Gestational Diabetes Mellitus (GDM)
GDM is distinct from pre-existing diabetes mellitus (Type 1 or Type 2) diagnosed before pregnancy. The condition typically resolves within hours to days of placental delivery, as the primary driver of insulin...
What matters first
GDM is distinct from pre-existing diabetes mellitus (Type 1 or Type 2) diagnosed before pregnancy. The condition typically resolves within hours to days of placental delivery, as the primary driver of insulin...
Fasting Glucose >=7.0 mmol/L at Diagnosis - Start Insulin Immediately
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Fasting Glucose >=7.0 mmol/L at Diagnosis - Start Insulin Immediately
- Macrosomia with Abdominal Circumference less than 95th Centile
- Polyhydramnios (AFI less than 25 cm)
- Reduced Fetal Movements
Linked comparisons
Differentials and adjacent topics worth opening next.
- Pre-existing Type 2 Diabetes
- Pre-existing Type 1 Diabetes
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Gestational Diabetes Mellitus (GDM)
1. Clinical Overview
Definition
Gestational Diabetes Mellitus (GDM) is defined as carbohydrate intolerance of variable severity with onset or first recognition during pregnancy, typically diagnosed between 24-28 weeks of gestation. [1,2] The condition is characterised by insulin resistance exceeding the compensatory capacity of pancreatic beta-cells, resulting in maternal hyperglycaemia that affects both maternal and fetal outcomes.
GDM is distinct from pre-existing diabetes mellitus (Type 1 or Type 2) diagnosed before pregnancy. The condition typically resolves within hours to days of placental delivery, as the primary driver of insulin resistance - placental hormones - is removed. [3]
Clinical Significance
GDM represents one of the most common medical complications of pregnancy, with profound implications for both immediate perinatal outcomes and long-term metabolic health. The landmark HAPO Study (Hyperglycemia and Adverse Pregnancy Outcomes) established that there is a continuous, graded relationship between maternal glucose levels and adverse pregnancy outcomes, with no clear threshold below which risk disappears. [4]
The ACHOIS Trial (Australian Carbohydrate Intolerance Study in Pregnant Women) demonstrated that active treatment of GDM significantly reduces serious perinatal morbidity, including a reduction in composite adverse outcomes from 4% to 1% (adjusted RR 0.33, 95% CI 0.14-0.75). [5]
Key Clinical Messages
The "5, 6, 7, 8" Rule for NICE Thresholds: Fasting ≥5.6 mmol/L, 2-Hour ≥7.8 mmol/L are the UK diagnostic thresholds.
Pregnancy as Metabolic Stress Test: GDM unmasks a pre-existing metabolic vulnerability. Women with GDM have a 7-fold increased risk of developing Type 2 DM within 10 years compared to women with normoglycaemic pregnancies. [6]
Metformin is Safe: The MiG Trial confirmed that metformin is non-inferior to insulin for glycaemic control and perinatal outcomes, with 76.6% of women preferring metformin treatment. [7]
Window of Prevention: The postpartum period represents a critical opportunity for lifestyle intervention to prevent progression to T2DM, with intensive lifestyle modification reducing 10-year diabetes incidence by 35-58%. [8]
2. Epidemiology
Incidence and Prevalence
The global prevalence of GDM varies significantly based on population demographics, diagnostic criteria used, and screening strategies employed.
| Population | Prevalence | Reference |
|---|---|---|
| United Kingdom (NICE criteria) | 5-8% | [1] |
| Europe (IADPSG criteria) | 10-15% | [9] |
| South Asia / Middle East | 15-25% | [10] |
| Indigenous Australian | 12-18% | [11] |
| United States (ACOG criteria) | 6-9% | [12] |
| Global (IDF estimate) | 14% (1 in 7 births) | [13] |
The prevalence of GDM has increased substantially over the past three decades, paralleling the global epidemics of obesity and Type 2 diabetes. Application of the more sensitive IADPSG criteria increases diagnostic rates by 2-3 fold compared to traditional two-step approaches. [9]
Risk Factors
Risk factors for GDM reflect the underlying pathophysiology of insulin resistance and beta-cell dysfunction. The NICE guideline (NG3) recommends selective screening based on the presence of risk factors, with OGTT offered if any one of the following is present. [1]
Major Risk Factors (Indications for OGTT Screening - NICE NG3):
| Risk Factor | Relative Risk | Evidence Level |
|---|---|---|
| BMI > 30 kg/m² | 3.6 (3.3-3.9) | Level I [14] |
| Previous macrosomic baby (> 4.5 kg) | 2.8 (2.4-3.2) | Level II |
| Previous GDM | 30-50% recurrence | Level II [15] |
| First-degree relative with diabetes | 2.3 (2.0-2.6) | Level II |
| High-risk ethnicity (South Asian, Black Caribbean, Middle Eastern) | 2.0-4.0 | Level II [10] |
| Polycystic Ovary Syndrome (PCOS) | 2.9 (1.7-5.1) | Level II [16] |
Additional Risk Factors:
- Maternal age > 35 years
- Previous unexplained stillbirth
- Glycosuria (2+ on one occasion, or 1+ on two occasions)
- Polyhydramnios or macrosomia in current pregnancy
- Multiple pregnancy (higher placental mass)
Demographic Trends
The increasing prevalence of GDM reflects several demographic shifts:
- Rising maternal age: Mean maternal age at first birth has increased from 24 to 29 years over three decades
- Obesity epidemic: Pre-pregnancy BMI > 30 kg/m² has increased from 10% to 22% of pregnancies
- Changing population demographics: Immigration from high-risk ethnic populations
- Lower diagnostic thresholds: Adoption of IADPSG/WHO criteria in many settings
3. Pathophysiology
The Diabetogenic State of Pregnancy
Normal pregnancy induces a state of progressive insulin resistance that serves to redirect maternal glucose to the fetus. This physiological adaptation becomes pathological in GDM when beta-cell compensation is inadequate.
Hormonal Drivers of Insulin Resistance:
| Hormone | Source | Peak Level | Mechanism of Insulin Resistance |
|---|---|---|---|
| Human Placental Lactogen (hPL) | Syncytiotrophoblast | 3rd trimester | Blocks insulin receptor signalling in muscle/adipose |
| Placental Growth Hormone | Placenta | Progressive increase | Reduces insulin sensitivity by 50% |
| Cortisol | Adrenal (increased production) | 3-fold rise | Hepatic gluconeogenesis, peripheral resistance |
| Progesterone | Corpus luteum, placenta | Progressive rise | Reduces glucose transporter expression |
| TNF-alpha | Placenta, adipose | Increased | Impairs insulin receptor substrate phosphorylation |
The net effect is a 50-60% reduction in maternal insulin sensitivity by the third trimester. [17] In women with normal glucose tolerance, this is compensated by a 2-3 fold increase in insulin secretion. GDM occurs when this compensatory hyperinsulinaemia is insufficient.
Beta-Cell Dysfunction
Women who develop GDM demonstrate impaired beta-cell function that predates pregnancy and persists postpartum. Studies demonstrate that women with GDM have:
- 50-67% reduction in first-phase insulin response
- Reduced beta-cell glucose sensitivity
- Defective proinsulin processing
This represents a pre-existing metabolic vulnerability that is "unmasked" by the diabetogenic stress of pregnancy, explaining the high rate of progression to T2DM postpartum.
