Hepatitis B (Adult)
Hepatitis B virus (HBV) is a partially double-stranded DNA hepatotropic virus of the Hepadnaviridae family that causes both acute and chronic liver disease. Unlike other hepatitis viruses, HBV establishes a persistent...
What matters first
Hepatitis B virus (HBV) is a partially double-stranded DNA hepatotropic virus of the Hepadnaviridae family that causes both acute and chronic liver disease. Unlike other hepatitis viruses, HBV establishes a persistent...
Acute Liver Failure (Fulminant Hepatitis)
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Acute Liver Failure (Fulminant Hepatitis)
- Hepatitis D Coinfection/Superinfection
- Reactivation with Immunosuppression (Rituximab, Chemotherapy)
- Decompensated Cirrhosis (Ascites, Encephalopathy, Variceal Bleeding)
Linked comparisons
Differentials and adjacent topics worth opening next.
- Hepatitis A
- Hepatitis C
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Hepatitis B (Adult)
1. Clinical Overview
At a Glance
Hepatitis B virus (HBV) is a partially double-stranded DNA hepatotropic virus of the Hepadnaviridae family that causes both acute and chronic liver disease. Unlike other hepatitis viruses, HBV establishes a persistent nuclear reservoir (covalently closed circular DNA, cccDNA) in hepatocytes, making complete viral eradication impossible with current antiviral therapy. [1,2]
Global Health Impact: HBV affects 296 million people globally with chronic infection and causes approximately 820,000 deaths annually from cirrhosis and hepatocellular carcinoma (HCC). It is the leading cause of HCC worldwide and was the first cancer for which a vaccine was developed (the "anti-cancer vaccine"). [3]
Clinical Paradigm: Management focuses on achieving "functional cure" (HBsAg loss with or without anti-HBs seroconversion) rather than complete viral eradication, preventing progression to cirrhosis and HCC, and interrupting transmission. [4]
Key Clinical Facts
| Feature | Detail |
|---|---|
| Incubation Period | 60-150 days (mean 90 days) |
| Chronicity Risk | Adults: less than 5%; Neonates: 90%; Children 1-5 |
| y: 25-50% | |
| Genome | Partially double-stranded relaxed circular DNA (rcDNA), 3.2kb |
| Nuclear Reservoir | cccDNA minichromosome (persistent, not targeted by antivirals) |
| Infectivity | Blood, semen, vaginal fluids (10-100x more infectious than HIV) |
| Natural Clearance | 95% of immunocompetent adults clear acute infection |
| HCC Risk | Occurs even in non-cirrhotic chronic HBV (unique among viral hepatitides) |
| Notifiable Disease | YES (statutory notification in UK, Australia, USA) |
2. Epidemiology
Global Burden and Distribution
Prevalence Patterns: [3,5]
- High Endemic (≥8% HBsAg+): Sub-Saharan Africa, East Asia, Pacific Islands
- "Dominant transmission: Vertical (mother-to-child)"
- "Lifetime risk of infection: > 60%"
- Intermediate Endemic (2-7%): Eastern Europe, Mediterranean, Middle East, Latin America
- Mixed transmission patterns
- Low Endemic (less than 2%): Western Europe, North America, Australia
- "Dominant transmission: Sexual, PWID (people who inject drugs)"
Genotype Distribution: [6] Eight genotypes (A-H) with geographic clustering:
- Genotype A: Northern Europe, North America, sub-Saharan Africa
- Genotype B & C: Asia-Pacific (C associated with higher HCC risk)
- Genotype D: Mediterranean, Middle East, India
- Genotype E: West Africa
- Genotype affects treatment response (Genotype A responds better to interferon)
Transmission Routes
| Route | Efficiency | High-Risk Groups |
|---|---|---|
| Vertical (Perinatal) | 90% if HBeAg+ mother | Babies born to HBsAg+ mothers, especially if HBV DNA > 200,000 IU/mL |
| Sexual | 20-30% in unvaccinated partners | MSM, commercial sex workers, multiple partners |
| Parenteral | Variable (high with needlestick) | PWID, healthcare workers, tattoos/piercings (unsterile), hemodialysis |
| Household | Low (horizontal child-to-child) | Sharing razors, toothbrushes in endemic areas |
| Blood Products | Historical (now rare) | Pre-1992 transfusions (before universal screening) |
NOT transmitted: Casual contact, sharing food/utensils, breastfeeding (if infant receives HBIG + vaccine), kissing (unless blood contact)
Demographics
Age and Acquisition: [7]
- Neonatal/early childhood infection → 90% chronic (immune tolerance)
- Age 1-5 years → 25-50% chronic
- Adults → less than 5% chronic (robust immune response)
- Older age at infection → lower chronicity but more severe acute hepatitis
Gender: Males have higher HCC risk at equivalent levels of viremia and cirrhosis stage
3. Aetiology and Pathophysiology
Virology and Genome Structure
Viral Classification: [1,2]
- Family: Hepadnaviridae (hepatotropic DNA viruses)
- Genus: Orthohepadnavirus
- Structure: 42nm enveloped virion (Dane particle)
- Genome: Partially double-stranded relaxed circular DNA (rcDNA), ~3.2 kilobases
Unique Features:
- Only human DNA virus using reverse transcription
- Replicates via an RNA intermediate (pregenomic RNA)
- Polymerase has reverse transcriptase activity (target for nucleos(t)ide analogues)
- Extremely compact genome with overlapping reading frames
Viral Components:
| Component | Function | Clinical Significance |
|---|---|---|
| HBsAg (Surface Antigen) | Envelope protein, viral entry | Marker of infection (acute or chronic); vaccine target |
| HBcAg (Core Antigen) | Nucleocapsid protein | Not detected in serum (remains in hepatocytes) |
| HBeAg (e Antigen) | Secreted non-structural protein | Marker of active replication and high infectivity |
| HBV DNA Polymerase | Reverse transcriptase | Viral replication; target for antivirals |
| HBx Protein | Transcriptional transactivator | Oncogenic potential (contributes to HCC) |
Replication Cycle and cccDNA Formation
Step-by-Step Lifecycle: [8]
-
Attachment and Entry:
- Virus binds to sodium taurocholate co-transporting polypeptide (NTPCP) receptor on hepatocytes
- Endocytosis into hepatocyte
-
Nuclear Transport and cccDNA Formation:
- Viral capsid transports rcDNA to nucleus
- Host DNA repair enzymes convert rcDNA → cccDNA (covalently closed circular DNA)
- cccDNA forms stable episomal "minichromosome" (persists for life of hepatocyte)
- Critical: cccDNA is NOT integrated into host genome (unlike HIV) but is extremely stable
-
Transcription:
- cccDNA transcribed by host RNA polymerase II
- Produces pregenomic RNA (pgRNA) and subgenomic RNAs
-
Reverse Transcription:
- pgRNA packaged with viral polymerase into capsid
- Reverse transcription of pgRNA → minus-strand DNA
- Partial synthesis of plus-strand DNA → rcDNA (explains partially double-stranded nature)
-
Viral Assembly and Release:
- Nucleocapsids acquire envelope (HBsAg) at ER/Golgi
- Secretion of complete virions
- Alternatively: Capsids recycled to nucleus to amplify cccDNA pool (early infection)
Why cccDNA Matters Clinically: [9]
- Nuclear cccDNA reservoir is NOT eliminated by current antivirals (nucleos(t)ide analogues only block polymerase)
- Explains why HBsAg loss (functional cure) is rare with antiviral therapy (~1-3% per year)
- Reservoir allows viral reactivation if immunosuppression occurs (e.g., chemotherapy)
- Half-life of infected hepatocytes: 10-100 days
- Complete eradication requires either immune-mediated clearance of all infected hepatocytes OR novel agents targeting cccDNA
Immunopathogenesis
Acute Infection in Immunocompetent Adults: [10]
The liver injury in HBV is primarily immune-mediated, not direct cytopathic effect:
-
Innate Immunity (Initial 1-2 weeks):
- HBV is "stealth virus" — poor interferon response (HBV polymerase suppresses innate immunity)
- NK cells and Kupffer cells provide limited initial control
-
Adaptive Immunity (Weeks 4-12):
- CD8+ T cells (cytotoxic): Recognize HBV peptides on MHC-I → kill infected hepatocytes
- Robust, multispecific CD8+ response → hepatocyte destruction → acute hepatitis (elevated ALT, jaundice)
- CD4+ T cells (helper): Support antibody production and CD8+ function
- Antibody response:
- Anti-HBc IgM appears first (weeks 4-6)
- Anti-HBs appears later (weeks 12-24) after HBsAg clearance → neutralizing immunity
-
Outcome in 95% of Adults:
- Complete viral control, HBsAg clearance, anti-HBs seroconversion
- However, cccDNA may persist in small numbers of hepatocytes → "occult HBV" (HBsAg negative, anti-HBc positive, low/undetectable HBV DNA)
Chronic Infection (Immune Tolerance/Exhaustion): [11]
Chronicity develops when adaptive immune response is:
- Absent (neonates: immature immune system recognizes HBsAg as "self")
- Weak (children: partial tolerance)
- Exhausted (overwhelming viral load)
T-Cell Exhaustion: [12]
- Chronic antigen exposure → upregulation of inhibitory receptors (PD-1, CTLA-4, TIM-3)
- CD8+ T cells become "exhausted" → unable to clear infection
- Rationale for checkpoint inhibitor research in chronic HBV
Natural History: The Four Phases
Phase Model of Chronic HBV: [13]
Patients typically progress through phases (though not all patients experience all phases):
| Phase | HBeAg | HBV DNA | ALT | Liver Histology | Duration | Clinical Significance |
|---|---|---|---|---|---|---|
| 1. Immune Tolerant | Positive | Very High (> 10⁷ IU/mL) | Normal | Minimal inflammation | 10-40 years (if vertically acquired) | Low HCC risk; Treatment generally not indicated |
| 2. Immune Active (HBeAg+) | Positive | High (> 2×10⁴ IU/mL) | Elevated | Moderate-severe inflammation/fibrosis | Variable (years) | High risk of progression to cirrhosis; Treatment indicated |
| 3. Inactive Carrier | Negative (Anti-HBe+) | Low (less than 2000 IU/mL) | Normal | Minimal inflammation | Decades (may be lifelong) | Low progression risk; Annual monitoring; Treatment NOT indicated |
| 4. HBeAg-Negative Chronic Hepatitis | Negative (Anti-HBe+) | Fluctuating (usually > 2000) | Elevated (fluctuating) | Moderate-severe inflammation | Variable | Precore/core promoter mutants; High progression risk; Treatment indicated |
Phase Details:
Exam Detail: Phase 1: Immune Tolerant (HBeAg-Positive Chronic Infection)
- Typically vertically acquired infection
- Massive viral replication (HBV DNA often > 10⁸ IU/mL)
- Normal ALT despite high viremia (immune system "tolerates" virus)
- Minimal or no liver inflammation/fibrosis on biopsy
- HBeAg positive, anti-HBe negative
- Low risk of disease progression during this phase
- Management: Monitor 6-12 monthly; treatment generally deferred (unless cirrhosis, family history of HCC, or age > 40 with elevated ALT)
- Duration: Can last 2-4 decades in those infected perinatally
Phase 2: Immune Active HBeAg-Positive (HBeAg-Positive Chronic Hepatitis)
- Immune system "wakes up" and attacks infected hepatocytes
- Elevated ALT (> 30 U/L males, > 19 U/L females)
- HBV DNA remains high (> 2×10⁴ IU/mL)
- Active necroinflammation on histology
- Flares: Periods of high ALT (may be confused with acute hepatitis)
