Hyperparathyroidism (Adult)
Hyperparathyroidism encompasses a spectrum of disorders characterised by excessive parathyroid hormone (PTH) secretion, ... MRCP exam preparation.
What matters first
Hyperparathyroidism encompasses a spectrum of disorders characterised by excessive parathyroid hormone (PTH) secretion, ... MRCP exam preparation.
Severe Hypercalcaemia (less than 3.5 mmol/L) → Cardiac Arrhythmia Risk
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Severe Hypercalcaemia (less than 3.5 mmol/L) → Cardiac Arrhythmia Risk
- Acute Pancreatitis
- Hypercalcaemic Crisis (Confusion/Coma)
- Neck Hematoma Post-operatively → Airway Obstruction
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Familial Hypocalciuric Hypercalcaemia
- Hypercalcaemia of Malignancy
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Hyperparathyroidism (Adult)
1. Overview
Hyperparathyroidism encompasses a spectrum of disorders characterised by excessive parathyroid hormone (PTH) secretion, leading to disturbances in calcium homeostasis. The condition is classified into three distinct forms: primary (autonomous gland dysfunction), secondary (physiological compensation), and tertiary (autonomous hyperfunction following prolonged secondary disease). Primary hyperparathyroidism (PHPT) is the most common cause of hypercalcaemia in the outpatient setting, with an estimated prevalence of 1-4 per 1000 in the general population. [1,2] In contrast, malignancy-associated hypercalcaemia predominates in the inpatient setting.
The clinical presentation of hyperparathyroidism has evolved dramatically over recent decades. Historically, patients presented with the classical tetrad of "bones, stones, abdominal groans, and psychic moans," reflecting advanced disease with skeletal manifestations, nephrolithiasis, gastrointestinal symptoms, and neuropsychiatric disturbances. Contemporary series indicate that 80-85% of patients with PHPT are now classified as "asymptomatic," detected incidentally through routine biochemical screening. [3,4] However, detailed assessment often reveals subtle symptoms including fatigue, cognitive impairment, and bone pain that improve following parathyroidectomy.
Secondary hyperparathyroidism develops as a compensatory response to chronic hypocalcaemia, most commonly secondary to vitamin D deficiency or chronic kidney disease (CKD). In CKD, progressive nephron loss impairs phosphate excretion and calcitriol synthesis, stimulating PTH secretion. Tertiary hyperparathyroidism represents autonomous parathyroid hyperfunction that develops after prolonged secondary stimulation, typically in advanced CKD or following renal transplantation. [5]
Understanding the pathophysiology, diagnostic approach, and management strategies for each form of hyperparathyroidism is essential for endocrinologists, nephrologists, surgeons, and general practitioners.
2. Epidemiology
Primary Hyperparathyroidism
Prevalence and Incidence
Primary hyperparathyroidism affects approximately 0.3-1% of the general population, with higher prevalence (2-3%) in postmenopausal women. [1,6] The annual incidence is estimated at 66 per 100,000 in women and 25 per 100,000 in men in developed countries. The introduction of automated multichannel biochemical analysers in the 1970s led to a marked increase in detection of asymptomatic cases.
| Demographic | Value | Source |
|---|---|---|
| Overall prevalence | 0.3-1% of general population | [1] |
| Postmenopausal women prevalence | 2-3% | [6] |
| Female:Male ratio | 3-4:1 | [2] |
| Peak age of onset | 50-70 years | [3] |
| Annual incidence (women) | 66 per 100,000 | [7] |
| Annual incidence (men) | 25 per 100,000 | [7] |
Risk Factors
- Female sex: 3-4 fold increased risk
- Age: Incidence increases with age, peaking in the sixth to seventh decades
- Prior neck irradiation: 2-3 fold increased risk [8]
- Lithium therapy: Associated with hypercalcaemia in 10-15% of long-term users [9]
- Familial syndromes: Multiple Endocrine Neoplasia (MEN) types 1 and 2A, familial isolated hyperparathyroidism
- Thiazide diuretics: May unmask latent hyperparathyroidism
Secondary Hyperparathyroidism
Secondary hyperparathyroidism affects the majority of patients with advanced CKD. In patients with estimated glomerular filtration rate (eGFR) less than 60 mL/min/1.73m², the prevalence of elevated PTH exceeds 50%, rising to > 80% in those with eGFR less than 30 mL/min/1.73m². [10] Vitamin D deficiency, another major cause, is highly prevalent globally, affecting an estimated 1 billion people worldwide.
Tertiary Hyperparathyroidism
Tertiary hyperparathyroidism develops in approximately 1-5% of patients with long-standing secondary hyperparathyroidism, particularly those on dialysis for > 5 years or following renal transplantation. [5]
3. Aetiology and Pathophysiology
Aetiology by Type
Primary Hyperparathyroidism
The underlying pathology differs across cases:
- Solitary parathyroid adenoma: 80-85% of cases [11]
- Multiglandular hyperplasia: 10-15% of cases
- Parathyroid carcinoma: less than 1% of cases
- Multiple adenomas: 3-5% of cases
Genetic Causes
- MEN1 syndrome: Autosomal dominant disorder (menin gene mutation on chromosome 11q13) associated with parathyroid hyperplasia, pancreatic neuroendocrine tumours, and pituitary adenomas. Parathyroid involvement occurs in > 90% by age 50. [12]
- MEN2A syndrome: Autosomal dominant disorder (RET proto-oncogene mutation) characterised by medullary thyroid carcinoma, phaeochromocytoma, and parathyroid hyperplasia (20-30% of cases).
- Familial isolated hyperparathyroidism (FIHP): Mutations in MEN1, CASR, or CDC73 genes
- Hyperparathyroidism-jaw tumour syndrome: CDC73 gene mutations, associated with increased risk of parathyroid carcinoma
Acquired Causes
- Sporadic adenoma formation: Clonal proliferation with loss of heterozygosity at multiple loci
- Prior neck irradiation: Latency period of 20-40 years
- Lithium therapy: Shifts calcium-sensing receptor (CaSR) set-point, leading to hypercalcaemia
Secondary Hyperparathyroidism
Primary Causes
-
Chronic kidney disease (most common in developed countries):
- Phosphate retention → hypocalcaemia
- Impaired 1α-hydroxylase activity → reduced calcitriol synthesis
- Skeletal resistance to PTH action
- Increased FGF-23 levels suppressing calcitriol
-
Vitamin D deficiency (most common globally):
- Inadequate dietary intake
- Reduced sun exposure
- Malabsorption (coeliac disease, inflammatory bowel disease, bariatric surgery)
- Chronic liver disease (impaired 25-hydroxylation)
-
Calcium malabsorption:
- Intestinal diseases (coeliac disease, Crohn's disease)
- Post-gastrectomy/bariatric surgery
- Chronic pancreatitis
Tertiary Hyperparathyroidism
Develops from prolonged secondary hyperparathyroidism with:
- Monoclonal parathyroid cell proliferation
- Loss of sensitivity to calcium and vitamin D suppression
- Autonomous PTH secretion despite normal/elevated serum calcium
- Typically occurs after years of dialysis or following renal transplantation [5]
Pathophysiology
Normal Parathyroid Hormone Function
PTH is an 84-amino acid polypeptide secreted by parathyroid chief cells in response to hypocalcaemia. The calcium-sensing receptor (CaSR) on parathyroid cells detects ionised calcium levels and regulates PTH secretion through a negative feedback loop.
PTH Actions
- Bone: Stimulates osteoclastic bone resorption, releasing calcium and phosphate
- Kidney:
- Increases calcium reabsorption in distal convoluted tubule
- Decreases phosphate reabsorption in proximal tubule
- Stimulates 1α-hydroxylase, converting 25(OH)D to active 1,25(OH)₂D
- Intestine (indirect): Active vitamin D increases intestinal calcium absorption
Exam Detail: #### Molecular Mechanisms in Primary Hyperparathyroidism
Loss of Normal Feedback Regulation
In adenomatous tissue, several mechanisms lead to autonomous PTH secretion:
-
Reduced CaSR expression: Decreased receptor density on parathyroid cells reduces sensitivity to calcium, shifting the set-point for PTH suppression to higher calcium concentrations [13]
-
Altered vitamin D receptor signalling: Reduced expression of vitamin D receptors (VDR) in hyperplastic/adenomatous tissue diminishes calcitriol's suppressive effect on PTH synthesis
-
Cell cycle dysregulation: Overexpression of cyclin D1 (PRAD1 gene on chromosome 11q13) drives parathyroid cell proliferation in a subset of adenomas. PTH gene rearrangement brings the PTH gene under control of cyclin D1 promoter.