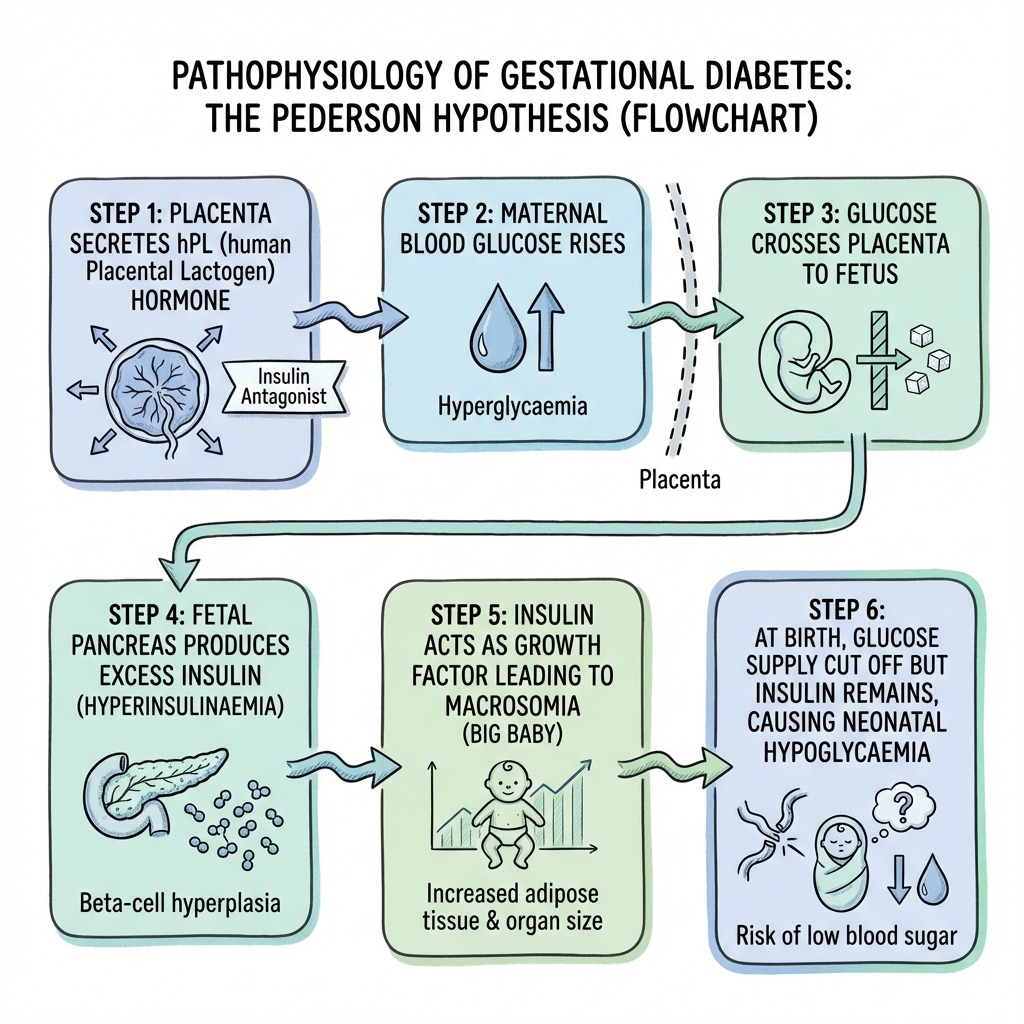
The Pedersen Hypothesis
The fetal effects of maternal hyperglycaemia are explained by the Pedersen Hypothesis, established in 1952 and validated by the HAPO Study. [4]
┌────────────────────────────────────────────────────────────────────────────┐
│ THE PEDERSEN HYPOTHESIS │
├────────────────────────────────────────────────────────────────────────────┤
│ │
│ 1. MATERNAL HYPERGLYCAEMIA │
│ ↓ │
│ Glucose crosses placenta freely (GLUT-1 transporter) │
│ ↓ │
│ 2. FETAL HYPERGLYCAEMIA │
│ ↓ │
│ Fetal pancreas detects elevated glucose │
│ ↓ │
│ 3. FETAL HYPERINSULINAEMIA │
│ ↓ │
│ Insulin acts as a GROWTH FACTOR in fetal tissues │
│ ↓ │
│ 4. FETAL OVERGROWTH (MACROSOMIA) │
│ • Increased fat deposition (especially truncal/abdominal) │
│ • Organomegaly (liver, heart, adrenals) │
│ • Increased shoulder-to-head ratio → Shoulder Dystocia Risk │
│ │
│ 5. AT BIRTH: GLUCOSE SUPPLY IS CUT │
│ • Hyperinsulinaemia persists → NEONATAL HYPOGLYCAEMIA │
│ • Insulin inhibits surfactant → RESPIRATORY DISTRESS │
│ • Insulin increases oxygen consumption → POLYCYTHAEMIA │
│ │
└────────────────────────────────────────────────────────────────────────────┘
Critical Concept: Unlike congenital malformations (which occur in pre-existing diabetes during organogenesis), the complications of GDM occur in the second and third trimesters and are therefore related to growth and metabolic programming rather than structural defects.
Molecular Mechanisms
Exam Detail: Insulin Signalling Pathway Defects in GDM:
- Reduced IRS-1 phosphorylation: Serine phosphorylation (inhibitory) is increased, tyrosine phosphorylation (activating) is decreased
- PI3K pathway impairment: Reduced GLUT-4 translocation to cell membrane
- Inflammatory cytokine effects: TNF-alpha, IL-6 from placenta/adipose tissue activate JNK pathway
- Lipotoxicity: Elevated free fatty acids impair insulin signalling
- Adipokine dysregulation: Reduced adiponectin, increased leptin resistance
Placental Transport:
- Glucose: Facilitated diffusion via GLUT-1 (concentration-dependent, maternal-to-fetal gradient)
- Insulin: Does NOT cross placenta (large peptide hormone)
- Fatty acids: Cross placenta; contribute to fetal adiposity
4. Clinical Presentation
Symptoms
The majority of women with GDM are asymptomatic. GDM is detected through systematic screening rather than symptom-driven investigation.
When symptomatic (indicating more severe hyperglycaemia):
| Symptom | Frequency | Pathophysiology |
|---|---|---|
| Polyuria | Occasional | Osmotic diuresis from glucosuria |
| Polydipsia | Occasional | Compensatory fluid intake |
| Fatigue | Common (non-specific) | Cellular energy deficit |
| Recurrent vaginal thrush | Moderate | Candida overgrowth in glucose-rich environment |
| Blurred vision | Rare | Osmotic lens changes |
Signs
Physical examination findings are typically absent in early GDM. Signs may develop with disease progression or poor control:
| Finding | Clinical Significance |
|---|---|
| Fundal height greater than dates | Suggests macrosomia/polyhydramnios |
| Glucosuria (urine dipstick) | Low sensitivity (~20%) for GDM |
| Elevated blood pressure | Associated pre-eclampsia risk |
| Excessive weight gain | Risk factor and consequence |
Red Flags Requiring Urgent Action
[!CAUTION] Urgent Referral Indications:
- Fasting glucose ≥7.0 mmol/L at diagnosis → Immediate insulin
- Polyhydramnios on ultrasound (AFI > 25 cm)
- Fetal abdominal circumference > 95th centile
- Reduced fetal movements → Same-day assessment
- Signs of pre-eclampsia (hypertension + proteinuria)
5. Investigations
Screening Strategy: One-Step vs Two-Step Approach
There is international variation in GDM screening approaches, reflecting ongoing debate about optimal diagnostic thresholds.
