Lupus Nephritis
Lupus nephritis (LN) represents one of the most serious manifestations of systemic lupus erythematosus (SLE), characteri... MRCP exam preparation.
What matters first
Lupus nephritis (LN) represents one of the most serious manifestations of systemic lupus erythematosus (SLE), characteri... MRCP exam preparation.
Rapidly progressive renal failure
5 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Rapidly progressive renal failure
- Nephrotic syndrome with active sediment
- Acute kidney injury in SLE
- Declining complement levels with rising anti-dsDNA
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- IgA Nephropathy
- Membranoproliferative Glomerulonephritis
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Lupus Nephritis
1. Overview
Lupus nephritis (LN) represents one of the most serious manifestations of systemic lupus erythematosus (SLE), characterized by immune complex-mediated glomerular injury that can lead to progressive renal impairment and end-stage renal disease (ESRD). [1] The condition results from deposition of autoantibodies and immune complexes in the glomeruli, triggering an inflammatory cascade that damages renal parenchyma and impairs kidney function. [2]
The clinical significance of lupus nephritis cannot be overstated: it affects 40-50% of patients with SLE during their disease course, often presenting within the first year of SLE diagnosis, and represents a major determinant of morbidity and mortality in this patient population. [3,4] Early recognition and aggressive treatment are essential, as untreated proliferative lupus nephritis can progress to ESRD in 10-20% of patients despite modern immunosuppressive therapies. [5]
Management has evolved significantly over the past two decades, with landmark trials establishing mycophenolate mofetil (MMF) as non-inferior to cyclophosphamide for induction therapy, and newer agents including voclosporin and belimumab providing additional therapeutic options for refractory disease. [6,7,8] Renal biopsy remains the gold standard for diagnosis and classification, with the International Society of Nephrology/Renal Pathology Society (ISN/RPS) 2003 classification system guiding treatment decisions. [9]
Key Clinical Messages
- Mandatory biopsy: All SLE patients with proteinuria > 500 mg/day, active urinary sediment, or unexplained decline in renal function require renal biopsy for ISN/RPS classification
- Treatment stratification: ISN/RPS Class I/II requires supportive care only; Class III/IV (proliferative) requires aggressive immunosuppression; Class V (membranous) is treated as Class III/IV if nephrotic-range proteinuria present
- Universal hydroxychloroquine: All lupus nephritis patients should receive hydroxychloroquine unless contraindicated, as it reduces flare rates and improves long-term outcomes
- Monitoring intensity: Serial monitoring of anti-dsDNA antibodies and complement (C3/C4) levels correlates with disease activity and guides treatment adjustments
2. Epidemiology
Incidence and Prevalence
Lupus nephritis develops in 40-50% of patients with SLE during the course of their disease, with the majority of cases presenting within the first year of SLE diagnosis. [3,4] The cumulative incidence approaches 60% when including subclinical disease detected on protocol biopsies. [10] Approximately 10-30% of patients with lupus nephritis will progress to ESRD despite treatment, with 5-year renal survival rates of 80-90% in contemporary cohorts receiving optimal immunosuppressive therapy. [5,11]
| Epidemiological Parameter | Value | Source |
|---|---|---|
| Prevalence in SLE patients | 40-50% | [3,4] |
| Cumulative incidence (including subclinical) | Up to 60% | [10] |
| Presentation within 1st year of SLE | 50-70% | [4] |
| 10-year ESRD risk | 10-20% | [5] |
| 5-year renal survival | 80-90% | [11] |
| Recurrence post-transplant | 2-5% | [12] |
Demographics and Risk Factors
The demographic profile of lupus nephritis mirrors that of SLE, with striking disparities based on ethnicity, sex, and age. Women are affected 9 times more frequently than men, reflecting the female predominance of SLE. [1] Peak incidence occurs between ages 20-40 years, coinciding with peak SLE incidence. [13]
High-Risk Populations:
Ethnicity represents the most significant risk factor for both development and severity of lupus nephritis:
- African American patients: 2-3 times higher incidence of lupus nephritis compared to Caucasians; present with more severe disease (predominantly Class III/IV); higher progression to ESRD (3-4 fold increased risk) [14,15]
- Hispanic/Latino patients: Intermediate risk between African American and Caucasian populations; increased prevalence of Class III/IV disease [14]
- Asian patients: Particularly East Asian (Chinese, Korean, Japanese) populations show 2-fold increased risk of lupus nephritis; higher rates of Class III/IV disease compared to Caucasians [16]
- Caucasian patients: Lowest incidence and severity; more likely to present with Class V (membranous) lupus nephritis [14]
Additional Risk Factors:
- Male sex (when SLE present): Higher risk of renal involvement and more aggressive disease course [17]
- Younger age at SLE diagnosis (less than 30 years): Associated with increased risk of developing lupus nephritis [13]
- Anti-dsDNA antibody positivity: Strong association with lupus nephritis development and activity [18]
- Hypocomplementemia: Low C3/C4 levels correlate with active nephritis [18]
- Family history of SLE or lupus nephritis: Genetic predisposition [1]
- Socioeconomic factors: Lower socioeconomic status associated with worse outcomes (likely related to access to care and medication adherence) [15]
Temporal Trends
Modern immunosuppressive regimens have improved outcomes substantially compared to historical cohorts. The 10-year renal survival has improved from approximately 45% in the 1970s to 80-90% in contemporary series. [11] However, progression to ESRD remains a significant problem, particularly in high-risk ethnic groups and those with delayed diagnosis or inadequate treatment response. [5,15]
3. Aetiology and Pathophysiology
Fundamental Immunopathology
Lupus nephritis represents a Type III hypersensitivity reaction characterized by immune complex deposition in glomerular structures, triggering complement activation and inflammatory cell recruitment that results in glomerular injury. [2] The fundamental pathogenic mechanism involves loss of immune tolerance to self-antigens, production of autoantibodies (particularly anti-double-stranded DNA), formation of circulating immune complexes, glomerular deposition, and local inflammatory injury. [1,2]
Autoantibody Production and Immune Complex Formation
The pathogenesis begins with aberrant B-cell activation and production of autoantibodies, most notably anti-dsDNA antibodies, which show the strongest correlation with lupus nephritis activity. [18] These antibodies form immune complexes either in circulation (circulating immune complexes) or in situ within the kidney (planted antigen hypothesis). [2]
Key Pathogenic Autoantibodies:
- Anti-dsDNA antibodies: Directly correlate with disease activity; can cross-react with glomerular antigens including α-actinin and annexin II [18]
- Anti-C1q antibodies: Associated with proliferative lupus nephritis and complement consumption [19]
- Anti-nucleosome antibodies: Precede clinical lupus nephritis and may predict renal flares [1]
- Anti-Smith antibodies: Specific for SLE but do not correlate strongly with renal disease activity [1]
Molecular Mechanisms of Glomerular Injury:
The pathophysiology of lupus nephritis involves multiple integrated mechanisms:
1. Immune Complex Deposition:
- Circulating immune complexes (preformed antigen-antibody complexes) deposit in glomeruli based on charge, size, and haemodynamic factors
- In situ immune complex formation occurs when antibodies bind to "planted" glomerular antigens or to intrinsic glomerular basement membrane components
- Subendothelial deposition (seen in Class III/IV) causes proliferative changes
- Subepithelial deposition (seen in Class V) causes membranous nephropathy pattern
2. Complement Activation:
- Immune complexes activate classical complement pathway (C1q → C4 → C3)
- C3a and C5a act as anaphylatoxins, recruiting inflammatory cells
- Membrane attack complex (C5b-9) causes direct podocyte and endothelial injury
- Consumption of C3 and C4 results in hypocomplementemia, a biomarker of active disease [18,19]
3. Inflammatory Cell Infiltration:
- Neutrophils release reactive oxygen species and proteases
- Macrophages produce pro-inflammatory cytokines (IL-1, IL-6, TNF-α)
- T cells (particularly Th1 and Th17 subsets) contribute to inflammation
- Dendritic cells present autoantigens, perpetuating immune activation [2]
4. Cytokine and Chemokine Networks:
- Type I interferons (IFN-α/β) drive B-cell activation and autoantibody production
- B-lymphocyte stimulator (BLyS/BAFF) promotes B-cell survival (target of belimumab)
- IL-6, IL-17, and TNF-α amplify inflammation
- Chemokines (MCP-1, RANTES) recruit inflammatory cells to glomeruli [1,2]
5. Podocyte Injury:
- Direct antibody-mediated injury to podocyte foot processes
- Complement-mediated damage via C5b-9 complex
- Results in proteinuria due to disruption of glomerular filtration barrier [2]
6. Endothelial Dysfunction:
- Immune complex deposition in subendothelial space damages endothelium
- Contributes to glomerular capillary thrombosis
- Impairs renal perfusion and promotes ischaemic injury [2]
7. Tubulointerstitial Inflammation:
- Secondary to glomerular injury and proteinuria
- Tubulointerstitial inflammation correlates strongly with long-term renal outcomes
- Tubular atrophy and interstitial fibrosis are poor prognostic indicators [9]
8. Genetic Susceptibility:
- HLA associations (HLA-DR2, HLA-DR3) increase lupus nephritis risk
- Complement deficiencies (C1q, C4, C2) strongly predispose to SLE and lupus nephritis
- FcγR polymorphisms affect immune complex clearance
- Polymorphisms in apoptosis-related genes contribute to impaired clearance of apoptotic material [1]
ISN/RPS Classification and Pathological Features
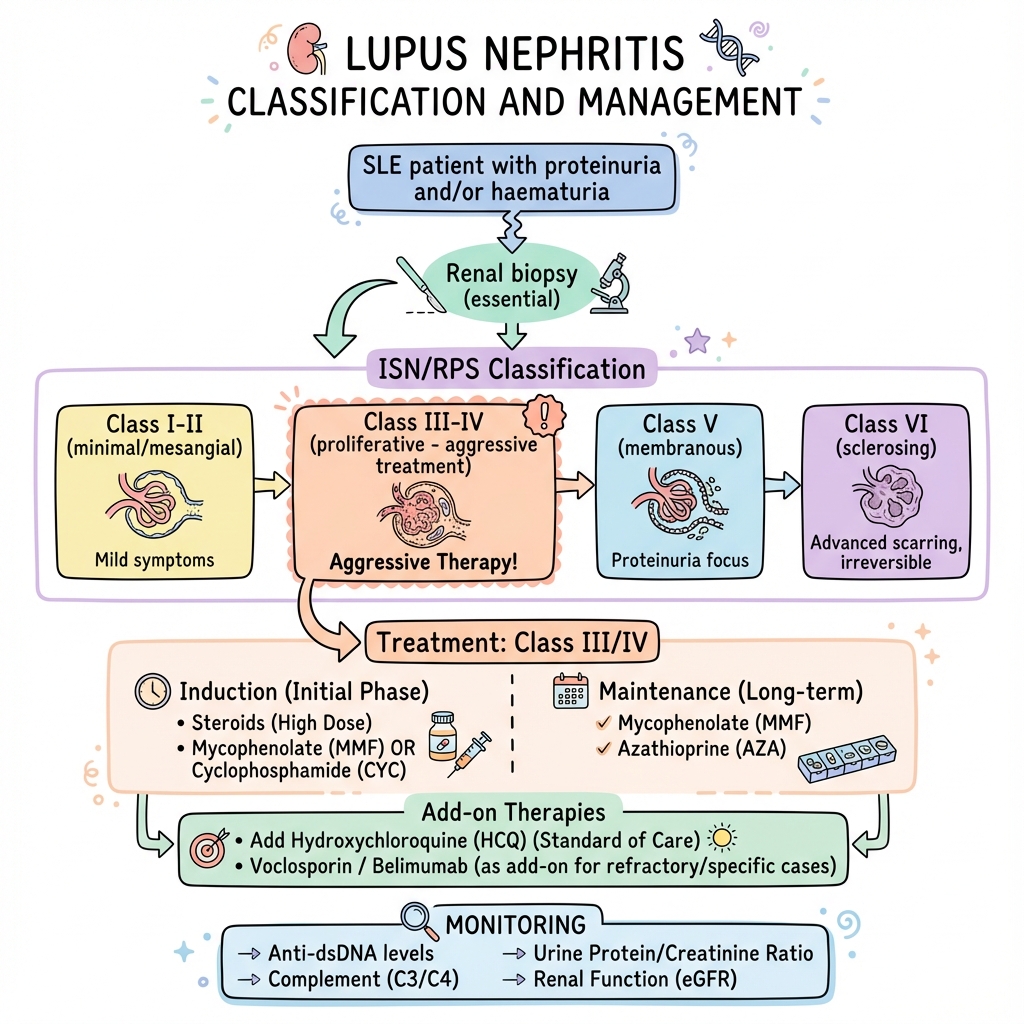
The ISN/RPS 2003 classification system categorizes lupus nephritis into six classes based on histopathological findings, with critical treatment implications. [9] This classification superseded earlier WHO classifications and provides both diagnostic and prognostic information.