-
Clonal proliferation: Loss of heterozygosity at tumour suppressor loci (MEN1, HRPT2/CDC73) permits uncontrolled cell growth
Biochemical Consequences
- Hypercalcaemia: Elevated PTH increases bone resorption, renal calcium reabsorption, and (via calcitriol) intestinal absorption
- Hypophosphataemia: PTH-mediated phosphaturia exceeds phosphate release from bone
- Elevated or inappropriately normal PTH: The key diagnostic finding—PTH should be suppressed in the presence of hypercalcaemia
- Metabolic acidosis (mild): Enhanced renal bicarbonate excretion
- Elevated bone turnover markers: Increased alkaline phosphatase, C-telopeptide, P1NP
Pathophysiology in Secondary Hyperparathyroidism (CKD)
Progressive Renal Dysfunction
-
Phosphate retention (GFR less than 60 mL/min):
- Direct stimulation of PTH secretion
- Increased FGF-23 secretion from bone
- FGF-23 suppresses calcitriol synthesis and increases phosphaturia
-
Calcitriol deficiency:
- Reduced 1α-hydroxylase expression in diseased kidneys
- FGF-23-mediated suppression
- Loss of negative feedback on PTH gene transcription
- Reduced intestinal calcium absorption → hypocalcaemia
-
Hypocalcaemia:
- Reduced calcitriol-mediated intestinal absorption
- Hyperphosphataemia-induced calcium precipitation
- Skeletal resistance to PTH action
-
Parathyroid gland hyperplasia:
- Reduced CaSR and VDR expression in hyperplastic glands
- Reduced sensitivity to calcium and calcitriol suppression
- Progressive parathyroid cell proliferation
-
Skeletal resistance:
- Downregulation of PTH receptors in bone
- Uraemic toxins impair osteoblast function
- "Relative" PTH resistance requires higher levels to maintain normocalcaemia
CKD-Mineral and Bone Disorder (CKD-MBD)
The constellation of biochemical abnormalities (hyperphosphataemia, hypocalcaemia, elevated PTH, FGF-23, reduced calcitriol), bone disease (renal osteodystrophy), and vascular calcification represents CKD-MBD. [10]
Osteitis Fibrosa Cystica
In severe, prolonged hyperparathyroidism (primary or secondary), excessive osteoclastic activity produces:
- Subperiosteal bone resorption (phalanges, distal clavicles)
- Bone cysts and "brown tumours" (haemorrhagic, cystic lesions)
- "Salt and pepper" skull appearance on radiographs
- Rugger jersey spine (sclerosis of vertebral endplates)
This pattern is now rare in primary disease due to early detection but may still occur in untreated secondary/tertiary hyperparathyroidism.
4. Clinical Presentation
Primary Hyperparathyroidism
Historical "Classical" Presentation
Prior to the era of automated biochemistry, patients presented with florid symptoms:
"Bones, Stones, Abdominal Groans, and Psychic Moans"
-
Bones:
- Osteitis fibrosa cystica (now rare)
- Brown tumours (giant cell reparative granulomas)
- Bone pain and pathological fractures
- Osteoporosis and osteopenia
-
Stones:
- Nephrolithiasis (calcium oxalate or calcium phosphate stones) in 15-20% [14]
- Nephrocalcinosis
- Recurrent urolithiasis
-
Abdominal Groans:
- Constipation (calcium-induced reduced gut motility)
- Nausea and anorexia
- Peptic ulcer disease (calcium stimulates gastrin secretion)
- Acute and chronic pancreatitis
-
Psychic Moans:
- Depression and anxiety
- Cognitive impairment ("brain fog")
- Fatigue and lethargy
- In severe cases: confusion, psychosis
Contemporary "Asymptomatic" Presentation
Modern series report 80-85% of patients are "asymptomatic" at diagnosis. However, systematic assessment reveals:
Subtle Symptoms
- Neurocognitive: Impaired concentration, memory difficulties, fatigue (reported in 40-60%)
- Musculoskeletal: Vague bone pain, proximal muscle weakness
- Neuropsychiatric: Depression (prevalence 30-40% vs 10-15% in controls) [15]
- Gastrointestinal: Mild constipation, dyspepsia
- Renal: Polyuria and polydipsia (PTH-induced nephrogenic diabetes insipidus)
Studies demonstrate improvement in quality of life, fatigue, and cognitive function following parathyroidectomy in "asymptomatic" patients, suggesting these symptoms are under-recognised. [16]
Target Organ Involvement
Even in asymptomatic disease:
- Osteoporosis: Present in 15-30%; predominantly affects cortical bone (distal radius, femoral neck)
- Reduced bone mineral density: T-score < -2.5 in approximately 25% [3]
- Silent kidney stones: Detected on imaging in 10-15%
- Cardiovascular effects: Hypertension (40-60%), left ventricular hypertrophy, valvular calcification
- Cardiovascular mortality: Increased standardised mortality ratio (1.5-2.0) even in mild disease [17]
Physical Examination Findings
- Neck: Usually unremarkable (adenomas typically less than 1-2 cm and located posteriorly/inferiorly). A palpable neck mass suggests thyroid pathology or, rarely, parathyroid carcinoma.
- Blood pressure: Hypertension in 40-60% (mechanisms include vascular stiffness, increased renin secretion, impaired endothelial function)
- Neuromuscular: Proximal muscle weakness, hyporeflexia (in severe hypercalcaemia)
- Band keratopathy: Calcium deposition in corneal limbus (rare, seen only in chronic, severe hypercalcaemia)
- Chondrocalcinosis: Calcium pyrophosphate deposition in joints
Parathyroid Crisis (Hypercalcaemic Crisis)
A rare, life-threatening complication (serum calcium typically > 3.5-4.0 mmol/L):
Clinical Features
- Severe dehydration (polyuria with inadequate replacement)
- Nausea, vomiting, abdominal pain
- Cardiac arrhythmias
- Acute kidney injury (pre-renal and intrinsic)
- Pancreatitis
Triggers: Dehydration, immobilisation, intercurrent illness
Secondary Hyperparathyroidism
Clinical Presentation in CKD
Symptoms relate to underlying renal failure and mineral-bone disorder:
- Bone pain (renal osteodystrophy)
- Pruritus (hyperphosphataemia)
- Muscle weakness
- Vascular calcification (asymptomatic until complications develop)
- Fractures (adynamic bone disease or osteitis fibrosa)
Serum calcium is typically low or normal; PTH is elevated.
Clinical Presentation in Vitamin D Deficiency
- Often asymptomatic
- Bone pain and muscle weakness (osteomalacia)
- Proximal myopathy (difficulty rising from chair, climbing stairs)
- Increased fracture risk
Serum calcium may be low or low-normal; PTH is elevated.
Tertiary Hyperparathyroidism
Develops in long-standing dialysis patients or following renal transplantation:
- Hypercalcaemia (key distinguishing feature from secondary)
- Persistent bone pain
- Pruritus
- Soft tissue and vascular calcification
- Calciphylaxis (rare, severe complication with skin necrosis)
5. Differential Diagnosis
Hypercalcaemia has numerous causes; the differential depends on PTH levels and clinical context.
