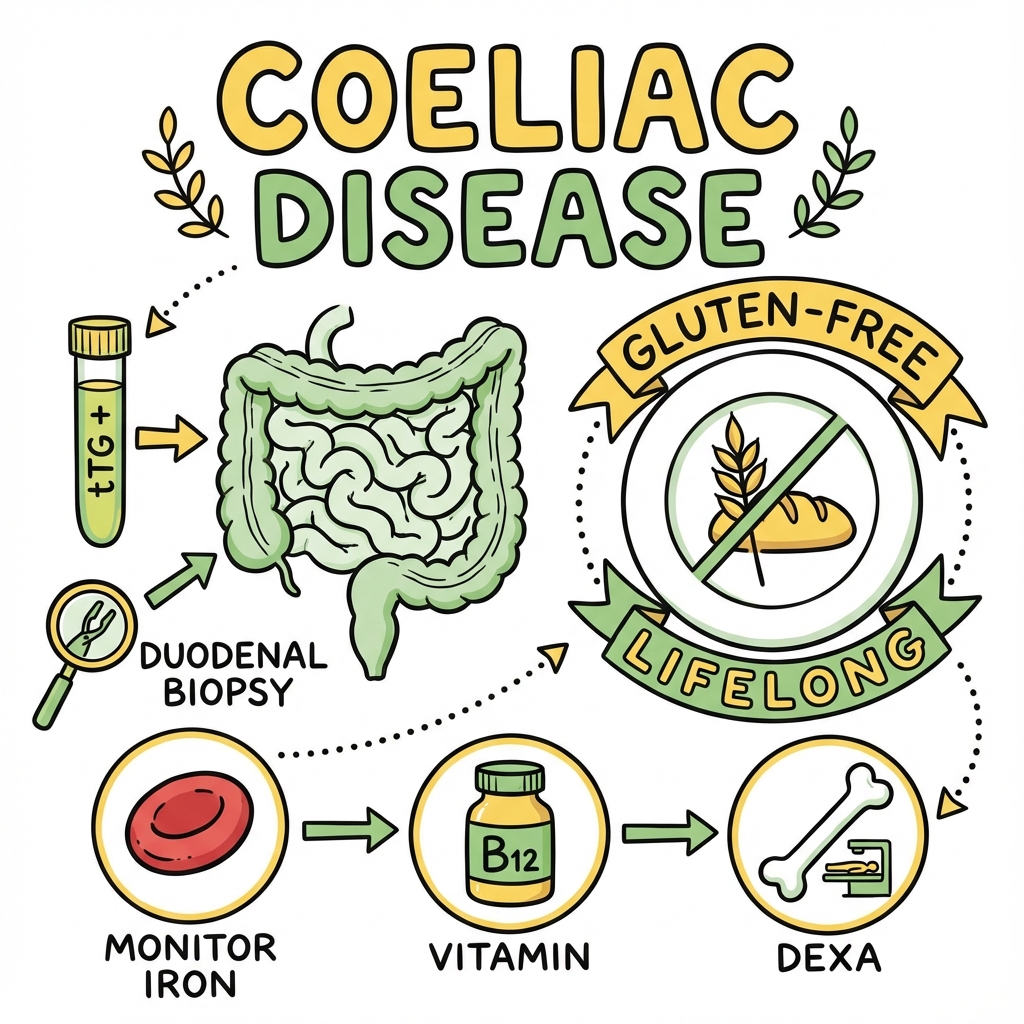
Coeliac Disease (Adult)
Coeliac disease is a chronic, immune-mediated enteropathy triggered by the ingestion of dietary gluten in genetically su... MRCP exam preparation.
What matters first
Coeliac disease is a chronic, immune-mediated enteropathy triggered by the ingestion of dietary gluten in genetically su... MRCP exam preparation.
Refractory coeliac disease (persistent symptoms despite strict GFD less than 12 months)
9 Jan 2025
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Refractory coeliac disease (persistent symptoms despite strict GFD less than 12 months)
- Unexplained weight loss despite confirmed GFD compliance
- Gastrointestinal bleeding or perforation
- Enteropathy-associated T-cell lymphoma (EATL)
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Non-Coeliac Gluten Sensitivity
- Tropical Sprue
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Coeliac Disease (Adult)
1. Topic Overview
Summary
Coeliac disease is a chronic, immune-mediated enteropathy triggered by the ingestion of dietary gluten in genetically susceptible individuals who carry the HLA-DQ2 or HLA-DQ8 haplotype. [1,2] The disease is characterised by a T-cell mediated inflammatory response to gluten-derived peptides, particularly gliadin, leading to small intestinal mucosal damage with villous atrophy, crypt hyperplasia, and intraepithelial lymphocytosis. [3] This results in malabsorption of macro- and micronutrients with consequent nutritional deficiencies.
The global prevalence is approximately 1% of the population, though the majority of cases remain undiagnosed due to the heterogeneous clinical presentation. [4] While classical coeliac disease presents with gastrointestinal symptoms including chronic diarrhoea, steatorrhoea, and weight loss, the majority of adult patients now present with non-classical or "silent" disease manifesting as iron deficiency anaemia, osteoporosis, elevated transaminases, or neurological symptoms. [5]
Diagnosis requires serological testing with tissue transglutaminase IgA antibodies (tTG-IgA) as the first-line investigation, followed by confirmatory duodenal biopsy demonstrating villous atrophy while the patient is consuming a gluten-containing diet. [6] Treatment is a strict, lifelong gluten-free diet (GFD) which leads to clinical, serological, and histological remission in the majority of patients. [7] Untreated coeliac disease is associated with significant morbidity including osteoporotic fractures, infertility, and a two- to four-fold increased risk of malignancy, particularly enteropathy-associated T-cell lymphoma (EATL). [8]
Key Facts
| Domain | Key Information |
|---|---|
| Definition | Chronic immune-mediated enteropathy triggered by dietary gluten in HLA-DQ2/DQ8-positive individuals |
| Prevalence | ~1% worldwide; diagnosed:undiagnosed ratio approximately 1:5-7 (iceberg phenomenon) |
| Genetics | HLA-DQ2 (90-95%) or HLA-DQ8 (5-10%) required but not sufficient; first-degree relatives have 10-15% risk |
| Primary Serology | tTG-IgA (sensitivity 95-98%, specificity 94-98%); must check total IgA level |
| Confirmatory Test | Duodenal biopsy showing villous atrophy (Marsh 3) with increased intraepithelial lymphocytes |
| Treatment | Strict lifelong gluten-free diet (less than 20 ppm gluten) |
| Complications | Osteoporosis, EATL, hyposplenism, refractory coeliac disease, dermatitis herpetiformis |
Clinical Pearls
"Must Be Eating Gluten" Rule: Both serology AND biopsy are only valid if the patient is currently consuming a gluten-containing diet (minimum 6 weeks, ideally 6-8 weeks of ≥10g gluten/day). Recommending a GFD before investigations are complete causes false negatives and diagnostic difficulty. [6]
IgA Deficiency Trap: Selective IgA deficiency occurs in 2-3% of coeliac patients (10-15 times more common than in the general population). If total IgA is low (less than 0.07 g/L), tTG-IgA will be falsely negative. Request IgG-based tests: tTG-IgG or deamidated gliadin peptide IgG (DGP-IgG). [9]
Think Beyond the Gut: The majority of adult patients present with extraintestinal manifestations rather than classical GI symptoms. Screen for coeliac in unexplained iron deficiency anaemia, osteoporosis/osteomalacia, persistently elevated transaminases, recurrent miscarriage, or neurological symptoms including peripheral neuropathy and ataxia. [5]
Biopsy Orientation Matters: At least four duodenal biopsies (two from D1 and two from D2) are required, as villous atrophy can be patchy. Proper orientation of biopsies is essential for accurate histological assessment. [10]
Why This Matters Clinically
Coeliac disease represents one of the most common autoimmune conditions globally, yet remains significantly underdiagnosed due to its protean manifestations. [4] Early diagnosis and treatment with a gluten-free diet prevents serious long-term complications including osteoporotic fractures, infertility, and malignancy. [8] The disease has significant implications for quality of life, requiring lifelong dietary modification and ongoing monitoring. Understanding the pathophysiology, diagnostic approach, and management of coeliac disease is essential for all clinicians, particularly those in gastroenterology, primary care, and internal medicine.