- Goal: Achieve HBeAg seroconversion (HBeAg loss + anti-HBe gain)
- Risk: Progression to cirrhosis (especially if prolonged or repeated flares)
- Management: Treatment indicated to prevent fibrosis progression
Phase 3: Inactive Carrier (HBeAg-Negative Chronic Infection)
- Follows successful immune control or HBeAg seroconversion
- HBeAg negative, anti-HBe positive, HBsAg positive (low levels)
- HBV DNA low or undetectable (less than 2000 IU/mL)
- Normal ALT
- Minimal liver disease
- Prognosis: Generally favorable; low risk of HCC (but NOT zero — still higher than general population)
- Risk: 4-20% may revert to active hepatitis (especially if immunosuppression or HDV superinfection)
- Management: Monitor ALT + HBV DNA 6-12 monthly; treat only if reactivation
Phase 4: HBeAg-Negative Chronic Hepatitis
- Develops from immune active phase OR reactivation of inactive carrier
- Due to precore mutation (G1896A) or basal core promoter mutation
- Mutations prevent HBeAg production but virus still replicates
- The "sneaky" virus (replicates without HBeAg marker)
- HBeAg negative, anti-HBe positive
- HBV DNA fluctuating (often > 2000 IU/mL, can be very high)
- ALT fluctuating (may have normal ALT periods even with ongoing disease)
- Geographic: More common in Mediterranean, Middle East, Asia (Genotype D, C)
- Prognosis: More aggressive; higher risk of cirrhosis and HCC
- Management: Treatment indicated; difficult to assess treatment endpoints (no HBeAg to lose)
Special Populations:
Occult Hepatitis B: [14]
- HBsAg negative, anti-HBc positive, HBV DNA detectable (usually very low less than 200 IU/mL)
- cccDNA persists in hepatocytes
- Clinical significance:
- Can transmit via transfusion/transplant
- Risk of reactivation with immunosuppression
- May contribute to HCC in anti-HBc+ individuals
4. Clinical Presentation
Acute Hepatitis B
Incubation: 60-150 days (mean 90 days) [15]
Clinical Course:
-
Prodrome (1-2 weeks):
- Anorexia, nausea, vomiting
- Malaise, fatigue
- Fever (low-grade), myalgia, arthralgia
- Right upper quadrant discomfort
- Urticaria/rash (10-20% — immune complex deposition)
- Serum sickness-like syndrome: Arthralgia/arthritis, urticaria (before jaundice)
-
Icteric Phase (Variable, 2-12 weeks):
- Jaundice (scleral icterus → generalized)
- Dark urine (conjugated bilirubinuria)
- Pale stools (reduced bile excretion)
- Pruritus
- Hepatomegaly (tender)
- Resolution: Gradual improvement over weeks to months
-
Recovery:
- Symptoms resolve (weeks to months)
- ALT normalizes
- HBsAg clearance, anti-HBs seroconversion
Anicteric Hepatitis: 70% of adults (especially young); mild/absent symptoms; diagnosed only if screened
Severity Spectrum:
- Mild: Fatigue, mild jaundice, full recovery
- Moderate: Significant symptoms, bilirubin 100-200 µmol/L
- Severe: Bilirubin > 200 µmol/L, prolonged PT, hypoalbuminemia
- Fulminant Hepatic Failure: [16]
- Encephalopathy within 8 weeks of symptom onset
- INR > 1.5 (coagulopathy)
- Hypoglycemia, renal failure, cerebral edema
- Mortality 50-90% without liver transplant
- "Risk factors: HDV coinfection, older age, delayed presentation"
- HBV accounts for less than 1% of acute liver failure in Western countries but higher in endemic areas
Chronic Hepatitis B
Asymptomatic Phase (Most Common): [17]
- Majority have NO symptoms for decades
- Detected incidentally (screening, blood donation, insurance medicals)
- May have mild fatigue (often attributed to other causes)
Symptomatic Chronic Disease:
- Fatigue (most common symptom)
- Right upper quadrant discomfort
- Arthralgia (extrahepatic manifestation)
Signs of Advanced Disease/Cirrhosis:
- Stigmata of chronic liver disease:
- Palmar erythema
- Spider naevi (> 5 is pathological)
- Gynecomastia (men), testicular atrophy
- Loss of axillary/pubic hair
- Leukonychia (white nails), clubbing
- Parotid enlargement
- Dupuytren's contracture
- Portal hypertension:
- Splenomegaly
- Ascites
- Caput medusae (dilated periumbilical veins)
- Hemorrhoids
- Hepatic decompensation:
- Jaundice
- Hepatic encephalopathy (confusion, asterixis, coma)
- Variceal bleeding (hematemesis, melena)
- Spontaneous bacterial peritonitis (fever, abdominal pain in ascites)
- Hepatorenal syndrome
Extrahepatic Manifestations (Immune-Complex Mediated) [18]
| Manifestation | Pathophysiology | Clinical Features | Frequency |
|---|---|---|---|
| Polyarteritis Nodosa (PAN) | HBsAg-anti-HBs immune complexes → medium vessel vasculitis | Fever, weight loss, myalgia, neuropathy (mononeuritis multiplex), abdominal pain, hypertension, renal involvement | 1-5% of chronic HBV (more common with acute HBV) |
| Membranous Glomerulonephritis | Immune complex deposition in glomeruli | Nephrotic syndrome (proteinuria, hypoalbuminemia, edema), normal BP/renal function initially | Most common renal manifestation; more in children |
| Mixed Cryoglobulinemia | Cryoglobulin deposition | Purpura, arthralgia, weakness, peripheral neuropathy | Rare (HCV more common cause) |
| Gianotti-Crosti Syndrome | Lymphocytic vasculitis | Papular acrodermatitis (face, buttocks, extensor limbs in children), lymphadenopathy | Children, acute HBV |
| Seronegative Arthritis | Immune complex arthritis | Symmetric polyarthritis (small joints), may mimic RA but RF negative | Acute HBV prodrome |
5. Differential Diagnosis
Acute Hepatitis
| Condition | Distinguishing Features | Key Tests |
|---|---|---|
| Hepatitis A | Fecal-oral, travel history, shorter incubation (15-50d), never chronic | Anti-HAV IgM |
| Hepatitis C | Parenteral transmission, usually asymptomatic acute phase, 70% chronic | Anti-HCV, HCV RNA |
| Hepatitis D | Only with HBV (requires HBsAg), severe/fulminant course | Anti-HDV IgM, HDV RNA |
| Hepatitis E | Fecal-oral, pregnant women (high mortality), self-limiting | Anti-HEV IgM |
| EBV/CMV Hepatitis | Systemic features (fever, lymphadenopathy, pharyngitis, atypical lymphocytes) | EBV VCA IgM, CMV IgM, heterophile Ab |
| Drug-Induced Liver Injury (DILI) | Medication history (paracetamol, herbal supplements, antibiotics) | Temporal relationship, RUCAM score |
| Autoimmune Hepatitis | Young/middle-aged females, hypergammaglobulinemia, interface hepatitis | ANA, SMA, anti-LKM, elevated IgG |
| Alcoholic Hepatitis | AST:ALT > 2:1, history of heavy alcohol use, MCV elevated | AST/ALT ratio, GGT, CDT |
| Ischemic Hepatitis | "Shock liver," very high transaminases (> 1000), rapid improvement | History of hypotension/cardiac event |
| Wilson's Disease | Young patients, hemolytic anemia, Kayser-Fleischer rings, low ceruloplasmin | Ceruloplasmin, 24h urinary copper, KF rings |
| Acute Budd-Chiari | Hepatomegaly, ascites, abdominal pain | Doppler USS (hepatic vein thrombosis) |
Chronic Hepatitis
| Condition | Distinguishing Features | Key Tests |
|---|---|---|
| Hepatitis C | Often asymptomatic, PWID, no vaccine | Anti-HCV, HCV RNA |
| NAFLD/NASH | Metabolic syndrome (obesity, T2DM, dyslipidemia), normal or mildly elevated ALT | Ultrasound (steatosis), FibroScan, liver biopsy |
| Autoimmune Hepatitis | Female predominance, hypergammaglobulinemia, responds to steroids | ANA, SMA, anti-LKM, elevated IgG, interface hepatitis |
| Primary Biliary Cholangitis | Middle-aged women, pruritus, cholestatic pattern (ALP>ALT) | Anti-mitochondrial antibody (AMA) |
| Primary Sclerosing Cholangitis | IBD association, cholestatic pattern, "beaded" ducts | MRCP (bile duct strictures), pANCA |
| Hemochromatosis | Diabetes, arthropathy, cardiomyopathy, slate-grey skin | Ferritin, transferrin saturation, HFE gene |
| Alpha-1 Antitrypsin Deficiency | Emphysema (young), neonatal hepatitis, PAS+ globules on biopsy | α1-AT level, phenotype (PiZZ) |
6. Investigations
Serological Diagnosis and Interpretation
Initial Screening: [19]
| Test | What It Detects | Interpretation |
|---|---|---|
| HBsAg | Surface antigen | Positive = Current infection (acute or chronic) |
| Anti-HBc Total | Core antibody (IgM + IgG) | Marker of exposure (natural infection, NOT vaccine) |
| Anti-HBs | Surface antibody | Positive = Immunity (vaccine or recovery) |
Reflex Tests (If HBsAg Positive):
| Test | Purpose | Interpretation |
|---|---|---|
| Anti-HBc IgM | Differentiate acute vs chronic | Positive in acute (less than 6 months) or flare; negative in chronic |
| HBeAg | Replication marker | Positive = high infectivity, active replication |
| Anti-HBe | Response to HBeAg | Positive = HBeAg seroconversion (lower infectivity) |
| HBV DNA (Quantitative PCR) | Viral load | Guides treatment decisions (see below) |
Serology Pattern Recognition (The "Exam Nightmare")
Exam Detail: Classic Patterns:
| HBsAg | Anti-HBc Total | Anti-HBc IgM | Anti-HBs | HBeAg | Interpretation | Action |
|---|---|---|---|---|---|---|
| Neg | Neg | Neg | Neg | N/A | Susceptible (Never exposed, not vaccinated) | Vaccinate |
| Neg | Neg | Neg | Pos | N/A | Immune (Vaccine) | No action (check titer if high risk and > 10 years since vaccine) |
| Neg | Pos | Neg | Pos | N/A | Immune (Natural — Past Infection) | No action; cured |
| Pos | Pos | Pos | Neg | +/− | Acute Infection (Or flare of chronic) | Confirm with repeat in 6 months; supportive care; monitor for fulminant failure |
| Pos | Pos | Neg | Neg | Pos | Chronic Infection (HBeAg+) | Check HBV DNA, ALT, assess for treatment |
| Pos | Pos | Neg | Neg | Neg | Chronic Infection (HBeAg−) | Check HBV DNA, ALT (may be inactive carrier or HBeAg-negative CHB) |
| Neg | Pos | Neg | Neg | N/A | Isolated Anti-HBc (See below) | Check HBV DNA; consider occult HBV or false positive |
| Pos | Neg | Neg | Neg | N/A | Very Early Acute (Anti-HBc not yet developed) OR False Positive HBsAg | Retest in 2-4 weeks |
The "Isolated Anti-HBc" Conundrum (HBsAg Negative, Anti-HBc Positive, Anti-HBs Negative): [20]
Possible explanations:
- "Window Period": Acute resolving infection (HBsAg cleared, anti-HBs not yet appeared)
- Check anti-HBc IgM (positive), ALT (elevated)
- Repeat serology in 2-4 weeks (anti-HBs should appear)
- Remote Infection: Anti-HBs waned below detectable levels
- Anti-HBc IgM negative, normal ALT
- Immune memory intact (booster vaccine dose → rapid anamnestic response)
- Occult Hepatitis B: HBsAg below detection but virus persists
- Check HBV DNA (may be low-level positive)
- Risk of reactivation with immunosuppression
- False Positive: Cross-reactivity in assay
- Repeat with different assay
Clinical Approach:
- Check HBV DNA
- If DNA negative: Give single vaccine dose → check anti-HBs in 4 weeks (should rise if immune memory present)
- If DNA positive: Occult HBV → monitor as chronic HBV (especially if immunosuppression planned)
Key Point for Exams: Core antibody (anti-HBc) is NEGATIVE in vaccinated individuals (vaccine contains only HBsAg, not core). If anti-HBc positive → natural infection (past or present), NOT vaccine response.