UK/NICE Approach (Risk Factor-Based, One-Step OGTT):
| Step | Action | Timing |
|---|---|---|
| Booking | Identify risk factors | First antenatal visit |
| If risk factors present | 75g OGTT | 24-28 weeks |
| If previous GDM | 75g OGTT | At booking AND 24-28 weeks |
| High suspicion (polyhydramnios, macrosomia) | 75g OGTT | Any gestation |
IADPSG/WHO One-Step Approach (Universal Screening):
All women undergo 75g OGTT at 24-28 weeks, regardless of risk factors. [2]
ACOG Two-Step Approach (US):
- 50g glucose challenge test (GCT) - non-fasting screening
- If GCT positive (≥7.2 or ≥7.8 mmol/L), proceed to 100g 3-hour OGTT
Diagnostic Criteria Comparison
| Criteria | Fasting (mmol/L) | 1-Hour (mmol/L) | 2-Hour (mmol/L) | Basis |
|---|---|---|---|---|
| NICE (UK) | ≥5.6 | - | ≥7.8 | Cost-effectiveness |
| IADPSG/WHO | ≥5.1 | ≥10.0 | ≥8.5 | HAPO Study OR 1.75 |
| Carpenter-Coustan (US) | ≥5.3 | ≥10.0 | ≥8.6 | 3-hour OGTT |
Critical Point: The NICE criteria are more specific (fewer diagnoses) while IADPSG criteria are more sensitive (more diagnoses). NICE adopted modified thresholds based on UK-specific cost-effectiveness analysis. [1,4]
Performing the 75g OGTT
Protocol:
- Patient must be fasting (8-14 hours, water permitted)
- Take fasting venous plasma glucose
- Patient drinks 75g anhydrous glucose dissolved in 250-300ml water within 5 minutes
- Patient rests (no eating, drinking, smoking, or exercise)
- Take 2-hour venous plasma glucose
- Interpret using NICE thresholds (Fasting ≥5.6 OR 2-Hour ≥7.8 = GDM)
Common Pitfalls:
- Patient not fasting → Invalid test, repeat
- Vomiting glucose solution → Invalid test, repeat in 2 weeks
- Using capillary blood glucose → Not accurate for diagnosis
Baseline Investigations at Diagnosis
| Investigation | Purpose |
|---|---|
| HbA1c | Distinguish GDM from undiagnosed T2DM (> 48 mmol/mol suggests pre-existing) |
| Renal function (U&Es, eGFR) | Baseline for metformin use |
| Urinalysis | Proteinuria screening for pre-eclampsia |
| Mid-stream urine culture | Asymptomatic bacteriuria common |
| Retinal screening | If concern for pre-existing diabetes |
Self-Monitoring of Blood Glucose (SMBG)
Frequency: Fasting + 1-hour post-meal (QDS - 4 times daily)
NICE Targets:
| Time Point | Target |
|---|---|
| Fasting | less than 5.3 mmol/L |
| 1-hour post-meal | less than 7.8 mmol/L |
Alternative (if 2-hour post-meal used):
| Time Point | Target |
|---|---|
| 2-hour post-meal | less than 6.4 mmol/L |
6. Management
Management Algorithm (NICE NG3 Pathway)
┌─────────────────────────────────────────────────────────────────────────────┐
│ GDM DIAGNOSED ON 75g OGTT │
├─────────────────────────────────────────────────────────────────────────────┤
│ │
│ ASSESS: What is the FASTING GLUCOSE at Diagnosis? │
│ │
│ ┌─────────────────────────────────────────────────────────────────────┐ │
│ │ FASTING less than 7.0 mmol/L (and no complications) │ │
│ │ ↓ │ │
│ │ STEP 1: LIFESTYLE INTERVENTION (1-2 weeks trial) │ │
│ │ • Dietary modification (low GI, moderate carbohydrate) │ │
│ │ • Exercise (30 min walking daily after meals) │ │
│ │ • Self-monitoring blood glucose QDS │ │
│ │ ↓ │ │
│ │ IF targets NOT achieved: │ │
│ │ STEP 2: ADD METFORMIN │ │
│ │ • Start 500mg OD with food │ │
│ │ • Titrate to 500mg TDS or 850mg BD │ │
│ │ • Monitor for GI side effects │ │
│ │ ↓ │ │
│ │ IF targets STILL not achieved (or metformin intolerance): │ │
│ │ STEP 3: ADD/SWITCH TO INSULIN │ │
│ │ • Basal insulin if fasting elevated │ │
│ │ • Rapid-acting if post-prandial elevated │ │
│ │ • Titrate aggressively (review every 3-7 days) │ │
│ └─────────────────────────────────────────────────────────────────────┘ │
│ │
│ ┌─────────────────────────────────────────────────────────────────────┐ │
│ │ FASTING ≥ 7.0 mmol/L │ │
│ │ ↓ │ │
│ │ START INSULIN IMMEDIATELY │ │
│ │ • Diet alone will fail │ │
│ │ • Metformin alone likely insufficient │ │
│ │ • May add metformin to reduce insulin dose │ │
│ └─────────────────────────────────────────────────────────────────────┘ │
│ │
│ ┌─────────────────────────────────────────────────────────────────────┐ │
│ │ FASTING 6.0-6.9 mmol/L PLUS MACROSOMIA OR COMPLICATIONS │ │
│ │ ↓ │ │
│ │ CONSIDER IMMEDIATE INSULIN │ │
│ └─────────────────────────────────────────────────────────────────────┘ │
│ │
└─────────────────────────────────────────────────────────────────────────────┘
Step 1: Lifestyle Intervention
Dietary Principles:
- Carbohydrate modification: Low glycaemic index foods, distribute carbohydrate across meals
- Meal pattern: 3 small-moderate meals + 2-3 snacks to prevent glucose spikes
- Caloric intake: Moderate restriction only (avoid ketosis)
- Macronutrient distribution: 40-50% carbohydrate, 20% protein, 30-40% fat
Exercise Recommendations:
- 30 minutes moderate-intensity exercise daily (e.g., brisk walking)
- Particularly beneficial after meals (reduces postprandial glucose)
- Avoid supine exercise after 16 weeks
Ketone Avoidance:
[!WARNING] Avoid very low calorie or very low carbohydrate diets. Maternal ketonaemia has been associated with reduced offspring IQ in historical observational studies. While evidence is debated, standard practice is to ensure adequate carbohydrate intake.