2. Epidemiology
Incidence & Prevalence
The epidemiology of coeliac disease has been transformed by serological screening, revealing a much higher prevalence than historically appreciated. [4]
| Parameter | Value | Notes |
|---|---|---|
| Global Prevalence | 1.0-1.4% | Based on serological screening studies |
| Prevalence by Serology | 1.4% (95% CI: 1.1-1.7%) | Systematic review of 96 studies [4] |
| Prevalence by Biopsy | 0.7% (95% CI: 0.5-0.9%) | Lower due to incomplete diagnostic workup |
| Incidence | 15-25 per 100,000 person-years | Increasing due to improved detection |
| Diagnosed:Undiagnosed Ratio | 1:5 to 1:7 | "Iceberg phenomenon" |
Regional Variation:
- Highest prevalence in Europe, North America, Middle East, and North Africa
- Lower prevalence in East Asia (related to lower wheat consumption and HLA-DQ2/DQ8 frequency)
- Saharawi population (Western Sahara) has the highest reported prevalence globally (5.6%)
- Prevalence increasing in developing countries with "westernisation" of diet [4]
Demographics
| Factor | Details |
|---|---|
| Age at Diagnosis | Bimodal distribution: early childhood (8-12 months after weaning) and adulthood (40-60 years); can present at any age |
| Sex Distribution | Female:Male ratio 2-3:1 in diagnosed cases; serological studies suggest true ratio closer to 1.5:1 |
| Ethnicity | More common in populations of European descent; lower prevalence in East Asian and Sub-Saharan African populations |
| Socioeconomic Status | Higher detection rates in higher socioeconomic groups (access to healthcare) |
Risk Factors
Genetic Factors (Essential but Not Sufficient):
| Genetic Factor | Frequency | Relative Risk |
|---|---|---|
| HLA-DQ2.5 (DQA105:01/DQB102:01) | 90-95% of patients | OR 7-10 |
| HLA-DQ8 (DQA103/DQB103:02) | 5-10% of patients | OR 3-5 |
| HLA-DQ2.2 (DQA102:01/DQB102:02) | Rare; requires additional DQB1*02 | OR 2-3 |
| HLA-DQ2/DQ8 Negative | less than 1% of patients | Very rare; question diagnosis |
| Non-HLA Genes | ~40 loci identified | IL2, IL21, CTLA4, CCR3 |
Family History:
| Relationship | Risk of Coeliac Disease |
|---|---|
| First-degree relative affected | 10-15% (OR 8-10) |
| Monozygotic twin | 75-85% concordance |
| Dizygotic twin | 10-20% concordance |
| Second-degree relative | 2-5% |
Associated Conditions (Screen for Coeliac Disease):
| Condition | Prevalence of Coeliac in This Group | Recommendation |
|---|---|---|
| Type 1 Diabetes Mellitus | 3-8% | Screen at diagnosis and periodically |
| Autoimmune Thyroid Disease | 2-5% | Screen if symptomatic |
| Down Syndrome | 5-12% | Screen at age 2-3 years and periodically |
| Turner Syndrome | 2-5% | Screen periodically |
| Williams Syndrome | 5-10% | Screen periodically |
| Selective IgA Deficiency | 8-10% | Use IgG-based serology |
| First-Degree Relatives | 10-15% | Screen if symptomatic; discuss asymptomatic |
| Autoimmune Hepatitis | 3-6% | Screen at diagnosis |
| Sjögren Syndrome | 2-5% | Screen if GI symptoms |
| Dermatitis Herpetiformis | 100% (by definition) | All have underlying coeliac |
3. Pathophysiology
Overview of Disease Mechanism
Coeliac disease results from a complex interplay between genetic susceptibility (HLA-DQ2/DQ8), environmental trigger (dietary gluten), and an aberrant immune response leading to intestinal damage. [2,3] The pathophysiology involves both adaptive immunity (CD4+ T-cell mediated) and innate immunity (intraepithelial lymphocyte activation).
Step-by-Step Pathogenic Mechanism
Exam Detail: Step 1: Gluten Ingestion and Partial Digestion
Gluten is a storage protein found in wheat, barley, and rye. It comprises two main fractions:
- Gliadin (alcohol-soluble; prolamins) — immunogenic fraction
- Glutenin (alcohol-insoluble)
Gliadin is rich in proline and glutamine residues, making it resistant to complete digestion by gastric, pancreatic, and brush border enzymes. Partially digested gliadin peptides, particularly the 33-mer peptide from α-gliadin, remain intact in the intestinal lumen and are highly immunogenic. [3]
Key immunogenic peptides:
- α-gliadin 57-73: Primary T-cell epitope
- α-gliadin 33-mer (p56-88): Contains multiple overlapping epitopes
- γ-gliadin and ω-gliadin peptides: Additional epitopes
Step 2: Epithelial Permeability and Peptide Translocation
Gliadin peptides cross the intestinal epithelium via:
- Paracellular transport: Gliadin triggers zonulin release from enterocytes, increasing tight junction permeability [11]
- Transcellular transport: Retro-transcytosis via secretory IgA-CD71 (transferrin receptor) pathway
- Direct epithelial damage: Gliadin peptides (particularly p31-43) directly activate innate immune responses in enterocytes, inducing IL-15 production
Zonulin Pathway:
- Gliadin binds to CXCR3 on enterocytes
- Triggers MyD88-dependent zonulin release
- Zonulin disrupts tight junction proteins (ZO-1, occludin)
- Increases paracellular permeability
- Allows larger gliadin peptides to access lamina propria [11]
Step 3: Deamidation by Tissue Transglutaminase (tTG)
Tissue transglutaminase (tTG2), a ubiquitous enzyme in the lamina propria, plays a central role:
- Deamidation: tTG converts glutamine residues in gliadin peptides to negatively charged glutamic acid
- Enhanced HLA binding: Deamidated gliadin peptides have dramatically increased affinity for HLA-DQ2/DQ8 molecules (10-fold increase)
- Crosslinking: tTG can crosslink gliadin to itself, creating tTG-gliadin complexes
This deamidation step explains why antibodies to tTG (anti-tTG) are generated and serve as the primary diagnostic marker. [3]
Step 4: Antigen Presentation and Adaptive Immune Response
Deamidated gliadin peptides are presented by antigen-presenting cells (dendritic cells, macrophages, B-cells) via HLA-DQ2 or HLA-DQ8 molecules to CD4+ T-helper cells in the lamina propria.