Virological Monitoring
HBV DNA Quantification (Real-Time PCR): [21]
| HBV DNA Level | Clinical Significance |
|---|---|
| Undetectable (less than 10 IU/mL) | Excellent viral suppression on treatment OR inactive carrier |
| Low (10-2000 IU/mL) | Inactive carrier phase; treatment usually not indicated |
| Intermediate (2000-20,000 IU/mL) | "Grey zone"; assess ALT, liver stiffness, consider treatment if inflammation |
| High (> 20,000 IU/mL) | Active replication; treatment indicated if ALT elevated or cirrhosis |
| Very High (> 200,000 IU/mL) | Immune tolerant phase OR high-risk for vertical transmission in pregnancy |
Genotyping:
- Not routinely required
- Useful if considering interferon therapy (Genotype A responds best)
- May guide HCC surveillance intensity (Genotype C highest risk)
Liver Biochemistry and Function
| Test | Finding in HBV | Interpretation |
|---|---|---|
| ALT/AST | Elevated (variable, 50-500+) | Hepatocyte necroinflammation; ALT>AST typically |
| AST:ALT Ratio | less than 1 usually | If > 1 → suspect cirrhosis or alcohol |
| ALP/GGT | Normal or mildly elevated | If disproportionately elevated → consider cholestatic liver disease |
| Bilirubin | Normal (chronic) or elevated (acute/decompensated) | Conjugated hyperbilirubinemia |
| Albumin | Normal (early) or low (cirrhosis) | Synthetic function marker |
| PT/INR | Normal (early) or prolonged (cirrhosis/acute liver failure) | Coagulation factor synthesis |
| Platelets | Normal or low (cirrhosis + portal hypertension) | Thrombocytopenia suggests splenomegaly/cirrhosis |
Treatment Thresholds (EASL 2017): [22]
- ALT Upper Limit Normal (ULN): 30 U/L (males), 19 U/L (females)
- Treatment considered if ALT >ULN and HBV DNA > 2000 IU/mL
Assessment of Liver Fibrosis
Non-Invasive Methods: [23]
| Method | Principle | Interpretation | Advantages | Limitations |
|---|---|---|---|---|
| FibroScan (Transient Elastography) | Measures liver stiffness (kPa) | less than 7 kPa: F0-F1 (no/mild fibrosis); 7-9.5: F2; 9.5-12.5: F3; > 12.5: F4 (cirrhosis) | Non-invasive, quick, reproducible | Obesity, ascites, narrow intercostal spaces reduce success |
| APRI (AST-to-Platelet Ratio Index) | (AST/ULN) × 100 / Platelet count | less than 0.5: Low fibrosis; > 1.5: Cirrhosis likely | Cheap, widely available | Less accurate in intermediate range |
| FIB-4 | Age × AST / (Platelet × √ALT) | less than 1.45: Low fibrosis; > 3.25: Advanced fibrosis | Simple calculation | Age-dependent |
| ELF Score (Enhanced Liver Fibrosis) | PIIINP, HA, TIMP-1 | less than 7.7: No fibrosis; > 11.3: Severe | Validated in NAFLD, HBV | Requires specialized lab |
Liver Biopsy: [24]
- Indications: Uncertain diagnosis, guide treatment if non-invasive markers equivocal, assess inflammation grade
- Staging: Metavir, Ishak, or Batts-Ludwig scoring
- Limitations: Invasive, sampling error, inter-observer variability
- Increasingly replaced by non-invasive methods
HCC Surveillance
AFP (Alpha-Fetoprotein): [25]
- Sensitivity 60%, specificity 80% (not ideal as sole test)
- Cutoff: > 20 ng/mL concerning; > 200 ng/mL highly suspicious
- Can be elevated in active hepatitis (false positive)
Ultrasound Abdomen:
- Sensitivity: 45-84% for HCC detection (operator-dependent, body habitus)
- Detects focal lesions, cirrhosis nodularity, portal hypertension features
CT/MRI:
- Used for characterization of USS abnormalities
- Arterial enhancement + washout on delayed phase → characteristic HCC pattern
Surveillance Protocol (See Management section)
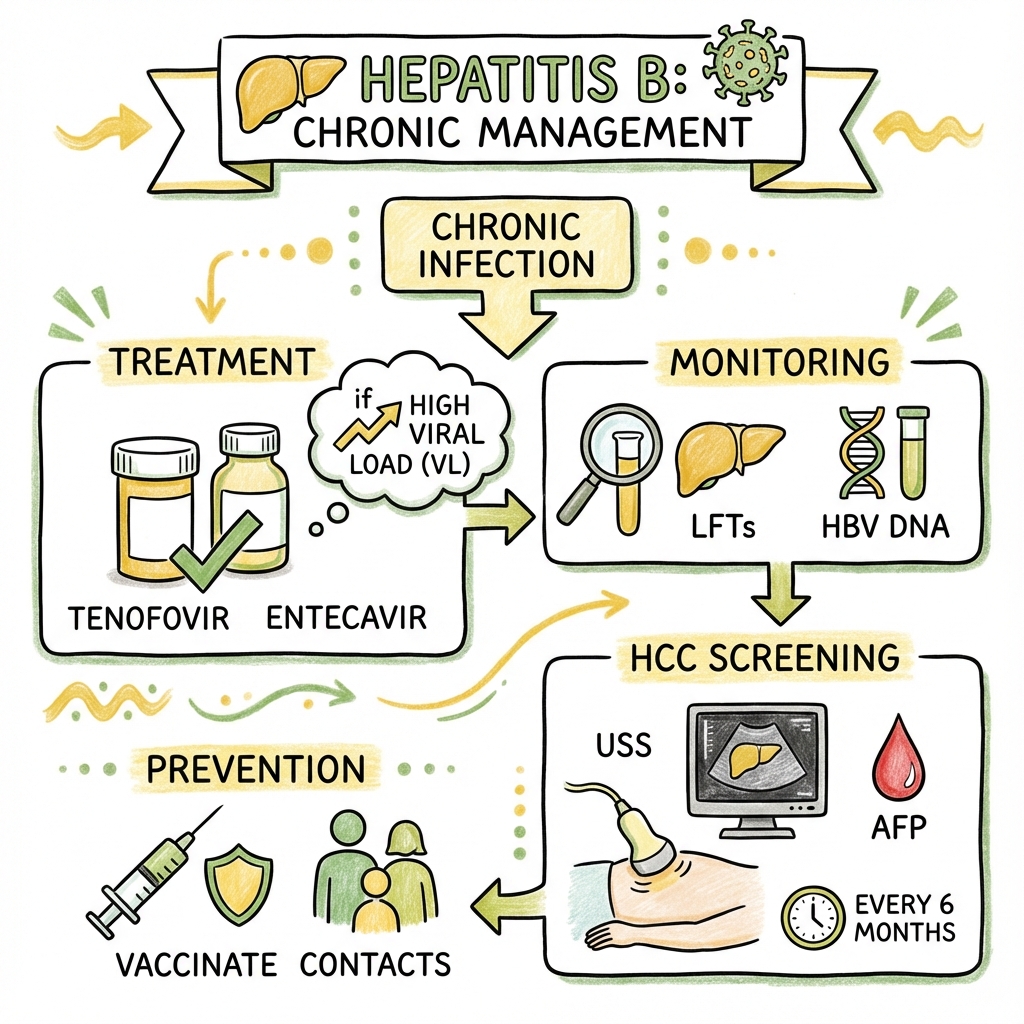
7. Management
Overview: Goals of Therapy
Primary Goals: [4,22]
- Prevent progression: Stop fibrosis → cirrhosis progression
- Prevent HCC: Suppress HBV DNA to reduce oncogenic pressure
- Achieve functional cure: HBsAg loss ± anti-HBs seroconversion (ideal but rare with NAs)
- Prevent transmission: Reduce infectivity
- Improve survival: Reduce liver-related mortality
"Functional Cure" vs "Complete Cure":
- Functional Cure: HBsAg loss (with or without anti-HBs seroconversion); HBV DNA undetectable
- Achieved in ~1-3% per year on nucleos(t)ide analogues (NAs)
- Up to 30% with peginterferon in selected patients
- Complete Cure: Elimination of cccDNA (currently impossible with existing therapies)
Treatment Indications (When to Treat)
Exam Detail: EASL 2017 Guidelines: [22]
Treat if ANY of the following:
-
Cirrhosis (Any Stage):
- All patients with cirrhosis should be treated regardless of HBV DNA level or ALT
- Goal: Prevent decompensation and HCC
- Exception: Decompensated cirrhosis → refer for transplant + antiviral bridge
-
Non-Cirrhotic with Active Disease:
- HBV DNA > 2000 IU/mL AND
- ALT >ULN (> 30 U/L males, > 19 U/L females) AND/OR
- Moderate-severe inflammation on histology (or FibroScan equivalent)
-
Special Indications (Regardless of ALT/DNA):
- Family history of HCC (first-degree relative)
- Extrahepatic manifestations (PAN, GN)
- HBV/HIV coinfection (treat as per HIV guidelines)
- Immunosuppressive therapy planned (see Reactivation Prevention)
- Acute liver failure
- Pregnancy with high viral load (> 200,000 IU/mL → treat third trimester to prevent vertical transmission)
DO NOT Treat (Monitor Instead):
-
Immune Tolerant Phase: HBeAg+, HBV DNA > 10⁷, normal ALT, age less than 30, no fibrosis
- "Rationale: Treatment unlikely to be effective; low risk of progression at this stage"
- "Exception: Age > 40 (even with normal ALT, consider treatment due to higher HCC risk)"
-
Inactive Carrier: HBsAg+, HBeAg−, anti-HBe+, HBV DNA less than 2000, normal ALT, no fibrosis
- "Management: Monitor ALT + HBV DNA every 6-12 months (risk of reactivation)"
Antiviral Therapy: Nucleos(t)ide Analogues (NAs)
Mechanism: Inhibit HBV DNA polymerase (reverse transcriptase) → block viral replication
Key NA Agents: [26,27]
| Drug | Potency | Resistance Barrier | Advantages | Disadvantages | Dosing |
|---|---|---|---|---|---|
| Entecavir (ETV) | High | High | Potent, low resistance (1% at 5y in treatment-naïve) | Cannot use if prior lamivudine resistance (cross-resistance) | 0.5mg PO daily (1mg if lamivudine-resistant) |
| Tenofovir Disoproxil Fumarate (TDF) | High | High | Potent, zero resistance to date, safe in pregnancy | Renal toxicity (Fanconi syndrome, AKI), bone density loss | 300mg PO daily |
| Tenofovir Alafenamide (TAF) | High | High | Same efficacy as TDF, less renal/bone toxicity | More expensive, not yet proven safe in pregnancy | 25mg PO daily |
| Lamivudine (LAM) | Moderate | Low | Cheap | High resistance (70% at 5 years) — now obsolete as monotherapy | 100mg PO daily |
| Adefovir (ADV) | Low | Moderate | Active against LAM-resistant strains | Weak, nephrotoxic — rarely used now | 10mg PO daily |
First-Line Therapy: [22]
- Preferred: Entecavir or Tenofovir (TDF/TAF)
- Choice between ETV vs TDF/TAF:
- "Entecavir: If renal impairment, osteoporosis, older age"
- "TDF: If pregnancy (category B; established safety data)"
- "TAF: If renal impairment, osteoporosis, elderly, but more costly"
Treatment Duration: [28]
- Cirrhotic patients: Lifelong (stopping risks decompensation)
- Non-cirrhotic HBeAg+ patients: Minimum 12 months after HBeAg seroconversion, then consider stopping if:
- HBV DNA undetectable
- Consolidation therapy ≥12 months post-seroconversion
- Close monitoring (risk of relapse 50%)
- Non-cirrhotic HBeAg− patients: Usually indefinite (no clear endpoint)
- May consider stopping if HBsAg loss achieved (rare)
- Stopping criteria (non-cirrhotic only):
- HBsAg loss (with or without anti-HBs seroconversion) = functional cure
- Close monitoring after stopping (ALT + HBV DNA monthly × 3, then 3-monthly × 12 months)
Monitoring On Treatment:
- Baseline: FBC, U&E, LFT, HBV DNA, HBsAg quantification (if available)
- During treatment:
- "ALT, HBV DNA: Every 3 months in first year, then every 6 months"
- "HBeAg/anti-HBe (if HBeAg+ at baseline): Every 6-12 months"
- "Renal function (if on TDF): Every 6-12 months"
- "Bone density scan (if on TDF and risk factors): Baseline and annually"
- HCC surveillance: Continue 6-monthly USS + AFP (even with undetectable HBV DNA)
Treatment Response Definitions:
- Virological response: HBV DNA undetectable (less than 10-20 IU/mL depending on assay sensitivity)
- Biochemical response: ALT normalization
- Serological response: HBeAg loss + anti-HBe gain (if HBeAg+ at baseline)
- Complete response: HBsAg loss ± anti-HBs seroconversion (functional cure)
Expected Outcomes with First-Line NAs: [29]
- HBV DNA undetectable at 1 year: 70-80%
- HBeAg seroconversion at 5 years: 30-40% (HBeAg+ patients)
- HBsAg loss at 5 years: 2-5%
Resistance and Treatment Failure:
- Virological breakthrough: > 1 log₁₀ increase in HBV DNA after initial response (check adherence first)
- Resistance with ETV/TDF/TAF: Virtually zero with adherence