Hypercalcaemia with Elevated or Inappropriately Normal PTH
| Condition | Key Distinguishing Features | Diagnostic Tests |
|---|---|---|
| Primary hyperparathyroidism | Most common outpatient cause; chronic, mild-moderate hypercalcaemia | High/normal PTH, high calcium, low phosphate, urine calcium elevated |
| Familial hypocalciuric hypercalcaemia (FHH) | Benign genetic condition; lifelong mild hypercalcaemia; family history | Calcium:creatinine clearance ratio less than 0.01, genetic testing (CASR mutation) |
| Tertiary hyperparathyroidism | History of CKD or dialysis; long-standing secondary hyperparathyroidism | CKD history, very high PTH (often > 500 pg/mL) |
| Lithium-induced hyperparathyroidism | History of bipolar disorder/lithium use; may persist after lithium cessation | Medication history, elevated PTH |
Hypercalcaemia with Suppressed PTH
| Condition | Key Distinguishing Features |
|---|---|
| Malignancy | Most common cause of inpatient hypercalcaemia; often severe, acute; known cancer or constitutional symptoms |
| Vitamin D toxicity | History of excessive supplementation; elevated 25(OH)D levels (> 150 ng/mL) |
| Granulomatous disease | Sarcoidosis, tuberculosis; extra-renal calcitriol production; elevated 1,25(OH)₂D |
| Thyrotoxicosis | Hyperthyroid symptoms; increased bone turnover |
| Milk-alkali syndrome | Excessive calcium and alkali ingestion (antacids, calcium supplements) |
| Thiazide diuretics | Usually mild; resolves with cessation |
Exam Detail: ### Critical Distinction: FHH vs PHPT
Familial Hypocalciuric Hypercalcaemia (FHH) is a benign autosomal dominant condition caused by inactivating mutations in the calcium-sensing receptor (CASR) gene. It mimics PHPT biochemically but requires no treatment. Parathyroidectomy is ineffective and inappropriate.
Distinguishing Features
| Feature | PHPT | FHH |
|---|---|---|
| Inheritance | Usually sporadic | Autosomal dominant (family history) |
| Age of onset | Adulthood (peak 50-70 years) | Present from birth/childhood |
| Serum calcium | 2.65-3.5 mmol/L | 2.65-3.0 mmol/L (mild elevation) |
| Urinary calcium | Usually elevated (> 200 mg/24h) | Low (less than 100 mg/24h) |
| CCCR | > 0.02 | less than 0.01 (key test) |
| PTH | Elevated or inappropriately normal | Mildly elevated or normal |
| Response to parathyroidectomy | Curative | No effect |
| Complications | Osteoporosis, stones, fractures | None (benign) |
Calcium:Creatinine Clearance Ratio (CCCR)
CCCR = (Urine calcium × Serum creatinine) / (Serum calcium × Urine creatinine)
- CCCR less than 0.01: FHH (do NOT operate)
- CCCR > 0.02: PHPT
- CCCR 0.01-0.02: Indeterminate (genetic testing for CASR mutations may be required)
Clinical Pearl: Always calculate CCCR or measure 24-hour urinary calcium before referring for parathyroid surgery. Missing FHH results in unnecessary surgery with no benefit and potential harm.
6. Investigations
Biochemical Diagnosis
Initial Testing
Serum Calcium (Corrected or Ionised)
- Corrected calcium = measured calcium + 0.02 × (40 - albumin in g/L)
- Ionised calcium is more accurate but less widely available
- Hypercalcaemia: corrected calcium > 2.60 mmol/L (> 10.4 mg/dL)
- Repeat testing: Essential to confirm persistent hypercalcaemia (exclude spurious results, dehydration)
Parathyroid Hormone (PTH)
- Second- or third-generation immunoassays measuring intact PTH (1-84)
- Interpretation:
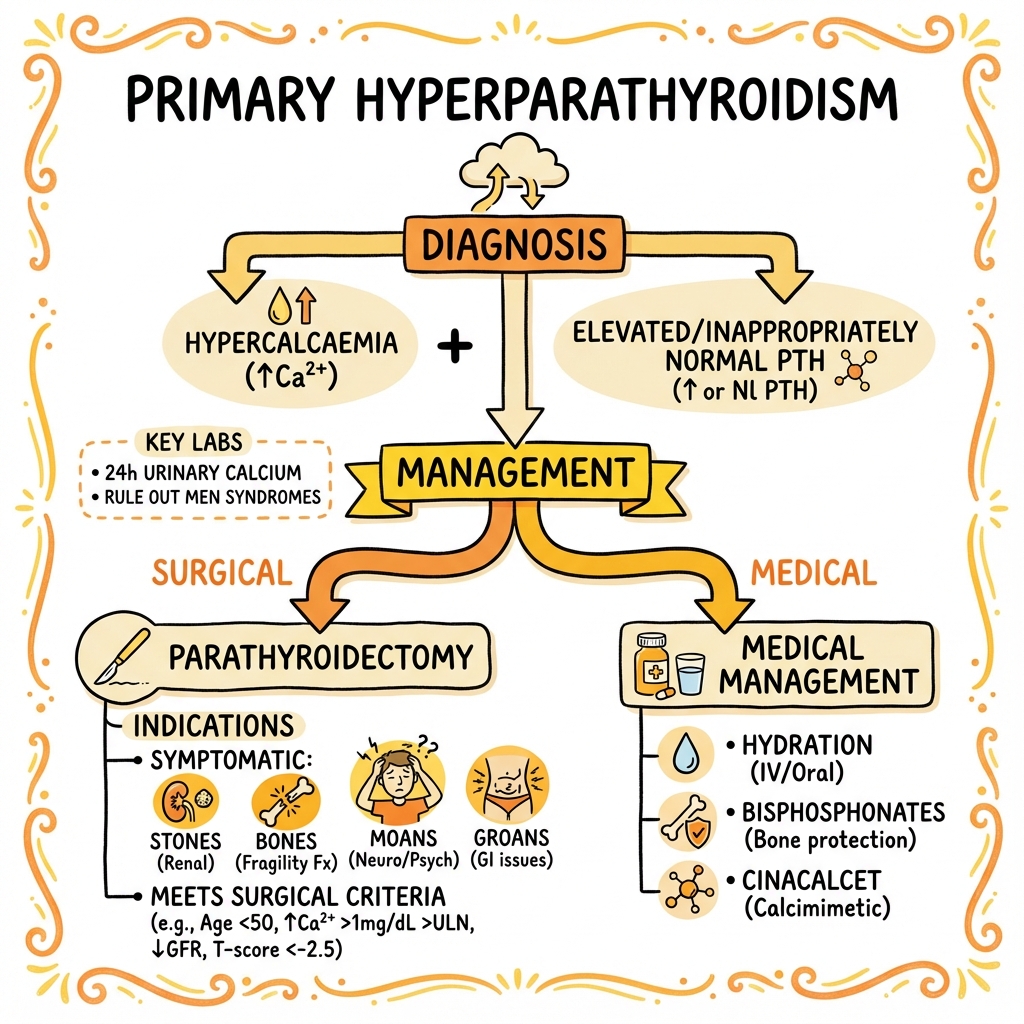
- "PHPT: PTH elevated (> 6.9 pmol/L or > 65 pg/mL) OR inappropriately normal (within reference range despite hypercalcaemia)"
- Key concept: In hypercalcaemia, PTH should be fully suppressed (less than 1.0 pmol/L). A "normal" PTH of 4.5 pmol/L in the presence of calcium 2.85 mmol/L is diagnostic of PHPT.
Phosphate
- Typically low or low-normal in PHPT (PTH-induced phosphaturia)
- Elevated in CKD-associated secondary hyperparathyroidism
Renal Function
- Serum creatinine and eGFR
- Essential for staging CKD in secondary hyperparathyroidism
- PHPT may cause nephrocalcinosis and renal impairment
25-Hydroxyvitamin D [25(OH)D]
- Many PHPT patients are vitamin D deficient (25-40%)
- Vitamin D deficiency exacerbates PTH elevation in PHPT
- Safe to replete vitamin D pre-operatively; reduces risk of post-operative hungry bone syndrome [18]
- Target 25(OH)D > 50 nmol/L (> 20 ng/mL) prior to parathyroidectomy
Alkaline Phosphatase (ALP)
- May be elevated (bone isoform) in PHPT with high bone turnover
- Markedly elevated in severe disease
Exclusion of FHH (Mandatory Before Surgery)
24-Hour Urinary Calcium
- Collect 24-hour urine for calcium and creatinine
- Measure concurrent serum calcium and creatinine
- Calculate CCCR (see above)
Interpretation:
- Urinary calcium less than 100 mg/24h with CCCR less than 0.01 suggests FHH
- Consider genetic testing for CASR, GNA11, AP2S1 mutations if CCCR borderline
Additional Tests for Secondary Hyperparathyroidism
- Serum calcium: Low or normal
- Phosphate: Usually elevated in CKD
- 25(OH)D: Assess for vitamin D deficiency
- eGFR: Stage CKD
- FGF-23: Elevated in CKD (specialist test)
Imaging for Localisation (Pre-operative)
Imaging is performed after biochemical diagnosis to guide surgical approach. It is NOT required for diagnosis.
First-Line Localisation Studies
Technetium-99m Sestamibi Scintigraphy (MIBI Scan)
- Mechanism: Sestamibi is taken up by mitochondria-rich parathyroid adenomas and washes out more slowly than thyroid tissue
- Protocol: Dual-phase imaging (early and delayed images at 2-3 hours)
- Sensitivity: 80-90% for single adenomas; lower for multiglandular disease [19]
- Specificity: 85-95%
- Advantages: Functional imaging; detects ectopic glands (mediastinum)
- Limitations: Reduced sensitivity in small adenomas (less than 500 mg), hyperplasia, concurrent thyroid nodules
Neck Ultrasound
- Sensitivity: 70-80% (operator-dependent)
- Advantages: Non-invasive, low cost, no radiation, can assess thyroid simultaneously
- Limitations: Cannot detect mediastinal glands; inferior parathyroids difficult to visualise; false positives (thyroid nodules, lymph nodes)
Combined Sestamibi + Ultrasound
- Concordant positive results (both identify same adenoma): 95% positive predictive value for successful minimally invasive parathyroidectomy (MIP) [20]
Second-Line Imaging
4-Dimensional Computed Tomography (4D-CT)
- Indication: Failed first-line localisation, persistent/recurrent disease, re-operative surgery
- Protocol: Multi-phase CT with arterial, venous, and delayed phases; parathyroid adenomas show rapid contrast uptake and washout
- Sensitivity: 85-90% in experienced centres
- Advantages: Superior anatomic detail; detects ectopic glands
- Disadvantages: Radiation exposure, cost, requires expertise in interpretation
MRI (Magnetic Resonance Imaging)
- Indication: Alternative to CT in young patients or pregnancy; ectopic adenoma localisation
- Sensitivity: 75-85%
Selective Venous Sampling for PTH
- Indication: Persistent disease after failed parathyroidectomy with negative imaging
- Technique: Catheterisation of neck and mediastinal veins with PTH sampling to regionalise hyperfunctioning gland
- Invasive, specialist technique
Exam Detail: #### Imaging Pitfalls
False Negatives
- Small adenomas (less than 300 mg)
- Multiglandular disease (sensitivity of sestamibi drops to 30-60%)
- Ectopic locations (intrathyroidal, carotid sheath, mediastinum)
False Positives
- Thyroid nodules (appear "hot" on sestamibi or mimic adenomas on US/CT)
- Lymph nodes
- Oesophagus (can mimic posterior mediastinal adenoma)
Key Point: Negative imaging does NOT exclude PHPT. Bilateral neck exploration by an experienced surgeon achieves > 95% cure rate even without pre-operative localisation.
Assessment of Target Organ Involvement
Bone Mineral Density (BMD)
Dual-Energy X-ray Absorptiometry (DEXA) Scan
-
Assess BMD at three sites:
- Lumbar spine (L1-L4)
- Femoral neck
- Distal one-third radius (cortical bone) – preferentially affected in PHPT
-
Interpretation:
- "T-score ≤-2.5: Osteoporosis (surgical indication)"
- "T-score -1.0 to -2.5: Osteopenia"
- "T-score >-1.0: Normal"
-
PHPT preferentially affects cortical bone; distal radius often shows lowest T-score
Renal Imaging
Indications: Assess for nephrolithiasis or nephrocalcinosis
- Renal ultrasound: First-line; detects stones > 5 mm
- Non-contrast CT abdomen/pelvis: Gold standard for stone detection; detects stones as small as 1-2 mm
- Plain radiograph (KUB): Low sensitivity (misses radiolucent stones)
Vertebral Fracture Assessment (VFA)
- Low-dose lateral spine imaging to detect vertebral compression fractures
- Can be performed during DEXA scan
- Identifies asymptomatic vertebral fractures (present in 10-15% of PHPT patients)
7. Classification and Severity Stratification
Classification by Aetiology
| Type | Pathophysiology | Calcium | PTH | Phosphate |
|---|---|---|---|---|
| Primary | Autonomous parathyroid overactivity (adenoma, hyperplasia, carcinoma) | ↑ | ↑ or inappropriately normal | ↓ or low-normal |
| Secondary | Compensatory response to chronic hypocalcaemia (CKD, vitamin D deficiency) | ↓ or normal | ↑ | ↑ (in CKD) or ↓ (vitamin D deficiency) |
| Tertiary | Autonomous hyperfunction after prolonged secondary hyperparathyroidism | ↑ | ↑↑ | Variable |
| Normocalcaemic PHPT | Mild, early PHPT; exclusion of secondary causes | Normal | ↑ | Normal or ↓ |
Primary Hyperparathyroidism: Symptomatic vs Asymptomatic
Symptomatic PHPT
Presence of one or more of:
- Nephrolithiasis
- Osteitis fibrosa cystica, pathological fracture
- Hypercalcaemia-related symptoms (polyuria, bone pain, cognitive impairment)
- Hypercalcaemic crisis
Asymptomatic PHPT
- No classical symptoms
- Incidental biochemical finding
- May have subtle neurocognitive symptoms not initially recognised
8. Management
Management strategies differ by type and severity of hyperparathyroidism.
Primary Hyperparathyroidism
Surgical Management: Parathyroidectomy
Indications for Surgery
Based on Fourth International Workshop on Asymptomatic PHPT (2014) and updated guidelines: [4,21]
Definite Indications:
- Age less than 50 years (progressive disease likely; long-term complications)
- Serum calcium > 0.25 mmol/L (> 1 mg/dL) above upper limit of normal
- Example: If ULN is 2.60 mmol/L, calcium > 2.85 mmol/L indicates surgery
- eGFR less than 60 mL/min/1.73m² (attributable to PHPT)
- Osteoporosis (T-score ≤-2.5 at any site: spine, hip, or distal radius)
- Vertebral fracture on imaging
- 24-hour urinary calcium > 400 mg/day (increased stone risk)
- Nephrolithiasis or nephrocalcinosis (history or imaging evidence)
- Symptomatic disease (bone pain, fractures, hypercalcaemia symptoms)
Relative Indications:
- Patient preference for surgery over long-term monitoring
- Inability to comply with monitoring (geographical, social factors)
- Worsening BMD or biochemistry on surveillance
Pre-operative Optimisation
- Vitamin D repletion: Target 25(OH)D > 50 nmol/L to reduce post-operative hypocalcaemia risk
- Hydration: Maintain adequate fluid intake; avoid dehydration
- Localisation imaging: Sestamibi and/or ultrasound if planning MIP
- Exclude FHH: Mandatory CCCR or 24-hour urinary calcium
Surgical Techniques
| Technique | Indications | Cure Rate | Complications |
|---|---|---|---|
| Minimally Invasive Parathyroidectomy (MIP) | Concordant imaging localising single adenoma | 95-98% | Lower than bilateral exploration; shorter operative time, smaller scar |
| Bilateral Neck Exploration | Multiglandular disease, negative/discordant imaging, MEN syndromes, parathyroid carcinoma | 95-98% | Recurrent laryngeal nerve injury (1-2%), permanent hypoparathyroidism (1-3%) |
Intra-operative Adjuncts
- Intra-operative PTH (IOPTH) monitoring: PTH measured before and 10-15 minutes after adenoma removal. > 50% drop from baseline indicates successful removal of all hyperfunctioning tissue (Miami criterion). [22]
- Frozen section histology: Limited utility; cannot reliably distinguish adenoma from hyperplasia or normal tissue
- Gamma probe: Radio-guided surgery using technetium-99m sestamibi injection pre-operatively
Post-operative Care
-
Monitor serum calcium: Check within 6 hours post-operatively, then daily for 2-3 days
-
Hungry bone syndrome: Severe hypocalcaemia (calcium less than 2.0 mmol/L) develops in 10-15% post-parathyroidectomy, especially if:
- High pre-operative PTH (> 200 pg/mL)
- Elevated alkaline phosphatase
- Large adenoma (> 2 cm)
- Vitamin D deficiency
- "Management: High-dose calcium (calcium carbonate 1-2 g TDS) + active vitamin D (calcitriol 0.5-2 mcg daily). May require IV calcium gluconate."
-
Recurrent laryngeal nerve monitoring: Assess voice post-operatively
-
Haematoma: Rare but potentially life-threatening (airway compromise); presents as neck swelling, stridor, dyspnoea
Long-term Outcomes
- Cure rate: 95-98% in experienced centres [11]
- Recurrence: 1-5% at 10 years (higher in familial syndromes)
- Bone density: Increases by 10-15% in first year, predominantly at cortical sites [23]
- Quality of life: Significant improvement in fatigue, bone pain, cognitive symptoms [16]
- Fracture risk: Reduced by approximately 50% after parathyroidectomy
Medical (Non-Surgical) Management
Indications:
- Patient refuses surgery
- Medical comorbidities preclude anaesthesia
- Monitoring criteria not met (asymptomatic, less than 50 years, normal BMD, mild hypercalcaemia)
Monitoring Protocol for Asymptomatic PHPT (Non-operative)
| Parameter | Frequency |
|---|---|
| Serum calcium | Every 6-12 months |
| Serum creatinine/eGFR | Annually |
| DEXA scan (spine, hip, distal radius) | Every 1-2 years |
| Renal imaging (if prior stones) | As clinically indicated |
Criteria for conversion to surgery during monitoring:
- Development of indications listed above (symptoms, worsening BMD, rising calcium, declining renal function)
Pharmacological Options
| Medication | Mechanism | Indications | Efficacy | Limitations |
|---|---|---|---|---|
| Cinacalcet | Calcimimetic; allosteric modulator of calcium-sensing receptor; reduces PTH secretion | Unfit for surgery, parathyroid carcinoma | Lowers serum calcium by 0.5-1.0 mmol/L; does NOT improve BMD | GI side effects (nausea 30%), expensive, does not address underlying adenoma |
| Bisphosphonates (e.g., alendronate) | Inhibit osteoclastic bone resorption | Osteoporosis in PHPT if surgery contraindicated | Improves BMD by 5-10% at spine and hip | Does not lower calcium or PTH; long-term safety in PHPT uncertain |
| Denosumab | RANKL inhibitor; reduces bone resorption | Alternative to bisphosphonates | Improves BMD; no effect on calcium or PTH | Requires ongoing treatment; rebound effect on cessation |
Conservative Measures
- Hydration: Maintain adequate fluid intake (2-3 L/day) to reduce stone risk
- Avoid thiazide diuretics: Can exacerbate hypercalcaemia
- Avoid high-calcium diet: Restrict calcium intake to 800-1000 mg/day (not severely restrictive)
- Avoid immobilisation: Weight-bearing exercise to preserve bone density
- Monitor vitamin D: Replete if deficient; concerns about exacerbating hypercalcaemia are unfounded [18]
Secondary Hyperparathyroidism
Management focuses on treating the underlying cause and controlling PTH levels.
Vitamin D Deficiency-Related Secondary Hyperparathyroidism
Treatment:
-
Vitamin D repletion:
- Cholecalciferol (vitamin D3) or ergocalciferol (vitamin D2)
- Loading dose: 50,000 IU weekly for 8-12 weeks
- Maintenance: 1000-2000 IU daily
- Target 25(OH)D > 50 nmol/L (> 20 ng/mL), ideally > 75 nmol/L
-
Calcium supplementation (if dietary intake inadequate):
- Calcium carbonate or citrate 500-1000 mg daily
-
Monitor response:
- Recheck 25(OH)D, PTH, calcium at 3 months
- PTH should normalise with adequate vitamin D repletion
CKD-Related Secondary Hyperparathyroidism
KDIGO (Kidney Disease: Improving Global Outcomes) Guidelines [10]
Goals:
- PTH target: 2-9 times upper limit of normal in dialysis patients (individualized)
- Serum phosphate: Within normal range
- Serum calcium: Within normal range
- Avoid hypercalcaemia and hyperphosphataemia (increase vascular calcification risk)
Stepwise Management:
Step 1: Dietary Phosphate Restriction
- Limit phosphate intake to 800-1000 mg/day
- Avoid processed foods, cola drinks, dairy products
Step 2: Phosphate Binders
- Calcium-based binders (calcium carbonate, calcium acetate): First-line; take with meals
- Non-calcium-based binders (sevelamer, lanthanum): Preferred if hypercalcaemia or high calcium-phosphate product
- Titrate to achieve normal serum phosphate
Step 3: Active Vitamin D Analogues
- Calcitriol (1,25-dihydroxyvitamin D): 0.25-0.5 mcg daily
- Alfacalcidol (1-alpha-hydroxyvitamin D): 0.25-1 mcg daily
- Paricalcitol (synthetic analogue): Less hypercalcaemia risk
- Suppress PTH synthesis; monitor calcium and phosphate closely (risk of hypercalcaemia)
Step 4: Calcimimetics
- Cinacalcet: Indicated if PTH remains elevated (> 500 pg/mL) despite above measures
- Dose: 30-180 mg daily
- Monitor serum calcium (risk of hypocalcaemia); adjust dose accordingly
Step 5: Parathyroidectomy
-
Indications:
- Severe, refractory hyperparathyroidism (PTH > 800 pg/mL despite medical therapy)
- Hypercalcaemia refractory to management
- Calciphylaxis
- Intractable bone pain, fractures
- Progressive soft tissue calcification
-
Surgical options:
- Subtotal parathyroidectomy (3.5 glands removed)
- Total parathyroidectomy with auto-transplantation (forearm or sternocleidomastoid)
Tertiary Hyperparathyroidism
Medical Management:
- Often refractory to medical therapy
- Cinacalcet may provide temporary control
Surgical Management:
- Parathyroidectomy is usually required
- Performed after renal transplantation once graft function stable (usually > 6 months post-transplant)
- Subtotal or total parathyroidectomy with auto-transplantation
Management of Hypercalcaemic Crisis
Emergency Management (Calcium > 3.5 mmol/L with symptoms)
Immediate Measures:
-
Aggressive IV hydration:
- 0.9% normal saline 200-300 mL/hour initially (adjust based on cardiac status)
- Goal: Restore intravascular volume, increase renal calcium excretion
- Monitor fluid balance, urine output
-
Loop diuretics (furosemide 20-40 mg IV):
- Only AFTER adequate rehydration
- Increases renal calcium excretion
- Avoid in hypovolaemia (worsens hypercalcaemia)
-
Bisphosphonates:
- Zoledronic acid 4 mg IV over 15 minutes (single dose)
- Pamidronate 60-90 mg IV over 2-4 hours
- Inhibit osteoclastic bone resorption
- Effect: Calcium decreases over 2-4 days; lasts weeks
-
Calcitonin (if very severe):
- 4 IU/kg IM/SC every 12 hours
- Rapid onset (4-6 hours) but tachyphylaxis develops (48-72 hours)
- Modest efficacy (lowers calcium by 0.25-0.5 mmol/L)
-
Dialysis:
- Consider in severe, refractory hypercalcaemia with renal failure
- Low-calcium dialysate
Definitive Treatment:
- Urgent parathyroidectomy once stabilised (within 24-72 hours)
9. Complications
Disease-Related Complications
Primary Hyperparathyroidism
| Complication | Incidence | Mechanism | Management |
|---|---|---|---|
| Osteoporosis and fractures | 15-30% have osteoporosis | Chronic PTH-mediated bone resorption | DEXA monitoring; parathyroidectomy; bisphosphonates if surgery contraindicated |
| Nephrolithiasis | 15-20% | Hypercalciuria, hypercalcaemia | Hydration; treat underlying PHPT; surgical removal of large stones |
| Nephrocalcinosis | 5-10% | Chronic hypercalciuria and hypercalcaemia | Parathyroidectomy; monitor renal function |
| Chronic kidney disease | 10-15% develop CKD stage 3 or higher | Nephrocalcinosis, recurrent stones, chronic hypercalcaemia | Parathyroidectomy halts progression |
| Cardiovascular disease | Increased mortality (SMR 1.5-2.0) | Hypertension, LVH, vascular calcification, arrhythmias | Blood pressure control; parathyroidectomy may improve CV outcomes |
| Hypercalcaemic crisis | less than 1% | Severe dehydration, immobilisation, intercurrent illness | Emergency management (see above); urgent parathyroidectomy |
| Pancreatitis | 1-7% | Hypercalcaemia-induced pancreatic enzyme activation | Treat pancreatitis; definitive treatment with parathyroidectomy |
| Peptic ulcer disease | 10-15% (higher in MEN1) | Calcium-stimulated gastrin secretion | PPI therapy; parathyroidectomy |
| Neuropsychiatric symptoms | 30-60% | Uncertain; possibly direct neuronal effects of hypercalcaemia | Improve with parathyroidectomy |
Secondary Hyperparathyroidism (CKD)
| Complication | Mechanism |