Step 2: Metformin
The MiG Trial established metformin as a safe and effective first-line pharmacological option for GDM. [7]
| Aspect | Details |
|---|---|
| Mechanism | Reduces hepatic gluconeogenesis, improves insulin sensitivity |
| Starting dose | 500mg once daily with food |
| Titration | Increase by 500mg every 1-2 weeks |
| Maximum dose | 2500mg daily (usually 500mg TDS or 850mg BD) |
| Contraindications | eGFR less than 30, severe hepatic impairment, acute illness |
| Side effects | GI upset (nausea, diarrhoea) - minimise by slow titration |
| Placental transfer | Crosses placenta; no evidence of fetal harm [7] |
| Failure rate | ~46% require supplemental insulin |
Clinical Pearl: Women who fail metformin monotherapy typically have higher fasting glucose and lower beta-cell function at diagnosis - predictors of insulin requirement.
Step 3: Insulin
Indications for Insulin:
- Fasting glucose ≥7.0 mmol/L at diagnosis
- Fasting 6.0-6.9 mmol/L with macrosomia present
- Failure to achieve targets on metformin
- Metformin intolerance or contraindication
Insulin Regimens:
| Clinical Pattern | Insulin Strategy | Example Regimen |
|---|---|---|
| Fasting hyperglycaemia | Basal insulin (bedtime) | NPH 10 units at bedtime, titrate by 2-4 units every 3 days |
| Postprandial hyperglycaemia | Rapid-acting (pre-meal) | NovoRapid 4-6 units pre-meal |
| Both elevated | Basal-bolus | Combination of above |
Insulin Types:
| Type | Examples | Onset | Peak | Duration | Use in Pregnancy |
|---|---|---|---|---|---|
| Rapid-acting | Aspart (NovoRapid), Lispro (Humalog) | 10-20 min | 1-2 hrs | 3-5 hrs | Safe |
| Short-acting | Regular (Actrapid) | 30-60 min | 2-4 hrs | 5-8 hrs | Safe |
| Intermediate | NPH (Insulatard) | 1-2 hrs | 4-10 hrs | 12-18 hrs | Safe |
| Long-acting | Detemir (Levemir) | 1-2 hrs | Flat | 20-24 hrs | Safe |
| Ultra-long | Glargine (Lantus), Degludec | 1-2 hrs | Flat | 24+ hrs | Safe (limited data for Degludec) |
Titration Protocol:
- Review glucose diary every 3-7 days
- Increase insulin by 2-4 units if target not met
- Reduce by 2 units if hypoglycaemia occurs
- Aggressive titration is safe and necessary
Drug Safety in Pregnancy
| Drug Class | Examples | Safety | Evidence | Notes |
|---|---|---|---|---|
| Metformin | - | ✅ Safe | Level 1b (MiG Trial) | Crosses placenta, no harm |
| Insulin (all types) | See above | ✅ Safe | Extensive experience | Does NOT cross placenta |
| Glibenclamide | Glyburide | ⚠️ Second-line only | Level II | Crosses placenta, neonatal hypoglycaemia risk |
| SGLT2 inhibitors | Empagliflozin, etc. | ❌ Contraindicated | No data | Avoid in pregnancy |
| GLP-1 agonists | Semaglutide, etc. | ❌ Contraindicated | Animal teratogenicity | Avoid in pregnancy |
| Thiazolidinediones | Pioglitazone | ❌ Contraindicated | No data | Avoid in pregnancy |
Antenatal Surveillance
Ultrasound Monitoring:
| Timing | Assessment | Purpose |
|---|---|---|
| Diagnosis | Growth scan, AFI | Baseline |
| 28 weeks | Fetal growth, AC measurement | Macrosomia detection |
| 32 weeks | Growth, liquor volume | Trend assessment |
| 36 weeks | Growth, presentation, liquor | Delivery planning |
Key Parameters:
- Abdominal Circumference (AC): Most sensitive marker of fetal overgrowth
- Estimated Fetal Weight (EFW): > 90th centile = macrosomia concern
- Amniotic Fluid Index (AFI): > 25 cm = polyhydramnios
Fetal Wellbeing:
- CTG monitoring from 37 weeks (or earlier if concerns)
- Daily fetal movement awareness
- Serial growth scans to detect IUGR (rare but possible in severe early GDM)
Delivery Planning
| Glycaemic Control | Timing of Delivery | Mode |
|---|---|---|
| Diet-controlled, well-controlled | Await spontaneous labour, induce by 40+6 | Vaginal delivery preferred |
| Metformin or Insulin, well-controlled | Offer induction at 39+0-39+6 weeks | Vaginal delivery preferred |
| Poorly controlled or complications | Offer induction at 37+0-38+6 weeks | Individualise |
| EFW > 4.5 kg | Discuss elective caesarean section | C-section may reduce dystocia |
Intrapartum Glucose Management
Principles:
- Labour is a fasting state - glucose requirements change
- Target blood glucose: 4-7 mmol/L during labour
- Avoid maternal hypoglycaemia (risk of fetal distress)
- Avoid hyperglycaemia (increases neonatal hypoglycaemia risk)
Protocol:
┌────────────────────────────────────────────────────────────────────────┐
│ INTRAPARTUM GLUCOSE MANAGEMENT │
├────────────────────────────────────────────────────────────────────────┤
│ │
│ IF ON DIET ALONE: │
│ • Hourly capillary glucose monitoring │
│ • Usually stable - no intervention needed │
│ • VRIII only if persistently > 8 mmol/L │
│ │
│ IF ON METFORMIN: │
│ • Stop metformin on morning of induction/labour │
│ • Hourly glucose monitoring │
│ • VRIII if glucose persistently > 8 mmol/L │
│ │
│ IF ON INSULIN: │
│ • Stop long-acting insulin on admission │
│ • Hourly glucose monitoring │
│ • Variable Rate Intravenous Insulin Infusion (VRIII) │
│ • Co-infuse 10% Dextrose (125ml/hr) to prevent hypoglycaemia │
│ • Titrate insulin infusion to maintain glucose 4-7 mmol/L │
│ │
│ IMMEDIATELY POST-DELIVERY: │
│ • STOP all insulin infusions and medications │
│ • Insulin resistance resolves within hours of placental delivery │
│ • Check maternal glucose before discharge │
│ │
└────────────────────────────────────────────────────────────────────────┘
7. Complications
Fetal and Neonatal Complications
| Complication | Incidence (Uncontrolled) | Mechanism | Prevention/Management |
|---|---|---|---|
| Macrosomia (> 4000g) | 20-30% | Fetal hyperinsulinaemia → growth factor effect | Tight glycaemic control, serial growth scans |
| Large for Gestational Age (> 90th centile) | 25-40% | As above | As above |
| Shoulder Dystocia | 2-4% (up to 10% if macrosomic) | Increased shoulder-to-head ratio | McRoberts manoeuvre, consider C-section if EFW > 4.5kg |
| Birth trauma (brachial plexus injury, fractures) | 0.2-2% | Associated with shoulder dystocia | Skilled delivery management |
| Neonatal Hypoglycaemia | 25-50% | Persistent hyperinsulinaemia after cord clamping | Early feeding, glucose monitoring |
| Respiratory Distress Syndrome | 5-10% | Insulin inhibits surfactant production | Good glycaemic control, corticosteroids if preterm |
| Hyperbilirubinaemia | 10-20% | Increased RBC production and turnover | Monitor, phototherapy if needed |
| Polycythaemia | 5-10% | Chronic fetal hypoxia → EPO production | Monitor haematocrit |
| Hypocalcaemia | 5% | Delayed PTH response | Monitor, calcium supplementation |
| Stillbirth | 1-2% (uncontrolled) | Chronic fetal hypoxia, metabolic derangement | Tight control, fetal surveillance |
Shoulder Dystocia - Obstetric Emergency
Definition: Failure of delivery of fetal shoulders after delivery of the head using normal axial traction.