- If resistance develops: Add alternative NA (e.g., TDF if on ETV, or vice versa)
Pegylated Interferon Alpha (PegIFN-α)
Mechanism: [30]
- Immunomodulatory (enhances T-cell response)
- Antiviral (direct inhibition of viral replication)
- Antiproliferative
Advantages:
- Finite duration: 48 weeks (vs indefinite NA therapy)
- Higher rates of HBsAg loss: 3-7% at end of treatment (vs 1-3% with NAs at 1 year)
- No resistance
- Possible sustained off-treatment response
Disadvantages:
- Severe side effects:
- Flu-like symptoms (fever, myalgia, headache)
- Bone marrow suppression (neutropenia, thrombocytopenia)
- Psychiatric effects (depression, suicidal ideation)
- Thyroid dysfunction (hypo/hyperthyroidism)
- Autoimmune complications
- Subcutaneous injection (weekly)
- Contraindications: Decompensated cirrhosis, pregnancy, psychiatric disease, autoimmune disease, severe cytopenias
Regimen:
- PegIFN-α2a: 180 µg SC weekly × 48 weeks
Patient Selection (Who Benefits Most): [31]
- Young patients (less than 40 years) wanting finite therapy
- HBeAg-positive chronic hepatitis (better response than HBeAg-negative)
- Genotype A or B (A responds best)
- Low baseline HBV DNA (less than 10⁶ IU/mL)
- High baseline ALT (> 2× ULN) → suggests active immune response
- Female gender
- No cirrhosis (contraindicated in decompensated cirrhosis)
Stopping Rules: [32]
- HBeAg+ patients: If HBsAg > 20,000 IU/mL at week 12 → stop (unlikely to respond)
- HBeAg− patients: If HBsAg decline less than 1 log₁₀ at week 12 → stop
Combination PegIFN + NA:
- No proven benefit over monotherapy
- Not routinely recommended
Emerging/Novel Therapies
Investigational Approaches: [33]
- Entry inhibitors: Bulevirtide (blocks NTCP receptor) — approved in EU for HDV
- Capsid assembly modulators: Disrupt nucleocapsid formation
- cccDNA targeting: CRISPR/Cas9, TALENs (gene editing)
- Immune checkpoint inhibitors: Anti-PD-1 (restore T-cell function)
- Therapeutic vaccines: Boost anti-HBV immune responses
- RNA interference (RNAi): siRNA targeting HBV transcripts
Goal: Increase rates of functional cure (HBsAg loss) beyond current 1-3% with NAs
8. HCC Surveillance and Management
Rationale for Surveillance
HCC Risk in Chronic HBV: [34]
- Annual incidence: 0.2-0.6% (non-cirrhotic), 2-5% (cirrhotic)
- Unique feature: HCC can develop in non-cirrhotic HBV (unlike HCV where cirrhosis is prerequisite)
- Risk factors for HCC in HBV:
- Cirrhosis (greatest risk)
- Male gender
- Age > 40
- Asian/African ethnicity
- Family history of HCC (first-degree relative)
- Genotype C
- High HBV DNA (> 2000 IU/mL)
- Persistent ALT elevation
- HBeAg positivity
- Coinfection (HCV, HDV, HIV)
- Aflatoxin exposure
Evidence for Surveillance: [35]
- Surveillance detects HCC at earlier stage (Milan criteria eligibility)
- Earlier detection → curative treatment (resection, ablation, transplant)
- RCT in China: 6-monthly AFP + USS reduced HCC mortality by 37%
Surveillance Protocol
Who to Surveil: [22,36]
Universal Screening (All):
- All patients with cirrhosis (regardless of etiology, even if HBV DNA undetectable on treatment)
Risk-Based Screening (Non-Cirrhotic HBV):
- Asian males ≥40 years
- Asian females ≥50 years
- Africans ≥20 years
- Family history of HCC (any age)
- Persistent HBV DNA > 2000 IU/mL with elevated ALT
Frequency: Every 6 months (USS ± AFP)
Modality:
- Ultrasound abdomen (sensitivity 45-84% for early HCC)
- Look for focal lesions, nodules, vascular changes
- AFP (alpha-fetoprotein):
- Sensitivity ~60%, specificity ~80% (suboptimal alone)
- "Cutoff: > 20 ng/mL warrants further imaging"
- Can be elevated in active hepatitis (false positive)
- Consider CT/MRI in high-risk or if USS inadequate (obesity, cirrhosis with nodularity)
Abnormal Findings:
- Nodule less than 1 cm: Repeat USS in 3 months
- Nodule 1-2 cm: Multiphasic CT or MRI (arterial phase hyperenhancement + venous/delayed washout = HCC)
- Nodule > 2 cm: As above; if typical HCC pattern → diagnose without biopsy
- AFP > 200 ng/mL + liver mass: Highly suspicious for HCC even if imaging equivocal
9. Special Clinical Scenarios
9.1 Pregnancy and Vertical Transmission Prevention
Background: [37]
- Vertical transmission is the leading cause of chronic HBV globally (90% chronicity if acquired perinatally)
- Transmission occurs primarily during delivery (blood/fluid exposure), NOT in utero
- Risk factors for transmission:
- Maternal HBeAg positivity (90% transmission if untreated)
- High maternal HBV DNA (> 200,000 IU/mL = high risk; > 1,000,000 = very high)
Antenatal Screening:
- Universal HBsAg screening at first antenatal visit (8-12 weeks) in all countries
- If HBsAg positive → check HBeAg, anti-HBe, HBV DNA, LFTs
Management:
| Maternal HBV DNA | Management | Rationale |
|---|---|---|
| less than 200,000 IU/mL | No maternal antiviral; neonatal immunoprophylaxis only | Low risk of transmission with HBIG + vaccine |
| ≥200,000 IU/mL | Tenofovir (TDF) from 28-32 weeks gestation until delivery (can continue if breastfeeding) | Reduce maternal viral load → reduce transmission risk to less than 5% |
Neonatal Immunoprophylaxis (All HBsAg+ mothers): [38]
- HBIG (Hepatitis B Immunoglobulin): 0.5 mL IM within 12 hours of birth (ideally less than 6 hours)
- HBV vaccine: 10 µg IM within 24 hours of birth
- Vaccine schedule: 0, 1, 6 months (complete series)
- Check infant: Anti-HBs and HBsAg at 9-12 months
- "Goal: Anti-HBs > 10 mIU/mL, HBsAg negative (95-99% efficacy)"
Breastfeeding: Safe if infant receives HBIG + vaccine [39]
Antiviral Safety in Pregnancy:
- Tenofovir (TDF): Category B; extensive safety data (from HIV literature)
- Lamivudine: Category C; generally safe
- Telbivudine: Studied in HBV pregnancy (but discontinued in many countries)
- Entecavir: Category C; limited data (avoid unless already on treatment)
- PegIFN: Contraindicated (teratogenic)
9.2 Reactivation with Immunosuppression
Definition: [40]
- Increase in HBV DNA ≥2 log₁₀ (100-fold) from baseline
- Or reappearance of HBsAg in previously HBsAg-negative, anti-HBc-positive patient
Clinical Spectrum:
- Asymptomatic biochemical hepatitis (mild ALT rise)
- Symptomatic acute hepatitis (jaundice, high ALT)
- Fulminant hepatic failure (mortality 5-50% without treatment)
High-Risk Immunosuppressive Agents: [41]
| Risk Level | Agents | Reactivation Risk (Untreated) |
|---|---|---|
| High (> 10%) | Anti-CD20 (Rituximab), Ofatumumab, Obinutuzumab; High-dose corticosteroids (≥20mg prednisone ≥4 weeks); Anthracyclines (Doxorubicin) | 20-50% |
| Moderate (1-10%) | Moderate-dose steroids; TNF-α inhibitors (Infliximab, Adalimumab); Cytotoxic chemotherapy (non-anthracycline); Tyrosine kinase inhibitors (Imatinib) | 1-10% |
| Low (less than 1%) | Azathioprine; 6-Mercaptopurine; Methotrexate; Intra-articular steroids | less than 1% |
"The Rituximab Rule": [42]
- Rituximab (anti-CD20) has highest reactivation risk (30-50% if HBsAg+, 5-10% if HBsAg− but anti-HBc+)
- Mechanism: B-cell depletion → loss of anti-HBs antibody → uncontrolled HBV replication from cccDNA reservoir
- Reactivation can occur 6-12 months after stopping rituximab (delayed)
Pre-Treatment Screening (MANDATORY before any immunosuppression): [43]
Screen ALL patients for:
- HBsAg
- Anti-HBc (total)
- Anti-HBs
Management Based on Screening:
| Serology | Risk | Management |
|---|---|---|
| HBsAg Positive | High | Prophylactic antiviral (TDF or ETV) starting before immunosuppression, continue during + ≥12 months after stopping (≥18 months if rituximab) |
| HBsAg Neg, Anti-HBc Pos (Occult HBV) | Moderate | Prophylactic antiviral if high-risk agent (rituximab, anthracyclines); Monitor (monthly ALT + HBV DNA × 3, then 3-monthly) if moderate/low-risk agent |
| HBsAg Neg, Anti-HBc Neg, Anti-HBs Pos (Vaccinated or immune) | Very Low | Monitor; consider booster vaccine if anti-HBs less than 10 mIU/mL |
| All Negative (Susceptible) | N/A | Vaccinate before immunosuppression if time permits |
Prophylaxis Duration: [22]
- During immunosuppression: Continue antiviral throughout
- After stopping immunosuppression:
- "Standard agents: ≥12 months"
- "Rituximab/anti-CD20: ≥18 months (delayed reactivation risk)"
- Monitor HBV DNA every 3 months after stopping prophylaxis
Established Reactivation:
- Start antiviral immediately (TDF or ETV)
- Do NOT stop chemotherapy if oncologically critical (treat through)
- Monitor for hepatic decompensation
9.3 Hepatitis D Virus (HDV) Coinfection/Superinfection
Background: [44]
- HDV is a "defective" RNA virus requiring HBsAg for assembly and transmission
- Can only infect individuals with HBV (cannot exist alone)
- Two scenarios:
- Coinfection: Simultaneous acute HBV + HDV (usually self-limiting like HBV alone, but more severe)
- Superinfection: Acute HDV on top of chronic HBV (high risk of rapid progression to cirrhosis)
Epidemiology:
- 5-10% of chronic HBV patients have HDV (15-20 million globally)