|---|---|
| Renal osteodystrophy | High-turnover bone disease (osteitis fibrosa), low-turnover disease (adynamic bone), osteomalacia |
| Vascular calcification | Elevated calcium-phosphate product, uraemia, inflammation |
| Cardiovascular mortality | Increased risk from vascular calcification, LVH |
| Calciphylaxis | Rare, severe complication; ischaemic skin necrosis due to small-vessel calcification; 50-80% mortality |
| Soft tissue calcification | Periarticular, visceral deposits |
Surgical Complications
| Complication | Incidence | Prevention | Management |
|---|---|---|---|
| Persistent hyperparathyroidism | 2-5% | Thorough exploration; IOPTH monitoring | Re-exploration after localisation with 4D-CT or venous sampling |
| Recurrent hyperparathyroidism | 1-5% at 10 years | Accurate diagnosis; identify multiglandular disease/MEN syndromes | Re-operation; assess for familial syndromes |
| Hypoparathyroidism (permanent) | 1-3% after bilateral exploration; less than 1% after MIP | Preserve at least one gland or parathyroid remnant; meticulous surgical technique | Lifelong calcium and active vitamin D supplementation |
| Hungry bone syndrome | 10-15% (higher if severe PHPT, vitamin D deficiency, elevated ALP) | Pre-operative vitamin D repletion | Calcium carbonate 1-2 g TDS + calcitriol 0.5-2 mcg daily; monitor calcium closely |
| Recurrent laryngeal nerve injury | 1-2% (transient); less than 1% (permanent) | Nerve identification and preservation during dissection; intra-operative nerve monitoring | Transient: observation; Permanent: voice therapy, possible medialisation procedures |
| Neck haematoma | less than 1% | Meticulous haemostasis | Emergency evacuation if airway compromise; return to theatre |
| Wound infection | less than 1% | Sterile technique; prophylactic antibiotics | Antibiotics; wound care |
10. Prognosis
Primary Hyperparathyroidism
Surgical Outcomes
- Cure rate: 95-98% with experienced parathyroid surgeon [11]
- Recurrence: 1-5% at 10 years (sporadic disease); higher in MEN syndromes (10-20%)
- Bone mineral density: Increases by 10-15% in first year post-operatively, predominantly at cortical sites (distal radius, femoral neck). Improvement continues over 5-10 years. [23]
- Fracture risk: Reduced by approximately 50% following parathyroidectomy
- Quality of life: Significant improvement in fatigue, bone pain, cognitive function, and depression scores in 60-80% of patients [16]
- Cardiovascular outcomes: Some studies suggest improved blood pressure control and reduced LVH after parathyroidectomy; cardiovascular mortality benefit remains debated
Non-Surgical (Observational) Outcomes
Natural history studies in asymptomatic PHPT:
- Progression: Approximately 25-30% develop surgical indications over 10-15 years (worsening BMD, rising calcium, stones, symptoms) [24]
- Stable disease: 30-40% remain stable without intervention
- Spontaneous resolution: Rare
- Mortality: Increased cardiovascular mortality (standardised mortality ratio 1.5-2.0) even in mild disease [17]
Secondary Hyperparathyroidism
Prognosis depends on underlying cause:
- Vitamin D deficiency: Excellent prognosis with vitamin D repletion; PTH normalises in 3-6 months
- CKD-related: Progressive disease paralleling renal decline; associated with increased cardiovascular mortality and fracture risk. Effective management of CKD-MBD improves outcomes.
Tertiary Hyperparathyroidism
- Parathyroidectomy usually curative
- Post-operative adynamic bone disease may develop (especially if total parathyroidectomy without auto-transplantation)
11. Prevention and Screening
Primary Hyperparathyroidism
No established screening programme in general population.
Targeted screening recommended in:
- MEN1 or MEN2A gene carriers: Annual calcium and PTH from age 8-10 years (MEN1) or age 16 years (MEN2A)
- First-degree relatives of FIHP: Consider periodic calcium screening
- History of neck irradiation: Periodic calcium monitoring (latency period 20-40 years)
Secondary Hyperparathyroidism
Prevention strategies:
- Vitamin D supplementation: Public health measures in at-risk populations (elderly, housebound, dark skin in northern latitudes)
- CKD monitoring: Regular PTH, calcium, phosphate monitoring in CKD stages 3-5 (per KDIGO guidelines)
- Dietary phosphate control: Early in CKD course
12. Key Guidelines and Evidence
Major Society Guidelines
| Guideline | Organisation | Year | Key Recommendations |
|---|---|---|---|
| Management of Asymptomatic PHPT | Fourth International Workshop on PHPT | 2014 | Surgical indications: age less than 50, calcium > 2.85 mmol/L, T-score ≤-2.5, eGFR less than 60, stones/nephrocalcinosis [4] |
| Hyperparathyroidism (primary): diagnosis, assessment and initial management | NICE (NG132) | 2019 | Offer parathyroidectomy to all suitable patients; refer to specialist endocrine surgery unit [21] |
| KDIGO Clinical Practice Guideline for CKD-MBD | KDIGO | 2017 | PTH targets, phosphate control, use of vitamin D analogues and calcimimetics [10] |
| Surgical Management of PHPT | American Association of Endocrine Surgeons | 2016 | Guidelines on pre-operative localisation, intra-operative PTH monitoring, surgical techniques [20] |
Landmark Evidence
1. Surgery vs Observation in Asymptomatic PHPT
Randomised controlled trials demonstrate superior outcomes with parathyroidectomy:
- Bollerslev et al. (2007): Surgery improved BMD and quality of life vs observation at 2 years [25]
- Ambrogini et al. (2007): Parathyroidectomy improved cardiovascular parameters and neuropsychological function [26]
2. Minimally Invasive vs Bilateral Neck Exploration
- Udelsman et al. (2011): MIP with IOPTH monitoring achieved equivalent cure rates to bilateral exploration with shorter operative time and lower complications in single-adenoma disease [22]
3. Cinacalcet in PHPT
- Peacock et al. (2011): Cinacalcet reduced serum calcium but did NOT improve BMD in asymptomatic PHPT [27]
4. Vitamin D Repletion in PHPT
- Shah et al. (2012): Vitamin D repletion in PHPT patients is safe; does not significantly worsen hypercalcaemia and reduces post-operative hypocalcaemia risk [18]
5. Long-term Observation
- Silverberg et al. (2007): 15-year follow-up of observational cohort showed progression in 37%, with worsening BMD most common [24]
13. Exam-Focused Content
Common Exam Questions (MRCP, FRACP)
Question 1: A 58-year-old woman is found to have a serum calcium of 2.82 mmol/L (normal 2.20-2.60) and PTH of 5.2 pmol/L (normal 1.6-6.9). What is the diagnosis?
Answer: Primary hyperparathyroidism. The key is recognising that PTH is inappropriately normal. In the presence of hypercalcaemia, PTH should be fully suppressed (less than 1.0 pmol/L). A "normal" PTH in this context indicates autonomous PTH secretion, consistent with PHPT.
Question 2: Before referring a patient for parathyroid surgery, what essential investigation must be performed?
Answer: Measurement of 24-hour urinary calcium excretion and calculation of calcium:creatinine clearance ratio (CCCR) to exclude familial hypocalciuric hypercalcaemia (FHH). CCCR less than 0.01 suggests FHH, which is a benign condition that does not respond to surgery.
Question 3: A 45-year-old woman undergoes parathyroidectomy for PHPT. On post-operative day 1, she develops perioral tingling and carpopedal spasm. Serum calcium is 1.85 mmol/L. What is the diagnosis and management?
Answer: Hungry bone syndrome. This is severe post-operative hypocalcaemia resulting from avid uptake of calcium into previously PTH-suppressed bone. Management includes high-dose oral calcium (calcium carbonate 1-2 g TDS) and active vitamin D (calcitriol 0.5-2 mcg daily). Severe symptomatic cases may require IV calcium gluconate.
Question 4: What are the indications for parathyroidectomy in asymptomatic primary hyperparathyroidism?
Answer (per Fourth International Workshop criteria):
- Age less than 50 years
- Serum calcium > 0.25 mmol/L above upper limit of normal
- eGFR less than 60 mL/min/1.73m²
- Osteoporosis (T-score ≤-2.5 at any site)
- Vertebral fracture
- 24-hour urinary calcium > 400 mg/day
- Nephrolithiasis or nephrocalcinosis
Question 5: Distinguish primary, secondary, and tertiary hyperparathyroidism biochemically.
Answer:
| Type | Calcium | PTH | Phosphate | Clinical Context |
|---|---|---|---|---|
| Primary | ↑ | ↑ or inappropriately normal | ↓ | Sporadic or familial; parathyroid adenoma/hyperplasia |
| Secondary | ↓ or normal | ↑ | ↑ (CKD) or ↓ (vitamin D deficiency) | CKD, vitamin D deficiency, malabsorption |
| Tertiary | ↑ | ↑↑ | Variable | History of long-standing CKD/dialysis or post-renal transplant |
Viva Points
Viva Point: Opening Statement: "Hyperparathyroidism is a disorder of excessive parathyroid hormone secretion, classified into primary, secondary, and tertiary forms. Primary hyperparathyroidism results from autonomous parathyroid gland dysfunction, most commonly due to a solitary adenoma, and is the most common cause of hypercalcaemia in the outpatient setting, with a prevalence of approximately 1 in 1000."
Key Facts to Mention:
- Prevalence: 0.3-1% of population; 2-3% in postmenopausal women
- Aetiology: 80-85% solitary adenoma; 10-15% hyperplasia; less than 1% carcinoma
- Modern presentation: 80-85% "asymptomatic" at diagnosis (incidental biochemical finding)
- Classical presentation: "Bones, stones, abdominal groans, psychic moans"
- Diagnostic biochemistry: Elevated or inappropriately normal PTH in presence of hypercalcaemia; hypophosphataemia
- Critical exclusion: FHH (CCCR less than 0.01) before surgery
- Management: Parathyroidectomy is definitive treatment; cure rate 95-98%; specific indications in asymptomatic disease per Fourth International Workshop
- Surgical complications: Recurrent laryngeal nerve injury (1-2%), hungry bone syndrome (10-15%), persistent/recurrent disease (2-5%)
Common Mistakes
❌ Mistakes that fail candidates:
-
Assuming "normal" PTH excludes PHPT: A PTH within the reference range is inappropriately normal in the setting of hypercalcaemia and confirms PHPT.
-
Forgetting to exclude FHH: Failure to measure urinary calcium or calculate CCCR before referral for surgery. Operating on FHH patients is ineffective and exposes them to surgical risk without benefit.
-
Advising strict calcium restriction: Modern guidelines do NOT recommend severe calcium restriction; normal dietary calcium (800-1000 mg/day) is appropriate.
-
Withholding vitamin D in PHPT: Vitamin D deficiency is common in PHPT and should be treated. Concerns about exacerbating hypercalcaemia are unfounded; vitamin D repletion reduces post-operative hungry bone syndrome.
-
Ordering imaging before biochemical confirmation: Imaging (sestamibi, ultrasound) is for localisation, not diagnosis. Diagnosis is biochemical (calcium + PTH).
-
Confusing secondary and tertiary hyperparathyroidism: Secondary has low/normal calcium; tertiary has elevated calcium (key distinguishing feature).
-
Missing surgical indications in "asymptomatic" PHPT: Candidates often miss age less than 50, osteoporosis (T-score ≤-2.5), or renal impairment (eGFR less than 60) as indications for surgery.
-
Inappropriate use of cinacalcet: Cinacalcet lowers calcium but does NOT improve bone density; it is not a substitute for surgery in operable candidates.
Model Viva Answers
Q: A 52-year-old woman presents with fatigue and is found to have calcium 2.88 mmol/L, PTH 6.5 pmol/L, phosphate 0.72 mmol/L. Describe your approach.
A: "This presentation is consistent with primary hyperparathyroidism. The key diagnostic feature is the inappropriately normal PTH in the presence of hypercalcaemia—PTH should be fully suppressed if calcium is elevated.
My initial approach would be to confirm the diagnosis by repeating serum calcium (corrected for albumin or ideally ionised calcium), PTH, and checking renal function and 25-hydroxyvitamin D levels. I would also check serum phosphate, which is typically low or low-normal in PHPT.
Next, I must exclude familial hypocalciuric hypercalcaemia (FHH) by measuring 24-hour urinary calcium and calculating the calcium:creatinine clearance ratio. A ratio less than 0.01 would indicate FHH, which is benign and does not require surgery.
Once PHPT is confirmed, I would assess for surgical indications per the Fourth International Workshop criteria: age less than 50, calcium > 2.85 mmol/L, osteoporosis, renal impairment, nephrolithiasis, or symptomatic disease. I would order a DEXA scan and renal ultrasound or CT to assess for osteoporosis and kidney stones.
Given her age of 52 and calcium of 2.88 mmol/L (> 0.25 above normal), she meets criteria for parathyroidectomy. I would optimise her by repleting vitamin D if deficient, then refer to an endocrine surgeon. Pre-operative localisation with sestamibi and ultrasound would guide the surgical approach—either minimally invasive parathyroidectomy if a single adenoma is localised, or bilateral neck exploration.
Post-operatively, I would monitor serum calcium closely for hungry bone syndrome and ensure adequate calcium and vitamin D supplementation."
Q: How does secondary hyperparathyroidism in CKD develop?
A: "Secondary hyperparathyroidism in chronic kidney disease develops through multiple mechanisms driven by progressive nephron loss.
First, phosphate retention occurs as GFR declines below 60 mL/min, leading to hyperphosphataemia. This directly stimulates PTH secretion and also triggers increased FGF-23 release from bone.
Second, calcitriol deficiency results from two processes: reduced 1-alpha-hydroxylase activity in diseased kidneys, and FGF-23-mediated suppression of this enzyme. Calcitriol normally suppresses PTH gene transcription via the vitamin D receptor on parathyroid cells; its deficiency removes this negative feedback, leading to increased PTH synthesis.
Third, hypocalcaemia develops due to reduced intestinal calcium absorption (from calcitriol deficiency) and precipitation of calcium with phosphate. This directly stimulates the calcium-sensing receptors on parathyroid cells to increase PTH secretion.
Fourth, parathyroid gland hyperplasia develops over time, with progressive parathyroid cell proliferation. The hyperplastic glands show reduced expression of both calcium-sensing receptors and vitamin D receptors, making them less responsive to suppression by calcium and calcitriol. This sets the stage for eventual autonomous function.
Finally, skeletal resistance to PTH develops in uraemia, creating a vicious cycle where higher PTH levels are required to maintain normocalcaemia, further driving parathyroid hyperplasia.
The net result is elevated PTH with low or normal calcium—in contrast to primary hyperparathyroidism, which presents with elevated PTH and elevated calcium. Management focuses on controlling phosphate with dietary restriction and binders, repleting vitamin D with active analogues like calcitriol, and using calcimimetics like cinacalcet if PTH remains refractory. Parathyroidectomy is reserved for severe, refractory cases."
14. Patient and Layperson Explanation
What are parathyroid glands?
You have four tiny parathyroid glands (each about the size of a grain of rice) located behind your thyroid gland in your neck. Despite the similar name, they are completely separate from the thyroid and have a different function. Their job is to regulate calcium levels in your blood by producing parathyroid hormone (PTH).
What is hyperparathyroidism?
Hyperparathyroidism means the parathyroid glands are making too much PTH. There are three types:
-
Primary: One of the glands develops a small, benign growth (adenoma) and starts working overtime, producing too much PTH. This pulls calcium out of your bones and puts it into your blood.
-
Secondary: Your parathyroid glands are working harder to compensate for low calcium levels, usually caused by kidney disease or vitamin D deficiency.
-
Tertiary: After years of secondary hyperparathyroidism (usually from kidney disease), the glands become "stuck" in overdrive and keep producing too much PTH even when calcium levels are normal or high.
What symptoms might I have?
Many people have no obvious symptoms and the condition is found on routine blood tests. Others may experience:
- Fatigue and weakness
- Bone pain or fractures
- Kidney stones
- Constipation, nausea, or stomach pain
- Difficulty concentrating or memory problems
- Depression or mood changes
Do I need an operation?