HLA-DQ2 Molecule:
- Preferentially binds negatively charged peptides at positions P4, P6, P7
- Deamidation creates optimal binding motif
- Present in 90-95% of coeliac patients
- DQ2.5 (cis or trans) confers highest risk
HLA-DQ8 Molecule:
- Alternative presenter in DQ2-negative patients
- Binds deamidated gliadin with different position preferences
- Present in 5-10% of patients
Step 5: T-Cell Activation and Inflammatory Cascade
Activated gluten-specific CD4+ T-cells drive the pathological response:
Cytokine Production:
- IFN-γ: Principal pro-inflammatory cytokine; activates macrophages, induces epithelial damage
- IL-21: Amplifies Th1 response; enhances IFN-γ production; activates NK and cytotoxic cells
- IL-17: Contributes to inflammatory milieu
- TNF-α: Pro-inflammatory; epithelial damage
B-Cell Activation: B-cells recognise tTG-gliadin complexes through their B-cell receptor, receive T-cell help, and differentiate into plasma cells producing:
- Anti-tTG antibodies (diagnostic)
- Anti-endomysial antibodies (EMA) — recognise tTG in smooth muscle
- Anti-deamidated gliadin peptide (DGP) antibodies
Step 6: Innate Immune Activation and Intraepithelial Lymphocytosis
Parallel to the adaptive response, innate immunity contributes to epithelial damage:
IL-15 Axis:
- Gliadin peptide p31-43 directly activates enterocytes to produce IL-15
- IL-15 acts on intraepithelial lymphocytes (IELs) expressing NKG2D receptor
- Stressed enterocytes upregulate MICA/MICB (NKG2D ligands)
- NKG2D-MICA interaction triggers cytotoxic killing of enterocytes by IELs
Intraepithelial Lymphocyte (IEL) Changes:
- Normal: less than 25 IELs per 100 enterocytes
- Coeliac: > 25 IELs per 100 enterocytes (Marsh 1+)
- IELs are predominantly CD3+ CD8+ γδ T-cells and cytotoxic αβ T-cells
- In refractory coeliac disease type 2: aberrant IEL clone (CD3+ CD8- with clonal TCR rearrangement)
Step 7: Mucosal Damage — Villous Atrophy
The combined effects of adaptive and innate immune activation result in:
- Crypt hyperplasia: Increased epithelial proliferation in crypts (compensatory)
- Villous atrophy: Loss of villous height from apoptosis of mature enterocytes
- Reduced absorptive surface: Results in malabsorption
- Inflammatory infiltrate: Lymphocytes, plasma cells in lamina propria
Marsh Classification of Histological Changes
The Marsh classification (modified Marsh-Oberhuber) grades the severity of mucosal damage: [10]
| Grade | IELs (per 100 enterocytes) | Crypts | Villi | Clinical Interpretation |
|---|---|---|---|---|
| Marsh 0 | less than 25 | Normal | Normal | Normal; rules out active coeliac |
| Marsh 1 | > 25 | Normal | Normal | Infiltrative; non-specific (infections, drugs, NCGS) |
| Marsh 2 | > 25 | Hyperplastic | Normal | Infiltrative-hyperplastic; suggestive of coeliac |
| Marsh 3a | > 25 | Hyperplastic | Partial atrophy | Definite coeliac disease |
| Marsh 3b | > 25 | Hyperplastic | Subtotal atrophy | Definite coeliac disease |
| Marsh 3c | > 25 | Hyperplastic | Total (flat mucosa) | Severe coeliac disease |
Important Notes:
- Marsh 3 is required for definitive diagnosis in adults
- Marsh 1-2 requires positive serology + clinical correlation for diagnosis
- Patchy distribution: at least 4 biopsies required (2 from D1, 2 from D2)
- Proper orientation essential; tangential cuts mimic villous atrophy
Consequences of Villous Atrophy
| Malabsorbed Nutrient | Location of Absorption | Clinical Consequence |
|---|---|---|
| Iron | Duodenum/proximal jejunum | Iron deficiency anaemia (microcytic) |
| Folate | Proximal jejunum | Macrocytic anaemia, neural tube defects |
| Calcium | Duodenum/jejunum | Osteomalacia, osteoporosis, hypocalcaemia |
| Vitamin D | Jejunum | Osteomalacia, secondary hyperparathyroidism |
| Vitamin B12 | Terminal ileum | Macrocytic anaemia (less common; ileum often spared) |
| Fat-soluble vitamins (A, D, E, K) | Jejunum | Varied manifestations; coagulopathy (vit K) |
| Zinc | Jejunum | Dermatitis, impaired wound healing |
| Carbohydrates/Fats | Jejunum/ileum | Steatorrhoea, weight loss, diarrhoea |
4. Clinical Presentation
Clinical Phenotypes
Coeliac disease presentation has evolved, with non-classical presentations now predominating in adults: [5]
| Phenotype | Characteristics | Frequency in Adults |
|---|---|---|
| Classical | Chronic diarrhoea, steatorrhoea, weight loss, malabsorption | 20-30% |
| Non-Classical | Extraintestinal manifestations ± mild GI symptoms | 50-60% |
| Silent/Asymptomatic | Positive serology and biopsy; no symptoms | 10-20% |
| Potential/Latent | Positive serology; normal biopsy (Marsh 0-1) | Variable |
Gastrointestinal Symptoms (Classical Presentation)
| Symptom | Frequency | Mechanism |
|---|---|---|
| Chronic diarrhoea | 40-60% | Malabsorption, osmotic effect |
| Steatorrhoea | 30-40% | Fat malabsorption; pale, bulky, malodorous stools |
| Abdominal bloating | 50-70% | Carbohydrate malabsorption, bacterial fermentation |
| Flatulence | 40-60% | Carbohydrate fermentation |
| Abdominal pain/discomfort | 30-50% | Distension, inflammation |
| Weight loss | 30-50% | Malabsorption |
| Nausea and vomiting | 10-20% | Less common |
| Constipation | 10-20% | Paradoxically; can occur |
Extraintestinal Manifestations (Non-Classical Presentation)
Exam Detail: Haematological:
| Manifestation | Frequency | Mechanism |
|---|---|---|
| Iron deficiency anaemia | 20-40% | Most common presenting feature; reduced iron absorption |
| Folate deficiency | 10-20% | Proximal jejunal malabsorption |
| Vitamin B12 deficiency | 5-10% | Less common (ileum often spared) |
| Mixed deficiency anaemia | Variable | Combined iron and folate/B12 deficiency |
| Functional hyposplenism | 30-50% | Reduced splenic function; Howell-Jolly bodies |
Metabolic Bone Disease:
| Manifestation | Frequency | Notes |
|---|---|---|
| Osteopenia | 40-70% | Reduced bone density by DEXA |
| Osteoporosis | 20-30% | T-score ≤-2.5; increased fracture risk |
| Osteomalacia | 5-10% | Vitamin D deficiency; bone pain |
| Secondary hyperparathyroidism | 20-30% | Response to hypocalcaemia |
| Fractures | Increased risk | Hip, spine, wrist; 40% increased risk [12] |
Hepatic:
| Finding | Frequency | Notes |
|---|---|---|
| Elevated transaminases | 15-25% | Usually mild; AST/ALT less than 5x ULN |
| Cryptogenic hepatitis | Rare | Diagnosis of exclusion; resolves with GFD |
| Autoimmune hepatitis | Associated | Screen coeliac patients with AIH |
Dermatological:
| Condition | Relationship | Features |
|---|---|---|
| Dermatitis herpetiformis | 10-25% of coeliac patients | Intensely pruritic papulovesicular rash on extensor surfaces (elbows, knees, buttocks); granular IgA deposits at dermal papillae; pathognomonic for coeliac |
| Alopecia areata | Associated | Autoimmune hair loss |
| Angular stomatitis | Nutritional | Iron/B12 deficiency |
| Aphthous ulceration | 10-20% | Recurrent mouth ulcers |
Neurological:
| Manifestation | Frequency | Notes |
|---|---|---|
| Gluten ataxia | 10-15% of idiopathic ataxia | Cerebellar ataxia; anti-gliadin antibodies; MRI may show cerebellar atrophy |
| Peripheral neuropathy | 5-10% | Sensory > motor; symmetric |
| Epilepsy with occipital calcifications | Rare | Associated syndrome |
| Cognitive impairment ("brain fog") | Common | Subjective; improves with GFD |
| Depression/anxiety | Increased | Mechanism unclear |
Reproductive:
| Manifestation | Notes |
|---|---|
| Delayed menarche | In undiagnosed childhood coeliac |
| Recurrent miscarriage | 2-3x increased risk; improves with GFD |
| Subfertility | Both male and female |
| Intrauterine growth restriction | Improved with GFD |
| Low birth weight | In untreated mothers |
Other:
| Manifestation | Mechanism |
|---|---|
| Fatigue | Multifactorial; anaemia, malnutrition, inflammation |
| Dental enamel defects | Developing teeth affected (childhood exposure) |
| Short stature | Childhood coeliac diagnosis; catch-up growth with GFD |
| Arthralgia/arthritis | Inflammatory; resolves with GFD |
Signs on Examination
General Inspection:
- May appear entirely normal
- Pallor (anaemia)
- Low body mass index or weight loss (severe cases)
- Short stature (undiagnosed childhood coeliac)
- Muscle wasting (severe malabsorption)
Specific Signs:
| Sign | Location/Technique | Significance |
|---|---|---|
| Pallor | Conjunctivae, palms, nail beds | Anaemia (iron, folate, B12) |
| Glossitis | Smooth, erythematous tongue | Iron or B12 deficiency |
| Angular stomatitis | Cracks at mouth corners | Iron or B12 deficiency |
| Aphthous ulcers | Oral mucosa | Recurrent; nutritional deficiency |