- Endemic: Mediterranean, Middle East, Central Asia, Amazon basin, West Africa
- Transmission: Parenteral (PWID), sexual (less efficient than HBV)
Clinical Features:
- Coinfection: More severe acute hepatitis, higher risk of fulminant failure (2-20% vs less than 1% with HBV alone)
- Superinfection: Rapid progression to cirrhosis (70-80% develop cirrhosis within 5-10 years vs 15-30% with HBV alone)
- Suspect if: Sudden deterioration in stable chronic HBV, severe acute hepatitis with HBsAg+
Diagnosis:
- Anti-HDV (total IgG + IgM): Screening test
- HDV RNA: Confirmatory, quantitative (indicates active replication)
- Pattern in superinfection: HBV DNA often suppressed by HDV (HDV dominant)
Treatment: [45]
- PegIFN-α: 48-96 weeks (only licensed therapy; 25-30% achieve HDV RNA suppression)
- Bulevirtide: Entry inhibitor (blocks NTCP receptor); approved in EU 2020; 180 µg SC daily
- NAs (TDF/ETV): Suppress HBV but NOT effective against HDV; still give to prevent HBV flare and maintain HBsAg (HDV needs it)
Prognosis:
- Worse than HBV monoinfection (faster fibrosis, higher HCC risk)
9.4 HIV/HBV Coinfection
Epidemiology: [46]
- 5-15% of HIV patients have chronic HBV (shared transmission routes)
- HBV is leading cause of liver-related mortality in HIV era of effective ART
Impact of HIV on HBV:
- Accelerated fibrosis progression
- Higher HBV DNA levels (impaired immune control)
- Lower rates of HBsAg clearance
- Higher risk of HCC
Management:
- Treat HBV as part of ART regimen
- Preferred ART: Tenofovir (TDF or TAF) + Emtricitabine (FTC) or Lamivudine (3TC) (dual HBV/HIV activity)
- Never use lamivudine or emtricitabine alone (rapid HBV resistance)
- If stopping ART: Continue TDF monotherapy to prevent HBV flare (HBV needs lifelong suppression)
10. Vaccination
Prophylactic Vaccination (Primary Prevention)
Vaccine Composition: [47]
- Recombinant HBsAg protein (produced in yeast)
- Adjuvanted with aluminum hydroxide
- Does NOT contain live virus, HBcAg, or HBV DNA → cannot cause infection
Standard Schedule:
- 0, 1, 6 months (three doses)
- Dose: 10-20 µg HBsAg (depends on formulation)
- Route: IM (deltoid in adults, anterolateral thigh in infants)
Accelerated Schedules:
- 0, 1, 2 months + booster at 12 months (for rapid protection, e.g., travel, occupational)
- 0, 7, 21 days + booster at 12 months (ultra-rapid, e.g., post-exposure)
Target Populations (Universal in many countries; selective in others): [48]
- Universal infant vaccination (WHO recommendation; implemented in > 95% countries)
- High-risk adults:
- Healthcare workers
- PWID
- MSM
- Dialysis patients
- Household/sexual contacts of HBsAg+ individuals
- Travelers to endemic areas
- HIV-positive individuals
- Chronic liver disease (any cause)
Checking Response:
- Anti-HBs titer 1-2 months after final dose
- Protective level: ≥10 mIU/mL (ideally > 100 for long-term protection)
Vaccine Response Rates: [49]
- Healthy adults: 90-95%
- Older age (> 60 years): 70-80%
- Dialysis, HIV, obesity, smoking: 50-70%
Non-Responders (Anti-HBs less than 10 mIU/mL after full course):
- 5-10% of healthy adults
- Action:
- Check if already immune: Test HBsAg and anti-HBc (may have occult infection)
- Repeat full course (0, 1, 6 months) with double dose (40 µg)
- Check anti-HBs again
- If still no response → consider intradermal route or accept non-responder status
- Non-responders and exposure: Give HBIG for post-exposure prophylaxis
Duration of Protection:
- Immunological memory persists ≥20-30 years (even if anti-HBs wanes below 10 mIU/mL)
- Booster NOT routinely recommended in immunocompetent individuals
- Exception: Healthcare workers, dialysis patients (check anti-HBs annually; booster if less than 10 mIU/mL)
Post-Exposure Prophylaxis (PEP)
Indications: [50]
- Needlestick/sharps injury from HBsAg+ source
- Sexual exposure (unprotected sex with HBsAg+ partner)
- Percutaneous/mucosal exposure to blood (splash to eyes/mouth, human bite)
- Neonates of HBsAg+ mothers (see Pregnancy section)
Management by Vaccination Status:
| Recipient Status | Source HBsAg Status | Action |
|---|---|---|
| Vaccinated, known responder (Anti-HBs ≥10) | HBsAg+ | No action (protected) |
| Vaccinated, non-responder (Anti-HBs less than 10) | HBsAg+ | HBIG (0.06 mL/kg IM) ASAP (within 24h, up to 7 days) + Vaccine booster |
| Unvaccinated | HBsAg+ | HBIG (0.06 mL/kg IM) ASAP + Vaccine series (0, 1, 6 months; give first dose simultaneously at different site) |
| Unvaccinated | Unknown/HBsAg− | Vaccine series (0, 1, 6 months) |
| Vaccinated, unknown response | HBsAg+ | Check recipient anti-HBs STAT; if less than 10 → give HBIG + booster; if ≥10 → no action |
Timing: [51]
- HBIG: Ideally within 24 hours (efficacy decreases after 48-72 hours; may give up to 7 days)
- Vaccine: Simultaneously with HBIG (different site) or ASAP
Follow-Up:
- Check HBsAg, anti-HBc at 3 and 6 months (to detect infection if PEP failed)
11. Prognosis and Complications
Natural History and Outcomes
Acute HBV in Adults: [15]
- 95% clear infection (HBsAg loss, anti-HBs seroconversion)
- less than 5% progress to chronic HBV
- less than 1% develop fulminant hepatic failure (higher with HDV coinfection, older age)
Chronic HBV: [52]
Inactive Carrier (Favorable):
- Cirrhosis risk: less than 10% over lifetime
- HCC risk: ~0.1-0.2% per year
- Normal life expectancy if no progression
Active Chronic Hepatitis (Untreated):
- Cirrhosis: Develops in 15-40% over 5-20 years
- Decompensation (if cirrhotic): 20-30% at 5 years
- HCC (if cirrhotic): 2-5% per year
- Liver-related mortality: 40-50% at 5 years (decompensated cirrhosis)
Impact of Antiviral Treatment: [53]
- Regression of fibrosis: 40-70% show histological improvement after 5 years of viral suppression
- Cirrhosis regression: Documented in 30-40% with sustained virological suppression
- HCC risk reduction: 50-70% reduction (but NOT eliminated — surveillance still required)
- Decompensation risk reduction: 70-80% (in compensated cirrhosis)
- Survival: Improved liver-related survival (HR 0.3-0.5 for mortality vs untreated)
Hepatocellular Carcinoma (HCC)
Incidence: [34]
- Non-cirrhotic chronic HBV: 0.2-0.6% per year
- Cirrhotic HBV: 2-5% per year (15-20% cumulative at 5 years)
Unique Feature: HBV can cause HCC without cirrhosis (unlike HCV)
- Mechanism: HBx protein (oncogenic), integration of HBV DNA into host genome (insertional mutagenesis)
Risk Stratification (PAGE-B Score, mPAGE-B): [54]
- Age, gender, platelet count (simple risk calculator)
- Low-risk (less than 10 points) vs high-risk (> 18 points)
Outcomes:
- Early-stage HCC (Milan criteria): 5-year survival 50-70% with curative treatment (resection/transplant/ablation)
- Advanced HCC: Median survival less than 1 year
Complications of Cirrhosis
Portal Hypertension:
- Varices (esophageal, gastric): Bleeding risk 10-15% per year
- Ascites: Median survival 2 years after first onset (if untreated)
- Hepatorenal syndrome: Mortality 90% without transplant
- Spontaneous bacterial peritonitis: Mortality 20-40%
Hepatic Encephalopathy:
- West Haven Grade 1-4
- Precipitants: GI bleeding, infection, dehydration, constipation
Hepatocellular Dysfunction:
- Coagulopathy (reduced factor synthesis)
- Hypoalbuminemia
- Hypoglycemia
Liver Transplantation:
- Indicated for decompensated cirrhosis (Child-Pugh B/C, MELD > 15)
- HBV recurrence post-transplant: Prevented with HBIG + long-term NA (recurrence less than 5%)
12. Prevention and Public Health
Population-Level Interventions
Universal Infant Vaccination: [55]
- WHO target: 90% coverage globally by 2030
- Timing: Birth dose within 24 hours (critical to prevent vertical transmission) + 3-dose series
- Impact: Countries with universal infant vaccination (since 1990s) have seen > 90% reduction in chronic HBV prevalence in children
Screening Programs:
- Antenatal screening: Universal HBsAg screening in pregnancy
- Blood donors: Universal HBsAg screening (since 1970s in developed countries)
- High-risk populations: MSM, PWID, migrants from endemic areas, dialysis patients
Harm Reduction (PWID):
- Needle exchange programs
- Opioid substitution therapy
- Vaccination
Sexual Health:
- Condom promotion
- Partner notification and vaccination
- PrEP for HIV (tenofovir-based) provides dual HBV protection
Elimination Goals (WHO 2030)
Global Targets: [56]
- 90% reduction in new chronic infections
- 65% reduction in mortality from viral hepatitis
- 90% of chronic infections diagnosed
- 80% of eligible patients treated
Challenges:
- Low diagnosis rates (10% globally know their status)
- Limited access to antivirals in low-resource settings
- Lack of curative therapy (functional cure rates low with NAs)
13. Examination Focus
History Taking (OSCE Station)
Key Questions:
Risk Factors:
- "Have you ever had a blood transfusion, especially before 1992?"