Class I - Minimal Mesangial Lupus Nephritis:
- Normal glomeruli on light microscopy
- Mesangial immune deposits on immunofluorescence or electron microscopy
- No clinical renal manifestations typically
- Treatment: None required; manage extrarenal SLE manifestations
Class II - Mesangial Proliferative Lupus Nephritis:
- Mesangial hypercellularity or matrix expansion on light microscopy
- Mesangial immune deposits on immunofluorescence
- May present with isolated haematuria or mild proteinuria
- Treatment: Supportive care; low-dose corticosteroids if symptomatic
Class III - Focal Lupus Nephritis:
- Involvement of less than 50% of glomeruli with endocapillary or extracapillary proliferation
- Subdivided into Class III (A) - active lesions; Class III (A/C) - active and chronic lesions; Class III (C) - chronic lesions only
- Subendothelial immune deposits
- Presents with haematuria, proteinuria (often nephrotic range), and declining renal function
- Treatment: Aggressive immunosuppression (induction + maintenance regimen)
Class IV - Diffuse Lupus Nephritis:
- Involvement of ≥50% of glomeruli with endocapillary or extracapillary proliferation
- Subdivided into Class IV-S (segmental lesions) and Class IV-G (global lesions)
- Further subdivided into IV (A), IV (A/C), IV (C) based on activity/chronicity
- Most common severe form of lupus nephritis
- Presents with nephritic syndrome (haematuria, hypertension, AKI) often with nephrotic-range proteinuria
- Treatment: Aggressive immunosuppression (induction + maintenance regimen)
Class V - Membranous Lupus Nephritis:
- Global or segmental subepithelial immune deposits
- Can occur alone (pure Class V) or in combination with Class III/IV
- Presents with nephrotic syndrome
- Treatment: If nephrotic-range proteinuria, treat as Class III/IV; if subnephrotic, may observe with supportive care
Class VI - Advanced Sclerosing Lupus Nephritis:
- ≥90% of glomeruli globally sclerosed
- Represents end-stage kidney disease
- Treatment: Supportive care; prepare for renal replacement therapy; consider transplant evaluation
Immunofluorescence and Electron Microscopy Findings:
Immunofluorescence microscopy demonstrates the characteristic "full house" pattern in lupus nephritis, consisting of:
- IgG (dominant immunoglobulin)
- IgA
- IgM
- C3
- C1q (particularly specific for lupus nephritis; seen in > 90% of proliferative cases) [9]
The presence of all major immunoreactants is highly suggestive of lupus nephritis, although not absolutely pathognomonic. The absence of C1q deposition makes lupus nephritis diagnosis questionable.
Electron microscopy reveals:
- Subendothelial deposits (Class III/IV): Located between endothelium and glomerular basement membrane; correlate with active proliferative disease
- Subepithelial deposits (Class V): Located between podocytes and GBM; create "spike and dome" appearance
- Mesangial deposits (all classes): Expansion of mesangial matrix
- Tubuloreticular inclusions: Electron-dense structures within endothelial cells; associated with type I interferon exposure (seen in 50% of lupus nephritis but also in HIV nephropathy) [9]
Activity and Chronicity Indices:
The NIH activity and chronicity indices score histological features to predict treatment response and prognosis:
Activity Index (0-24):
- Endocapillary proliferation
- Neutrophil/monocyte infiltration
- Fibrinoid necrosis (×2)
- Cellular crescents (×2)
- Hyaline deposits
- Interstitial inflammation
Chronicity Index (0-12):
- Glomerular sclerosis
- Fibrous crescents
- Tubular atrophy
- Interstitial fibrosis
High activity index (> 12) suggests potential treatment responsiveness; high chronicity index (> 4) predicts poor renal outcomes and reduced treatment responsiveness. [9]
4. Clinical Presentation
Patterns of Renal Involvement
Lupus nephritis presents with diverse clinical manifestations ranging from asymptomatic urinary abnormalities to rapidly progressive glomerulonephritis. The clinical presentation correlates imperfectly with histological class, necessitating renal biopsy for definitive classification. [1,3]
Symptoms
Many patients with lupus nephritis are asymptomatic, with renal involvement detected only on routine screening urinalysis. [3] When symptoms occur, they typically reflect the syndrome caused by glomerular injury:
Nephrotic Syndrome Features:
- Peripheral oedema (ankle, periorbital, presacral)
- Frothy urine (due to heavy proteinuria)
- Weight gain (fluid retention)
- Dyspnoea (pulmonary oedema in severe cases)
- Fatigue
Nephritic Syndrome Features:
- Cola- or tea-coloured urine (macroscopic haematuria)
- Reduced urine output (oliguria)
- Headaches (hypertension)
- Nausea and vomiting (uraemia)
Systemic SLE Features:
- Constitutional symptoms: Fatigue, fever, weight loss
- Arthralgia and myalgia
- Rash (malar rash, discoid lesions, photosensitivity)
- Serositis (pleuritic chest pain, pericarditis)
- Neuropsychiatric manifestations
Signs
General Examination:
- Hypertension (present in 60-80% of Class III/IV lupus nephritis) [3]
- Peripheral oedema (pitting ankle/leg oedema)
- Periorbital oedema
- Ascites (severe nephrotic syndrome)
- Pleural effusions
- Pulmonary oedema (severe fluid overload)
Extrarenal SLE Manifestations:
- Skin: Malar rash, discoid rash, photosensitivity, oral ulcers, alopecia
- Joints: Non-erosive arthritis (hands, wrists, knees)
- Serositis: Pleural or pericardial rub
- Neurological: Altered mental status, seizures, psychosis
- Haematological: Pallor (anaemia), petechiae (thrombocytopenia)
Clinical Syndromes
| Clinical Syndrome | Features | Typical Histology | Frequency |
|---|---|---|---|
| Isolated urinary abnormalities | Microscopic haematuria, subnephrotic proteinuria, normal renal function | Class II or early Class III | 30-40% |
| Nephrotic syndrome | Proteinuria > 3.5 g/day, hypoalbuminemia, oedema, hyperlipidaemia | Class V or Class III/IV with membranous features | 40-50% |
| Nephritic syndrome | Haematuria, hypertension, AKI, subnephrotic proteinuria | Class III/IV | 20-30% |
| Mixed nephrotic-nephritic | Features of both syndromes | Class III/IV or combined Class III/IV + V | 30-40% |
| Rapidly progressive GN | Rapid decline in renal function (days-weeks), oliguria, severe hypertension | Class III/IV with crescents | 5-10% |
| Chronic kidney disease | Slowly progressive renal impairment, bland sediment | Class VI or chronic Class III/IV | 10-20% |
Red Flag Features
Immediate Nephrology Referral Required:
- Rapidly rising serum creatinine (> 25% increase over baseline)
- Nephrotic-range proteinuria (> 3.5 g/day or UPCR > 350 mg/mmol)
- Active urinary sediment (RBC casts, dysmorphic RBCs)
- Acute kidney injury (creatinine > 1.5× baseline)
- Combination of declining complement + rising anti-dsDNA + worsening proteinuria
- Resistant hypertension
- Oliguria or anuria
Pregnancy Considerations
Lupus nephritis significantly complicates pregnancy, with increased risks of:
- Maternal: Lupus flare (30-50%), preeclampsia (20-30%), worsening renal function, thromboembolic events
- Fetal: Preterm delivery (50%), intrauterine growth restriction, fetal loss (10-20%), neonatal lupus [20]
Active lupus nephritis is a contraindication to pregnancy; disease should be quiescent for at least 6 months before conception. Pregnancy management requires multidisciplinary care (nephrology, rheumatology, maternal-fetal medicine). [20]
5. Differential Diagnosis
The presence of glomerulonephritis in a patient with known or suspected SLE strongly suggests lupus nephritis, but alternative diagnoses must be considered, particularly when serological features are atypical or when additional clinical features suggest alternative pathology. [1,3]
Primary Glomerulonephritides
IgA Nephropathy:
- Most common primary GN worldwide
- Presents with episodic macroscopic haematuria (often following upper respiratory infection)
- Persistent microscopic haematuria between episodes
- Nephrotic syndrome uncommon (10-15%)
- Immunofluorescence: Dominant IgA deposition (not "full house")
- Normal complement levels (unlike lupus nephritis)
- No extrarenal SLE features
- Renal biopsy distinguishes definitively [9]
Membranoproliferative Glomerulonephritis (MPGN):
- Presents with nephrotic-nephritic features (similar to lupus nephritis Class III/IV)
- Low C3 with normal C4 in complement-mediated MPGN (versus low C3 and C4 in lupus nephritis)
- Absence of anti-dsDNA antibodies
- May be secondary to chronic infections (hepatitis C, hepatitis B), monoclonal gammopathy, or complement dysregulation
- Histology: Mesangial proliferation, GBM duplication ("tram-tracking"), subendothelial deposits
- Immunofluorescence: C3 dominant (not "full house")
Post-Infectious Glomerulonephritis:
- Follows streptococcal or other bacterial infection by 1-3 weeks
- Acute onset nephritic syndrome
- Low C3 with normal C4 (versus both low in lupus nephritis)
- Absent anti-dsDNA antibodies
- Self-limited course; complement normalizes within 8-12 weeks
- Histology: Diffuse proliferative GN with subepithelial "humps"
Minimal Change Disease:
- Presents with pure nephrotic syndrome (no haematuria, normal renal function initially)
- Normal complement levels
- Negative autoantibodies
- Histology: Normal glomeruli on light microscopy; podocyte foot process effacement on electron microscopy
- No immune complex deposition
- Excellent response to corticosteroids alone
Focal Segmental Glomerulosclerosis (FSGS):
- Nephrotic syndrome with variable microscopic haematuria
- Often accompanied by hypertension and renal impairment
- Normal complement levels
- Negative autoantibodies
- Histology: Segmental sclerosis and hyalinosis
- Immunofluorescence: IgM and C3 in sclerotic segments (not "full house")
Systemic Vasculitides
ANCA-Associated Vasculitis (Granulomatosis with Polyangiitis, Microscopic Polyangiitis):
- Rapidly progressive GN with nephritic syndrome
- Often accompanied by pulmonary haemorrhage (pulmonary-renal syndrome)
- Normal complement levels (key distinguishing feature from lupus nephritis)
- Positive PR3-ANCA or MPO-ANCA (ANCA-positive in 90%)
- Absent anti-dsDNA antibodies
- Histology: Pauci-immune crescentic GN (minimal immune deposits on immunofluorescence)
- May have upper respiratory involvement (epistaxis, sinusitis, saddle nose deformity in GPA)
Anti-GBM Disease (Goodpasture Syndrome):
- Rapidly progressive GN, often with pulmonary haemorrhage
- Normal complement levels
- Positive anti-GBM antibodies
- Linear IgG deposition along GBM on immunofluorescence (pathognomonic)
- Requires urgent plasmapheresis
Henoch-Schönlein Purpura (IgA Vasculitis):
- Usually in children or young adults
- Palpable purpura on lower extremities and buttocks
- Abdominal pain, arthralgia
- GN with IgA-dominant immunofluorescence
- Normal complement levels
Other Autoimmune Conditions
Mixed Connective Tissue Disease:
- Features of SLE, systemic sclerosis, polymyositis
- High titre anti-U1 RNP antibodies
- Can cause membranous nephropathy or proliferative GN
- Distinguished by antibody profile and extrarenal features
Cryoglobulinemic Vasculitis:
- Often associated with hepatitis C infection
- Palpable purpura, arthralgias, peripheral neuropathy
- Low C4 with normal or low C3 (reversed pattern from lupus nephritis)
- Positive cryoglobulins
- Histology: MPGN pattern with intracapillary thrombi
Thrombotic Microangiopathy
Thrombotic Thrombocytopenic Purpura (TTP):
- Can occur in SLE patients
- Microangiopathic haemolytic anaemia, thrombocytopenia, AKI, neurological symptoms, fever
- Schistocytes on blood film
- Severely reduced ADAMTS13 activity (less than 10%)
- Requires urgent plasmapheresis
Antiphospholipid Syndrome-Associated Nephropathy:
- Thrombotic microangiopathy affecting kidneys
- Positive antiphospholipid antibodies (anticardiolipin, anti-β2-glycoprotein I, lupus anticoagulant)
- Histology: Glomerular capillary thrombosis, vascular occlusion
- Treated with anticoagulation rather than immunosuppression
| Differential Diagnosis | Key Distinguishing Features | Complement | Specific Antibodies |
|---|---|---|---|
| IgA Nephropathy | Episodic macroscopic haematuria, IgA-dominant immunofluorescence | Normal | None |
| MPGN | Low C3 with normal C4, no extrarenal SLE features | C3↓ C4 normal | Variable |
| ANCA Vasculitis | Pauci-immune GN, pulmonary-renal syndrome | Normal | PR3-ANCA or MPO-ANCA |
| Anti-GBM Disease | Linear IgG on immunofluorescence, pulmonary haemorrhage | Normal | Anti-GBM |
| Post-infectious GN | Recent infection, self-limited, subepithelial humps | C3↓ C4 normal | ASO, anti-DNase B |
| Lupus Nephritis | Full-house immunofluorescence, extrarenal SLE features | C3↓ C4↓ | Anti-dsDNA, anti-C1q |
6. Investigations
A systematic approach to investigation is essential for diagnosing lupus nephritis, classifying disease severity, guiding treatment decisions, and monitoring response to therapy. [1,3]
Initial Screening Investigations
All patients with confirmed or suspected SLE should undergo regular screening for renal involvement:
Urinalysis (Dipstick and Microscopy):
- Frequency: Every 3 months in stable SLE; monthly if high risk or recent nephritis
- Findings suggesting lupus nephritis:
- Proteinuria (trace to 4+)
- Haematuria (microscopic or macroscopic)
- RBC casts (highly specific for glomerulonephritis)
- WBC casts (inflammatory GN)
- Granular casts (tubular injury)
- Dysmorphic RBCs (glomerular origin) [3]
Urine Protein Quantification:
- Spot urine protein-to-creatinine ratio (UPCR): Preferred for routine monitoring
- "Normal: less than 15 mg/mmol (less than 150 mg/g)"
- "Nephrotic range: > 350 mg/mmol (> 3500 mg/g or > 3.5 g/day)"
- 24-hour urine protein collection: Gold standard but cumbersome; rarely required if accurate UPCR available
- Urine albumin-to-creatinine ratio (ACR): May underestimate total proteinuria if significant tubular proteinuria present [3]
Renal Function Tests:
- Serum creatinine: Baseline and trend over time
- Estimated GFR (eGFR): Using CKD-EPI equation (preferred) or MDRD
- Blood urea nitrogen (BUN)
- Serum electrolytes (potassium, sodium, bicarbonate)
- Trends more important than single values; rising creatinine or falling eGFR suggests active nephritis
Complete Blood Count:
- Anaemia: Normochromic normocytic (chronic disease) or microangiopathic haemolytic anaemia (TTP/HUS)
- Leukopenia: Active SLE
- Thrombocytopenia: Active SLE or TTP/antiphospholipid syndrome
- Eosinophilia: Drug-induced interstitial nephritis (if on medications)
Metabolic Panel:
- Hypoalbuminemia: Nephrotic syndrome (less than 30 g/L)
- Hyperlipidaemia: Nephrotic syndrome
- Hypocalcemia, hyperphosphataemia: Renal impairment
Serological Investigations
Autoantibody Profile:
| Antibody | Sensitivity for Lupus Nephritis | Specificity | Clinical Significance |
|---|---|---|---|
| Anti-dsDNA | 70-90% | High | Correlates with disease activity; rising titres predict flare [18] |
| Anti-nuclear antibody (ANA) | > 95% | Low | Screening test; if negative, SLE unlikely |
| Anti-Smith (anti-Sm) | 20-30% | Very high | Specific for SLE but does not correlate with renal activity |
| Anti-C1q | 40-60% | Moderate | Associated with proliferative LN and disease activity [19] |
| Anti-nucleosome | 60-80% | Moderate | May predict renal flares |
| Anti-ribosomal P | 10-20% | High | Associated with neuropsychiatric SLE |
| Anticardiolipin/anti-β2GP1 | Variable | Moderate | Antiphospholipid syndrome; thrombosis risk |
| Lupus anticoagulant | Variable | Moderate | Antiphospholipid syndrome |
Complement Levels:
Low complement levels (C3 and C4) are characteristic of active lupus nephritis due to complement consumption by immune complexes. [18]
- C3: Normal 90-180 mg/dL; low in 70-90% of active proliferative lupus nephritis
- C4: Normal 10-40 mg/dL; low in 80-95% of active proliferative lupus nephritis
- Both C3 and C4 low: Strongly suggests lupus nephritis (versus isolated C3 deficiency in MPGN or post-infectious GN)
- Trend over time correlates with disease activity; rising complement suggests treatment response
- Genetic complement deficiencies (C1q, C4, C2) predispose to SLE
Renal Biopsy: Gold Standard Investigation
Renal biopsy is mandatory for definitive diagnosis, ISN/RPS classification, assessment of activity versus chronicity, and treatment planning. [9]
Indications for Renal Biopsy in SLE Patients:
Absolute indications:
- Proteinuria > 500 mg/day (UPCR > 50 mg/mmol)
- Proteinuria of any degree with active urinary sediment (RBC casts, dysmorphic RBCs)
- Unexplained rise in serum creatinine (> 25% increase from baseline or > 0.3 mg/dL acute rise)
- Combination of proteinuria + haematuria (even if both mild)
Relative indications:
- Persistent proteinuria 150-500 mg/day to exclude ISN/RPS Class I/II versus early Class III
- Suspected lupus nephritis flare despite adequate immunosuppression (to guide escalation)
- Atypical features raising concern for alternative diagnosis
Contraindications to Renal Biopsy:
- Uncontrolled hypertension (typically > 160/100 mmHg)
- Bleeding diathesis (uncorrected coagulopathy, thrombocytopenia less than 50,000, recent anticoagulation)
- Solitary functioning kidney (relative contraindication)
- Renal abscess or active pyelonephritis
- Hydronephrosis or anatomical abnormality
- Uncooperative patient unable to lie flat and still
Biopsy Techniques:
- Percutaneous native kidney biopsy under ultrasound or CT guidance (preferred)
- Transjugular renal biopsy (if contraindications to percutaneous approach)
- Minimum 10-15 glomeruli required for adequate assessment; ideally > 20 glomeruli
Histopathological Assessment:
- Light microscopy: ISN/RPS class, activity index, chronicity index
- Immunofluorescence: "Full house" pattern (IgG, IgA, IgM, C3, C1q)
- Electron microscopy: Location of immune deposits (subendothelial, subepithelial, mesangial), tubuloreticular inclusions
Renal Biopsy Interpretation Pearls:
The renal biopsy report should provide:
- ISN/RPS Class: Dictates initial treatment strategy
- Activity Index (0-24): High activity (> 12) suggests aggressive disease requiring intensive immunosuppression
- Chronicity Index (0-12): High chronicity (> 4) predicts poor treatment response and worse long-term renal outcomes
- Percentage of Glomeruli with:
- Global sclerosis (permanent damage)
- Cellular or fibrous crescents (acute severe injury)
- Fibrinoid necrosis (active inflammation)
- Tubulointerstitial Features:
- Tubular atrophy percentage
- Interstitial fibrosis percentage
- Interstitial inflammation severity
- Tubular atrophy and interstitial fibrosis strongly predict progression to ESRD [9]
- Vascular Changes:
- Arteriosclerosis (hypertensive damage)
- Thrombotic microangiopathy (antiphospholipid syndrome, TTP)
Repeat Biopsy Indications:
- Incomplete response to treatment after 12 months (to reassess histological class and activity/chronicity)
- Suspected flare with rising proteinuria and/or declining renal function (to distinguish true flare from chronic damage)
- Development of new clinical features suggesting class transition (e.g., Class V developing nephritic features suggesting concurrent Class III/IV)
Additional Investigations
Renal Imaging:
- Renal ultrasound: Assess kidney size, echogenicity, exclude obstruction
- Normal to increased kidney size in acute lupus nephritis
- Small echogenic kidneys suggest chronic disease (Class VI)
- Doppler assessment of renal blood flow if concern for renal artery stenosis or thrombosis
Cardiovascular Risk Assessment:
- Lipid profile: Hyperlipidaemia in nephrotic syndrome; SLE patients have increased CV risk
- Blood pressure monitoring: Hypertension common in lupus nephritis
- ECG: Baseline before hydroxychloroquine (rare retinopathy risk)
Infection Screening Before Immunosuppression:
- Hepatitis B surface antigen, core antibody, surface antibody
- Hepatitis C antibody (and RNA if positive)
- HIV testing (particularly in high-risk populations)
- Tuberculosis screening: Interferon-gamma release assay (QuantiFERON, T-SPOT) or tuberculin skin test
- Strongyloides serology (in endemic areas)
Bone Health:
- Vitamin D level (often deficient in SLE; required for bone health)
- Baseline DEXA scan (before long-term corticosteroid therapy)
Monitoring Investigations During Treatment
Frequent Monitoring (Monthly During Induction, Every 3 Months During Maintenance):
- UPCR or 24-hour urine protein
- Serum creatinine and eGFR
- Anti-dsDNA antibodies and complement (C3, C4)
- Urinalysis with microscopy
- Complete blood count (monitor for cytopenias from immunosuppression)
Medication-Specific Monitoring:
- Mycophenolate: CBC (leukopenia, anaemia), GI symptoms
- Cyclophosphamide: CBC (profound leukopenia risk), urinalysis (haemorrhagic cystitis), fertility counselling
- Calcineurin inhibitors (voclosporin, tacrolimus, ciclosporin): Serum drug levels, blood pressure, nephrotoxicity monitoring
- Hydroxychloroquine: Annual ophthalmology examination (retinopathy screening)
7. Classification and Staging
The ISN/RPS 2003 classification system is the internationally accepted standard for classifying lupus nephritis based on renal biopsy findings. [9] This classification guides treatment decisions and provides prognostic information.
ISN/RPS 2003 Classification
| Class | Name | Histological Features | Clinical Presentation | Treatment Approach |
|---|---|---|---|---|
| Class I | Minimal Mesangial LN | Normal glomeruli on light microscopy; mesangial immune deposits on IF/EM | Usually asymptomatic; normal urinalysis or minimal abnormalities | No specific therapy; treat extrarenal SLE |
| Class II | Mesangial Proliferative LN | Mesangial hypercellularity or matrix expansion; mesangial immune deposits | Microscopic haematuria, subnephrotic proteinuria; normal renal function | Supportive care; low-dose steroids if symptomatic |
| Class III | Focal LN (less than 50% glomeruli) | Endocapillary or extracapillary proliferation in less than 50% glomeruli; subendothelial deposits | Haematuria, proteinuria (often nephrotic), HTN, declining GFR | Aggressive immunosuppression (induction + maintenance) |
| Class IV | Diffuse LN (≥50% glomeruli) | Endocapillary or extracapillary proliferation in ≥50% glomeruli; subendothelial deposits | Nephrotic-nephritic syndrome, HTN, AKI | Aggressive immunosuppression (induction + maintenance) |
| Class V | Membranous LN | Global or segmental subepithelial immune deposits; may coexist with III/IV | Nephrotic syndrome; variable haematuria and renal function | Treat as Class III/IV if nephrotic; observe if subnephrotic |
| Class VI | Advanced Sclerosing LN | ≥90% globally sclerosed glomeruli | CKD with bland sediment, HTN | Supportive care; prepare for RRT |
Class III and IV Subclassification
Active (A) versus Chronic (C) Lesions:
-
Class III (A) or IV (A): Active lesions predominate
- "Features: Endocapillary proliferation, neutrophil infiltration, fibrinoid necrosis, cellular crescents, wire-loop deposits"
- High activity index, low chronicity index
- Best treatment response potential
-
Class III (A/C) or IV (A/C): Mixed active and chronic lesions
- "Features: Active lesions plus glomerular sclerosis, fibrous crescents, tubular atrophy, interstitial fibrosis"
- Moderate activity and chronicity indices
- Intermediate treatment response
-
Class III (C) or IV (C): Chronic lesions predominate
- "Features: Glomerulosclerosis, fibrous crescents, tubular atrophy, interstitial fibrosis without active inflammation"
- Low activity index, high chronicity index
- Poor treatment response; immunosuppression may not be beneficial
Segmental (S) versus Global (G) Lesions (Class IV only):
- Class IV-S: Segmental (less than half of glomerular tuft) endocapillary or extracapillary proliferation in ≥50% of glomeruli
- Class IV-G: Global (more than half of glomerular tuft) endocapillary or extracapillary proliferation in ≥50% of glomeruli
- Class IV-G generally has worse prognosis than Class IV-S
Combined Classes
Class III/IV + V: Proliferative lupus nephritis with concurrent membranous features (subepithelial deposits)
- Treat with aggressive immunosuppression as for pure Class III/IV
- May have more pronounced nephrotic syndrome
Class II + V: Mesangial proliferative with membranous features
- Intermediate phenotype; treatment based on severity of proteinuria
Clinical-Pathological Correlation
The histological class does not always correlate perfectly with clinical presentation:
- Class III/IV typically presents with nephrotic-nephritic syndrome but can have isolated proteinuria
- Class V typically presents with nephrotic syndrome but may have minimal proteinuria if early or treated
- Asymptomatic patients can have Class III/IV on protocol biopsy (rare)
This underscores the necessity of renal biopsy for accurate classification and treatment planning. [9]
8. Management
Management of lupus nephritis requires a multipronged approach combining immunosuppressive therapy targeted at the histological class, supportive care to manage complications, and long-term monitoring to detect and treat relapses. [6,7,8]
General Principles
- Histology-driven treatment: ISN/RPS class determines immunosuppression intensity
- Induction-maintenance paradigm: Intensive immunosuppression for 3-6 months (induction) followed by prolonged lower-intensity therapy (maintenance)
- Universal interventions: Hydroxychloroquine and RAAS blockade for all patients unless contraindicated
- Multidisciplinary care: Nephrology, rheumatology, pharmacy collaboration
- Address cardiovascular risk: Control hypertension, hyperlipidaemia, smoking cessation
Class I and II: Minimal to Mesangial Proliferative LN
Treatment Strategy: No specific immunosuppression required for renal disease; supportive care
Management:
- Treat extrarenal SLE manifestations as indicated
- Hydroxychloroquine 200-400 mg daily (reduces SLE flares) [1]
- Monitor for progression: UPCR, creatinine every 3 months
- Low-dose corticosteroids (prednisolone 5-10 mg daily) if symptomatic (persistent haematuria, mild proteinuria)
- RAAS blockade (ACE inhibitor or ARB) if proteinuria > 0.5 g/day or hypertension
Prognosis: Excellent; rarely progresses to proliferative disease
Class III and IV: Proliferative Lupus Nephritis
Class III and IV lupus nephritis require aggressive immunosuppression to prevent progression to ESRD. [6,7] The treatment paradigm consists of induction therapy (3-6 months) to rapidly control inflammation, followed by maintenance therapy (minimum 3-5 years) to sustain remission and prevent flares.
Induction Therapy
Goal: Achieve complete or partial renal response within 12 months
- Complete response: UPCR less than 50 mg/mmol (less than 500 mg/g), normal or near-normal serum creatinine, inactive urinary sediment
- Partial response: ≥50% reduction in UPCR, stabilization or improvement in renal function
First-Line Options:
Option 1: Mycophenolate Mofetil (MMF) + Corticosteroids (Preferred)
Evidence base: ALMS trial demonstrated MMF non-inferior to cyclophosphamide for induction, with better tolerability [6]
Regimen:
- Mycophenolate mofetil: 2-3 g/day orally in divided doses (1000-1500 mg twice daily)
- "Target dose: 3 g/day for proliferative LN if tolerated"
- Reduce dose if GI intolerance (diarrhoea, nausea) or leukopenia
- Mycophenolic acid (enteric-coated) 1440-2160 mg/day is alternative formulation
- Corticosteroids:
- IV methylprednisolone 500-1000 mg daily for 3 days (pulse therapy) at initiation, OR
- Oral prednisolone starting at 0.5-1 mg/kg/day (maximum 60 mg/day)
- Taper to ≤7.5 mg/day by 6 months
- Minimize cumulative steroid exposure to reduce toxicity
Duration: 6 months
Advantages: Better tolerated than cyclophosphamide; preserves fertility; no haemorrhagic cystitis risk
Disadvantages: Teratogenic (requires reliable contraception); GI side effects common
Option 2: Cyclophosphamide + Corticosteroids
Evidence base: Historical gold standard; Euro-Lupus trial showed low-dose Euro-Lupus regimen non-inferior to high-dose NIH regimen [7]
Regimens:
- Low-dose Euro-Lupus protocol (preferred for Caucasian patients, lower toxicity):
- IV cyclophosphamide 500 mg every 2 weeks for 6 doses (total 3 g over 12 weeks)
- Followed by maintenance therapy (azathioprine or MMF)
- High-dose NIH protocol (may be preferred for African American patients or severe disease):
- IV cyclophosphamide 0.5-1.0 g/m² monthly for 6 months
- Adjust dose for renal function and WBC nadir
- Corticosteroids: As per MMF regimen above
Duration: 3-6 months
Advantages: Rapid, potent immunosuppression; long track record of efficacy
Disadvantages:
- Haemorrhagic cystitis (requires mesna prophylaxis and hydration)
- Ovarian failure (30-50% risk if age > 30; lower if younger)
- Profound leukopenia risk (monitor CBC weekly)
- Infections
- Malignancy risk (bladder cancer, haematological)
- Teratogenic (contraception mandatory)
Choice of MMF versus Cyclophosphamide:
- Most centres now prefer MMF as first-line due to better tolerability and equivalent efficacy [6]
- Cyclophosphamide reserved for: severe disease (crescentic GN, rapidly progressive GN), African American ethnicity (may respond better), or MMF intolerance/failure
Add-On Therapies for Induction
Voclosporin + MMF + Corticosteroids:
Evidence base: AURORA trial (2021) and AURORA-2 extension study showed voclosporin added to MMF/steroids improved complete response rates compared to MMF/steroids alone [8]
Regimen:
- Voclosporin 23.7 mg orally twice daily (dose-adjusted for CYP3A4 interactions)
- Plus MMF 2 g/day (lower dose than MMF monotherapy)
- Plus low-dose corticosteroids (rapid taper to ≤2.5 mg/day by 16 weeks)
Approval: FDA-approved 2021 for lupus nephritis based on AURORA data [8]
Advantages: Higher complete response rate (40% vs 23% with MMF alone); allows steroid minimization
Disadvantages:
- Expensive; requires therapeutic drug monitoring
- Calcineurin inhibitor nephrotoxicity potential (monitor serum creatinine, blood pressure)
- CYP3A4 interactions (avoid strong CYP3A4 inhibitors/inducers)
Indication: Consider for severe Class III/IV or after previous treatment failure
Belimumab + Standard Therapy:
Evidence base: BLISS-LN trial showed belimumab added to MMF or cyclophosphamide + steroids improved response rates compared to standard therapy alone [8]
Regimen:
- Belimumab 10 mg/kg IV at weeks 0, 2, 4, then every 4 weeks, OR
- Belimumab 200 mg SC weekly
- Plus standard induction (MMF or cyclophosphamide + steroids)
Mechanism: Anti-BLyS (B-lymphocyte stimulator) monoclonal antibody; reduces B-cell survival and autoantibody production
Advantages: Improved complete response (43% vs 32% with standard therapy alone); reduced renal flare risk
Disadvantages: Expensive; IV infusion burden; risk of infusion reactions
Indication: Severe or refractory Class III/IV lupus nephritis
Voclosporin Mechanism and Pharmacology:
Voclosporin is a novel calcineurin inhibitor (CNI) structurally related to ciclosporin but with key pharmacological advantages:
Mechanism of Action:
- Binds to cyclophilin, forming complex that inhibits calcineurin
- Calcineurin inhibition prevents dephosphorylation of NFAT (nuclear factor of activated T-cells)
- NFAT cannot translocate to nucleus → reduced IL-2 and other cytokine production
- Suppresses T-cell activation and proliferation
- Also stabilizes podocyte cytoskeleton, reducing proteinuria
Advantages over Ciclosporin:
- More predictable pharmacokinetics (less inter-patient variability)
- Less CYP3A4 metabolism variability
- Potentially less nephrotoxic (debated; requires long-term data)
- Dosed twice daily without need for routine therapeutic drug monitoring (though monitoring serum creatinine essential)
AURORA Trial Design and Results:
- Phase 3 RCT: 357 patients with Class III, IV, or V (±III/IV) lupus nephritis
- Intervention: Voclosporin 23.7 mg BID + MMF 2 g/day + steroids vs. placebo + MMF 2 g/day + steroids
- Primary endpoint (52 weeks): Complete renal response (UPCR ≤0.5 mg/mg, eGFR ≥60 or no decline > 20%, rapid steroid taper)
- Results: Voclosporin group 40.8% vs. control 22.5% achieved complete response (pless than 0.001)
- AURORA-2 (extension): Sustained benefit to 3 years [8]
Belimumab Mechanism:
Belimumab is a fully human IgG1λ monoclonal antibody targeting BLyS (B-lymphocyte stimulator), also known as BAFF (B-cell activating factor):
Mechanism of Action:
- BLyS is a cytokine produced by myeloid cells that promotes B-cell survival and differentiation
- SLE patients have elevated BLyS levels
- Belimumab binds soluble BLyS, preventing interaction with B-cell receptors (BR3, TACI, BCMA)
- Reduces B-cell survival, leading to decreased autoantibody production and immune complex formation
BLISS-LN Trial Design and Results:
- Phase 3 RCT: 448 patients with Class III or IV (±V) lupus nephritis
- Intervention: Belimumab 10 mg/kg IV monthly + standard therapy (MMF or CYC) vs. placebo + standard therapy
- Primary endpoint (104 weeks): Primary efficacy renal response (UPCR ≤0.7, eGFR ≥60 or no decline > 20%, no rescue therapy)
- Results: Belimumab group 43% vs. control 32% (OR 1.6, p=0.03)
- Secondary endpoints: Reduced risk of renal-related events, improved complete response rate [8]
Maintenance Therapy
Goal: Sustain remission, prevent flares, minimize cumulative immunosuppression toxicity
First-Line Maintenance (After Successful Induction):
Mycophenolate Mofetil:
- Dose: 1-2 g/day (lower than induction dose)
- Duration: Minimum 3-5 years; lifelong if recurrent flares
- Evidence: MAINTAIN Nephritis trial showed MMF superior to azathioprine for maintaining remission [6]
Alternative: Azathioprine:
- Dose: 1.5-2.5 mg/kg/day orally
- Duration: Minimum 3-5 years
- Indication: If MMF intolerance or pregnancy planning (azathioprine safer in pregnancy than MMF)
- Evidence: Effective but inferior to MMF for preventing relapses [6]
Low-Dose Corticosteroids:
- Prednisolone ≤7.5 mg/day
- Attempt to taper to ≤5 mg/day or discontinue if sustained remission
- Balance maintenance benefit against cumulative steroid toxicity (infection, osteoporosis, diabetes, cardiovascular disease)
Add-On Maintenance:
Belimumab or voclosporin may be continued during maintenance phase if used in induction, particularly if high-risk features (African American ethnicity, previous relapse, persistently active serology).
Duration of Maintenance Therapy
- Minimum 3-5 years after achieving complete remission
- Consider longer duration (5-10 years or indefinite) if:
- Recurrent relapses
- African American or Hispanic ethnicity (higher relapse risk)
- Class IV-G disease
- Persistently positive anti-dsDNA or low complement despite clinical remission
- Gradual taper and discontinuation only after sustained remission (> 3 years) with close monitoring
Treatment of Refractory or Relapsing Disease
Definitions:
- Primary refractory: No response (complete or partial) after 12 months of induction therapy
- Relapse: Worsening proteinuria (doubling of UPCR or increase to nephrotic range), declining eGFR (> 25% decline), or active urinary sediment after achieving response
Management Options:
-
Switch induction agent:
- If on MMF → switch to cyclophosphamide
- If on cyclophosphamide → switch to MMF (or tacrolimus/ciclosporin)
-
Add voclosporin or belimumab to standard therapy
-
Calcineurin inhibitors (tacrolimus, ciclosporin):
- Tacrolimus 0.05-0.1 mg/kg/day (target level 5-10 ng/mL)
- Ciclosporin 3-5 mg/kg/day (target level 100-200 ng/mL)
- Evidence: Small RCTs suggest efficacy, particularly for membranous LN [1]
- Requires monitoring for nephrotoxicity, hypertension, hyperglycaemia
-
Rituximab (anti-CD20 monoclonal antibody):
- Dose: 1000 mg IV on days 1 and 15, OR 375 mg/m² weekly ×4
- Evidence: LUNAR trial (rituximab + MMF) did not meet primary endpoint, but post-hoc analyses and observational data suggest benefit in refractory cases [1]
- Indication: Refractory disease after failure of standard agents
-
Repeat renal biopsy: Essential to guide treatment escalation
- Confirms true relapse (activity) versus chronic damage (high chronicity index)
- May reveal class transformation (e.g., Class V developing concurrent Class III/IV)
Class V: Membranous Lupus Nephritis
Treatment Strategy: Risk-stratified based on proteinuria severity
Nephrotic-Range Proteinuria (UPCR > 350 mg/mmol or > 3.5 g/day):
- Treat as Class III/IV: Induction with MMF or cyclophosphamide + steroids, followed by maintenance
- Consider calcineurin inhibitors (tacrolimus, ciclosporin) as alternative or add-on therapy
- Voclosporin particularly effective for Class V based on AURORA trial subgroup [8]
Subnephrotic Proteinuria (UPCR less than 350 mg/mmol):
- Observation with supportive care (RAAS blockade, hydroxychloroquine)
- Low-dose corticosteroids (prednisolone 0.5 mg/kg/day tapering over 6 months)
- Monitor closely; escalate to immunosuppression if proteinuria worsens or renal function declines
Class V + III/IV (Combined):
- Treat as pure Class III/IV (aggressive immunosuppression)
Class VI: Advanced Sclerosing LN
Treatment Strategy: Supportive care; no role for immunosuppression
Management:
- Manage chronic kidney disease complications: Anaemia (erythropoietin-stimulating agents, iron), bone mineral disease (phosphate binders, vitamin D), metabolic acidosis
- Blood pressure control (target less than 130/80 mmHg)
- Prepare for renal replacement therapy: Vascular access planning, transplant evaluation
- Treat extrarenal SLE as needed
- Low-dose hydroxychloroquine continued for SLE management
Renal Replacement Therapy:
- Haemodialysis, peritoneal dialysis, or kidney transplantation
- Transplantation outcomes excellent if SLE quiescent for 6-12 months pre-transplant
- Recurrence of lupus nephritis in allograft: 2-5% [12]
Universal Adjunctive Therapies (All Classes with Proteinuria)
Hydroxychloroquine:
- Dose: 200-400 mg/day (≤5 mg/kg/day actual body weight to reduce retinopathy risk)
- Indication: All lupus nephritis patients unless contraindicated
- Evidence: Reduces SLE flares, renal flares, thrombosis risk, and improves long-term survival [1]
- Monitoring: Annual ophthalmology examination for retinopathy
- Contraindications: Retinopathy, G6PD deficiency (relative)
RAAS Blockade (ACE Inhibitor or ARB):
- Indication: All patients with proteinuria > 0.5 g/day or hypertension
- Evidence: Reduces proteinuria independently of blood pressure effect; nephroprotective [1]
- Examples: Ramipril 5-10 mg daily, enalapril 10-20 mg daily, losartan 50-100 mg daily
- Monitor: Serum creatinine and potassium (risk of hyperkalemia, particularly if eGFR less than 30)
- Avoid in pregnancy (teratogenic)
Blood Pressure Control:
- Target: less than 130/80 mmHg (lower if tolerated)
- First-line: ACE inhibitor or ARB (as above)
- Add-on agents: Calcium channel blockers (amlodipine), beta-blockers, diuretics
Lipid Management:
- Statin therapy if LDL > 3.0 mmol/L or cardiovascular risk factors
- Particularly important in nephrotic syndrome (hyperlipidaemia)
Antiplatelet Therapy:
- Low-dose aspirin (75-100 mg daily) if antiphospholipid antibodies positive or high cardiovascular risk
- Not routinely recommended in all lupus nephritis
Bone Health:
- Calcium (1000-1500 mg daily) + vitamin D (800-2000 IU daily) supplementation
- Baseline and periodic DEXA scans (monitor for steroid-induced osteoporosis)
- Bisphosphonate (alendronate 70 mg weekly) if high fracture risk (T-score -2.5 or less or fragility fracture)
Infection Prophylaxis:
- Pneumocystis jirovecii pneumonia (PCP) prophylaxis:
- "Indication: During induction therapy with cyclophosphamide or high-dose steroids + MMF"
- "Agent: Trimethoprim-sulfamethoxazole 480-960 mg three times weekly or daily"
- "Alternative: Dapsone (if sulfa allergy), atovaquone"
Contraception (Women of Childbearing Potential):
- Mandatory during MMF or cyclophosphamide therapy (highly teratogenic)
- Barrier methods, progesterone-only contraception, or IUD preferred
- Combined oral contraceptive pills relatively contraindicated in SLE (thrombosis risk, especially if antiphospholipid antibodies present)
- Counsel on fertility preservation before cyclophosphamide (GnRH agonist co-administration, oocyte cryopreservation)
Treatment Monitoring
Frequency:
- Monthly during induction therapy
- Every 3 months during maintenance therapy
- More frequently if concerns about disease flare or medication toxicity
Parameters:
- UPCR or 24-hour urine protein
- Serum creatinine and eGFR
- Urinalysis with microscopy
- Anti-dsDNA antibodies and complement (C3, C4)
- Complete blood count (monitor for cytopenias)
- Medication-specific monitoring (e.g., voclosporin levels, cyclophosphamide WBC nadir)
Definitions of Treatment Response:
| Response Category | Definition (at 12 months) |
|---|---|
| Complete Response | UPCR less than 50 mg/mmol (less than 0.5 mg/mg), serum creatinine normal or less than 115% of baseline, inactive urinary sediment |
| Partial Response | ≥50% reduction in UPCR (if nephrotic at baseline, UPCR less than 300 mg/mmol), serum creatinine ≤125% of baseline |
| No Response | Failure to achieve partial or complete response |
| Relapse | After achieving response: UPCR increase to > 100 mg/mmol with doubling from remission value, or ≥25% decline in eGFR, or nephritic sediment recurrence |
Special Populations
Pregnancy:
- Active lupus nephritis is contraindication to pregnancy
- Disease should be quiescent for ≥6 months before conception
- Medication adjustments:
- "Safe: Hydroxychloroquine (continue), azathioprine, low-dose prednisolone, tacrolimus"
- "Unsafe: MMF (stop 6 weeks before conception), cyclophosphamide (stop 3 months before), methotrexate, ACE inhibitors/ARBs"
- Close monitoring throughout pregnancy (monthly clinic visits, UPCR, creatinine, anti-dsDNA, complement)
- High-risk obstetric care (maternal-fetal medicine) [20]
Elderly Patients:
- Lower immunosuppression doses to reduce infection risk
- More aggressive supportive care (blood pressure, bone health)
- Consider comorbidities when selecting agents
Children and Adolescents:
- Lupus nephritis more aggressive in paediatric-onset SLE
- Treatment regimens similar to adults (adjusted for weight/BSA)
- Growth monitoring (steroid effects)
- Transition planning to adult services
9. Complications
Lupus nephritis is associated with significant complications arising from the disease itself, its treatment, and the underlying systemic nature of SLE. [1,5,11]
Renal Complications
End-Stage Renal Disease (ESRD):
- Incidence: 10-20% of lupus nephritis patients progress to ESRD requiring dialysis or transplantation despite treatment [5]
- Risk factors:
- African American or Hispanic ethnicity (3-4 fold increased risk) [14,15]
- Delayed diagnosis or treatment
- Class IV-G disease (global proliferative)
- High chronicity index on biopsy (> 4)
- Baseline renal impairment (eGFR less than 60 at diagnosis)
- Persistent nephrotic-range proteinuria despite treatment
- Poor medication adherence
- Prevention: Early diagnosis, aggressive treatment, close monitoring, addressing socioeconomic barriers to care
Chronic Kidney Disease (CKD):
- Even without progression to ESRD, many patients develop CKD stages 3-4
- Complications: Anaemia, bone mineral disease, hypertension, cardiovascular disease
- Management: Nephroprotective strategies (RAAS blockade), CKD complication management, slow progression
Renal Flares:
- Incidence: 50-70% of patients experience ≥1 renal flare during long-term follow-up despite maintenance therapy [1]
- Triggers: Medication non-adherence, infections, pregnancy, discontinuation of immunosuppression
- Detection: Rising UPCR, declining eGFR, active sediment, rising anti-dsDNA, falling complement
- Management: Re-biopsy if severe or uncertain; re-induction therapy; optimize maintenance regimen; address adherence
Thrombotic Microangiopathy:
- Associated with antiphospholipid syndrome in SLE
- Presentation: AKI, microangiopathic haemolytic anaemia, thrombocytopenia, neurological symptoms
- Management: Differentiate from TTP (ADAMTS13 activity); anticoagulation; consider plasmapheresis or eculizumab
Cardiovascular Complications
Hypertension:
- Prevalence: 60-80% of Class III/IV lupus nephritis patients [3]
- Causes: Renal disease, corticosteroid therapy, calcineurin inhibitors, intrinsic SLE vasculopathy
- Target: less than 130/80 mmHg
- Management: RAAS blockade first-line; add calcium channel blockers, beta-blockers, diuretics as needed
Accelerated Atherosclerosis:
- SLE patients have 5-10 fold increased cardiovascular disease risk compared to age-matched controls [1]
- Mechanisms: Chronic inflammation, dyslipidaemia (particularly in nephrotic syndrome), hypertension, corticosteroid therapy
- Prevention: Statin therapy, blood pressure control, smoking cessation, aspirin (if antiphospholipid antibodies)
Thromboembolic Events:
- Increased risk due to:
- Antiphospholipid antibodies (20-30% of SLE patients) [1]
- Nephrotic syndrome (hypercoagulability due to urinary loss of antithrombin III)
- Inflammatory state
- Manifestations: Deep vein thrombosis, pulmonary embolism, stroke, myocardial infarction
- Prevention: Anticoagulation if antiphospholipid syndrome; mobilization; consider prophylactic anticoagulation in nephrotic syndrome if albumin less than 20 g/L
Infectious Complications
Increased Infection Risk:
- Causes: Immunosuppressive therapy (cyclophosphamide, MMF, corticosteroids, belimumab), complement deficiency, uraemia (if CKD)
- Common infections:
- "Bacterial: Urinary tract infections, pneumonia (Streptococcus pneumoniae, Haemophilus influenzae), skin/soft tissue infections"
- "Opportunistic: Pneumocystis jirovecii pneumonia (PCP), cytomegalovirus, fungal infections (Candida, Aspergillus)"
- "Viral: Herpes zoster reactivation (particularly with MMF/cyclophosphamide), influenza"
- Prevention:
- PCP prophylaxis during intensive immunosuppression
- "Vaccinations (before starting immunosuppression if possible): Pneumococcal (PCV13 and PPSV23), influenza (annual), hepatitis B"
- Avoid live vaccines during immunosuppression (MMR, varicella, yellow fever)
- Prompt treatment of infections
Sepsis:
- Leading cause of death in lupus nephritis patients [11]
- High index of suspicion in immunosuppressed patient with fever
- Aggressive investigation and empirical broad-spectrum antibiotics
Pregnancy-Related Complications
Maternal Complications:
- Lupus flare (30-50% risk during pregnancy or postpartum period) [20]
- Renal flare (higher risk if active disease at conception)
- Preeclampsia (20-30% risk; difficult to distinguish from lupus nephritis flare)
- Thromboembolic events (if antiphospholipid syndrome)
- Need for emergent delivery due to maternal deterioration
Fetal Complications:
- Preterm delivery (50%)
- Intrauterine growth restriction (30%)
- Fetal loss (miscarriage 10-20%, stillbirth 2-5%)
- Neonatal lupus (congenital heart block if anti-Ro/anti-La antibodies present; photosensitive rash) [20]
Management: Multidisciplinary high-risk obstetric care; close monitoring; safe medication continuation (hydroxychloroquine, azathioprine, prednisolone); delivery planning
Malignancy
Increased Cancer Risk:
- Mechanisms: Chronic immunosuppression (particularly cyclophosphamide), SLE-related immune dysregulation
- Specific malignancies:
- Bladder cancer (cyclophosphamide exposure; risk increases with cumulative dose)
- Haematological malignancies (non-Hodgkin lymphoma, leukaemia)
- Cervical cancer (HPV; inadequate immune surveillance)
- Surveillance: Age-appropriate cancer screening; haematuria investigation (cystoscopy if cyclophosphamide exposure); cervical screening
Medication-Specific Complications
Corticosteroids:
- Short-term: Hyperglycaemia, hypertension, infection, psychiatric disturbance, insomnia, weight gain
- Long-term: Osteoporosis, avascular necrosis (hip, knee), cataracts, glaucoma, cushingoid features, diabetes mellitus, cardiovascular disease
Mycophenolate Mofetil:
- Gastrointestinal: Diarrhoea, nausea, abdominal pain (20-30%)
- Haematological: Leukopenia, anaemia
- Teratogenicity (absolute contraindication in pregnancy)
Cyclophosphamide:
- Haemorrhagic cystitis (10-20%; reduced with mesna and hydration)
- Gonadal toxicity: Ovarian failure (30-50% if age > 30; lower if younger), azoospermia
- Profound leukopenia (monitor CBC weekly)
- Malignancy (bladder cancer, leukaemia)
- Alopecia (reversible)
Calcineurin Inhibitors (Voclosporin, Tacrolimus, Ciclosporin):
- Nephrotoxicity (acute and chronic; requires monitoring)
- Hypertension
- Neurotoxicity (tremor, headache)
- Hyperglycaemia (new-onset diabetes after transplantation)
- Gingival hyperplasia, hirsutism (ciclosporin)
Hydroxychloroquine:
- Retinopathy (rare; less than 1% if dose ≤5 mg/kg/day and annual screening)
- Gastrointestinal upset
- Skin hyperpigmentation
Belimumab:
- Infusion reactions (headache, nausea)
- Infections
- Depression (monitor mental health)
Bone Health
Osteoporosis:
- Risk factors: Corticosteroid therapy (dose and duration dependent), chronic kidney disease, vitamin D deficiency
- Screening: Baseline DEXA scan before long-term corticosteroids; repeat every 1-2 years
- Prevention: Calcium and vitamin D supplementation; bisphosphonates if T-score -2.5 or less or fragility fracture
Avascular Necrosis (Osteonecrosis):
- Typically affects hip (femoral head), less commonly knee, shoulder
- Risk factors: High-dose or long-term corticosteroids, antiphospholipid antibodies
- Presentation: Hip or groin pain; may progress to collapse and require arthroplasty
- Diagnosis: MRI (sensitive; detects early changes before X-ray abnormalities)
10. Prognosis
The prognosis of lupus nephritis has improved dramatically over the past 50 years due to advances in immunosuppressive therapy, earlier diagnosis, and better supportive care. [11]
Renal Survival
| Outcome | Rate | Notes |
|---|---|---|
| 5-year renal survival | 80-90% | Contemporary cohorts with optimal therapy [11] |
| 10-year renal survival | 70-80% | Significant improvement from 45% in 1970s [11] |
| 20-year renal survival | 60-70% | Long-term data emerging [5] |
| Progression to ESRD | 10-20% | Despite treatment; higher in certain populations [5] |
| Renal relapse rate | 50-70% | At least one flare during long-term follow-up [1] |
Patient Survival
- 5-year patient survival: > 90% in most contemporary series [11]
- 10-year patient survival: 85-90%
- Leading causes of death: Infection (40-50%), cardiovascular disease (25-30%), active SLE (10-15%), malignancy (5-10%) [11]
Prognostic Factors
Poor Prognostic Indicators:
- Histological:
- Class IV-G (global) versus Class IV-S (segmental)
- High chronicity index (> 4) on biopsy [9]
- Tubulointerstitial fibrosis and tubular atrophy > 25%
- Crescents in > 50% of glomeruli
- Clinical:
- African American, Hispanic, or Asian ethnicity [14,15]
- Baseline renal impairment (eGFR less than 60 ml/min/1.73m²)
- Nephrotic-range proteinuria (> 3.5 g/day) at presentation
- Delayed diagnosis (> 6 months from symptom onset)
- Hypertension at presentation
- Male sex [17]
- Laboratory:
- Persistently low complement despite treatment
- Persistently elevated anti-dsDNA despite treatment
- Lack of complete response by 12 months
- Socioeconomic:
- Low socioeconomic status (access to care, medication adherence) [15]
- Uninsured or underinsured status
- Medication non-adherence
Good Prognostic Indicators:
- Class I, II, or V (pure) disease
- Low chronicity index (less than 2)
- Early diagnosis and treatment initiation
- Complete response to induction therapy by 6-12 months
- Caucasian ethnicity (compared to African American)
- Good medication adherence
- Access to multidisciplinary nephrology/rheumatology care
Response to Treatment
Complete Response:
- Achieved in 40-60% of patients by 12 months with modern regimens (MMF or cyclophosphamide + steroids) [6,7]
- Complete response by 12 months strongly predicts long-term renal survival
- Higher rates with add-on voclosporin (40% vs 23%) or belimumab (43% vs 32%) [8]
Partial Response:
- Achieved in additional 20-30% of patients
- Partial response better than no response but inferior to complete response for long-term outcomes
- May convert to complete response with continued therapy beyond 12 months
No Response (Refractory Disease):
- 10-20% of patients fail to achieve even partial response
- Requires treatment escalation (switch agents, add voclosporin/belimumab, consider rituximab)
- Repeat biopsy essential to guide further management
Long-Term Outcomes
Renal Function:
- Patients achieving complete response: Stable renal function in 80-90%
- Patients with partial response: 50-70% stable; 20-30% progressive CKD
- Patients with no response: High risk of progression to ESRD
Quality of Life:
- Significantly impaired compared to general population
- Contributors: Fatigue (multifactorial: disease activity, anaemia, uraemia), chronic pain (arthralgia), medication side effects (corticosteroids), psychological burden (depression, anxiety), employment disability
Transplantation Outcomes:
- Renal transplantation is viable option for ESRD due to lupus nephritis
- Outcomes: 5-year graft survival 80-85%, similar to other causes of ESRD [12]
- Recurrence of lupus nephritis in allograft: 2-5% (low) [12]
- Recommendations: SLE should be quiescent for 6-12 months pre-transplant; continue hydroxychloroquine post-transplant; standard immunosuppression (tacrolimus, MMF, prednisolone) treats both allograft rejection and SLE
11. Key Guidelines and Evidence
Major Guidelines
KDIGO 2021 Glomerulonephritis Guidelines:
- Comprehensive evidence-based recommendations for lupus nephritis management
- Emphasizes renal biopsy for classification
- Recommends MMF or cyclophosphamide + corticosteroids for Class III/IV induction
- Supports hydroxychloroquine for all patients
- Target blood pressure less than 120/80 mmHg if proteinuria
- PMID: 34556256 [1]
EULAR/ERA-EDTA 2019 Recommendations:
- European consensus on lupus nephritis management
- Similar recommendations to KDIGO
- Emphasizes multidisciplinary care
- Addresses voclosporin and belimumab as emerging therapies
ACR 2012 Guidelines (Older but Foundational):
- American College of Rheumatology recommendations
- Established MMF and cyclophosphamide as equivalent first-line options
- Hydroxychloroquine universal recommendation
Landmark Trials
| Trial | Year | Intervention | Key Finding | Reference |
|---|---|---|---|---|
| ALMS | 2009 | MMF vs CYC induction | MMF non-inferior to CYC for induction; better tolerated | [6] |
| MAINTAIN | 2010 | MMF vs AZA maintenance | MMF superior to AZA for preventing relapses | [6] |
| Euro-Lupus | 2002 | Low-dose vs high-dose CYC | Low-dose CYC (Euro-Lupus protocol) non-inferior to high-dose NIH protocol | [7] |
| AURORA | 2021 | Voclosporin + MMF vs MMF | Voclosporin improved complete response rate (41% vs 23%) | [8] |
| BLISS-LN | 2020 | Belimumab + standard vs standard | Belimumab improved primary efficacy renal response (43% vs 32%) | [8] |
12. Examination Focus
High-Yield Exam Topics
For MRCP and Nephrology Exams:
- ISN/RPS classification system (Classes I-VI, treatment implications)
- Indications for renal biopsy in SLE patients
- "Full house" immunofluorescence pattern
- MMF versus cyclophosphamide (ALMS trial, non-inferiority)
- Voclosporin and belimumab mechanisms and trial data (AURORA, BLISS-LN)
- Universal therapies: Hydroxychloroquine and RAAS blockade
- Anti-dsDNA and complement monitoring (C3, C4)
- Complete versus partial response definitions
- Pregnancy in lupus nephritis (risks, safe medications)
- Complications: ESRD risk (10-20%), infection, cardiovascular disease
Viva Approach
Opening Statement:
"Lupus nephritis is immune complex-mediated glomerulonephritis occurring in 40-50% of patients with systemic lupus erythematosus. It is classified using the ISN/RPS 2003 system based on renal biopsy findings into six classes, which guide treatment decisions. Class III and IV proliferative lupus nephritis require aggressive immunosuppression with induction and maintenance therapy, while Class I and II require supportive care only."
Structured Approach to Case Scenario:
If presented with a patient with suspected lupus nephritis:
-
Confirm SLE diagnosis:
- Clinical criteria (malar rash, arthritis, serositis, etc.)
- Serological criteria (ANA, anti-dsDNA, anti-Sm, complement)
-
Assess for renal involvement:
- Proteinuria (UPCR), haematuria, declining renal function
- Active urinary sediment (RBC casts, dysmorphic RBCs)
-
Arrange renal biopsy:
- Indications: Proteinuria > 500 mg/day, active sediment, rising creatinine
- Obtain ISN/RPS class, activity/chronicity indices
-
Initiate treatment based on biopsy class:
- Class I/II: Supportive care, hydroxychloroquine
- Class III/IV: Induction (MMF 2-3 g/day or cyclophosphamide) + corticosteroids, followed by maintenance (MMF 1-2 g/day for ≥3-5 years)
- Class V: If nephrotic, treat as Class III/IV
- Class VI: Supportive care, prepare for RRT
-
Universal adjuncts:
- Hydroxychloroquine 200-400 mg/day
- ACE inhibitor or ARB for proteinuria and blood pressure control
-
Monitor response:
- Monthly: UPCR, creatinine, anti-dsDNA, C3/C4
- Target: Complete response by 12 months (UPCR less than 50 mg/mmol, normal creatinine)
Examiner Questions and Model Answers:
Q: What is the ISN/RPS classification and why is it important?
A: "The ISN/RPS 2003 classification categorizes lupus nephritis into six classes based on histopathology. Class I is minimal mesangial, Class II is mesangial proliferative, Class III is focal proliferative affecting less than 50% of glomeruli, Class IV is diffuse proliferative affecting 50% or more, Class V is membranous, and Class VI is advanced sclerosing. This classification is critical because it determines treatment intensity: Class I and II require supportive care only, Class III and IV require aggressive immunosuppression with induction and maintenance regimens, Class V is treated as Class III/IV if nephrotic-range proteinuria is present, and Class VI requires preparation for renal replacement therapy."
Q: What are the treatment options for Class III/IV lupus nephritis?
A: "First-line induction therapy consists of either mycophenolate mofetil 2-3 grams daily or cyclophosphamide—using the low-dose Euro-Lupus protocol of 500 mg IV every 2 weeks for six doses—combined with corticosteroids, initially IV methylprednisolone or high-dose oral prednisolone tapered to 7.5 mg daily or less by 6 months. The ALMS trial demonstrated MMF non-inferior to cyclophosphamide with better tolerability, so MMF is now preferred first-line at most centers. After achieving response, maintenance therapy with MMF 1-2 grams daily or azathioprine continues for a minimum of 3-5 years. Newer add-on therapies include voclosporin—a calcineurin inhibitor shown in the AURORA trial to improve complete response rates to 41% versus 23% with MMF alone—and belimumab, an anti-BLyS monoclonal antibody that improved response in the BLISS-LN trial. Hydroxychloroquine and RAAS blockade are universal adjuncts for all patients."
Q: How do you monitor treatment response and when would you consider treatment failure?
A: "I would monitor monthly during induction with urine protein-to-creatinine ratio, serum creatinine and eGFR, urinalysis with microscopy, anti-dsDNA antibodies, and complement levels C3 and C4. The goal is to achieve complete response by 12 months, defined as UPCR less than 50 mg/mmol, serum creatinine normal or less than 115% of baseline, and inactive urinary sediment. Partial response is a ≥50% reduction in UPCR with creatinine ≤125% of baseline. Primary refractory disease means no response after 12 months of induction therapy. In this scenario, I would arrange a repeat renal biopsy to confirm true active disease versus chronic damage, then consider switching immunosuppressive agents—such as from MMF to cyclophosphamide or vice versa—or adding voclosporin or belimumab. Relapse after achieving response is defined by doubling of UPCR or increase to nephrotic range, ≥25% decline in eGFR, or recurrence of active sediment, and similarly requires repeat biopsy and treatment intensification."
Q: What are the major complications of lupus nephritis and its treatment?
A: "The major disease-related complications are progression to end-stage renal disease in 10-20% of patients despite treatment, renal flares in 50-70% during long-term follow-up, hypertension, thromboembolic events particularly if antiphospholipid antibodies are present, and pregnancy complications including preeclampsia and fetal loss. Treatment-related complications include infections due to immunosuppression—sepsis is a leading cause of death—for which we use PCP prophylaxis during intensive immunosuppression. Cyclophosphamide causes hemorrhagic cystitis requiring mesna prophylaxis, gonadal toxicity with ovarian failure in 30-50% of women over age 30, and long-term malignancy risk particularly bladder cancer. Mycophenolate causes GI side effects and is highly teratogenic. Corticosteroids cause infection, osteoporosis, avascular necrosis, diabetes, and cardiovascular disease. Calcineurin inhibitors like voclosporin risk nephrotoxicity and hypertension requiring close monitoring."
Q: How do you manage pregnancy in a patient with lupus nephritis?
A: "Active lupus nephritis is a contraindication to pregnancy, so disease must be quiescent for at least 6 months before conception. Pre-pregnancy planning is essential with multidisciplinary involvement from nephrology, rheumatology, and maternal-fetal medicine. Medications must be adjusted: mycophenolate must be stopped 6 weeks before conception, cyclophosphamide 3 months before, and ACE inhibitors or ARBs discontinued due to teratogenicity. Safe medications to continue are hydroxychloroquine—which reduces flare risk and should absolutely be continued—azathioprine, low-dose prednisolone, and tacrolimus if needed. During pregnancy, the patient requires monthly monitoring with UPCR, creatinine, anti-dsDNA, and complement levels to detect flares, which occur in 30-50% of pregnancies. Distinguishing lupus nephritis flare from preeclampsia is challenging and may require complement levels, anti-dsDNA trends, and clinical judgment. Fetal risks include preterm delivery in 50%, intrauterine growth restriction in 30%, and fetal loss in 10-20%. If anti-Ro or anti-La antibodies are present, there is risk of neonatal lupus including congenital heart block requiring fetal echocardiography surveillance."
Common Exam Mistakes to Avoid
❌ Failing to perform renal biopsy when indicated (proteinuria > 500 mg/day) and attempting to treat empirically
❌ Treating Class I/II with aggressive immunosuppression (not required; supportive care sufficient)
❌ Omitting hydroxychloroquine from the treatment regimen (universal indication unless contraindicated)
❌ Not monitoring complement and anti-dsDNA during treatment (key biomarkers of activity)
❌ Assuming low complement is specific for lupus nephritis (also occurs in post-infectious GN, MPGN, cryoglobulinemia)
❌ Forgetting pregnancy planning and contraception counseling (MMF and cyclophosphamide highly teratogenic)
❌ Failing to provide PCP prophylaxis during intensive immunosuppression
❌ Not assessing response to treatment by 12 months (complete response strongly predicts long-term outcomes)
❌ Confusing ISN/RPS classes (particularly Class III less than 50% vs Class IV ≥50% glomerular involvement)
❌ Assuming ANCA vasculitis when complement is normal (ANCA-associated vasculitis does not consume complement)
Key Phrases for Exams
- "Lupus nephritis requires renal biopsy for ISN/RPS classification to guide treatment."
- "The full-house immunofluorescence pattern (IgG, IgA, IgM, C3, C1q) is characteristic."
- "Class III/IV proliferative lupus nephritis requires induction plus maintenance immunosuppression."
- "Mycophenolate mofetil is non-inferior to cyclophosphamide for induction (ALMS trial) with better tolerability."
- "Hydroxychloroquine is universal for all lupus nephritis patients to reduce flares."
- "Anti-dsDNA and low complement (C3/C4) correlate with disease activity."
- "Voclosporin improved complete response to 41% versus 23% in the AURORA trial."
- "Belimumab targets BLyS (B-lymphocyte stimulator) to reduce B-cell survival."
- "Complete response by 12 months predicts long-term renal survival."
- "10-20% progress to ESRD despite optimal treatment, higher in African American patients."
References
-
Kidney Disease: Improving Global Outcomes (KDIGO). Clinical Practice Guideline for Glomerulonephritis. Kidney Int Suppl. 2021;11(2):e1-e143. doi:10.1016/j.kisu.2021.05.001
-
Anders HJ, Saxena R, Zhao MH, Parodis I, Salmon JE, Mohan C. Lupus nephritis. Nat Rev Dis Primers. 2020;6(1):7. doi:10.1038/s41572-019-0141-9
-
Almaani S, Meara A, Rovin BH. Update on lupus nephritis. Clin J Am Soc Nephrol. 2017;12(5):825-835. doi:10.2215/CJN.05780616
-
Davidson A. What is damaging the kidney in lupus nephritis? Nat Rev Rheumatol. 2016;12(3):143-153. doi:10.1038/nrrheum.2015.159
-
Hanly JG, O'Keeffe AG, Su L, et al. The frequency and outcome of lupus nephritis: results from an international inception cohort study. Rheumatology (Oxford). 2016;55(2):252-262. doi:10.1093/rheumatology/kev311
-
Appel GB, Contreras G, Dooley MA, et al. Mycophenolate mofetil versus cyclophosphamide for induction treatment of lupus nephritis. J Am Soc Nephrol. 2009;20(5):1103-1112. doi:10.1681/ASN.2008101028
-
Houssiau FA, Vasconcelos C, D'Cruz D, et al. Immunosuppressive therapy in lupus nephritis: the Euro-Lupus Nephritis Trial, a randomized trial of low-dose versus high-dose intravenous cyclophosphamide. Arthritis Rheum. 2002;46(8):2121-2131. doi:10.1002/art.10461
-
Rovin BH, Teng YKO, Ginzler EM, et al. Efficacy and safety of voclosporin versus placebo for lupus nephritis (AURORA 1): a double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2021;397(10289):2070-2080. doi:10.1016/S0140-6736(21)00578-X
-
Weening JJ, D'Agati VD, Schwartz MM, et al. The classification of glomerulonephritis in systemic lupus erythematosus revisited. J Am Soc Nephrol. 2004;15(2):241-250. doi:10.1097/01.asn.0000108969.21691.5d
-
Appel GB, Pisetsky DS, Bomback AS. Lupus nephritis: pathology, pathogenesis and treatment. UpToDate. 2020. [Review Article]
-
Mejía-Vilet JM, Cordova-Sanchez BM, Arreola-Guerra JM, Morales-Buenrostro LE, Uribe-Uribe NO, Correa-Rotter R. Renal flare prediction and prognosis in lupus nephritis Hispanic patients. Lupus. 2016;25(3):315-324. doi:10.1177/0961203315606985
-
Goral S. Renal transplantation in patients with lupus nephritis. Transplant Rev (Orlando). 2014;28(1):1-8. doi:10.1016/j.trre.2013.10.001
-
Petri M, Orbai AM, Alarcón GS, et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 2012;64(8):2677-2686. doi:10.1002/art.34473
-
Feldman CH, Hiraki LT, Liu J, et al. Epidemiology and sociodemographics of systemic lupus erythematosus and lupus nephritis among US adults with Medicaid coverage, 2000-2004. Arthritis Rheum. 2013;65(3):753-763. doi:10.1002/art.37795
-
Lim SS, Bayakly AR, Helmick CG, Gordon C, Easley KA, Drenkard C. The incidence and prevalence of systemic lupus erythematosus, 2002-2004: The Georgia Lupus Registry. