Surface enterocyte changes: Loss of brush border, cuboidal rather than columnar cells
- Lamina propria inflammation: Increased plasma cells, lymphocytes
Villous height:crypt depth ratio:
- Normal: 3-5:1
- Partial villous atrophy (3a): 2-3:1
- Subtotal villous atrophy (3b): 1-2:1
- Total villous atrophy (3c): less than 1:1 or flat mucosa
Consequences of Villous Atrophy
| Consequence | Mechanism | Clinical Manifestation |
|---|---|---|
| Reduced absorptive surface | Loss of villi | Malabsorption of all nutrients |
| Iron deficiency | Duodenal absorption (Fe2+) | Microcytic anaemia (most common) |
| Folate deficiency | Jejunal absorption | Megaloblastic anaemia |
| Vitamin B12 deficiency | Terminal ileum (if extensive) | Megaloblastic anaemia, neuropathy |
| Calcium/Vitamin D malabsorption | Fat-soluble vitamin loss | Osteoporosis, osteomalacia |
| Fat malabsorption | Reduced bile salt absorption | Steatorrhoea |
| Lactase deficiency | Brush border enzyme loss | Secondary lactose intolerance |
| Hypoproteinaemia | Protein malabsorption | Oedema (rare, severe cases) |
4. Clinical Presentation
Coeliac disease has a highly variable clinical phenotype. The Oslo definitions classify presentations into distinct categories: [1]
Classification of Clinical Presentations
| Category | Definition | Proportion |
|---|---|---|
| Classical coeliac disease | Malabsorption syndrome: diarrhoea, steatorrhoea, weight loss | 30-40% |
| Non-classical coeliac disease | Symptomatic without malabsorption: anaemia, osteoporosis, fatigue | 50-60% |
| Subclinical (silent) coeliac disease | No apparent symptoms; detected by screening at-risk groups | 10-20% |
| Potential coeliac disease | Positive serology, normal biopsy, HLA-DQ2/DQ8+ | Variable |
Gastrointestinal Symptoms
| Symptom | Frequency | Characteristics |
|---|---|---|
| Chronic diarrhoea | 50-70% | Watery, voluminous; steatorrhoea in severe cases |
| Abdominal bloating/distension | 50-60% | Postprandial; improved with GFD |
| Abdominal pain | 40-50% | Diffuse, cramping; periumbilical |
| Flatulence | 40-50% | From carbohydrate fermentation |
| Nausea | 20-30% | Especially after gluten-rich meals |
| Steatorrhoea | 20-30% | Pale, bulky, foul-smelling, floating stools |
| Constipation | 10-20% | Increasingly recognised presentation |
| Weight loss | 30-40% | May be subtle or profound |
| Aphthous stomatitis | 10-20% | Recurrent oral ulcers |
Extraintestinal Manifestations
Exam Detail: #### Haematological | Manifestation | Frequency | Mechanism | |---------------|-----------|-----------| | Iron deficiency anaemia | 30-50% (most common adult presentation) | Duodenal iron malabsorption | | Folate deficiency | 20-40% | Jejunal folate malabsorption | | Vitamin B12 deficiency | 5-20% | Less common; terminal ileum usually spared | | Thrombocytosis | Variable | Reactive; iron deficiency | | Hyposplenism | 30-50% | Functional hyposplenism; mechanism unclear |
Bone and Metabolic
| Manifestation | Frequency | Mechanism |
|---|---|---|
| Osteopenia/osteoporosis | 30-40% at diagnosis | Calcium/vitamin D malabsorption; cytokine-mediated |
| Osteomalacia | 10% | Vitamin D deficiency |
| Pathological fractures | Increased risk (1.4x) | Low bone mineral density |
| Hypocalcaemia | 20% | Malabsorption |
| Secondary hyperparathyroidism | 20-30% | Compensatory to hypocalcaemia |
Neurological
| Manifestation | Frequency | Mechanism |
|---|---|---|
| Peripheral neuropathy | 5-10% | Nutritional (B12, copper) and/or autoimmune |
| Gluten ataxia | 2-5% | Autoimmune cerebellar degeneration; anti-gliadin antibodies |
| Headache/migraine | 10-20% | Unclear mechanism |
| Epilepsy | Increased | Especially with occipital calcifications |
| Brain fog/cognitive dysfunction | Common | Improves with GFD |
Reproductive
| Manifestation | Frequency | Mechanism |
|---|---|---|
| Infertility (female) | Increased (OR 3-4) | Improves with GFD |
| Recurrent miscarriage | Increased (OR 3-9) | Nutrient deficiency, autoimmune |
| Delayed menarche | Childhood-onset | Malnutrition |
| Amenorrhoea | Secondary | Nutritional |
| Intrauterine growth restriction | Increased | Maternal malabsorption |
| Male infertility | Increased | Zinc deficiency, autoimmune |
Dermatological
| Manifestation | Frequency | Mechanism |
|---|---|---|
| Dermatitis herpetiformis | 15-25% lifetime risk | Cutaneous IgA deposition |
| Alopecia areata | Increased | Autoimmune association |
| Psoriasis | Increased | Shared autoimmune pathways |
Hepatic
| Manifestation | Frequency | Mechanism |
|---|---|---|
| Elevated transaminases | 10-40% | "Coeliac hepatitis"; usually mild, reversible with GFD |
| Autoimmune hepatitis | 3-6% association | Shared autoimmunity |
| Primary biliary cholangitis | 3% association | Shared autoimmunity |
Dental
| Manifestation | Frequency | Mechanism |
|---|---|---|
| Dental enamel defects | 10-50% (if childhood onset) | Hypoplasia from malnutrition |
| Recurrent aphthous ulcers | 10-20% | Autoimmune/nutritional |
Associated Autoimmune Conditions
Coeliac disease associates with numerous autoimmune conditions, sharing HLA and non-HLA genetic risk loci: [18]
| Associated Condition | Prevalence in Coeliac Patients | Bidirectional? |
|---|---|---|
| Type 1 diabetes mellitus | 3-8% (screen all T1DM) | Yes |
| Autoimmune thyroiditis (Hashimoto's) | 2-5% | Yes |
| Autoimmune hepatitis | 3-6% | Yes |
| Dermatitis herpetiformis | 15-25% lifetime | DH = coeliac |
| Primary biliary cholangitis | 3% | Yes |
| Sjögren's syndrome | 3-5% | Yes |
| IgA nephropathy | Increased | Yes |
| Addison's disease | Increased | Yes |
| Rheumatoid arthritis | Increased | Yes |
| Systemic lupus erythematosus | Increased | Yes |
Red Flags Requiring Urgent Assessment
[!CAUTION] Urgent referral and investigation required for:
- Severe, unexplained weight loss or malnutrition
- Failure to respond to strict gluten-free diet after 6-12 months (refractory coeliac disease)
- New abdominal pain, mass, or lymphadenopathy (concern for EATL)
- Severe or widespread dermatitis herpetiformis
- Progressive neurological symptoms (ataxia, neuropathy)
- Recurrent small bowel obstruction
- GI bleeding without obvious cause
5. Clinical Examination
General Inspection
| Finding | Significance |
|---|---|
| Pallor | Anaemia (iron, folate, B12 deficiency) |
| Cachexia/weight loss | Severe malabsorption |
| Short stature | Childhood-onset untreated coeliac |
| Oedema | Hypoproteinaemia (rare, severe) |
| Angular stomatitis | Iron/B12/folate deficiency |
| Glossitis | Smooth, red tongue; iron/B12/folate deficiency |
Skin Examination
| Finding | Location | Description |
|---|---|---|
| Dermatitis herpetiformis | Extensor surfaces (elbows, knees, buttocks, shoulders, scalp) | Intensely pruritic, grouped vesicles on erythematous base; excoriations common |
| Bruising | Generalised | Vitamin K deficiency |
| Poor wound healing | Any | Protein/zinc malnutrition |
Abdominal Examination
| Finding | Significance |
|---|---|
| Abdominal distension | Bloating, gaseous distension |
| Hyperactive bowel sounds | Increased peristalsis |
| Mild generalised tenderness | Non-specific |
| Hepatomegaly | Fatty liver; rarely, autoimmune hepatitis |
| No specific masses | Masses suggest complication (lymphoma) |
Other Systems
| System | Finding | Significance |
|---|---|---|
| Nails | Koilonychia | Iron deficiency |
| Mouth | Aphthous ulcers | Recurrent; improves with GFD |
| Angular stomatitis | Nutrient deficiency | |
| Glossitis | Smooth tongue | |
| Dental enamel defects | Childhood onset | |
| Neurological | Peripheral neuropathy | Glove-and-stocking sensory loss |
| Cerebellar signs | Gluten ataxia (ataxia, nystagmus, dysarthria) | |
| Musculoskeletal | Bone tenderness | Osteomalacia |
| Proximal myopathy | Vitamin D deficiency | |
| Tetany | Hypocalcaemia |
6. Investigations
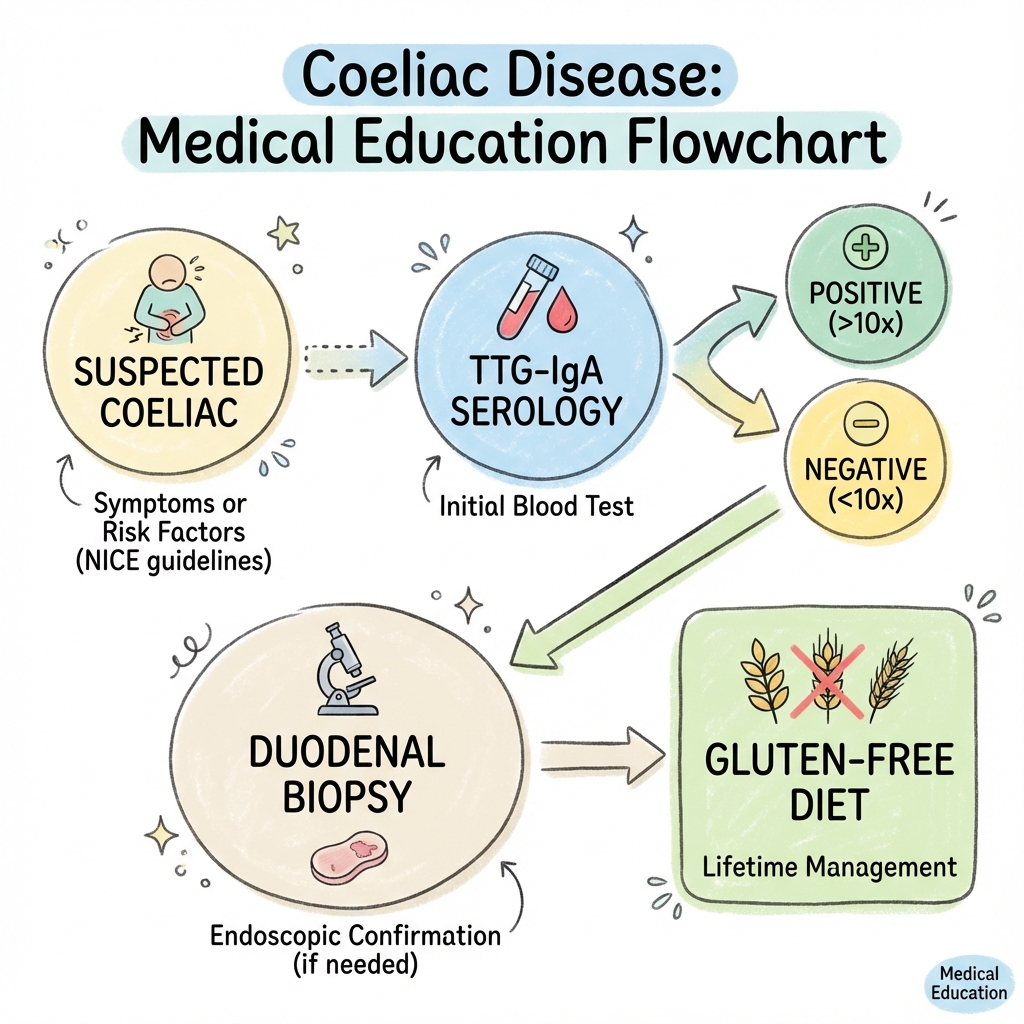
Diagnostic Algorithm
The diagnosis of coeliac disease requires serological testing followed by duodenal biopsy, both performed while the patient is consuming a gluten-containing diet. [5]
SUSPECTED COELIAC DISEASE
│
▼
┌───────────────────────────────────────────────────────────┐
│ STEP 1: SEROLOGICAL TESTING (while on gluten) │
│ • IgA-tTG (first-line) │
│ • Total IgA level (to exclude IgA deficiency) │
└───────────────────────────────────────────────────────────┘
│
┌──────────────┼──────────────┐
▼ ▼ ▼
IgA-tTG IgA-tTG IgA deficient
POSITIVE NEGATIVE (less than 0.07 g/L)
│ │ │
▼ │ ▼
┌─────────────────┐ │ ┌─────────────────────┐
│ Refer for │ │ │ Request IgG-tTG │
│ duodenal biopsy │ │ │ and/or IgG-DGP │
└─────────────────┘ │ └─────────────────────┘
│ │ │
▼ ▼ ▼
┌───────────────────────────────────────────────────────────┐
│ STEP 2: DUODENAL BIOPSY (≥4 D2 + 1-2 bulb biopsies) │
│ • Villous atrophy (Marsh 3a-3c) = DIAGNOSTIC │
│ • Marsh 1-2 alone: consider other causes │
└───────────────────────────────────────────────────────────┘
│
▼
┌─────────────────────────────────────────────────────┐
│ BIOPSY SHOWS MARSH 3 + POSITIVE SEROLOGY │
│ │ │
│ ▼ │
│ COELIAC DISEASE CONFIRMED │
│ • Initiate strict gluten-free diet │
│ • Essential: expert dietitian referral │
│ • Baseline nutritional assessment │
│ • DEXA scan │
│ • Discuss family screening │
│ • Pneumococcal vaccination │
│ • Follow-up serology at 6-12 months │
└─────────────────────────────────────────────────────┘
Serological Testing
First-Line: IgA Tissue Transglutaminase (IgA-tTG)
| Parameter | Value |
|---|---|
| Sensitivity | 95-98% [5] |
| Specificity | 95-98% [5] |
| Positive predictive value | 80-90% (population dependent) |
| Negative predictive value | > 99% |
Critical requirements:
- Patient must be consuming gluten (≥3g/day for ≥6 weeks, ideally 10g/day for 3+ months)
- Always check total IgA level concurrently (2-3% of coeliacs are IgA deficient)
- High titres (> 10x ULN) strongly predictive of coeliac disease
In IgA Deficiency (Total IgA less than 0.07 g/L)
Use IgG-based testing:
- IgG-tTG: Sensitivity 90-95%, specificity 95%
- IgG-DGP (deamidated gliadin peptide): Sensitivity 90-95%, specificity 95%
Other Serological Tests
| Test | Use | Sensitivity/Specificity |
|---|---|---|
| Anti-endomysial antibody (EMA-IgA) | Confirmatory; highly specific | 90-95% / 99% |
| IgA-DGP | Children less than 2 years; IgA deficiency | 85-95% / 90-95% |
| Anti-gliadin antibodies (AGA) | Obsolete; low specificity | Not recommended |
Exam Detail: False positives for tTG:
- Autoimmune diseases (SLE, RA)
- Chronic liver disease
- Heart failure
- Infections (rarely)
- Inflammatory bowel disease
False negatives for tTG:
- IgA deficiency (check total IgA!)
- Gluten-free diet prior to testing
- Immunosuppression
- Age less than 2 years (use DGP)
- Very early disease
HLA Typing (DQ2/DQ8)
| Application | Rationale |
|---|---|
| Exclude coeliac disease | Negative DQ2/DQ8 has > 99% NPV; essentially excludes coeliac |
| Family screening | HLA-negative relatives can be reassured |
| Uncertain diagnosis | Supports or refutes diagnosis when serology/biopsy equivocal |
| Already on GFD | Helps clarify prior to gluten challenge |
| Potential coeliac disease | Risk stratification for monitoring |
Important: Positive HLA-DQ2/DQ8 does NOT confirm coeliac disease (30-40% of population positive).
Duodenal Biopsy
Gold standard for diagnosis. [5]
| Requirement | Detail |
|---|---|
| Number of biopsies | Minimum 4 from D2 + 1-2 from duodenal bulb |
| Why multiple sites? | Patchy disease; bulb-only involvement in 9-13% |
| Gluten exposure | Must be on gluten-containing diet (≥6 weeks, ideally ≥3 months) |
| Histological criteria | Increased IELs (> 25/100 enterocytes) + villous atrophy (Marsh 3) |
Exam Detail: Biopsy technique:
- Oesophagogastroduodenoscopy (OGD)
- Multiple biopsies using standard forceps
- Orient biopsies correctly (villous surface up)
- Biopsies from D2 (4+) and duodenal bulb (1-2)
- Submit in formalin; specify "query coeliac disease"
Histopathological assessment:
- IEL count (CD3 immunostaining if needed)
- Villous:crypt ratio
- Crypt hyperplasia
- Surface enterocyte morphology
- Lamina propria inflammation
- Marsh-Oberhuber classification
Pitfalls in biopsy interpretation:
- Poor orientation
- Tangential sectioning
- Peptic duodenitis mimicking changes
- Inadequate number of biopsies
- Patient already on GFD
Other causes of villous atrophy (differential):
- Tropical sprue
- Giardiasis
- Common variable immunodeficiency
- Autoimmune enteropathy
- Drugs (olmesartan, mycophenolate)
- Crohn's disease
- Small bowel bacterial overgrowth
- HIV enteropathy
- Whipple's disease
- Eosinophilic enteritis
No-Biopsy Diagnosis (Selected Cases)
ESPGHAN 2020 guidelines (primarily paediatric, increasingly applied to adults): [5]
Biopsy may be omitted if ALL of the following criteria met:
- IgA-tTG > 10x upper limit of normal
- Positive EMA-IgA (on separate sample)
- Positive HLA-DQ2 or DQ8
- Symptomatic (classical or non-classical symptoms)
- Full understanding and acceptance by patient/family
BSG guidelines recommend confirmatory biopsy remains standard of care in adults.
Baseline Investigations at Diagnosis
| Investigation | Purpose |
|---|---|
| Full blood count | Anaemia (microcytic, macrocytic, or mixed) |
| Iron studies | Ferritin, serum iron, TIBC |
| Vitamin B12 | Deficiency common |
| Folate | Red cell folate preferred |
| Calcium, phosphate | Bone metabolism |
| 25-OH Vitamin D | Deficiency universal |
| PTH | Secondary hyperparathyroidism |
| Liver function tests | Elevated transaminases ("coeliac hepatitis") |
| Thyroid function (TSH) | Autoimmune thyroid disease association |
| DEXA scan | Osteoporosis screening (mandatory in adults) |
| HbA1c or glucose | T1DM screening if indicated |
7. Classification and Staging
Oslo Definitions (2012) [1]
| Term | Definition |
|---|---|
| Coeliac disease | Immune-mediated systemic disorder elicited by gluten in genetically susceptible individuals, characterised by presence of gluten-dependent antibodies and HLA-DQ2/DQ8, with enteropathy |
| Classical coeliac disease | Presents with signs/symptoms of malabsorption (diarrhoea, steatorrhoea, weight loss) |
| Non-classical coeliac disease | Symptomatic but without malabsorption (anaemia, osteoporosis, elevated LFTs, neurological symptoms) |
| Subclinical (silent) coeliac disease | Below threshold of clinical detection; identified by screening |
| Potential coeliac disease | Positive serology, normal biopsy; at risk of developing enteropathy |
| Refractory coeliac disease | Persistent symptoms and villous atrophy despite strict GFD for > 12 months |
| Latent coeliac disease | HLA-DQ2/DQ8+ with normal biopsy; had coeliac in past or will develop later |
Marsh-Oberhuber Classification
See Section 3 (Pathophysiology) for detailed classification.
Refractory Coeliac Disease Classification [21]
| Type | Intraepithelial Lymphocyte Phenotype | Prognosis |
|---|---|---|
| Type I (RCD-I) | Polyclonal, normal phenotype (CD3+CD8+) | Good; 80-90% 5-year survival |
| Type II (RCD-II) | Aberrant/clonal phenotype (CD3+CD8-, intracellular CD3+, surface CD3-) | Poor; 40-50% 5-year survival; precursor to EATL |
8. Management
Management Overview
CONFIRMED COELIAC DISEASE
│
┌───────────────────┼───────────────────┐
│ │ │
▼ ▼ ▼
DIETARY NUTRITIONAL MONITORING
TREATMENT SUPPORT & SCREENING
│ │ │
▼ ▼ ▼
┌──────────────┐ ┌─────────────────┐ ┌─────────────────┐
│ Strict │ │ Dietitian │ │ Serology │
│ gluten-free │ │ referral │ │ at 6-12 months │
│ diet │ │ (ESSENTIAL) │ │ │
│ │ │ │ │ Annual clinical │
│ Lifelong │ │ Nutritional │ │ review │
│ │ │ supplementation │ │ │
│ Education │ │ (iron, Ca, D) │ │ DEXA scan │
│ │ │ │ │ │
│ Support │ │ Bone health │ │ Family screening│
│ groups │ │ │ │ │
│ │ │ Vaccinations │ │ Consider repeat │
│ Coeliac UK │ │ │ │ biopsy if non- │
│ │ │ │ │ responsive │
└──────────────┘ └─────────────────┘ └─────────────────┘
Gluten-Free Diet (GFD)
The cornerstone of treatment is a strict, lifelong gluten-free diet. [7]
Foods to AVOID (Contain Gluten)
| Source | Examples |
|---|---|
| Wheat (including spelt, kamut, durum, semolina) | Bread, pasta, cereals, cakes, biscuits, pastries, couscous, bulgur |
| Barley | Beer, malt, malt extract, malt vinegar |
| Rye | Rye bread, rye crackers, some whiskeys |
| Contaminated oats | Regular commercial oats (cross-contaminated) |
| Hidden sources | Sauces, gravies, soups, processed foods, medications, communion wafers, some cosmetics (lipstick) |
Safe Foods (Gluten-Free)
| Category | Examples |
|---|---|
| Naturally gluten-free grains | Rice, corn/maize, quinoa, millet, buckwheat, amaranth, sorghum, teff |
| Protein sources | Fresh meat, poultry, fish, eggs, legumes (unprocessed) |
| Dairy | Milk, cheese, yoghurt (check additives in processed dairy) |
| Fruits and vegetables | All fresh fruits and vegetables |
| Fats | Oils, butter |
| Pure oats | Certified gluten-free oats (less than 20ppm); tolerated by 95% of coeliacs [14] |
| Labelled gluten-free products | Must contain less than 20ppm gluten (Codex Alimentarius standard) |
Exam Detail: #### Gluten Thresholds
| Standard | Definition |
|---|---|
| Gluten-free | less than 20 parts per million (ppm) gluten |
| Very low gluten | 20-100 ppm gluten |
| Daily tolerance | Most coeliacs tolerate less than 10mg gluten/day without mucosal damage |
| Detectable damage | Occurs with > 50mg/day gluten exposure |
Oats in Coeliac Disease [14]
- Pure, uncontaminated oats (certified less than 20ppm) are safe for 95% of coeliacs
- Introduce after initial GFD stabilisation (6-12 months)
- Limit to 50g dry oats/day
- 5% of patients develop avenin sensitivity (oat storage protein)
- Monitor clinically and serologically after introduction
Dietitian Referral (ESSENTIAL)
Expert dietitian input is MANDATORY for successful management. [7]
| Role of Dietitian | Details |
|---|---|
| Education | Gluten-containing foods, reading labels, hidden sources |
| Nutritional adequacy | Ensure balanced diet; GFD can be low in fibre, iron, B vitamins |
| Cross-contamination | Kitchen hygiene, eating out, travel |
| Lifestyle support | Social situations, schools/workplaces, travel |
| Monitoring adherence | Dietary assessment, troubleshooting non-response |
| Oat introduction | When and how to safely trial pure oats |
Nutritional Supplementation
| Deficiency | Recommended Treatment | Duration |
|---|---|---|
| Iron deficiency | Ferrous sulphate 200mg BD-TDS (or IV iron if malabsorption) | Until replete + 3 months |
| Folate deficiency | Folic acid 5mg daily | Until replete |
| Vitamin B12 deficiency | Hydroxocobalamin 1mg IM (loading then maintenance) | As per protocol |
| Vitamin D deficiency | Colecalciferol 800-4000 IU daily (higher doses if severe) | Lifelong if at risk |
| Calcium | 1000-1200 mg daily (diet + supplements) | Lifelong |
| Zinc | Zinc sulphate 45mg daily (if deficient) | Until replete |
Bone Health Management [13]
| Recommendation | Detail |
|---|---|
| DEXA scan at diagnosis | All adults; osteoporosis in 30-40% |
| Calcium/vitamin D | All patients: Ca 1000-1200mg/day, Vitamin D 800-2000 IU/day |
| Repeat DEXA | At 1-2 years on GFD if osteoporosis/osteopenia at baseline |
| Bisphosphonates | Consider if T-score ≤-2.5 and/or fragility fracture |
| Lifestyle | Weight-bearing exercise, smoking cessation, limit alcohol |
Vaccinations [7]
| Vaccine | Rationale |
|---|---|
| Pneumococcal vaccine | Functional hyposplenism (30-50% of coeliacs); PPV23 + PCV13 |
| Influenza (annual) | Recommended |
| Haemophilus influenzae type b | Consider in asplenic/hyposplenic patients |
| Meningococcal | Consider if travel/exposure risk |
Follow-Up and Monitoring
| Timepoint | Assessment | Action |
|---|---|---|
| At diagnosis | Baseline bloods, DEXA, dietitian referral | Establish treatment plan |
| 4-6 weeks | Initial dietitian review | Reinforce education; troubleshoot |
| 3-6 months | Symptom review; adherence assessment | Adjust plan as needed |
| 6-12 months | Repeat serology (tTG should fall > 50%) | Confirms dietary adherence |
| Annually | Clinical review; serology if indicated | Ongoing support |
| Non-response | Review adherence; repeat biopsy | Exclude refractory coeliac, other causes |
Exam Detail: #### Monitoring Response to GFD
Symptomatic response:
- GI symptoms typically improve within 2-4 weeks
- Fatigue, other symptoms over weeks to months
Serological response:
- tTG-IgA should decrease by ≥50% at 6-12 months
- May normalise by 12-24 months
- Persistent elevation suggests ongoing gluten exposure
Histological response:
- Mucosal healing occurs over 6-24 months
- May take longer in adults (1-2 years)
- Repeat biopsy not routinely required unless non-responsive
Definition of non-responsive coeliac disease (NRCD):
- Persistent symptoms and/or
- Persistently elevated serology and/or
- Persistent villous atrophy despite ostensibly strict GFD for ≥12 months
Causes of NRCD (in order of frequency):
- Ongoing gluten exposure (most common: 50-70%)
- Incorrect initial diagnosis (7-10%)
- Concomitant conditions (IBS, lactose intolerance, SIBO, microscopic colitis) (10-30%)
- Refractory coeliac disease Type I (less than 10%)
- Refractory coeliac disease Type II (less than 2%)
Special Populations
Pregnancy [22]
| Consideration | Recommendation |
|---|---|
| Pre-conception counselling | Ensure strict GFD; optimise nutrition |
| Folic acid | 400mcg (5mg if history of NTD) |
| Monitor nutritional status | Iron, B12, folate, calcium |
| Avoid gluten challenge | Do not perform during pregnancy |
| Breastfeeding | GFD safe and recommended |
| Offspring screening | HLA typing or serology if symptomatic |
Elderly
| Consideration | Recommendation |
|---|---|
| Higher index of suspicion | Often non-classical presentation |
| Nutrition priority | May already be malnourished |
| Bone health | Higher fracture risk |
| Polypharmacy | Check medications for gluten |
| Practical support | Help with food preparation |
9. Complications
Malignancy [8,15]
| Malignancy | Relative Risk | Notes |
|---|---|---|
| Enteropathy-associated T-cell lymphoma (EATL) | 6-30x | Rare but serious; Type I EATL from RCD-II |
| Non-Hodgkin lymphoma (other) | 2-6x | Various sites |
| Small bowel adenocarcinoma | 10-30x | Rare overall |
| Oesophageal squamous cell carcinoma | 2-3x | |
| Overall cancer mortality | 1.3-1.4x | Normalises with strict GFD after 3-5 years |
Exam Detail: #### Enteropathy-Associated T-Cell Lymphoma (EATL) [15]
| Feature | Detail |
|---|---|
| Type | Aggressive non-Hodgkin T-cell lymphoma arising from IELs |
| Classification | Type I (60%, HLA-linked, from RCD-II) and Type II (15%, not HLA-linked) |
| Presentation | Weight loss, abdominal pain, diarrhoea, obstruction, perforation |
| Risk factors | Older age at diagnosis, non-adherence to GFD, RCD-II, severe initial presentation |
| Diagnosis | CT/MRI, PET-CT, capsule endoscopy, deep enteroscopy with biopsy |
| Prognosis | Poor: 5-year survival 10-20% |
| Prevention | Strict lifelong GFD (reduces risk by 90%+) |
Warning signs:
- Unexplained weight loss
- New or worsening abdominal pain
- GI bleeding or obstruction
- B symptoms (fever, night sweats)
- Failure to respond to GFD (RCD-II)
Refractory Coeliac Disease (RCD) [21]
| Type | Characteristics | Management | Prognosis |
|---|---|---|---|
| RCD Type I | Normal IEL phenotype (polyclonal); exclusion of other causes | Corticosteroids (budesonide 9mg/day or prednisolone), azathioprine, infliximab (trial) | Good; 80-90% 5-year survival |
| RCD Type II | Aberrant IEL phenotype (clonal, CD3+CD8-); pre-lymphomatous | Cladribine, autologous stem cell transplant, clinical trials | Poor; 40-50% 5-year survival; 50% develop EATL within 5 years |
Investigation of suspected RCD:
- Confirm strict GFD adherence (dietitian assessment)
- Repeat duodenal biopsy (persistent villous atrophy)
- IEL immunophenotyping (flow cytometry)
- T-cell receptor gene rearrangement (clonality)
- Small bowel imaging (CT/MRI, capsule endoscopy)
- PET-CT if lymphoma suspected
Other Complications
| Complication | Incidence | Prevention/Management |
|---|---|---|
| Osteoporosis | 30-40% at diagnosis | DEXA scan; calcium/vitamin D; bisphosphonates if needed; strict GFD |
| Fractures | 1.4x increased risk | Bone health optimisation |
| Anaemia | 30-50% | Iron, folate, B12 replacement |
| Hyposplenism | 30-50% | Pneumococcal vaccination; awareness of infection risk |
| Infertility | OR 3-4x (female) | GFD improves fertility; optimise nutrition |
| Recurrent miscarriage | OR 3-9x | Strict GFD |
| Neurological | 5-10% | May be irreversible; strict GFD |
| Secondary autoimmune disease | Increased | Longer gluten exposure increases risk |
Dermatitis Herpetiformis [11]
| Feature | Detail |
|---|---|
| Definition | Cutaneous manifestation of coeliac disease with dermal IgA deposits |
| Presentation | Intensely pruritic papulovesicular eruption on extensor surfaces |
| Distribution | Elbows, knees, buttocks, shoulders, scalp, face |
| Diagnosis | Skin biopsy (perilesional) with direct immunofluorescence showing granular IgA at dermal papillae |
| Gut involvement | 100% have coeliac enteropathy on biopsy (often subclinical) |
| Treatment | Strict GFD (primary); dapsone for symptom control (50-150mg/day) |
| Response | Skin lesions may take 6-24 months to respond to GFD; dapsone provides rapid relief |
| Monitoring | G6PD screen before dapsone; FBC (haemolysis, methaemoglobinaemia) |
10. Prognosis and Outcomes
Response to Gluten-Free Diet [7]
| Parameter | Expected Response |
|---|---|
| Symptoms | Improve within 2-4 weeks |
| Serology | Normalises by 6-24 months |
| Histology | Mucosal healing 6-24 months (may take longer in adults) |
| Bone density | Improves over 1-3 years |
| Fertility | Improves with strict GFD |
| Neurological | Variable; may be irreversible |
Long-Term Outcomes [8]
| Outcome | Detail |
|---|---|
| Life expectancy | Normal with strict GFD adherence after 3-5 years |
| All-cause mortality | 1.2-1.4x increased if diagnosed (normalises with time on GFD) |
| Cancer mortality | 1.3x increased; primarily lymphoma; reduces with GFD |
| Cardiovascular mortality | Not increased |
| Quality of life | Improves significantly on GFD; dietary burden can be challenging |
Prognostic Factors
| Good Prognosis | Poor Prognosis |
|---|---|
| Early diagnosis | Late diagnosis (> 50 years) |
| Strict GFD adherence | Poor adherence |
| Good histological response | Persistent villous atrophy |
| No complications at diagnosis | RCD-II |
| Younger age | Malignancy at presentation |
11. Prevention and Screening
Primary Prevention
Currently, no proven strategies to prevent coeliac disease development. [19]
| Strategy | Evidence |
|---|---|
| Timing of gluten introduction | No clear benefit from delayed introduction |
| Breastfeeding | Not protective (PREVENT-CD, CELIPREV trials) |
| Amount of gluten | High intake may increase risk |
| Probiotics | Insufficient evidence |
Screening Recommendations
Who to screen:
| Group | Recommendation | Evidence |
|---|---|---|
| First-degree relatives | Offer serological testing | 10% prevalence |
| Type 1 diabetes | Screen at diagnosis and periodically | 3-8% prevalence |
| Autoimmune thyroid disease | Consider screening if symptomatic | 2-5% prevalence |
| Down syndrome | Screen | 5-12% prevalence |
| Turner syndrome | Screen | 4-8% prevalence |
| IgA deficiency | Screen (use IgG-based testing) | 10-15% prevalence |
| Unexplained iron deficiency anaemia | Screen | High yield |
| Unexplained osteoporosis | Screen | High yield |
| Unexplained elevated transaminases | Screen | High yield |
| Irritable bowel syndrome | Consider screening (NICE) | 4-7% prevalence in IBS |
12. Key Guidelines
BSG Guidelines (2021) [7]
Key recommendations:
- First-line test: IgA-tTG with total IgA
- Duodenal biopsy remains standard for adult diagnosis
- Minimum 4 biopsies from D2 + 1-2 from duodenal bulb
- Lifelong strict GFD as sole treatment
- Expert dietitian referral essential
- DEXA scan for all adults at diagnosis
- Annual follow-up recommended
- Pneumococcal vaccination for hyposplenism
NICE NG20 (2015, updated 2022) [23]
Key recommendations:
- Screen high-risk groups (IBS, Type 1 diabetes, autoimmune thyroid, first-degree relatives)
- Test IgA-tTG with total IgA
- Refer for biopsy if positive serology
- Do not start GFD before diagnosis confirmed
- Offer DEXA to adults at diagnosis
- Consider repeat biopsy if non-responsive
ACG Guidelines (2023) [17]
Key recommendations:
- IgA-tTG as first-line test
- Confirm diagnosis with duodenal biopsy (≥4 biopsies)
- Gluten-free diet is currently the only effective treatment
- Screen first-degree relatives, T1DM, autoimmune thyroid
- Monitor adherence with dietitian support
- Assess for refractory disease if non-responsive
ESPGHAN Guidelines (2020) [5]
Key recommendations:
- No-biopsy diagnosis acceptable in children with IgA-tTG > 10x ULN + positive EMA + HLA-DQ2/DQ8 + symptoms
- Adults: biopsy still recommended for diagnosis
- Total IgA essential to exclude deficiency
- HLA typing useful for exclusion
13. Exam-Focused Content
Common MRCP/FRACP Questions
- "What is the pathophysiology of coeliac disease?"
- "How would you investigate suspected coeliac disease?"
- "A patient with known coeliac disease is not responding to GFD. What are the causes and your approach?"
- "What complications are associated with coeliac disease?"
- "Who should be screened for coeliac disease?"
- "Describe the Marsh classification."
- "What is the significance of HLA-DQ2/DQ8 testing?"
- "How would you manage a patient with refractory coeliac disease?"
Viva Points
Viva Point: Opening statement: "Coeliac disease is a chronic immune-mediated enteropathy triggered by dietary gluten in genetically susceptible individuals carrying HLA-DQ2 or HLA-DQ8 haplotypes. It has a prevalence of approximately 1% worldwide and is characterised by villous atrophy on duodenal biopsy, positive anti-tissue transglutaminase antibodies, and clinical response to a strict gluten-free diet."
Key facts to articulate:
- Prevalence 1%; 5 undiagnosed for every diagnosed case
- HLA-DQ2 (90-95%) or HLA-DQ8 (5-10%) necessary but not sufficient
- Tissue transglutaminase deamidates gliadin → enhanced HLA-DQ2/DQ8 binding → CD4+ T-cell activation
- Diagnosis: IgA-tTG (sensitivity/specificity 95-98%) + duodenal biopsy (Marsh 3)
- Always check total IgA (2-3% IgA deficient)
- Treatment: strict lifelong GFD + expert dietitian
- Complications: osteoporosis, anaemia, EATL, refractory coeliac disease
- Strict GFD normalises mortality after 3-5 years
Model Answers
Q: A 35-year-old woman presents with fatigue and is found to have iron deficiency anaemia. How would you approach suspected coeliac disease?
A: "I would approach this systematically. Iron deficiency anaemia is the most common presentation of coeliac disease in adults, affecting 30-50% at diagnosis.
First, I would take a detailed history focusing on GI symptoms (diarrhoea, bloating, abdominal pain), extraintestinal features (fatigue, bone pain, skin rash), and family history of coeliac disease or autoimmunity.
For investigation, I would request IgA tissue transglutaminase (IgA-tTG) as the first-line serological test, along with total IgA level to exclude IgA deficiency. The patient must be consuming a gluten-containing diet for accurate results.
If IgA-tTG is positive, I would refer for upper GI endoscopy with duodenal biopsies – at least 4 from D2 and 1-2 from the duodenal bulb – looking for villous atrophy (Marsh 3 classification).
If confirmed, I would initiate a strict gluten-free diet with essential dietitian referral, arrange baseline nutritional investigations (ferritin, B12, folate, calcium, vitamin D, LFTs, TFTs), perform a DEXA scan for osteoporosis screening, ensure pneumococcal vaccination for hyposplenism, and discuss family screening.
Follow-up would include repeat serology at 6-12 months, which should show falling antibody titres, and annual review thereafter."
Q: What are the causes of non-responsive coeliac disease?
A: "Non-responsive coeliac disease is defined as persistent symptoms, elevated serology, or villous atrophy despite an ostensibly strict gluten-free diet for 12 months or more.
The causes, in order of frequency, are:
- Ongoing gluten exposure – the most common cause (50-70%), often inadvertent from hidden sources
- Incorrect initial diagnosis – other causes of villous atrophy such as tropical sprue, giardiasis, CVID
- Concomitant conditions – lactose intolerance, IBS, small intestinal bacterial overgrowth, microscopic colitis, exocrine pancreatic insufficiency
- Refractory coeliac disease Type I – persistent villous atrophy with polyclonal IELs despite strict GFD
- Refractory coeliac disease Type II – aberrant clonal IEL expansion; pre-lymphomatous condition with poor prognosis
My approach would be: confirm dietary adherence with expert dietitian assessment, repeat duodenal biopsy, test for other causes, and if RCD suspected, perform IEL immunophenotyping and TCR gene rearrangement studies."
Common Mistakes
❌ Mistakes that fail candidates:
- Testing serology when patient already on GFD (false negative)
- Not checking total IgA level (miss IgA-deficient patients)
- Taking insufficient biopsies (miss patchy disease)
- Not biopsying the duodenal bulb (bulb-only disease in 9-13%)
- Diagnosing coeliac without villous atrophy (Marsh 1-2 alone is insufficient)
- Not referring to expert dietitian
- Forgetting to screen for osteoporosis (DEXA)
- Forgetting pneumococcal vaccination (hyposplenism)
- Missing hidden gluten sources as cause of non-response
Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Irritable bowel syndrome | Normal serology, normal biopsy, symptom-based diagnosis (Rome IV) |
| Non-coeliac gluten sensitivity | Negative serology, normal biopsy, symptom response to GFD |
| Lactose intolerance | Lactose hydrogen breath test positive; often secondary in coeliac |
| Small intestinal bacterial overgrowth | Glucose/lactulose breath test positive; bloating predominant |
| Inflammatory bowel disease | Colonoscopy/ileoscopy with biopsies; inflammatory markers |
| Microscopic colitis | Normal colonoscopy but diagnostic colonic biopsies |
| Tropical sprue | Travel history; responds to antibiotics and folate |
| Giardiasis | Stool microscopy/antigen; travel history |
| Common variable immunodeficiency | Low immunoglobulins; recurrent infections |
| Autoimmune enteropathy | Anti-enterocyte antibodies; often severe |
| Drug-induced enteropathy | History of olmesartan, mycophenolate, other drugs |
14. References
-
Ludvigsson JF, Leffler DA, Bai JC, et al. The Oslo definitions for coeliac disease and related terms. Gut. 2013;62(1):43-52. doi:10.1136/gutjnl-2011-301346
-
Sollid LM, Jabri B. Triggers and drivers of autoimmunity: lessons from coeliac disease. Nat Rev Immunol. 2013;13(4):294-302. doi:10.1038/nri3407
-
Marsh MN. Gluten, major histocompatibility complex, and the small intestine. A molecular and immunobiologic approach to the spectrum of gluten sensitivity ('celiac sprue'). Gastroenterology. 1992;102(1):330-354. doi:10.1016/0016-5085(92)91819-P
-
Lebwohl B, Sanders DS, Green PHR. Coeliac disease. Lancet. 2018;391(10115):70-81. doi:10.1016/S0140-6736(17)31796-8
-
Husby S, Koletzko S, Korponay-Szabó I, et al. European Society Paediatric Gastroenterology, Hepatology and Nutrition Guidelines for Diagnosing Coeliac Disease 2020. J Pediatr Gastroenterol Nutr. 2020;70(1):141-156. doi:10.1097/MPG.0000000000002497
-
Chow MA, Lebwohl B, Reilly NR, et al. Immunoglobulin A deficiency in celiac disease. J Clin Gastroenterol. 2012;46(10):850-854. doi:10.1097/MCG.0b013e31824b2277
-
Ludvigsson JF, Bai JC, Biagi F, et al. Diagnosis and management of adult coeliac disease: guidelines from the British Society of Gastroenterology. Gut. 2014;63(8):1210-1228. doi:10.1136/gutjnl-2013-306578
-
Lebwohl B, Green PHR, Söderling J, et al. Association between celiac disease and mortality risk in a Swedish population. JAMA. 2020;323(13):1277-1285. doi:10.1001/jama.2020.1943
-
Singh P, Arora A, Strand TA, et al. Global prevalence of celiac disease: systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2018;16(6):823-836.e2. doi:10.1016/j.cgh.2017.06.037
-
Sollid LM, Markussen G, Ek J, et al. Evidence for a primary association of celiac disease to a particular HLA-DQ alpha/beta heterodimer. J Exp Med. 1989;169(1):345-350. doi:10.1084/jem.169.1.345
-
Salmi TT, Hervonen K, Kautiainen H, et al. Prevalence and incidence of dermatitis herpetiformis: a 40-year prospective study from Finland. Br J Dermatol. 2011;165(2):354-359. doi:10.1111/j.1365-2133.2011.10385.x
-
Fasano A, Berti I, Gerarduzzi T, et al. Prevalence of celiac disease in at-risk and not-at-risk groups in the United States: a large multicenter study. Arch Intern Med. 2003;163(3):286-292. doi:10.1001/archinte.163.3.286
-
Zanchetta MB, Longobardi V, Costa F, et al. Impaired bone microarchitecture improves after one year on gluten-free diet: a prospective longitudinal HRpQCT study in women with celiac disease. J Bone Miner Res. 2017;32(1):135-142. doi:10.1002/jbmr.2922
-
Aaltonen K, Laurikka P, Huhtala H, et al. The long-term consumption of oats in celiac disease patients is safe: a large cross-sectional study. Nutrients. 2017;9(6):611. doi:10.3390/nu9060611
-
Malamut G, Cellier C. Refractory celiac disease: epidemiology and clinical manifestations. Dig Dis. 2015;33(2):221-226. doi:10.1159/000369519
-
Catassi C, Gatti S, Fasano A. The new epidemiology of celiac disease. J Pediatr Gastroenterol Nutr. 2014;59(Suppl 1):S7-S9. doi:10.1097/01.mpg.0000450393.23156.59
-
Rubio-Tapia A, Hill ID, Semrad C, et al. American College of Gastroenterology Guidelines Update: Diagnosis and Management of Celiac Disease. Am J Gastroenterol. 2023;118(1):59-76. doi:10.14309/ajg.0000000000002075
-
Cosnes J, Cellier C, Viola S, et al. Incidence of autoimmune diseases in celiac disease: protective effect of the gluten-free diet. Clin Gastroenterol Hepatol. 2008;6(7):753-758. doi:10.1016/j.cgh.2007.12.022
-
Vriezinga SL, Auricchio R, Bravi E, et al. Randomized feeding intervention in infants at high risk for celiac disease. N Engl J Med. 2014;371(14):1304-1315. doi:10.1056/NEJMoa1404172
-
Jabri B, Sollid LM. T cells in celiac disease. J Immunol. 2017;198(8):3005-3014. doi:10.4049/jimmunol.1601693
-
Rubio-Tapia A, Murray JA. Classification and management of refractory coeliac disease. Gut. 2010;59(4):547-557. doi:10.1136/gut.2009.195131
-
Saccone G, Berghella V, Sarno L, et al. Celiac disease and obstetric complications: a systematic review and metaanalysis. Am J Obstet Gynecol. 2016;214(2):225-234. doi:10.1016/j.ajog.2015.09.080
-
National Institute for Health and Care Excellence. Coeliac disease: recognition, assessment and management. NICE guideline [NG20]. 2015 (updated 2022). https://www.nice.org.uk/guidance/ng20
-
Al-Toma A, Volta U, Auricchio R, et al. European Society for the Study of Coeliac Disease (ESsCD) guideline for coeliac disease and other gluten-related disorders. United European Gastroenterol J. 2019;7(5):583-613. doi:10.1177/2050640619844125
Last Updated: 2026-01-09
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Small Intestinal Anatomy and Physiology
- Immunology of the Gut
Differentials
Competing diagnoses and look-alikes to compare.
- Irritable Bowel Syndrome
- Non-Coeliac Gluten Sensitivity
- Inflammatory Bowel Disease
Consequences
Complications and downstream problems to keep in mind.
- Osteoporosis
- Enteropathy-Associated T-Cell Lymphoma