- "Do you inject drugs or have you in the past?"
- "Any tattoos or body piercings?"
- "Have you lived or traveled to areas where hepatitis B is common (Asia, Africa)?"
- "How many sexual partners have you had? Do you use condoms?"
- "Any family history of liver disease or hepatitis?"
- "Was your mother known to have hepatitis B?" (vertical transmission)
Symptoms (often none):
- "Do you feel more tired than usual?"
- "Any yellowing of eyes or skin?"
- "Any abdominal swelling or pain?"
- "Have you noticed confusion or drowsiness?" (encephalopathy)
- "Any bleeding problems (gums, bruising)?"
Complications:
- "Any joint pains or rashes?" (extrahepatic manifestations)
- "Any swelling of ankles or abdomen?" (ascites)
Social:
- "Do you drink alcohol? How much per week?"
- "Are you taking any regular medications or herbal supplements?"
- "Any vaccinations for hepatitis?"
ICE (Ideas, Concerns, Expectations):
- "What do you understand about hepatitis B?"
- "What are you most worried about?"
- "What would you like to know today?"
Examination Findings (Long Case/PACES)
Inspection:
- Jaundice (scleral icterus)
- Muscle wasting, cachexia
- Gynecomastia (men)
Hands:
- Palmar erythema
- Leukonychia (white nails)
- Clubbing
- Dupuytren's contracture
- Asterixis (flapping tremor — encephalopathy)
Face:
- Scleral icterus
- Parotid enlargement
- Kayser-Fleischer rings (exclude Wilson's — slit-lamp)
Chest:
- Spider naevi (> 5 pathological; count in distribution of SVC)
- Loss of axillary hair
Abdomen:
- Hepatomegaly: Palpate from RIF upwards; measure cm below costal margin; assess edge (smooth vs nodular), consistency (firm/hard in cirrhosis)
- Splenomegaly: Palpate from RIF towards LUQ; percuss Traube's space
- Ascites: Shifting dullness, fluid thrill (if gross ascites)
- Caput medusae: Dilated periumbilical veins (portal hypertension)
- Scars: Previous surgery/biopsy
Legs:
- Peripheral edema (hypoalbuminemia)
Other:
- Testicular atrophy (men)
- Loss of pubic hair
Complete Examination:
- "I would like to check temperature chart (SBP), urine dipstick (protein, blood), and perform a PR exam (melena)."
Viva Questions and Model Answers
Exam Detail: Q1: Interpret this serology: HBsAg negative, anti-HBc positive, anti-HBs negative.
A: This is isolated anti-HBc positivity. The differential includes:
- Window period of acute HBV (HBsAg cleared, anti-HBs not yet developed) — check anti-HBc IgM and ALT
- Remote infection with waning anti-HBs levels — patient has immune memory
- Occult hepatitis B — HBsAg below detection but virus persists; check HBV DNA
- False positive anti-HBc
I would check HBV DNA. If negative, I'd give a vaccine booster dose and check anti-HBs in 4 weeks — a rapid anamnestic response confirms past infection with immunity. If HBV DNA is positive, this is occult HBV and requires monitoring, especially if immunosuppression is planned.
Q2: A patient on chemotherapy for lymphoma was not screened for HBV beforehand. They develop acute hepatitis with ALT 800, bilirubin 120, and are found to be HBsAg positive, HBV DNA 10⁷ IU/mL. What has happened and how do you manage?
A: This is HBV reactivation triggered by chemotherapy-induced immunosuppression. The patient either had chronic HBV (missed due to lack of screening) or occult HBV (HBsAg negative but anti-HBc positive) that reactivated.
Mechanism: Chemotherapy (especially rituximab if used) suppresses the immune system, allowing dormant cccDNA in hepatocytes to replicate unchecked. When the immune system recovers, it attacks infected hepatocytes, causing immune-mediated hepatitis.
Immediate Management:
- Start antiviral therapy urgently: Tenofovir or entecavir (do NOT wait for confirmatory tests)
- Continue chemotherapy if oncologically critical (treat through the reactivation)
- Monitor closely: LFTs, coagulation (risk of acute liver failure)
- Consider HDV coinfection (can make reactivation more severe)
- Supportive care: IV fluids, avoid hepatotoxic drugs
- Transplant assessment if signs of acute liver failure (encephalopathy, INR > 1.5)
Prevention (for the future): This highlights the importance of universal HBV screening (HBsAg, anti-HBc, anti-HBs) before starting any immunosuppression. If HBsAg or anti-HBc positive, start prophylactic antivirals.
Q3: What are the treatment goals in chronic hepatitis B, and why can't we cure it?
A: The treatment goals are:
- Suppress HBV DNA to undetectable levels
- Normalize ALT (reduce necroinflammation)
- Prevent or reverse fibrosis and progression to cirrhosis
- Reduce HCC risk (though not eliminate)
- Achieve functional cure: HBsAg loss with or without anti-HBs seroconversion (ideal but rare — ~1-3% per year with NAs, up to 30% with peginterferon in selected patients)
Why we can't cure HBV: HBV establishes a stable nuclear cccDNA reservoir (covalently closed circular DNA) in hepatocytes. This episomal "minichromosome" is:
- NOT integrated into the host genome (so not targeted by integration-site therapies)
- NOT eliminated by current nucleos(t)ide analogues (which only block the polymerase/reverse transcriptase step of replication)
- Very stable with a long half-life (10-100 days per infected hepatocyte)
Complete cure would require either:
- Immune-mediated clearance of all infected hepatocytes (achievable in acute infection but rare in chronic due to T-cell exhaustion)
- Novel therapies targeting cccDNA (e.g., CRISPR/Cas9 gene editing, epigenetic silencing) — currently investigational
Therefore, current therapy aims for "functional cure" (HBsAg loss), which represents very low-level cccDNA with effective immune control, but virus can still reactivate if immunosuppression occurs.
Q4: A pregnant woman is HBsAg positive, HBeAg positive, HBV DNA 1,500,000 IU/mL. What is your management plan?
A: This patient has high viral load with HBeAg positivity, placing her baby at very high risk of vertical transmission (up to 90% without intervention).
Management Plan:
Antenatal:
- Antiviral therapy: Start tenofovir (TDF) 300mg daily at 28-32 weeks gestation (some start earlier if DNA > 10⁶)
- TDF is pregnancy category B with extensive safety data
- Goal: Reduce maternal HBV DNA to less than 200,000 IU/mL by delivery (reduces transmission risk to less than 5%)
- Monitor: Monthly HBV DNA and ALT
- Counsel: Explain rationale, safety, and neonatal plan
- Delivery: Normal vaginal delivery is safe (no evidence for C-section reducing transmission if prophylaxis given)
Neonatal (within 24 hours of birth):
- HBIG 0.5 mL IM within 12 hours (ideally within 6 hours)
- HBV vaccine 10 µg IM within 24 hours (different site from HBIG)
- Complete vaccine series: 0, 1, 6 months
Postnatal:
- Breastfeeding: Encouraged (safe if infant has received HBIG + vaccine)
- Maternal antivirals: Can continue TDF if breastfeeding (minimal secretion in breastmilk)
- If mother not requiring long-term treatment (non-cirrhotic, low DNA, normal ALT), can stop TDF after delivery and monitor
- Infant follow-up: Check HBsAg and anti-HBs at 9-12 months
- Goal: HBsAg negative, anti-HBs ≥10 mIU/mL (confirms protection)
Efficacy: With this protocol, transmission risk reduced from 90% to less than 5%.
Q5: When would you consider stopping antiviral therapy in chronic hepatitis B?
A: Stopping antiviral therapy is complex and depends on patient factors. General principles:
NEVER stop in:
- Cirrhosis (any stage) → lifelong treatment (risk of decompensation if stopped)
- Immunosuppressed patients → continue indefinitely
MAY consider stopping in NON-cirrhotic patients if:
Ideal ("Functional Cure"):
- HBsAg loss (with or without anti-HBs seroconversion)
- This occurs in ~1-3% per year on NAs
- Even after HBsAg loss, monitor 6-12 monthly (small risk of late relapse)
HBeAg-Positive Patients:
- HBeAg seroconversion (HBeAg loss + anti-HBe gain) PLUS
- Consolidation therapy for at least 12 months post-seroconversion PLUS
- HBV DNA undetectable PLUS
- Shared decision-making (explain 50% relapse risk)
HBeAg-Negative Patients:
- No clear endpoint → usually indefinite treatment
- Some guidelines suggest considering stopping after ≥3 years HBV DNA undetectable + normal ALT BUT high relapse risk (50-90%)
- Requires intensive monitoring if stopped
Stopping Protocol (if criteria met):
- Explain risks: Relapse risk ~50% (HBeAg+) to 90% (HBeAg−); small risk of severe flare
- Close monitoring:
- ALT + HBV DNA: Monthly × 3 months, then every 3 months × 12 months, then 6-monthly
- Retreat if:
- HBV DNA > 2000 IU/mL + ALT elevated
- Or clinical/biochemical hepatitis
My practice: I rarely stop in HBeAg-negative patients due to high relapse risk. I focus on achieving HBsAg loss (functional cure) as the endpoint.
Q6: Explain the difference between HBV genotypes and their clinical relevance.
A: There are 8 major HBV genotypes (A-H) with distinct geographic distribution and clinical implications:
Geographic Distribution:
- Genotype A: North America, Northern Europe, Sub-Saharan Africa
- Genotypes B & C: Asia-Pacific (China, Southeast Asia, Japan)
- Genotype D: Mediterranean, Middle East, India
- Genotype E: West Africa
Clinical Relevance:
-
HCC Risk:
- Genotype C > B in HCC risk (Asia-Pacific)
- Mechanism: Genotype C has mutations in basal core promoter → higher HBV DNA levels, delayed HBeAg seroconversion
-
Response to Interferon:
- Genotype A responds best to peginterferon (up to 40% HBeAg seroconversion)
- Genotype D poorest response (~20%)
- Genotype influences decision to offer interferon vs NAs
-
Natural History:
- Genotype C: Delayed HBeAg seroconversion (later in life → more cumulative liver damage)
- Genotype B: Earlier seroconversion but HCC can occur at younger age
-
Nucleos(t)ide Analogue Response:
- No significant difference — all genotypes respond equally to entecavir/tenofovir
Practical Use:
- Routine genotyping NOT required (doesn't change first-line NA choice)
- Consider if: Planning peginterferon therapy (Genotype A favors interferon)
- HCC surveillance: More intensive in Genotype C (though all chronic HBV need surveillance if high-risk)
14. Patient/Layperson Explanation
"What is Hepatitis B?"