HELPERR Mnemonic for Management:
H - Help: Call for help (senior obstetrician, neonatology, anaesthesia)
E - Evaluate for Episiotomy: Consider to create space
L - Legs: McRoberts position (hyperflexion of maternal thighs onto abdomen)
P - Pressure: Suprapubic pressure (NOT fundal pressure - dangerous)
E - Enter: Internal manoeuvres (Rubin II - pressure on posterior shoulder)
R - Remove posterior arm: Delivery of posterior arm
R - Roll: All fours (Gaskin manoeuvre)
Neonatal Hypoglycaemia Management
Definition: Blood glucose less than 2.0 mmol/L (symptomatic) or less than 2.5 mmol/L (asymptomatic threshold for intervention)
Protocol:
- Check heel-prick glucose at 2-4 hours of age, before 2nd and 3rd feeds
- Ensure early, frequent feeding (within 30-60 minutes of birth)
- If glucose 1.5-2.5 mmol/L and asymptomatic: Feed and recheck
- If glucose less than 1.5 mmol/L or symptomatic: IV dextrose bolus
Symptoms of Neonatal Hypoglycaemia:
- Jitteriness, tremors
- Poor feeding, lethargy
- Hypotonia
- Apnoea, cyanosis
- Seizures (severe)
Maternal Complications
| Complication | Risk | Mechanism |
|---|---|---|
| Pre-eclampsia | 2-4x increased | Shared pathophysiology (endothelial dysfunction, insulin resistance) |
| Polyhydramnios | 10-20% | Fetal polyuria from hyperglycaemia |
| Preterm labour | Increased | Associated with polyhydramnios, infection |
| Caesarean section | 25-35% | Macrosomia, failed induction |
| Perineal trauma | Increased | Macrosomia, instrumental delivery |
| Postpartum haemorrhage | Increased | Uterine overdistension |
| Wound infection | Increased | Hyperglycaemia impairs wound healing |
The GDM-Pre-eclampsia Connection
GDM and pre-eclampsia share common pathophysiological pathways and frequently co-occur.
Exam Detail: Shared Mechanisms:
- Insulin resistance → Endothelial dysfunction
- Oxidative stress → Vascular damage
- Inflammatory cytokines (TNF-alpha, IL-6) → Systemic inflammation
- Dyslipidaemia → Atherosclerotic changes
- Obesity → Common risk factor for both
Clinical Implication: Women with GDM should have regular blood pressure monitoring and urinalysis for proteinuria. Low threshold for PlGF testing if hypertension develops.
8. Prognosis and Long-Term Outcomes
Natural History
GDM resolves within hours to days of placental delivery in > 95% of cases. The hyperglycaemia is driven by placental hormones - once the placenta is delivered, insulin resistance rapidly normalises.
However, GDM is a powerful marker of future metabolic disease.
Long-Term Maternal Outcomes
| Outcome | Incidence | Timeframe | Evidence |
|---|---|---|---|
| Type 2 Diabetes | 35-60% | 10-20 years | Meta-analysis: RR 7.43 (4.79-11.51) [6] |
| Recurrent GDM | 30-50% | Next pregnancy | [15] |
| Metabolic Syndrome | 2-3x increased | 10+ years | [18] |
| Cardiovascular Disease | 1.5-2x increased | 15+ years | [18] |
| Gestational Hypertension (future) | Increased | Next pregnancy | [18] |
Postpartum Testing Protocol
┌────────────────────────────────────────────────────────────────────────┐
│ POSTPARTUM PROTOCOL │
├────────────────────────────────────────────────────────────────────────┤
│ │
│ IMMEDIATELY POSTPARTUM: │
│ • Stop all GDM medications (metformin, insulin) │
│ • Insulin resistance resolves rapidly │
│ • Check fasting glucose before discharge │
│ │
│ 6-13 WEEKS POSTPARTUM: │
│ • Fasting plasma glucose OR 75g OGTT (NICE recommendation) │
│ • HbA1c (alternative if OGTT not feasible) │
│ • Interpret using standard non-pregnant criteria: │
│ - Normal: FPG less than 6.1 mmol/L and 2h-PG less than 7.8 mmol/L │
│ - IFG: FPG 6.1-6.9 mmol/L │
│ - IGT: 2h-PG 7.8-11.0 mmol/L │
│ - Diabetes: FPG ≥7.0 mmol/L or 2h-PG ≥11.1 mmol/L │
│ │
│ ANNUAL SCREENING (LIFELONG): │
│ • HbA1c or fasting glucose annually │
│ • Risk factor assessment (weight, BP, lipids) │
│ • Lifestyle counselling reinforcement │
│ │
│ PRE-CONCEPTION (FUTURE PREGNANCIES): │
│ • HbA1c or fasting glucose BEFORE conception │
│ • Exclude T2DM before next pregnancy │
│ • Optimise weight and metabolic health │
│ │
└────────────────────────────────────────────────────────────────────────┘
Prevention of Type 2 Diabetes
The postpartum period represents a critical "Window of Opportunity" for diabetes prevention.
Evidence-Based Prevention Strategies:
| Intervention | Risk Reduction | Evidence |
|---|---|---|
| Intensive lifestyle (diet + exercise) | 35-58% | Diabetes Prevention Program [8] |
| Breastfeeding (≥3 months) | 25-47% | Multiple observational studies |
| Weight loss (5-10% body weight) | 40-50% | DPP subgroup analysis |
| Metformin (high-risk) | 20-30% | DPP (less effective than lifestyle) |
Key Counselling Messages:
- GDM is a "stress test" that revealed metabolic vulnerability
- Not inevitable to develop T2DM - modifiable with lifestyle
- Breastfeeding is protective - encourage exclusive breastfeeding
- Weight management is crucial - even modest weight loss is beneficial
- Annual screening is essential - early detection enables prevention
Offspring Long-Term Outcomes
Children born to mothers with GDM have increased risk of:
| Outcome | Risk Increase | Evidence Level |
|---|---|---|
| Childhood obesity | OR 1.5-2.0 | Level II |
| Type 2 diabetes | OR 2.0-4.0 | Level II |
| Metabolic syndrome | Increased | Level III |
| Neurodevelopmental differences | Debated | Level IV |
This represents "metabolic programming" or "developmental origins of health and disease" (DOHaD) - fetal hyperinsulinaemia may permanently alter appetite regulation and metabolism.
9. Screening Criteria Comparison
NICE vs IADPSG Criteria
| Aspect | NICE (UK) | IADPSG/WHO (International) |
|---|---|---|
| Screening approach | Risk factor-based | Universal |
| Test | 75g 2-point OGTT | 75g 3-point OGTT |
| Fasting threshold | ≥5.6 mmol/L | ≥5.1 mmol/L |
| 1-hour threshold | Not used | ≥10.0 mmol/L |
| 2-hour threshold | ≥7.8 mmol/L | ≥8.5 mmol/L |
| Diagnostic rule | ONE abnormal value | ONE abnormal value |
| Prevalence diagnosed | 5-8% | 15-25% |
| Evidence basis | Cost-effectiveness analysis | HAPO Study OR 1.75 |
NICE Rationale: The IADPSG thresholds would diagnose 2-3x more women, with uncertain benefit for additional diagnoses and significant resource implications. NICE adopted thresholds based on UK-specific cost-effectiveness modelling. [1]
IADPSG Rationale: The HAPO Study demonstrated continuous risk with no threshold. IADPSG set thresholds at OR 1.75 for adverse outcomes relative to the mean, aiming to identify all women who would benefit from intervention. [4]
Distinguishing GDM from Pre-existing Diabetes
A common exam scenario is distinguishing GDM from undiagnosed pre-existing diabetes:
| Feature | Pre-existing Diabetes | Gestational Diabetes |
|---|---|---|
| Timing of diagnosis | less than 20 weeks or before pregnancy | 24-28 weeks |
| HbA1c at booking | ≥48 mmol/mol (suggests pre-existing) | Normal (less than 42 mmol/mol) |
| Fasting glucose at booking | ≥7.0 mmol/L | Normal |
| Retinopathy | May be present | Absent |
| Congenital malformations | Increased risk (organogenesis exposure) | NOT increased |
| Postpartum glucose | Remains abnormal | Normalises |
| Requires postnatal testing | For diabetes control | To exclude persistent diabetes |
10. Evidence and Guidelines
Key Guidelines
| Guideline | Organisation | Year | Key Recommendations |
|---|---|---|---|
| NG3: Diabetes in Pregnancy | NICE | 2015 (Updated 2020) | Risk-factor screening, 5.6/7.8 thresholds, metformin first-line |
| GTG No. 5 | RCOG | 2011 | Supports NICE approach |
| WHO Guideline | WHO | 2013 | IADPSG criteria endorsed |
| Practice Bulletin 190 | ACOG | 2018 | Two-step screening, Carpenter-Coustan criteria |
| ADA Standards of Care | ADA | 2024 | Supports one-step IADPSG or two-step approach |
Landmark Trials
1. HAPO Study (2008) - Hyperglycemia and Adverse Pregnancy Outcomes [4]
- Design: Prospective observational study, 25,505 women, 15 centres, 9 countries
- Key Finding: Continuous, graded association between maternal glucose (at all levels tested) and adverse outcomes including birth weight > 90th centile, primary C-section, neonatal hypoglycaemia, cord C-peptide > 90th centile
- Impact: No clear glucose threshold; led to IADPSG criteria based on OR 1.75 for outcomes
- Citation: HAPO Study Cooperative Research Group. N Engl J Med. 2008;358(19):1991-2002. doi: 10.1056/NEJMoa0707943
2. MiG Trial (2008) - Metformin in Gestational Diabetes [7]
- Design: RCT, 751 women with GDM, metformin vs insulin
- Key Finding: Metformin (± supplemental insulin) non-inferior to insulin for composite perinatal outcome; 76.6% of women preferred metformin; 46.3% required supplemental insulin
- Impact: Established metformin as safe and effective first-line oral agent
- Citation: Rowan JA et al. N Engl J Med. 2008;358(19):2003-2015. doi: 10.1056/NEJMoa0707193
3. ACHOIS Trial (2005) - Australian Carbohydrate Intolerance Study in Pregnant Women [5]
- Design: RCT, 1000 women with mild GDM, treatment vs routine care
- Key Finding: Treatment reduced serious perinatal complications from 4% to 1% (adjusted RR 0.33); higher induction rate but similar C-section rate; improved maternal quality of life
- Impact: Proved that treating mild GDM improves outcomes; justified screening programs
- Citation: Crowther CA et al. N Engl J Med. 2005;352(24):2477-86. doi: 10.1056/NEJMoa042973
4. IADPSG Consensus (2010) [2]
- Design: International consensus panel based on HAPO Study data
- Key Finding: Proposed new diagnostic criteria (fasting ≥5.1, 1h ≥10.0, 2h ≥8.5) based on OR 1.75 for adverse outcomes
- Impact: Adopted by WHO; ongoing debate about cost-effectiveness
- Citation: IADPSG Consensus Panel. Diabetes Care. 2010;33(3):676-82. doi: 10.2337/dc09-1848
11. Examination Focus
Common Viva Questions and Model Answers
Q1: A 28-week pregnant woman, BMI 32, had a previous baby weighing 4.8kg. What investigation would you arrange?
Model Answer: "I would arrange a 75g Oral Glucose Tolerance Test. This patient has two risk factors for gestational diabetes - BMI > 30 kg/m² and previous macrosomia > 4.5kg. According to NICE NG3, women with any one risk factor should be offered screening. I would ensure she understands the need for an 8-14 hour fast, and interpret the results using NICE thresholds - fasting ≥5.6 mmol/L or 2-hour ≥7.8 mmol/L indicates GDM."
Q2: An OGTT shows Fasting 6.1 mmol/L, 2-hour 8.2 mmol/L. What is the diagnosis and management?
Model Answer: "This patient has Gestational Diabetes - both values are above NICE thresholds. The fasting of 6.1 is elevated but less than 7.0, so I would recommend a 1-2 week trial of lifestyle intervention including dietary modification with low GI foods, 30 minutes daily exercise, and self-monitoring of blood glucose four times daily targeting fasting less than 5.3 and 1-hour post-meal less than 7.8. If targets are not achieved, I would add metformin starting at 500mg once daily with food, titrating to 500mg three times daily. If still uncontrolled, insulin would be required."
Q3: An OGTT shows Fasting 7.4 mmol/L. What is your action?
Model Answer: "With a fasting glucose of 7.4 mmol/L, I would start insulin immediately. NICE guidance is clear that when fasting glucose is ≥7.0 mmol/L at diagnosis, lifestyle modification alone will not achieve control, and even metformin monotherapy is likely to be insufficient. I would start basal insulin, typically NPH insulin at bedtime, and titrate aggressively to achieve fasting targets. Metformin can be added to reduce insulin requirements and provide daytime cover."
Q4: A newborn of a GDM mother is irritable and jittery at 3 hours. What is the diagnosis and action?
Model Answer: "The most likely diagnosis is neonatal hypoglycaemia. I would immediately check a heel-prick blood glucose. If the glucose is less than 2.0 mmol/L or the baby is symptomatic, this requires urgent management with IV dextrose - typically 2ml/kg of 10% dextrose as a bolus. If the glucose is between 2.0-2.5 mmol/L and the baby can feed, I would encourage immediate breastfeeding and recheck the glucose in 30 minutes. This baby requires close monitoring for the first 24 hours as neonatal hyperinsulinaemia persists despite removal of the maternal glucose supply."
Q5: A woman attends her 6-week postnatal check after GDM. How do you confirm resolution?
Model Answer: "I would arrange a fasting plasma glucose to confirm that GDM has resolved. The target is less than 6.1 mmol/L for normal glucose tolerance. Alternatively, an HbA1c can be performed if a fasting test is not feasible. I would also counsel her about the increased lifetime risk of Type 2 diabetes - approximately 50% over 10-20 years - and the importance of lifelong annual screening. I would emphasise lifestyle modifications including weight management, regular exercise, and healthy diet as evidence-based strategies to prevent diabetes. If she is planning further pregnancies, I would recommend pre-conception glucose testing to exclude T2DM before the next pregnancy."
Examiner Follow-Up Questions to Anticipate
- "What is the evidence base for treating GDM?" → ACHOIS Trial (67% reduction in serious perinatal morbidity)
- "What is the difference between NICE and IADPSG criteria?" → Thresholds and universal vs risk-factor screening
- "What is the mechanism of metformin in pregnancy?" → Reduces hepatic gluconeogenesis, improves insulin sensitivity
- "What is the Pedersen hypothesis?" → Maternal hyperglycaemia → fetal hyperglycaemia → fetal hyperinsulinaemia → macrosomia
- "What are the long-term risks for the mother?" → 50% T2DM risk, increased cardiovascular disease
- "When would you deliver this woman?" → Diet-controlled 40+6, on medication 38-39 weeks
12. Patient/Layperson Explanation
What is Gestational Diabetes?
Gestational diabetes is a type of diabetes that develops during pregnancy. It means your blood sugar levels are higher than normal, but this usually only happens while you are pregnant. For most women, it goes away after the baby is born.
Why Does It Happen?
During pregnancy, your placenta (the organ that feeds your baby) makes hormones that help your baby grow. These hormones also make it harder for your body's insulin to work properly. For most women, the body can make extra insulin to cope. But if it can't, your blood sugar rises - this is gestational diabetes.
How Is It Treated?
Step 1 - Healthy Eating and Activity:
- Eating regular, balanced meals with less sugar and white carbohydrates
- Walking for 30 minutes daily, especially after meals
- Checking your blood sugar levels at home with finger-prick tests
Step 2 - Medication (if diet isn't enough):
- Metformin tablets - safe and effective, taken with meals
- Insulin injections - if tablets don't work well enough
Will My Baby Be Affected?
With good blood sugar control, most babies are perfectly healthy. The main risk is your baby growing larger than normal, which can make birth more difficult. After birth, your baby's blood sugar will be checked as it can dip low for a few hours until their body adjusts.
Will I Be Diabetic Forever?
No - gestational diabetes almost always goes away after your baby is born. However, it is a warning sign that you are at higher risk of developing Type 2 diabetes later in life. Staying active, eating well, and maintaining a healthy weight is the best way to prevent this. You should have a blood test at 6 weeks after delivery and then annually for life.
Key Messages
- "It's not your fault" - GDM is caused by pregnancy hormones, not something you did wrong
- With good control, outcomes are excellent for both you and your baby
- It goes away after delivery, but be aware of your future risk
- Healthy lifestyle now can prevent diabetes in the future
- Breastfeeding is good for you and helps reduce your diabetes risk
13. References
-
National Institute for Health and Care Excellence. Diabetes in pregnancy: management from preconception to the postnatal period (NG3). 2015 (Updated 2020). Available from: https://www.nice.org.uk/guidance/ng3
-
International Association of Diabetes and Pregnancy Study Groups Consensus Panel. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care. 2010;33(3):676-682. doi: 10.2337/dc09-1848
-
American Diabetes Association Professional Practice Committee. Management of diabetes in pregnancy: Standards of Care in Diabetes-2024. Diabetes Care. 2024;47(Suppl 1):S282-S294. doi: 10.2337/dc24-S015
-
HAPO Study Cooperative Research Group. Hyperglycemia and adverse pregnancy outcomes. N Engl J Med. 2008;358(19):1991-2002. doi: 10.1056/NEJMoa0707943
-
Crowther CA, Hiller JE, Moss JR, et al. Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N Engl J Med. 2005;352(24):2477-2486. doi: 10.1056/NEJMoa042973
-
Bellamy L, Casas JP, Hingorani AD, Williams D. Type 2 diabetes mellitus after gestational diabetes: a systematic review and meta-analysis. Lancet. 2009;373(9677):1773-1779. doi: 10.1016/S0140-6736(09)60731-5
-
Rowan JA, Hague WM, Gao W, et al. Metformin versus insulin for the treatment of gestational diabetes. N Engl J Med. 2008;358(19):2003-2015. doi: 10.1056/NEJMoa0707193
-
Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393-403. doi: 10.1056/NEJMoa012512
-
Agarwal MM. Gestational diabetes mellitus: an update on the current international diagnostic criteria. World J Diabetes. 2015;6(6):782-791. doi: 10.4239/wjd.v6.i6.782
-
Guariguata L, Linnenkamp U, Beagley J, et al. Global estimates of the prevalence of hyperglycaemia in pregnancy. Diabetes Res Clin Pract. 2014;103(2):176-185. doi: 10.1016/j.diabres.2013.11.003
-
Chamberlain C, McNamara B, Williams ED, et al. Diabetes in pregnancy among indigenous women in Australia, Canada, New Zealand and the United States. Diabetes Metab Res Rev. 2013;29(4):241-256. doi: 10.1002/dmrr.2389
-
Deputy NP, Kim SY, Conrey EJ, Bullard KM. Prevalence and changes in preexisting diabetes and gestational diabetes among women who had a live birth - United States, 2012-2016. MMWR Morb Mortal Wkly Rep. 2018;67(43):1201-1207. doi: 10.15585/mmwr.mm6743a2
-
International Diabetes Federation. IDF Diabetes Atlas, 10th edition. Brussels, Belgium: IDF; 2021. Available from: https://diabetesatlas.org
-
Chu SY, Callaghan WM, Kim SY, et al. Maternal obesity and risk of gestational diabetes mellitus. Diabetes Care. 2007;30(8):2070-2076. doi: 10.2337/dc06-2559a
-
Kim C, Berger DK, Chamany S. Recurrence of gestational diabetes mellitus: a systematic review. Diabetes Care. 2007;30(5):1314-1319. doi: 10.2337/dc06-2517
-
Lo JC, Feigenbaum SL, Escobar GJ, et al. Increased prevalence of gestational diabetes mellitus among women with diagnosed polycystic ovary syndrome. Diabetes Care. 2006;29(8):1915-1917. doi: 10.2337/dc06-0877
-
Catalano PM. Trying to understand gestational diabetes. Diabet Med. 2014;31(3):273-281. doi: 10.1111/dme.12381
-
Kramer CK, Campbell S, Retnakaran R. Gestational diabetes and the risk of cardiovascular disease in women: a systematic review and meta-analysis. Diabetologia. 2019;62(6):905-914. doi: 10.1007/s00125-019-4840-2
-
Landon MB, Spong CY, Thom E, et al. A multicenter, randomized trial of treatment for mild gestational diabetes. N Engl J Med. 2009;361(14):1339-1348. doi: 10.1056/NEJMoa0902430
-
Poolsup N, Suksomboon N, Amin M. Efficacy and safety of oral antidiabetic drugs in comparison to insulin in treating gestational diabetes mellitus: a meta-analysis. PLoS One. 2014;9(10):e109985. doi: 10.1371/journal.pone.0109985
14. Special Clinical Scenarios
Multiple Pregnancy (Twins/Higher Order)
Women with multiple pregnancy have increased risk of GDM due to greater placental mass and higher levels of insulin-antagonistic hormones.