| Dental enamel hypoplasia | Teeth (pitting, discoloration) | Childhood coeliac exposure |
| Clubbing | Fingers | Chronic malabsorption (rare) |
| Koilonychia | Spoon-shaped nails | Severe iron deficiency |
| Abdominal distension | Inspection, palpation | Gas accumulation |
| Dermatitis herpetiformis | Elbows, knees, buttocks, scalp | Pruritic vesicles, excoriations; pathognomonic |
| Peripheral oedema | Lower limbs | Hypoalbuminaemia (severe) |
| Bruising | Skin | Vitamin K deficiency |
| Tetany/Chvostek sign | Facial nerve percussion | Hypocalcaemia (severe) |
| Proximal myopathy | Power testing | Vitamin D deficiency |
Red Flags
[!CAUTION] Red Flags Requiring Urgent Attention:
- Refractory coeliac disease: Persistent symptoms and villous atrophy despite strict GFD for > 12 months
- Unexplained weight loss with documented dietary compliance
- New abdominal mass or palpable lymphadenopathy (lymphoma risk)
- Gastrointestinal bleeding: Haematemesis, melaena, overt rectal bleeding
- Bowel perforation or obstruction symptoms
- Rapid clinical deterioration after initial improvement on GFD
- Progressive neurological symptoms despite treatment
- Severe malnutrition or hypoalbuminaemia
5. Differential Diagnosis
Primary Differential Diagnoses
| Condition | Key Distinguishing Features | Investigation |
|---|---|---|
| Non-Coeliac Gluten Sensitivity (NCGS) | Negative serology, normal biopsy; symptoms with gluten; no definitive biomarker | Exclusion diagnosis; gluten challenge |
| Wheat Allergy | IgE-mediated; acute symptoms (urticaria, anaphylaxis); positive wheat-specific IgE | Skin prick test, specific IgE |
| Irritable Bowel Syndrome | Negative serology; normal biopsy; Rome IV criteria; no weight loss or anaemia | Clinical diagnosis; exclude coeliac |
| Tropical Sprue | Geographic history (tropical regions); responds to antibiotics and folate | Jejunal biopsy; antibiotic response |
| Small Intestinal Bacterial Overgrowth | Hydrogen breath test positive; responds to antibiotics | Glucose/lactulose breath test |
| Lactose Intolerance | Secondary to coeliac or primary; responds to lactose exclusion | Lactose hydrogen breath test |
| Giardiasis | Travel history; cysts/trophozoites on stool microscopy | Stool ova and parasites; giardia antigen |
| Whipple Disease | PAS-positive macrophages on biopsy; multisystem disease | Duodenal biopsy with PAS stain |
| Crohn Disease | Skip lesions; granulomas; transmural inflammation | Colonoscopy with ileoscopy; MR enterography |
| Autoimmune Enteropathy | Anti-enterocyte antibodies; severe diarrhoea; often childhood onset | Biopsy; anti-enterocyte antibodies |
| Common Variable Immunodeficiency | Recurrent infections; low immunoglobulins; nodular lymphoid hyperplasia | Immunoglobulin levels |
| Drug-Induced Enteropathy | Olmesartan, mycophenolate, NSAIDs; temporal relationship to drug | Drug history; biopsy; improvement with withdrawal |
Causes of Villous Atrophy Other Than Coeliac Disease
| Cause | Key Features |
|---|---|
| Tropical sprue | Geographic history; megaloblastic anaemia; responds to antibiotics |
| Autoimmune enteropathy | Anti-enterocyte antibodies; childhood onset |
| Common variable immunodeficiency | Hypogammaglobulinaemia; recurrent infections |
| Giardiasis | Stool testing positive; travel history |
| Whipple disease | PAS-positive macrophages; arthralgias; CNS involvement |
| HIV enteropathy | Known HIV; opportunistic infections |
| Intestinal lymphoma | Mass lesion; B symptoms |
| Radiation enteritis | Prior radiation therapy |
| Drug-induced (olmesartan) | Angiotensin receptor blocker use |
| Eosinophilic gastroenteritis | Eosinophilic infiltration; food allergies |
| Crohn disease | Patchy involvement; granulomas |
6. Investigations
Diagnostic Algorithm
SUSPECTED COELIAC DISEASE
↓
┌─────────────────────────────────────────────────────────┐
│ STEP 1: CONFIRM PATIENT IS EATING GLUTEN │
│ │
│ • Minimum 6 weeks of gluten-containing diet │
│ • ≥10g gluten/day (equivalent to 4 slices bread) │
│ • If already on GFD: gluten challenge required │
└─────────────────────────────────────────────────────────┘
↓
┌─────────────────────────────────────────────────────────┐
│ STEP 2: SEROLOGICAL TESTING │
│ │
│ • tTG-IgA (first-line) + Total IgA │
│ • If IgA deficient: tTG-IgG or DGP-IgG │
└─────────────────────────────────────────────────────────┘
↓
┌─────────┴─────────┐
↓ ↓
NEGATIVE POSITIVE
↓ ↓
Coeliac ┌──────────────────────────────────────┐
unlikely* │ STEP 3: DUODENAL BIOPSY │
│ │
│ • OGD with ≥4 biopsies │
│ • 2 from D1, 2 from D2 │
│ • Proper orientation for histology │
└──────────────────────────────────────┘
↓
┌──────────┴──────────┐
↓ ↓
MARSH 3 (VA+) MARSH 0-2 (No VA)
↓ ↓
┌──────────────────┐ ┌──────────────────────┐
│ COELIAC DISEASE │ │ Consider: │
│ CONFIRMED │ │ • Potential coeliac │
│ │ │ • HLA-DQ2/DQ8 testing │
│ Start GFD │ │ • Repeat biopsy later │
│ Dietitian referral│ │ • Alternative diagnoses│
└──────────────────┘ └──────────────────────┘
* Consider IgA deficiency, seronegative coeliac (rare), or
gluten exclusion at time of testing
Serological Testing
Exam Detail: First-Line: tTG-IgA (Tissue Transglutaminase IgA)
| Parameter | Value |
|---|---|
| Sensitivity | 95-98% |
| Specificity | 94-98% |
| Positive Predictive Value | 85-90% (depends on pretest probability) |
| Threshold | Usually > 10 U/mL (assay-dependent) |
Mechanism: Antibodies to tissue transglutaminase enzyme generated as part of the immune response to tTG-gliadin complexes.
False Positives:
- Autoimmune liver disease
- Inflammatory bowel disease
- Heart failure
- Other autoimmune conditions
- Transient in children with viral infections
False Negatives:
- IgA deficiency (2-3% of coeliac patients)
- Gluten-free diet at time of testing
- Young children (less than 2 years) — may not yet mount antibody response
- Immunosuppressive therapy
- Early/mild coeliac disease
Total IgA Level:
- MUST be checked with tTG-IgA
- Selective IgA deficiency (less than 0.07 g/L) occurs in 2-3% of coeliac patients
- If IgA deficient: use IgG-based serology
Second-Line Tests:
| Test | Sensitivity | Specificity | Use |
|---|---|---|---|
| EMA-IgA (Endomysial Antibodies) | 95-98% | 98-100% | Confirmatory; immunofluorescence; operator-dependent |
| DGP-IgG (Deamidated Gliadin Peptide IgG) | 90-95% | 95-98% | IgA deficiency; children less than 2 years |
| tTG-IgG | 85-90% | 95-98% | IgA deficiency |
| DGP-IgA | 90-95% | 95-98% | Alternative to tTG-IgA |
HLA-DQ2/DQ8 Testing:
| Indication | Result | Interpretation |
|---|---|---|
| Uncertain diagnosis | Positive | Does not confirm coeliac (30-40% of population positive) |
| Uncertain diagnosis | Negative | Essentially rules out coeliac (less than 1% risk) |
| Family screening | Negative | Very low risk; no further testing needed |
| Already on GFD | Any | Useful as HLA status unaffected by diet |
Serology in IgA Deficiency:
- Check total IgA with tTG-IgA
- If IgA less than 0.07 g/L: Request tTG-IgG and/or DGP-IgG
- Both IgG-based tests recommended for optimal sensitivity
Histological Confirmation (Duodenal Biopsy)
Procedure Requirements:
- Upper GI endoscopy (OGD)
- Minimum 4 biopsies: 2 from duodenal bulb (D1), 2 from second part of duodenum (D2)
- Additional biopsies recommended (up to 6) as lesions can be patchy
- Proper orientation by histopathologist essential [10]
Histological Features of Coeliac Disease:
| Feature | Description |
|---|---|
| Villous atrophy | Reduced villus:crypt ratio (less than 3:1 normally); severity graded by Marsh |
| Crypt hyperplasia | Elongated crypts (compensatory proliferation) |
| Increased IELs | > 25 IELs per 100 enterocytes; predominantly CD3+ CD8+ T-cells |
| Lamina propria inflammation | Increased plasma cells and lymphocytes |
| Surface enterocyte damage | Cuboidal rather than columnar; loss of brush border |
Immunohistochemistry (if needed):
- CD3 staining: Highlights IELs; count > 25/100 enterocytes
- γδ T-cell receptor staining: Increased in coeliac
- tTG deposits: Subepithelial deposits in early disease
Additional Investigations
Baseline Investigations at Diagnosis:
| Category | Tests | Purpose |
|---|---|---|
| Haematology | FBC, blood film | Anaemia, macrocytosis, hyposplenism (Howell-Jolly bodies) |
| Iron Studies | Ferritin, serum iron, TIBC | Iron deficiency (most common) |
| Haematinics | Folate, vitamin B12 | Deficiency states |
| Bone Profile | Calcium, phosphate, ALP, vitamin D, PTH | Metabolic bone disease |
| Liver Function | LFTs (AST, ALT, GGT, ALP, bilirubin) | Hepatic involvement (15-25%) |
| Thyroid Function | TSH, free T4 | Associated autoimmune thyroid disease |
| Coagulation | PT/INR | Vitamin K deficiency |
| Nutritional | Zinc, copper (if symptomatic) | Micronutrient deficiency |
Bone Mineral Density (DEXA Scan):
- Recommended for all adults at diagnosis
- T-score ≤-1.0: Osteopenia; ≤-2.5: Osteoporosis
- Repeat in 1-2 years if abnormal; assess response to GFD and supplementation [12]
Imaging:
- Not routinely required
- Small bowel MRI/CT enterography: If complications suspected (lymphoma, strictures, ulcerative jejunitis)
- Findings in coeliac: Jejunisation of ileum, ilealisation of jejunum, mesenteric lymphadenopathy
Diagnostic Criteria
NICE NG20 (2015) — Adult Diagnosis: [6]
Diagnosis of coeliac disease requires:
- Positive serology (tTG-IgA ± EMA) while on gluten-containing diet
- Duodenal biopsy demonstrating villous atrophy (Marsh 3)
ESPGHAN Criteria (Children — Not Adults):
- In children with tTG-IgA > 10× upper limit of normal, positive EMA, positive HLA-DQ2/DQ8, and symptoms: biopsy may be omitted
- This "no-biopsy" pathway is NOT recommended for adults
Seronegative Coeliac Disease:
- Rare (less than 5% of cases)
- Villous atrophy on biopsy with negative serology
- Exclude other causes of villous atrophy
- HLA-DQ2/DQ8 positive
- Response to GFD supports diagnosis
7. Management
Management Overview
CONFIRMED COELIAC DISEASE
↓
┌─────────────────────────────────────────────────────────┐
│ IMMEDIATE MANAGEMENT │
├─────────────────────────────────────────────────────────┤
│ 1. Dietitian Referral (Essential) │
│ 2. Start Gluten-Free Diet (GFD) │
│ 3. Correct Nutritional Deficiencies │
│ 4. DEXA Scan (Bone Mineral Density) │
│ 5. Vaccinations (Pneumococcal for hyposplenism) │
│ 6. Patient Education and Support │
└─────────────────────────────────────────────────────────┘
↓
┌─────────────────────────────────────────────────────────┐
│ ONGOING MONITORING │
├─────────────────────────────────────────────────────────┤
│ • Annual clinical review │
│ • Serology: tTG-IgA (should normalise 6-12 months) │
│ • FBC, ferritin, folate, B12, calcium, vitamin D, LFTs │
│ • Dietitian review as needed │
│ • DEXA repeat if osteopenia/osteoporosis detected │
└─────────────────────────────────────────────────────────┘
↓
┌─────────────────────────────────────────────────────────┐
│ IF NOT RESPONDING TO GFD (After 6-12 months) │
├─────────────────────────────────────────────────────────┤
│ 1. Assess dietary compliance (dietitian review) │
│ 2. Consider inadvertent gluten exposure │
│ 3. Repeat biopsy to assess histological response │
│ 4. Exclude alternative diagnoses │
│ 5. If truly refractory: Specialist referral │
└─────────────────────────────────────────────────────────┘
Gluten-Free Diet (GFD)
Principle: Complete, lifelong avoidance of gluten is the only effective treatment. [7]
Exam Detail: Gluten-Containing Cereals (MUST AVOID):
| Cereal | Prolamins | Common Foods |
|---|---|---|
| Wheat | Gliadin | Bread, pasta, cakes, biscuits, cereals, beer, soy sauce |
| Barley | Hordein | Beer, malt, malt vinegar |
| Rye | Secalin | Rye bread, rye-based cereals |
| Triticale | Mixed | Wheat-rye hybrid |
Controversial: Oats
- Pure, uncontaminated oats tolerated by most coeliac patients (> 95%)
- Avenin (oat prolamin) is immunogenic in a minority
- Cross-contamination with wheat is common in commercial oats
- Recommendation: Avoid initially; reintroduce pure oats after symptoms settle (if tolerated)
- Maximum 50-70g/day of pure, uncontaminated oats [13]
Naturally Gluten-Free Foods:
| Category | Safe Foods |
|---|---|
| Grains/Starches | Rice, corn (maize), potato, quinoa, buckwheat, millet, amaranth, teff, sorghum |
| Proteins | Fresh meat, fish, eggs, legumes, nuts, seeds |
| Dairy | Milk, cheese, butter, plain yogurt |
| Fruits/Vegetables | All fresh fruits and vegetables |
| Other | Pure spices, herbs, oils, vinegar (except malt) |
Gluten-Free Threshold:
- Codex Alimentarius standard: less than 20 ppm gluten to be labelled "gluten-free"
- This level is safe for the vast majority of coeliac patients
- Cross-contamination: Major concern; separate preparation surfaces, utensils [14]
Practical Challenges:
- Hidden gluten: Sauces, gravies, soups, processed foods, medications
- Eating out: Risk of cross-contamination
- Social situations: Difficult to maintain strict adherence
- Cost: Gluten-free products often more expensive
- Nutritional adequacy: Risk of low fibre, high fat if relying on processed GF foods
Prescribable Gluten-Free Foods (UK NHS):
- Gluten-free bread, flour, pasta
- Availability varies by region
- Prescription options being reduced in some areas
Dietitian Referral
Essential for all newly diagnosed patients. [7]
Dietitian Role:
- Education on gluten-free diet
- Label reading skills
- Avoiding cross-contamination
- Nutritional adequacy assessment
- Identifying hidden sources of gluten
- Practical meal planning
- Assessing compliance
- Addressing psychosocial impact
Follow-up:
- Initial intensive support (multiple sessions)
- Ongoing access as needed
- Particularly important if non-responsive or struggling with adherence
Nutritional Supplementation
Iron:
- If iron deficiency: Oral iron (ferrous sulphate 200mg TDS or equivalent)
- Check ferritin at 3-4 months
- If not responding: Consider IV iron, reassess compliance
- Usually corrects with GFD alone in mild deficiency
Calcium and Vitamin D:
- If deficient or osteopenia/osteoporosis:
- Calcium 1000-1200mg/day (diet + supplements)
- Vitamin D 800-2000 IU/day (colecalciferol)
- Correct severe deficiency with loading dose
- Bone-specific treatment (bisphosphonates) if osteoporosis and fracture risk
Folate and B12:
- Correct deficiency with supplementation
- Folate 5mg daily until corrected
- B12: Oral if dietary deficiency; IM if malabsorption/severe
Zinc and Copper:
- Replace if symptomatic deficiency
- Zinc supplementation if deficient (consider acrodermatitis enteropathica-like picture)
Vaccination
Pneumococcal Vaccination (Essential): [15]
- Functional hyposplenism in 30-50% of coeliac patients
- Increased risk of encapsulated organism infection (Streptococcus pneumoniae, Haemophilus influenzae, Neisseria meningitidis)
- Recommendation:
- "Pneumococcal polysaccharide vaccine (PPV23): All adults at diagnosis"
- "Pneumococcal conjugate vaccine (PCV13): Consider in high-risk"
- Booster every 5 years
Other Vaccinations:
- Haemophilus influenzae type b (Hib): If not vaccinated
- Meningococcal vaccine: Consider in high-risk
- Annual influenza vaccine: Recommended
- Ensure up-to-date with routine vaccinations
Follow-Up and Monitoring
Annual Review Components:
| Assessment | Purpose |
|---|---|
| Symptoms | Resolution of GI and extraintestinal symptoms |
| Dietary adherence | Dietitian review if concerns |
| Weight | Normalisation; watch for excess gain |
| tTG-IgA | Should normalise within 6-12 months |
| FBC | Anaemia resolution |
| Ferritin, folate, B12 | Haematinic restoration |
| Calcium, vitamin D, PTH | Bone health |
| LFTs | Transaminase normalisation |
| TSH | Screen for autoimmune thyroid disease |
Expected Response to GFD:
- Symptomatic improvement: Weeks to months
- Serological normalisation: 6-12 months (tTG-IgA)
- Histological recovery: Variable; may take 2-5 years in adults
- Complete mucosal healing occurs in 60-80% at 2 years
Repeat Biopsy:
- Not routinely required if clinical and serological response
- Consider if: Non-responsive, high-risk complications, unclear initial diagnosis
- Some centres repeat at 1-2 years to confirm healing
8. Refractory Coeliac Disease
Definition
Refractory coeliac disease (RCD) is defined as persistent or recurrent symptoms and villous atrophy despite strict adherence to a gluten-free diet for at least 12 months, in the absence of other causes. [16]
Prevalence: 1-2% of coeliac patients
Classification
| Type | IEL Phenotype | Prognosis | EATL Risk |
|---|---|---|---|
| RCD Type 1 | Normal CD3+ CD8+ IELs; polyclonal TCR | Good (5-year survival > 90%) | Low (less than 5%) |
| RCD Type 2 | Aberrant CD3+ CD8- IELs; clonal TCR rearrangement | Poor (5-year survival 40-60%) | High (30-50% progress to EATL) |
Assessment
Confirm True Refractory Disease:
- Verify strict dietary compliance (dietitian assessment, GIP testing)
- Exclude inadvertent gluten exposure
- Exclude other diagnoses (microscopic colitis, SIBO, lactose intolerance, pancreatic insufficiency)
- Repeat duodenal biopsy: Confirm persistent villous atrophy
- Review original diagnosis (was it definitely coeliac?)
Investigations for RCD:
- Small bowel MRI/CT enterography: Exclude lymphoma, strictures
- Capsule endoscopy: Assess small bowel mucosa
- IEL immunophenotyping: Flow cytometry for CD3, CD8, TCR
- TCR gene rearrangement: Clonality assessment (PCR)
- PET-CT: If lymphoma suspected
Management
RCD Type 1:
- Strict GFD reinforcement
- Nutritional support (TPN if severe)
- Immunosuppression:
- Budesonide (9mg daily; topically acting corticosteroid)
- Prednisolone (if budesonide insufficient)
- Azathioprine or 6-mercaptopurine (steroid-sparing)
- Prognosis: Generally good with treatment
RCD Type 2:
- Specialist centre management essential
- High risk of progression to EATL
- Treatment options:
- Corticosteroids (prednisolone)
- Azathioprine/6-MP
- Cladribine (purine analogue)
- Autologous stem cell transplantation (investigational)
- Anti-IL-15 antibodies (investigational)
- Close surveillance for lymphoma (regular imaging, PET-CT)
9. Complications
Nutritional Complications
| Complication | Frequency | Prevention/Management |
|---|---|---|
| Iron deficiency anaemia | 20-40% | Iron supplementation; GFD allows recovery |
| Folate deficiency | 10-20% | Folate supplementation |
| Vitamin B12 deficiency | 5-10% | B12 supplementation (oral or IM) |
| Osteopenia/Osteoporosis | 40-70% / 20-30% | DEXA screening; calcium, vitamin D, bisphosphonates if needed [12] |
| Vitamin D deficiency | 50-70% | Supplementation (colecalciferol) |
| Zinc deficiency | Variable | Zinc supplementation if symptomatic |
Malignancy
Enteropathy-Associated T-Cell Lymphoma (EATL): [8,17]
| Feature | Details |
|---|---|
| Risk | 6-10 fold increased in untreated coeliac; absolute risk still low (0.5-1%) |
| Association | Strongly associated with RCD Type 2 (30-50% progress to EATL) |
| Presentation | Abdominal pain, weight loss, diarrhoea, obstruction, perforation |
| Diagnosis | CT/MRI enterography, PET-CT, biopsy |
| Prognosis | Poor; 5-year survival 10-20% |
| Prevention | Strict GFD reduces risk; diagnosis and treatment of coeliac |
Other Malignancies:
| Malignancy | Relative Risk | Notes |
|---|---|---|
| Small bowel adenocarcinoma | 10-30x | Rare in absolute terms |
| Oesophageal squamous cell carcinoma | 2-3x | Modest increase |
| Oropharyngeal carcinoma | 2-3x | Modest increase |
| Non-Hodgkin lymphoma (other types) | 2-4x | Various subtypes |
| Overall cancer mortality | 1.3x | Increases with delayed diagnosis |
Protective Effect of GFD:
- Strict adherence to GFD reduces malignancy risk toward general population
- Risk reduction takes 3-5 years of strict adherence
Dermatitis Herpetiformis
Definition: Cutaneous manifestation of coeliac disease characterised by intensely pruritic papulovesicular eruption. [18]
| Feature | Details |
|---|---|
| Prevalence | 10-25% of coeliac patients; 100% have underlying coeliac |
| Distribution | Extensor surfaces: Elbows, knees, buttocks, shoulders, scalp |
| Morphology | Grouped papules, vesicles; often excoriated; symmetric |
| Histology | Neutrophilic microabscesses at dermal papillae |
| Direct Immunofluorescence | Granular IgA deposits at dermal papillae (pathognomonic) |
| Treatment | GFD (first-line); Dapsone for symptom control |
Dapsone:
- Rapid symptom relief (24-48 hours)
- Does not treat underlying enteropathy
- Dose: 50-150mg daily
- Side effects: Methaemoglobinaemia, haemolysis (check G6PD), agranulocytosis
- Aim to wean off once GFD controls symptoms (6-24 months)
Hyposplenism
Prevalence: 30-50% of coeliac patients
Mechanism: Lymphoid depletion, altered splenic function
Evidence of Hyposplenism:
- Howell-Jolly bodies on blood film (nuclear remnants in RBCs)
- Acanthocytes
- Target cells
- Reduced splenic size on imaging
Clinical Significance:
- Increased susceptibility to encapsulated organisms
- Risk of overwhelming post-splenectomy-like infection (OPSI)
- Organisms: Streptococcus pneumoniae, Haemophilus influenzae, Neisseria meningitidis
Management:
- Pneumococcal vaccination (PPV23) essential
- Consider Hib and meningococcal vaccines
- Patient education on infection risk
- Early antibiotic therapy for febrile illness
Neurological Complications
| Complication | Features | Management |
|---|---|---|
| Gluten ataxia | Cerebellar ataxia; gait and limb; positive anti-gliadin antibodies; MRI may show cerebellar atrophy | Strict GFD; may stabilise or improve; irreversible damage if delayed [19] |
| Peripheral neuropathy | Sensory > motor; symmetric; distal | GFD; may improve; symptomatic treatment |
| Epilepsy with occipital calcifications | Rare syndrome; seizures; bilateral occipital calcifications | Anti-epileptic drugs; GFD |
| Migraine | Increased frequency | May improve with GFD |
| Depression/Anxiety | Common | Multifactorial; may improve with GFD |
Other Complications
| Complication | Notes |
|---|---|
| Subfertility | Male and female; improves with GFD |
| Recurrent miscarriage | 2-3x increased risk |
| Adverse pregnancy outcomes | IUGR, low birth weight; improved with GFD |
| Lactose intolerance | Secondary; brush border damage; usually resolves with mucosal healing |
| Small intestinal bacterial overgrowth | Consider if symptoms persist despite GFD |
| Ulcerative jejunitis | Rare; multiple ulcers; may progress to EATL |
| Collagenous sprue | Subepithelial collagen deposition; poor prognosis |
10. Prognosis & Outcomes
Natural History
Untreated Coeliac Disease:
- Progressive malnutrition and nutritional deficiencies
- Increased morbidity: Osteoporotic fractures, anaemia, subfertility
- Increased mortality: Standardised mortality ratio (SMR) 1.2-1.4 [8]
- Malignancy risk: 2-4 fold increased (particularly EATL)
- Quality of life: Significantly impaired
Outcomes with Treatment (Gluten-Free Diet)
| Parameter | Expected Outcome |
|---|---|
| Symptomatic improvement | 70-80% within weeks to months |
| Serological normalisation | 6-12 months; tTG-IgA should become negative |
| Histological healing | 60-80% achieve Marsh 0-1 within 2-5 years; slower in adults than children |
| Bone density improvement | Significant improvement with GFD + calcium/vitamin D |
| Malignancy risk | Returns toward general population with 3-5 years strict GFD |
| Mortality | Normalises with adherence; elevated with non-adherence |
| Quality of life | Significantly improved; burden of dietary restriction |
Prognostic Factors
Good Prognosis:
- Early diagnosis (younger age at diagnosis in adults)
- Strict dietary compliance
- Classical presentation (often responds well)
- Serological and histological normalisation
- Absence of complications at diagnosis
- Good nutritional status
Poor Prognosis:
- Delayed diagnosis (older age, long duration of untreated disease)
- Refractory coeliac disease (particularly Type 2)
- Non-adherence to GFD
- Severe malnutrition at presentation
- Development of EATL or other malignancy
- Neurological complications (may be irreversible)
Adherence Rates
| Factor | Impact on Adherence |
|---|---|
| Dietitian support | Improved adherence |
| Coeliac society membership | Improved adherence |
| Asymptomatic (silent) disease | Lower adherence |
| Adolescents/young adults | Lower adherence |
| Social support | Improved adherence |
| Understanding of consequences | Improved adherence |
| Accessibility of GF products | Improved adherence |
Adherence Monitoring:
- Clinical assessment: Symptom recurrence
- Serological: Rising tTG-IgA suggests gluten exposure
- Dietitian interview: Detailed dietary history
- Gluten immunogenic peptides (GIP): Urine/stool testing detects recent gluten ingestion (research/emerging tool)
11. Prevention & Screening
Primary Prevention
No proven strategies to prevent coeliac disease development. Previous theories about timing of gluten introduction have not been supported by evidence. [20]
Factors NOT Shown to Prevent Coeliac Disease:
- Delayed introduction of gluten beyond 6 months
- Early introduction of gluten at 4 months
- Breastfeeding (may delay onset but does not prevent)
- Probiotics during infancy
Screening Recommendations
Who to Screen: [6]
| Group | Recommendation |
|---|---|
| Symptomatic individuals | All with compatible symptoms (GI or extraintestinal) |
| Type 1 Diabetes | At diagnosis and if symptoms develop |
| Autoimmune thyroid disease | If GI symptoms or unexplained anaemia |
| IBS diagnosis | Screen to exclude coeliac before diagnosing IBS |
| Down syndrome | Screen in childhood and if symptoms |
| Turner syndrome | Screen in childhood and if symptoms |
| First-degree relatives | Discuss; screen if symptomatic; consider if asymptomatic |
| Unexplained iron deficiency anaemia | Screen after excluding blood loss |
| Metabolic bone disease | Screen if unexplained osteoporosis/osteomalacia |
| Unexplained elevated transaminases | Screen as part of liver screen |
| Unexplained subfertility | Consider screening |
| Unexplained neurological symptoms | Ataxia, neuropathy; consider screening |
Population Screening:
- Not currently recommended
- Rationale: Majority of screen-detected cases are asymptomatic; benefit of GFD in silent disease uncertain; dietary burden
- May change as evidence evolves
12. Emerging Therapies & Research
Investigational Therapies
Exam Detail: | Therapy | Mechanism | Development Stage | |---------|-----------|-------------------| | Latiglutenase | Glutenase enzyme; digests gluten in stomach | Phase 2b; adjunct to GFD | | Larazotide acetate | Tight junction regulator; prevents zonulin-mediated permeability | Phase 3; reduces symptoms | | Nexvax2 | Peptide vaccine; induces tolerance to gluten | Discontinued 2019 (failed Phase 2) | | Anti-IL-15 antibodies | Blocks IL-15 axis; reduces IEL activation | Early phase for RCD | | Transglutaminase inhibitors | Prevents deamidation of gliadin | Preclinical | | Gluten sequestrants | Polymer binds gluten in gut; prevents absorption | Early phase | | CAR-T cells | Targeting aberrant IELs in RCD Type 2 | Investigational |
Key Points:
- No approved pharmacological alternatives to GFD
- Investigational therapies likely to be adjuncts, not replacements
- GFD will remain mainstay for foreseeable future
- RCD Type 2 has most urgent need for novel therapies
13. Key Guidelines
Major Guidelines
| Guideline | Organisation | Year | Key Recommendations |
|---|---|---|---|
| NG20: Coeliac Disease | NICE (UK) | 2015 | tTG-IgA first-line; biopsy for confirmation; annual review [6] |
| BSG Guidelines | British Society of Gastroenterology | 2014 | Comprehensive UK guidance on diagnosis and management [1] |
| ACG Clinical Guideline | American College of Gastroenterology | 2013 | US guidance; similar approach |
| ESPGHAN Guidelines | European Society for Paediatric GI | 2020 | Paediatric guidelines; no-biopsy pathway for children |
| WGO Global Guidelines | World Gastroenterology Organisation | 2016 | Global perspective; resource-adapted |
Summary of Key Recommendations
- Diagnosis: tTG-IgA + total IgA; confirm with duodenal biopsy while on gluten
- Treatment: Strict lifelong GFD; dietitian essential
- Monitoring: Annual review with serology and bloods
- Bone health: DEXA at diagnosis for adults
- Vaccination: Pneumococcal (hyposplenism)
- Screening: Target high-risk groups
- Refractory disease: Specialist referral; exclude RCD Type 2
14. Viva Preparation
Opening Statement
Viva Point: "Coeliac disease is a chronic, immune-mediated enteropathy triggered by dietary gluten in genetically susceptible individuals carrying HLA-DQ2 or DQ8. It affects approximately 1% of the population and causes small intestinal villous atrophy leading to malabsorption. Diagnosis requires positive serology with tTG-IgA and confirmatory duodenal biopsy while on a gluten-containing diet. Treatment is a strict lifelong gluten-free diet."
Common Viva Questions and Model Answers
Q: What is the pathophysiology of coeliac disease?
A: "Coeliac disease results from an immune response to gluten-derived peptides in genetically susceptible individuals. Gliadin peptides cross the intestinal epithelium and are deamidated by tissue transglutaminase, creating negatively charged peptides that bind strongly to HLA-DQ2 or DQ8 molecules. These are presented to CD4+ T-cells, triggering a Th1 response with IFN-gamma and IL-21 production. This leads to villous atrophy, crypt hyperplasia, and increased intraepithelial lymphocytes. B-cells produce anti-tTG and anti-endomysial antibodies. The innate immune response via IL-15 further amplifies epithelial damage."
Q: How would you diagnose coeliac disease?
A: "I would first ensure the patient is consuming a gluten-containing diet. I would request tTG-IgA as the first-line serological test along with total IgA level, as 2-3% of coeliac patients have IgA deficiency. If IgA deficient, I would use tTG-IgG or DGP-IgG. If serology is positive, I would arrange OGD with at least four duodenal biopsies, two from D1 and two from D2. Histology showing villous atrophy with increased intraepithelial lymphocytes, Marsh 3, confirms the diagnosis."
Q: A patient with coeliac disease is not responding to a gluten-free diet. How would you approach this?
A: "I would take a systematic approach. First, I would assess dietary compliance with detailed dietitian review, as inadvertent gluten exposure is the most common cause. If adherent, I would exclude other conditions such as lactose intolerance, microscopic colitis, small intestinal bacterial overgrowth, and pancreatic insufficiency. I would repeat serology; persistently elevated tTG suggests ongoing gluten exposure. I would repeat duodenal biopsy to confirm persistent villous atrophy. If truly refractory after 12 months of strict GFD, I would consider refractory coeliac disease and refer to a specialist centre for IEL phenotyping to distinguish Type 1 from Type 2, given the malignancy risk with Type 2."
Q: What are the complications of coeliac disease?
A: "Complications include nutritional deficiencies, particularly iron, folate, B12, calcium, and vitamin D, leading to anaemia and metabolic bone disease. Osteoporosis affects 20-30% and increases fracture risk. There is a 6-10 fold increased risk of enteropathy-associated T-cell lymphoma, particularly in untreated or refractory disease. Dermatitis herpetiformis is the skin manifestation. Hyposplenism occurs in 30-50%, increasing infection risk with encapsulated organisms. Neurological complications include gluten ataxia and peripheral neuropathy. There is increased risk of subfertility and adverse pregnancy outcomes. Strict GFD reduces most of these risks."