"Hepatitis B is a virus that affects your liver. It's spread through blood and body fluids — for example, through unprotected sex, sharing needles, or from mother to baby during birth. Many people catch it as children in countries where it's common, and their immune system doesn't fully clear it, so it stays in the body long-term."
"Do I have it for life?"
"If you've had the virus for more than 6 months, it's called 'chronic' hepatitis B. While we can't completely cure it with current medicines, we can control it very effectively with a daily tablet — similar to how we manage high blood pressure or diabetes. The goal is to keep the virus 'asleep' so it doesn't damage your liver."
"Will I need treatment?"
"Not everyone needs treatment straight away. It depends on how active the virus is and whether it's causing inflammation in your liver. We check this with blood tests (virus levels and liver function tests) and sometimes scans. If the virus is very quiet and your liver is healthy, we may just monitor you regularly. If it's active, we'd recommend starting a tablet to suppress it and protect your liver."
"What's the medication like?"
"The main tablets we use are called tenofovir or entecavir. They're very safe, taken once a day, and work by stopping the virus from multiplying. Side effects are uncommon. If you have cirrhosis (severe scarring of the liver), you'll need to take the medication for life. If you don't have cirrhosis, we may be able to stop it in the future if the virus becomes completely inactive — but we'd monitor you very closely."
"Can I pass it to my family?"
"Hepatitis B is not spread by casual contact — you can't pass it by hugging, sharing food, or using the same toilet. The main risks are through blood contact or unprotected sex. We strongly recommend that all your household members and sexual partners get vaccinated — the vaccine is very effective (over 90% protection) and is given as three injections over 6 months. Your partner should use condoms until they're fully vaccinated."
"What about having children?"
"If you're pregnant or planning pregnancy, we can protect your baby very effectively. If your virus levels are high, we'll give you a safe tablet (tenofovir) in the last 3 months of pregnancy. When the baby is born, they'll get a hepatitis B vaccination and a special antibody injection within 24 hours — this reduces the risk of transmission to less than 5%. Breastfeeding is safe once your baby has had their injections."
"Will I get liver cancer?"
"Having hepatitis B does slightly increase the risk of liver cancer compared to people without the virus — but the risk is still low, especially if your liver isn't scarred (cirrhosis). This is why we do regular monitoring with ultrasound scans and blood tests every 6 months. Taking medication to suppress the virus reduces the cancer risk by more than half. If we do find anything early, there are excellent treatments available."
"Can I drink alcohol?"
"Alcohol and hepatitis B together significantly increase the risk of cirrhosis and liver cancer — much more than either alone. I'd strongly advise avoiding alcohol completely, or limiting it to very occasional small amounts. Even moderate drinking can accelerate liver damage."
"What should I avoid?"
- "Tell all your doctors and dentists that you have hepatitis B — especially before any surgery or if you're starting chemotherapy or other strong medications (some can 'wake up' the virus)."
- "Don't share razors, toothbrushes, or nail clippers (tiny blood contact risk)."
- "If you inject drugs, never share needles."
- "Some herbal remedies and supplements can harm your liver — always check with your doctor before taking anything new."
"What's the outlook?"
"With modern treatment and monitoring, most people with hepatitis B live normal, healthy lives. The key is keeping the virus suppressed and having regular check-ups. If we keep your liver healthy and prevent scarring, your long-term outlook is excellent."
15. Key Guidelines and Evidence
Guidelines
| Guideline | Organization | Year | Key Recommendations |
|---|---|---|---|
| Hepatitis B Management | EASL (European Association for the Study of the Liver) | 2017 | First-line: ETV or TDF/TAF; treat if cirrhosis, HBV DNA > 2000 + elevated ALT, or HCC risk; HCC surveillance 6-monthly |
| Hepatitis B Guidance | AASLD (American Association for the Study of Liver Diseases) | 2018 | Similar to EASL; emphasizes reactivation screening before immunosuppression |
| Global Hepatitis Strategy | WHO | 2016-2021 | 90% reduction in new infections, 65% reduction in mortality by 2030; universal infant vaccination |
| Hepatitis B in Pregnancy | ACOG (American College of Obstetricians and Gynecologists) | 2020 | Universal antenatal screening; TDF if HBV DNA > 200,000 IU/mL from 28-32 weeks; neonatal HBIG + vaccine |
| Prevention of HBV Reactivation | CDC | 2016 | Screen all patients for HBsAg and anti-HBc before immunosuppression; prophylactic antivirals for HBsAg+ or anti-HBc+ on high-risk agents |
Landmark Trials and Studies
| Study | Year | Key Finding | Impact |
|---|---|---|---|
| Marcellin et al. (NEJM) | 2008 | Tenofovir for 48 weeks achieved 76% HBV DNA suppression, 70% ALT normalization, histological improvement in 72% | Established TDF as first-line therapy [26] |
| Chang et al. (Hepatology) | 2010 | 5-year TDF: 74% showed regression of cirrhosis on biopsy | Proved NAs can reverse fibrosis/cirrhosis [53] |
| REVEAL-HBV Study | 2006 | Linear relationship between HBV DNA level and HCC risk; HBV DNA > 2000 IU/mL significantly increases risk | Established viral load thresholds for treatment [34] |
| Liaw et al. (Gastroenterology) | 2004 | Antiviral therapy reduced HCC incidence by 51% (HR 0.49) | Proved treatment reduces HCC risk [57] |
| Loomba et al. (NEJM) | 2015 | Reactivation occurred in 20-50% of HBsAg+ patients on rituximab without prophylaxis; 5-10% in anti-HBc+ patients | Established reactivation risk, mandated screening [42] |
| Chan et al. (Lancet) | 2016 | Tenofovir in pregnancy (HBV DNA > 200,000) reduced vertical transmission from 18% to 5% | Standard of care for high viral load in pregnancy [37] |
| Zhang et al. (Lancet Oncology) | 2004 | Biannual surveillance with AFP + USS reduced HCC mortality by 37% in RCT | Proved effectiveness of HCC surveillance [35] |
| Buti et al. (J Hepatol) | 2015 | TAF non-inferior to TDF for efficacy, superior bone/renal safety at 96 weeks | TAF as alternative first-line for those at risk of TDF toxicity [27] |
16. Summary: High-Yield Points for Exams
Must-Know Facts:
- HBV is a partially dsDNA virus; cccDNA reservoir in nucleus makes cure impossible with current NAs
- Chronicity risk: 90% (neonates), less than 5% (adults)
- Serology: HBsAg = infection; anti-HBs = immunity; anti-HBc = exposure (NOT vaccine)
- Isolated anti-HBc: Window period, occult HBV, or remote infection (check HBV DNA)
- Natural history: 4 phases (immune tolerant → immune active → inactive carrier → HBeAg-negative CHB)
- First-line treatment: Entecavir or tenofovir (TDF/TAF)
- Treat if: Cirrhosis (any DNA/ALT) OR HBV DNA > 2000 + elevated ALT + inflammation
- HCC surveillance: 6-monthly USS + AFP in cirrhotics, high-risk non-cirrhotics
- Pregnancy (HBV DNA > 200,000): TDF from 28-32 weeks + neonatal HBIG + vaccine within 24 hours
- Reactivation: Screen HBsAg + anti-HBc before immunosuppression; prophylactic TDF/ETV if positive (especially rituximab)
- Vaccine: 0, 1, 6 months; check anti-HBs ≥10 mIU/mL; 5-10% non-responders (repeat with double dose)
- HDV superinfection: Rapid progression; treat with pegIFN or bulevirtide
- Extrahepatic: PAN, membranous GN (immune-complex mediated)
- Functional cure (HBsAg loss): 1-3% per year with NAs, up to 30% with pegIFN (selected patients)
- HCC can occur in non-cirrhotic HBV (unique vs HCV)
17. References
-
Seeger C, Mason WS. Molecular biology of hepatitis B virus infection. Virology. 2015;479-480:672-686. doi:10.1016/j.virol.2015.02.031
-
Nassal M. HBV cccDNA: viral persistence reservoir and key obstacle for a cure of chronic hepatitis B. Gut. 2015;64(12):1972-1984. doi:10.1136/gutjnl-2015-309809
-
WHO. Global hepatitis report 2024. World Health Organization; 2024. https://www.who.int/publications/i/item/9789240091672
-
Terrault NA, Lok ASF, McMahon BJ, et al. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance. Hepatology. 2018;67(4):1560-1599. doi:10.1002/hep.29800
-
Schweitzer A, Horn J, Mikolajczyk RT, Krause G, Ott JJ. Estimations of worldwide prevalence of chronic hepatitis B virus infection: a systematic review of data published between 1965 and 2013. Lancet. 2015;386(10003):1546-1555. doi:10.1016/S0140-6736(15)61412-X
-
Lin CL, Kao JH. Hepatitis B virus genotypes and variants. Cold Spring Harb Perspect Med. 2015;5(5):a021436. doi:10.1101/cshperspect.a021436
-
McMahon BJ. The natural history of chronic hepatitis B virus infection. Hepatology. 2009;49(5 Suppl):S45-S55. doi:10.1002/hep.22898
-
Seeger C, Zoulim F, Mason WS. Hepadnaviruses. In: Knipe DM, Howley PM, eds. Fields Virology. 7th ed. Wolters Kluwer; 2021:2185-2221.
-
Revill PA, Chisari FV, Block JM, et al. A global scientific strategy to cure hepatitis B. Lancet Gastroenterol Hepatol. 2019;4(7):545-558. doi:10.1016/S2468-1253(19)30119-0
-
Bertoletti A, Ferrari C. Adaptive immunity in HBV infection. J Hepatol. 2016;64(1 Suppl):S71-S83. doi:10.1016/j.jhep.2016.01.026
-
Guidotti LG, Chisari FV. Immunobiology and pathogenesis of viral hepatitis. Annu Rev Pathol. 2006;1:23-61. doi:10.1146/annurev.pathol.1.110304.100230
-
Schurich A, Pallett LJ, Jajbhay D, Wijngaarden J, Otano I, Gill US, et al. Distinct metabolic requirements of exhausted and functional virus-specific CD8 T cells in the same host. Cell Rep. 2016;16(5):1243-1252. doi:10.1016/j.celrep.2016.06.078
-
European Association for the Study of the Liver. EASL 2017 Clinical Practice Guidelines on the management of hepatitis B virus infection. J Hepatol. 2017;67(2):370-398. doi:10.1016/j.jhep.2017.03.021
-
Raimondo G, Locarnini S, Pollicino T, Levrero M, Zoulim F, Lok AS. Update of the statements on biology and clinical impact of occult hepatitis B virus infection. J Hepatol. 2019;71(2):397-408. doi:10.1016/j.jhep.2019.03.034
-
Liaw YF, Chu CM. Hepatitis B virus infection. Lancet. 2009;373(9663):582-592. doi:10.1016/S0140-6736(09)60207-5
-
Lee WM, Squires RH Jr, Nyberg SL, Doo E, Hoofnagle JH. Acute liver failure: summary of a workshop. Hepatology. 2008;47(4):1401-1415. doi:10.1002/hep.22177
-
Fattovich G, Bortolotti F, Donato F. Natural history of chronic hepatitis B: special emphasis on disease progression and prognostic factors. J Hepatol. 2008;48(2):335-352. doi:10.1016/j.jhep.2007.11.011
-
Cacoub P, Terrier B. Hepatitis B-related autoimmune manifestations. Rheum Dis Clin North Am. 2009;35(1):125-137. doi:10.1016/j.rdc.2009.03.006
-
Abara WE, Qaseem A, Schillie S, McMahon BJ, Harris AM; High Value Care Task Force of the American College of Physicians. Hepatitis B vaccination, screening, and linkage to care: best practice advice from the American College of Physicians and CDC. Ann Intern Med. 2017;167(11):794-804. doi:10.7326/M17-1106
-
Grob P, Jilg W, Bornhak H, et al. Serological pattern "anti-HBc alone": report on a workshop. J Med Virol. 2000;62(4):450-455.