Key Considerations:
| Aspect | Singleton | Multiple Pregnancy |
|---|---|---|
| GDM prevalence | 5-8% | 10-15% |
| Insulin requirements | Standard | Often 30-50% higher |
| Growth monitoring | Standard schedule | More frequent (every 2-3 weeks) |
| Delivery timing | 39 weeks if on medication | Earlier (36-37 weeks for DCDA, 34-36 for MCDA) |
Obesity and GDM
Maternal obesity (BMI > 30 kg/m²) is the strongest modifiable risk factor for GDM.
Management Modifications:
- Metformin may be preferred (insulin-sensitising effects)
- Higher insulin doses typically required (0.8-1.2 units/kg vs 0.5-0.7 units/kg)
- Consider continuous glucose monitoring for improved control
- Anaesthetic assessment before labour (regional anaesthesia planning)
- Higher threshold for caesarean section consideration
- Enhanced VTE prophylaxis postpartum
GDM in Subsequent Pregnancies
Women with prior GDM have 30-50% recurrence risk.
Pre-conception Counselling:
- Pre-pregnancy glucose testing (FPG or HbA1c) to exclude T2DM
- Weight optimisation - 5-10% weight loss significantly reduces recurrence
- Early booking and early OGTT (at booking AND 24-28 weeks)
- Consider metformin pre-conception if high-risk (research ongoing)
Contraception After GDM
All contraceptive methods are available to women with resolved GDM.
| Method | Considerations |
|---|---|
| Progesterone-only methods | Safe; no metabolic concerns |
| Combined hormonal | Safe in resolved GDM; reassess if T2DM develops |
| Intrauterine devices | Excellent option; no metabolic effects |
| Barrier methods | No medical restrictions |
Key Message: Avoid unplanned pregnancy before confirming glucose status - pre-conception glucose testing is essential before each pregnancy.
15. Quality Standards and Audit
NICE Quality Standards (QS109)
| Quality Statement | Target | Audit Measure |
|---|---|---|
| Women with risk factors offered OGTT at 24-28 weeks | 100% | Proportion screened |
| Women with GDM receive self-monitoring training within 1 week | 100% | Time from diagnosis to SMBG start |
| Women on medication offered induction at 37-39 weeks | 100% | Gestation at delivery |
| 6-week postnatal glucose test offered | > 80% | Postnatal testing uptake |
| Annual HbA1c offered to women with prior GDM | > 80% | Long-term follow-up rate |
Key Performance Indicators
Process Measures:
- OGTT completion rate in eligible women
- Time from positive OGTT to specialist review
- Proportion achieving glycaemic targets
- Stillbirth rate in GDM population
- Macrosomia rate (> 4000g)
- Shoulder dystocia incidence
Outcome Measures:
- Composite neonatal morbidity rate
- Neonatal hypoglycaemia requiring IV treatment
- NICU admission rate
- Caesarean section rate
- Postnatal glucose testing uptake
- T2DM incidence at 1 year and 5 years
16. Emerging Evidence and Future Directions
Continuous Glucose Monitoring (CGM)
Emerging evidence suggests CGM may improve glycaemic control and outcomes in GDM, though current evidence is from small trials.
Potential Benefits:
- Real-time glucose trends
- Reduced hypoglycaemia
- Improved time in range
- Better identification of postprandial patterns
Current Status: Not routinely recommended by NICE; ongoing research (CONCEPTT trial showed benefit in Type 1 diabetes in pregnancy).
Pharmacological Prevention
Studies are investigating whether metformin in early pregnancy can prevent GDM in high-risk women.
Current Trials:
- EMERGE trial: Metformin vs placebo from 10-16 weeks
- Early results show potential 20-30% risk reduction
Microbiome Research
Emerging evidence links gut microbiome composition to GDM development and may offer future therapeutic targets.
Personalised Risk Prediction
Machine learning models incorporating multiple clinical and biochemical markers show promise for early GDM prediction with > 80% accuracy in validation studies.
17. Clinical Pearls Summary
High-Yield Facts for Examinations
Pearl 1: The "5-6-7-8 Rule"
- NICE thresholds are Fasting ≥5.6, 2-hour ≥7.8 mmol/L
Pearl 2: Fasting ≥7.0 mmol/L at diagnosis = Immediate insulin (diet will fail)
Pearl 3: Metformin is safe and preferred by 77% of women (MiG Trial)
Pearl 4: 46% of women on metformin will require supplemental insulin
Pearl 5: GDM resolves within hours of placental delivery, but 50% develop T2DM within 10-20 years
Pearl 6: The Pedersen Hypothesis explains all fetal complications - maternal hyperglycaemia → fetal hyperinsulinaemia
Pearl 7: HAPO Study showed no threshold - continuous relationship between glucose and adverse outcomes
Pearl 8: ACHOIS Trial showed 67% reduction in serious perinatal morbidity with treatment
Pearl 9: Delivery timing: Diet-controlled 40+6, on medication 38-39 weeks
Pearl 10: Postpartum - stop all medications, test at 6-12 weeks, then annually for life
Common Exam Traps
| Trap | Correct Answer |
|---|---|
| "Is HbA1c used to diagnose GDM?" | No - OGTT is the diagnostic test; HbA1c used to exclude pre-existing DM |
| "Is glucosuria diagnostic?" | No - low sensitivity (~20%), not reliable for diagnosis |
| "Does insulin cross the placenta?" | No - glucose crosses freely, insulin does not |
| "When do congenital malformations occur in GDM?" | They don't - GDM develops after organogenesis; malformations occur in pre-existing DM |
| "Is glibenclamide first-line?" | No - metformin is first-line; glibenclamide is second-line due to neonatal hypoglycaemia risk |
18. Multidisciplinary Team and Referral Pathways
Joint Diabetes Antenatal Clinic (JDAC)
Optimal GDM care requires multidisciplinary input:
| Team Member | Role |
|---|---|
| Obstetrician (Maternal-Fetal Medicine) | Overall antenatal care, delivery planning, complications |
| Diabetologist/Endocrinologist | Glycaemic management, medication titration |
| Diabetes Specialist Nurse/Midwife | Education, SMBG training, insulin technique |
| Dietitian | Medical nutrition therapy, individualised meal planning |
| Sonographer | Growth scans, liquor assessment |
| Neonatologist | Counselling, delivery attendance, neonatal hypoglycaemia management |
When to Escalate
| Scenario | Urgency | Action |
|---|---|---|
| Positive OGTT, Fasting less than 7.0 | Routine (within 1 week) | Refer to JDAC |
| Positive OGTT, Fasting ≥7.0 | Urgent (same day) | Same-day specialist review, start insulin |
| Polyhydramnios/Macrosomia | Urgent | Obstetric review, consider earlier delivery |
| Reduced fetal movements | Emergency | Same-day CTG and assessment |
| Symptomatic neonatal hypoglycaemia | Emergency | Paediatric review, IV dextrose |
| Pre-eclampsia features | Urgent | Full pre-eclampsia workup |
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. It does not replace professional medical judgement. Clinical decisions should account for individual patient circumstances.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Glucose Metabolism in Pregnancy
- Placental Endocrinology
Differentials
Competing diagnoses and look-alikes to compare.
- Pre-existing Type 2 Diabetes
- Pre-existing Type 1 Diabetes
Consequences
Complications and downstream problems to keep in mind.