Common Mistakes to Avoid
❌ Mistakes That Fail Candidates:
- Testing serology or taking biopsy when patient already on gluten-free diet
- Forgetting to check total IgA level with tTG-IgA
- Not taking sufficient biopsies (minimum 4, from D1 and D2)
- Missing the diagnosis in patients with extraintestinal manifestations
- Forgetting pneumococcal vaccination for hyposplenism
- Not referring to dietitian at diagnosis
- Failing to consider refractory coeliac disease in non-responders
- Not screening for associated conditions (thyroid, diabetes)
- Missing red flags suggesting malignancy
15. Patient Information
What is Coeliac Disease?
Coeliac disease is a condition where your immune system reacts abnormally to gluten, a protein found in wheat, barley, and rye. When you eat gluten, your immune system attacks the lining of your small intestine, damaging it and making it difficult to absorb nutrients from food.
Why Does it Matter?
Without treatment, coeliac disease can cause:
- Anaemia (low iron levels) making you tired
- Weak bones (osteoporosis) increasing fracture risk
- Difficulty becoming pregnant
- Long-term, a slightly increased risk of some bowel cancers
The good news is that treatment is very effective. With a gluten-free diet, the gut heals and most people feel completely better.
How is it Treated?
Gluten-free diet for life:
- You need to completely avoid wheat, barley, and rye
- This means avoiding most breads, pasta, cereals, cakes, and biscuits unless specifically gluten-free
- Even small amounts of gluten can cause damage, even if you don't have symptoms
Safe foods include:
- Rice, potatoes, corn, quinoa
- Meat, fish, eggs
- Fruits and vegetables
- Milk, cheese, butter
You will receive:
- Referral to a dietitian who will teach you how to read food labels and avoid hidden gluten
- Blood tests to check for vitamin and mineral deficiencies
- A bone density scan to check bone health
- Vaccinations to protect against certain infections
What to Expect
- Symptoms often improve within weeks of starting a gluten-free diet
- Complete gut healing can take 1-2 years
- Annual blood tests and check-ups are important
- You will need to follow the diet for life
When to Seek Help
See your doctor if:
- Symptoms don't improve on the gluten-free diet
- You are losing weight unexpectedly
- You have new or worsening symptoms
- You are struggling to follow the diet
Support
- Coeliac UK (www.coeliac.org.uk) provides excellent resources and support
- Ask your GP about gluten-free foods available on prescription
16. References
-
Ludvigsson JF, Bai JC, Biagi F, et al. Diagnosis and management of adult coeliac disease: guidelines from the British Society of Gastroenterology. Gut. 2014;63(8):1210-1228. doi:10.1136/gutjnl-2013-306578
-
Sollid LM, Jabri B. Triggers and drivers of autoimmunity: lessons from coeliac disease. Nat Rev Immunol. 2013;13(4):294-302. doi:10.1038/nri3407
-
Abadie V, Sollid LM, Barreiro LB, Jabri B. Integration of genetic and immunological insights into a model of celiac disease pathogenesis. Annu Rev Immunol. 2011;29:493-525. doi:10.1146/annurev-immunol-040210-092915
-
Singh P, Arora A, Strand TA, et al. Global prevalence of celiac disease: systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2018;16(6):823-836.e2. doi:10.1016/j.cgh.2017.06.037
-
Caio G, Volta U, Sapone A, et al. Celiac disease: a comprehensive current review. BMC Med. 2019;17(1):142. doi:10.1186/s12916-019-1380-z
-
National Institute for Health and Care Excellence. Coeliac disease: recognition, assessment and management. NICE guideline [NG20]. 2015. Available at: https://www.nice.org.uk/guidance/ng20
-
Rubio-Tapia A, Hill ID, Kelly CP, Calderwood AH, Murray JA; American College of Gastroenterology. ACG clinical guidelines: diagnosis and management of celiac disease. Am J Gastroenterol. 2013;108(5):656-676. doi:10.1038/ajg.2013.79
-
Lebwohl B, Sanders DS, Green PHR. Coeliac disease. Lancet. 2018;391(10115):70-81. doi:10.1016/S0140-6736(17)31796-8
-
Villalta D, Tonutti E, Prause C, et al. IgG antibodies against deamidated gliadin peptides for diagnosis of celiac disease in patients with IgA deficiency. Clin Chem. 2010;56(3):464-468. doi:10.1373/clinchem.2009.128132
-
Marsh MN. Gluten, major histocompatibility complex, and the small intestine. A molecular and immunobiologic approach to the spectrum of gluten sensitivity ('celiac sprue'). Gastroenterology. 1992;102(1):330-354. doi:10.1016/0016-5085(92)91819-P
-
Fasano A. Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer. Physiol Rev. 2011;91(1):151-175. doi:10.1152/physrev.00003.2008
-
Zanchetta MB, Longobardi V, Bai JC. Bone and celiac disease. Curr Osteoporos Rep. 2016;14(2):43-48. doi:10.1007/s11914-016-0304-5
-
Comino I, Moreno Mde L, Real A, et al. The gluten-free diet: testing alternative cereals tolerated by celiac patients. Nutrients. 2013;5(10):4250-4268. doi:10.3390/nu5104250
-
Codex Alimentarius Commission. Standard for foods for special dietary use for persons intolerant to gluten. CODEX STAN 118-1979. Revised 2008.
-
Di Sabatino A, Rosado MM, Cazzola P, et al. Splenic hypofunction and the spectrum of autoimmune and malignant complications in celiac disease. Clin Gastroenterol Hepatol. 2006;4(2):179-186. doi:10.1016/S1542-3565(05)00982-1
-
Rubio-Tapia A, Murray JA. Classification and management of refractory coeliac disease. Gut. 2010;59(4):547-557. doi:10.1136/gut.2009.195131
-
Catassi C, Bearzi I, Holmes GK. Association of celiac disease and intestinal lymphomas and other cancers. Gastroenterology. 2005;128(4 Suppl 1):S79-S86. doi:10.1053/j.gastro.2005.02.027
-
Bolotin D, Petronic-Rosic V. Dermatitis herpetiformis. Part I. Epidemiology, pathogenesis, and clinical presentation. J Am Acad Dermatol. 2011;64(6):1017-1024. doi:10.1016/j.jaad.2010.09.777
-
Hadjivassiliou M, Sanders DS, Grünewald RA, Woodroofe N, Boscolo S, Aeschlimann D. Gluten sensitivity: from gut to brain. Lancet Neurol. 2010;9(3):318-330. doi:10.1016/S1474-4422(09)70290-X
-
Vriezinga SL, Auricchio R, Bravi E, et al. Randomized feeding intervention in infants at high risk for celiac disease. N Engl J Med. 2014;371(14):1304-1315. doi:10.1056/NEJMoa1404172
-
Husby S, Koletzko S, Korponay-Szabó I, et al. European Society Paediatric Gastroenterology, Hepatology and Nutrition Guidelines for Diagnosing Coeliac Disease 2020. J Pediatr Gastroenterol Nutr. 2020;70(1):141-156. doi:10.1097/MPG.0000000000002497
-
Green PHR, Cellier C. Celiac disease. N Engl J Med. 2007;357(17):1731-1743. doi:10.1056/NEJMra071600
-
Leffler DA, Green PHR, Fasano A. Extraintestinal manifestations of coeliac disease. Nat Rev Gastroenterol Hepatol. 2015;12(10):561-571. doi:10.1038/nrgastro.2015.131
-
Al-Toma A, Volta U, Auricchio R, et al. European Society for the Study of Coeliac Disease (ESsCD) guideline for coeliac disease and other gluten-related disorders. United European Gastroenterol J. 2019;7(5):583-613. doi:10.1177/2050640619844125
-
Ludvigsson JF, Montgomery SM, Ekbom A, Brandt L, Granath F. Small-intestinal histopathology and mortality risk in celiac disease. JAMA. 2009;302(11):1171-1178. doi:10.1001/jama.2009.1320
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate guidelines and specialists for patient care.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Small Bowel Physiology
- Immunology of the Gut
Differentials
Competing diagnoses and look-alikes to compare.
- Non-Coeliac Gluten Sensitivity
- Tropical Sprue
- Whipple Disease
Consequences
Complications and downstream problems to keep in mind.
- Osteoporosis
- Enteropathy-Associated T-cell Lymphoma