For primary hyperparathyroidism, surgery (parathyroidectomy) is the only cure and is recommended if:
- You are under 50 years old
- Your calcium level is quite high
- You have weak bones (osteoporosis)
- You have kidney stones or kidney problems
- You have symptoms affecting your quality of life
The operation takes 30-90 minutes and involves removing the overactive gland(s). Most people go home the same day or the next day. The success rate is very high (95-98%).
What if I don't have surgery?
If you don't meet criteria for surgery or prefer not to have it, your doctor will monitor your blood calcium, kidney function, and bone density regularly (every 6-12 months). About 25-30% of people eventually develop a reason to have surgery (worsening bones, rising calcium, kidney stones).
What are the risks of surgery?
Risks are low but include:
- Voice changes (1-2% risk of injury to the nerve controlling your vocal cords)
- Low calcium levels after surgery, requiring calcium and vitamin D tablets
- Bleeding or infection (rare)
What happens after surgery?
Most people feel much better within weeks to months. Bone density improves over the first year, fracture risk decreases, and many people notice improvement in energy, mood, and concentration. You'll need blood tests to monitor calcium levels for a few months after surgery.
15. References
-
Fraser WD. Hyperparathyroidism. Lancet. 2009;374(9684):145-158. doi:10.1016/S0140-6736(09)60507-9
-
Bilezikian JP, Bandeira L, Khan A, Cusano NE. Hyperparathyroidism. Lancet. 2018;391(10116):168-178. doi:10.1016/S0140-6736(17)31430-7
-
Silverberg SJ, Clarke BL, Peacock M, et al. Current issues in the presentation of asymptomatic primary hyperparathyroidism: proceedings of the Fourth International Workshop. J Clin Endocrinol Metab. 2014;99(10):3580-3594. doi:10.1210/jc.2014-1415
-
Bilezikian JP, Brandi ML, Eastell R, et al. Guidelines for the management of asymptomatic primary hyperparathyroidism: summary statement from the Fourth International Workshop. J Clin Endocrinol Metab. 2014;99(10):3561-3569. doi:10.1210/jc.2014-1413
-
Cozzolino M, Ketteler M, Zehnder D. The vitamin D system: a crosstalk between the heart and kidney. Eur J Heart Fail. 2010;12(10):1031-1041. doi:10.1093/eurjhf/hfq125
-
Yeh MW, Ituarte PH, Zhou HC, et al. Incidence and prevalence of primary hyperparathyroidism in a racially mixed population. J Clin Endocrinol Metab. 2013;98(3):1122-1129. doi:10.1210/jc.2012-4022
-
Wermers RA, Khosla S, Atkinson EJ, et al. Incidence of primary hyperparathyroidism in Rochester, Minnesota, 1993-2001: an update on the changing epidemiology of the disease. J Bone Miner Res. 2006;21(1):171-177. doi:10.1359/JBMR.050910
-
Schneider DF, Mazeh H, Lubner SJ, Jaume JC, Chen H. Cancer of the endocrine system. J Natl Compr Canc Netw. 2011;9(7):737-771. doi:10.6004/jnccn.2011.0066
-
McKnight RF, Adida M, Budge K, Stockton S, Goodwin GM, Geddes JR. Lithium toxicity profile: a systematic review and meta-analysis. Lancet. 2012;379(9817):721-728. doi:10.1016/S0140-6736(11)61516-X
-
Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int Suppl. 2017;7(1):1-59. doi:10.1016/j.kisu.2017.04.001
-
Udelsman R, Lin Z, Donovan P. The superiority of minimally invasive parathyroidectomy based on 1650 consecutive patients with primary hyperparathyroidism. Ann Surg. 2011;253(3):585-591. doi:10.1097/SLA.0b013e318208fed9
-
Thakker RV, Newey PJ, Walls GV, et al. Clinical practice guidelines for multiple endocrine neoplasia type 1 (MEN1). J Clin Endocrinol Metab. 2012;97(9):2990-3011. doi:10.1210/jc.2012-1230
-
Brown EM. Clinical lessons from the calcium-sensing receptor. Nat Clin Pract Endocrinol Metab. 2007;3(2):122-133. doi:10.1038/ncpendmet0388
-
Rejnmark L, Vestergaard P, Mosekilde L. Nephrolithiasis and renal calcifications in primary hyperparathyroidism. J Clin Endocrinol Metab. 2011;96(8):2377-2385. doi:10.1210/jc.2011-0569
-
Walker MD, McMahon DJ, Inabnet WB, et al. Neuropsychological features in primary hyperparathyroidism: a prospective study. J Clin Endocrinol Metab. 2009;94(6):1951-1958. doi:10.1210/jc.2008-2574
-
Bollerslev J, Jansson S, Mollerup CL, et al. Medical observation, compared with parathyroidectomy, for asymptomatic primary hyperparathyroidism: a prospective, randomized trial. J Clin Endocrinol Metab. 2007;92(5):1687-1692. doi:10.1210/jc.2006-1836
-
Vestergaard P, Mosekilde L, Brixen K, et al. Cardiovascular events before and after surgery for primary hyperparathyroidism. World J Surg. 2003;27(2):216-222. doi:10.1007/s00268-002-6541-z
-
Shah VN, Shah CS, Bhadada SK, Rao DS. Effect of 25 (OH) D replacements in patients with primary hyperparathyroidism (PHPT) and coexistent vitamin D deficiency on serum 25(OH) D, calcium and PTH levels: a meta-analysis and review of literature. Clin Endocrinol (Oxf). 2014;80(6):797-803. doi:10.1111/cen.12398
-
Hindié E, Ugur O, Fuster D, et al. 2009 EANM parathyroid guidelines. Eur J Nucl Med Mol Imaging. 2009;36(7):1201-1216. doi:10.1007/s00259-009-1131-z
-
Wilhelm SM, Wang TS, Ruan DT, et al. The American Association of Endocrine Surgeons Guidelines for Definitive Management of Primary Hyperparathyroidism. JAMA Surg. 2016;151(10):959-968. doi:10.1001/jamasurg.2016.2310
-
National Institute for Health and Care Excellence. Hyperparathyroidism (primary): diagnosis, assessment and initial management. NICE guideline [NG132]. Published May 2019. https://www.nice.org.uk/guidance/ng132
-
Carneiro DM, Solorzano CC, Nader MC, Ramirez M, Irvin GL 3rd. Comparison of intraoperative iPTH assay (QPTH) criteria in guiding parathyroidectomy: which criterion is the most accurate? Surgery. 2003;134(6):973-979. doi:10.1016/j.surg.2003.06.001
-
Cheng SP, Liu CL, Chen HH, Lee JJ, Liu TP, Yang TL. Prolonged hospital stay after parathyroidectomy for secondary hyperparathyroidism. World J Surg. 2009;33(1):72-79. doi:10.1007/s00268-008-9777-1
-
Silverberg SJ, Shane E, Jacobs TP, Siris ES, Gartenberg F, Seldin D, Clemens TL, Bilezikian JP. Nephrolithiasis and bone involvement in primary hyperparathyroidism. Am J Med. 1990;89(3):327-334. doi:10.1016/0002-9343(90)90345-I
-
Ambrogini E, Cetani F, Cianferotti L, et al. Surgery or surveillance for mild asymptomatic primary hyperparathyroidism: a prospective, randomized clinical trial. J Clin Endocrinol Metab. 2007;92(8):3114-3121. doi:10.1210/jc.2007-0219
-
Rubin MR, Bilezikian JP, McMahon DJ, et al. The natural history of primary hyperparathyroidism with or without parathyroid surgery after 15 years. J Clin Endocrinol Metab. 2008;93(9):3462-3470. doi:10.1210/jc.2007-1215
-
Peacock M, Bilezikian JP, Klassen PS, Guo MD, Turner SA, Shoback D. Cinacalcet hydrochloride maintains long-term normocalcemia in patients with primary hyperparathyroidism. J Clin Endocrinol Metab. 2005;90(1):135-141. doi:10.1210/jc.2004-0842
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate specialists and refer to current local and international guidelines for management decisions.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Calcium Homeostasis
- Parathyroid Hormone Physiology
Differentials
Competing diagnoses and look-alikes to compare.
- Familial Hypocalciuric Hypercalcaemia
- Hypercalcaemia of Malignancy
- Vitamin D Toxicity
Consequences
Complications and downstream problems to keep in mind.
- Osteoporosis
- Nephrolithiasis
- Hypercalcaemic Crisis
- Chronic Kidney Disease