-
Cornberg M, Wong VW, Locarnini S, Brunetto M, Janssen HLA, Chan HL. The role of quantitative hepatitis B surface antigen revisited. J Hepatol. 2017;66(2):398-411. doi:10.1016/j.jhep.2016.08.009
-
European Association for the Study of the Liver. EASL 2017 Clinical Practice Guidelines on the management of hepatitis B virus infection. J Hepatol. 2017;67(2):370-398. doi:10.1016/j.jhep.2017.03.021
-
European Association for Study of Liver; Asociacion Latinoamericana para el Estudio del Higado. EASL-ALEH Clinical Practice Guidelines: non-invasive tests for evaluation of liver disease severity and prognosis. J Hepatol. 2015;63(1):237-264. doi:10.1016/j.jhep.2015.04.006
-
Brunt EM, Janney CG, Di Bisceglie AM, Neuschwander-Tetri BA, Bacon BR. Nonalcoholic steatohepatitis: a proposal for grading and staging the histological lesions. Am J Gastroenterol. 1999;94(9):2467-2474. doi:10.1111/j.1572-0241.1999.01377.x
-
Tzartzeva K, Obi J, Rich NE, et al. Surveillance imaging and alpha fetoprotein for early detection of hepatocellular carcinoma in patients with cirrhosis: a meta-analysis. Gastroenterology. 2018;154(6):1706-1718.e1. doi:10.1053/j.gastro.2018.01.064
-
Marcellin P, Heathcote EJ, Buti M, et al. Tenofovir disoproxil fumarate versus adefovir dipivoxil for chronic hepatitis B. N Engl J Med. 2008;359(23):2442-2455. doi:10.1056/NEJMoa0802878
-
Buti M, Gane E, Seto WK, et al. Tenofovir alafenamide versus tenofovir disoproxil fumarate for the treatment of patients with HBeAg-negative chronic hepatitis B virus infection: a randomised, double-blind, phase 3, non-inferiority trial. Lancet Gastroenterol Hepatol. 2016;1(3):196-206. doi:10.1016/S2468-1253(16)30107-8
-
Papatheodoridis GV, Manolakopoulos S, Touloumi G, et al. Hepatocellular carcinoma risk in HBeAg-negative chronic hepatitis B patients with or without cirrhosis treated with entecavir: HepNet.Greece cohort. J Viral Hepat. 2017;24(10):868-876. doi:10.1111/jvh.12725
-
Chang TT, Liaw YF, Wu SS, et al. Long-term entecavir therapy results in the reversal of fibrosis/cirrhosis and continued histological improvement in patients with chronic hepatitis B. Hepatology. 2010;52(3):886-893. doi:10.1002/hep.23785
-
Janssen HLA, van Zonneveld M, Senturk H, et al. Pegylated interferon alfa-2b alone or in combination with lamivudine for HBeAg-positive chronic hepatitis B: a randomised trial. Lancet. 2005;365(9454):123-129. doi:10.1016/S0140-6736(05)17701-0
-
Sonneveld MJ, Hansen BE, Piratvisuth T, et al. Response-guided peginterferon therapy in hepatitis B e antigen-positive chronic hepatitis B using serum hepatitis B surface antigen levels. Hepatology. 2013;58(3):872-880. doi:10.1002/hep.26436
-
Rijckborst V, Hansen BE, Cakaloglu Y, et al. Early on-treatment prediction of response to peginterferon alfa-2a for HBeAg-negative chronic hepatitis B using HBsAg and HBV DNA levels. Hepatology. 2010;52(2):454-461. doi:10.1002/hep.23722
-
Revill PA, Chisari FV, Block JM, et al. A global scientific strategy to cure hepatitis B. Lancet Gastroenterol Hepatol. 2019;4(7):545-558. doi:10.1016/S2468-1253(19)30119-0
-
Chen CJ, Yang HI, Su J, et al; REVEAL-HBV Study Group. Risk of hepatocellular carcinoma across a biological gradient of serum hepatitis B virus DNA level. JAMA. 2006;295(1):65-73. doi:10.1001/jama.295.1.65
-
Zhang BH, Yang BH, Tang ZY. Randomized controlled trial of screening for hepatocellular carcinoma. J Cancer Res Clin Oncol. 2004;130(7):417-422. doi:10.1007/s00432-004-0552-0
-
Marrero JA, Kulik LM, Sirlin CB, et al. Diagnosis, staging, and management of hepatocellular carcinoma: 2018 practice guidance by the American Association for the Study of Liver Diseases. Hepatology. 2018;68(2):723-750. doi:10.1002/hep.29913
-
Pan CQ, Duan Z, Dai E, et al. Tenofovir to prevent hepatitis B transmission in mothers with high viral load. N Engl J Med. 2016;374(24):2324-2334. doi:10.1056/NEJMoa1508660
-
Lee C, Gong Y, Brok J, Boxall EH, Gluud C. Hepatitis B immunisation for newborn infants of hepatitis B surface antigen-positive mothers. Cochrane Database Syst Rev. 2006;(2):CD004790. doi:10.1002/14651858.CD004790.pub2
-
Shi Z, Yang Y, Wang H, et al. Breastfeeding of newborns by mothers carrying hepatitis B virus: a meta-analysis and systematic review. Arch Pediatr Adolesc Med. 2011;165(9):837-846. doi:10.1001/archpediatrics.2011.72
-
Perrillo RP, Gish R, Falck-Ytter YT. American Gastroenterological Association Institute technical review on prevention and treatment of hepatitis B virus reactivation during immunosuppressive drug therapy. Gastroenterology. 2015;148(1):221-244.e3. doi:10.1053/j.gastro.2014.10.038
-
Reddy KR, Beavers KL, Hammond SP, Lim JK, Falck-Ytter YT; American Gastroenterological Association Institute. American Gastroenterological Association Institute guideline on the prevention and treatment of hepatitis B virus reactivation during immunosuppressive drug therapy. Gastroenterology. 2015;148(1):215-219. doi:10.1053/j.gastro.2014.10.039
-
Loomba R, Liang TJ. Hepatitis B reactivation associated with immune suppressive and biological modifier therapies: current concepts, management strategies, and future directions. Gastroenterology. 2017;152(6):1297-1309. doi:10.1053/j.gastro.2017.02.009
-
Weinbaum CM, Williams I, Mast EE, et al; Centers for Disease Control and Prevention. Recommendations for identification and public health management of persons with chronic hepatitis B virus infection. MMWR Recomm Rep. 2008;57(RR-8):1-20.
-
Rizzetto M. Hepatitis D virus: introduction and epidemiology. Cold Spring Harb Perspect Med. 2015;5(7):a021576. doi:10.1101/cshperspect.a021576
-
Wedemeyer H, Yurdaydin C, Dalekos GN, et al; HIDIT Study Group. Peginterferon plus adefovir versus either drug alone for hepatitis delta. N Engl J Med. 2011;364(4):322-331. doi:10.1056/NEJMoa0912696
-
Thio CL, Seaberg EC, Skolasky R Jr, et al. HIV-1, hepatitis B virus, and risk of liver-related mortality in the Multicenter Cohort Study (MACS). Lancet. 2002;360(9349):1921-1926. doi:10.1016/s0140-6736(02)11913-1
-
Andre FE. Summary of safety and efficacy data on a yeast-derived hepatitis B vaccine. Am J Med. 1989;87(3A):14S-20S. doi:10.1016/0002-9343(89)90524-5
-
Schillie S, Vellozzi C, Reingold A, et al. Prevention of hepatitis B virus infection in the United States: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep. 2018;67(1):1-31. doi:10.15585/mmwr.rr6701a1
-
Hollinger FB, Liang TJ. Hepatitis B virus. In: Knipe DM, Howley PM, eds. Fields Virology. 6th ed. Lippincott Williams & Wilkins; 2013:2185-2221.
-
Schillie S, Murphy TV, Sawyer M, et al. CDC guidance for evaluating health-care personnel for hepatitis B virus protection and for administering postexposure management. MMWR Recomm Rep. 2013;62(RR-10):1-19.
-
Lok ASF, McMahon BJ. Chronic hepatitis B: update 2009. Hepatology. 2009;50(3):661-662. doi:10.1002/hep.23190
-
Fattovich G, Bortolotti F, Donato F. Natural history of chronic hepatitis B: special emphasis on disease progression and prognostic factors. J Hepatol. 2008;48(2):335-352. doi:10.1016/j.jhep.2007.11.011
-
Marcellin P, Gane E, Buti M, et al. Regression of cirrhosis during treatment with tenofovir disoproxil fumarate for chronic hepatitis B: a 5-year open-label follow-up study. Lancet. 2013;381(9865):468-475. doi:10.1016/S0140-6736(12)61425-1
-
Papatheodoridis G, Dalekos G, Sypsa V, et al. PAGE-B predicts the risk of developing hepatocellular carcinoma in Caucasians with chronic hepatitis B on 5-year antiviral therapy. J Hepatol. 2016;64(4):800-806. doi:10.1016/j.jhep.2015.11.035
-
World Health Organization. Hepatitis B vaccines: WHO position paper – July 2017. Wkly Epidemiol Rec. 2017;92(27):369-392.
-
World Health Organization. Global health sector strategy on viral hepatitis 2016-2021. Geneva: WHO; 2016. https://www.who.int/publications/i/item/WHO-HIV-2016.06
-
Liaw YF, Sung JJ, Chow WC, et al; Cirrhosis Asian Lamivudine Multicentre Study Group. Lamivudine for patients with chronic hepatitis B and advanced liver disease. N Engl J Med. 2004;351(15):1521-1531. doi:10.1056/NEJMoa033364
END OF ENHANCED TOPIC
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Liver Function Tests Interpretation
- Viral Hepatitis Overview
Differentials
Competing diagnoses and look-alikes to compare.
- Hepatitis A
- Hepatitis C
- Autoimmune Hepatitis
- Non-Alcoholic Fatty Liver Disease
Consequences
Complications and downstream problems to keep in mind.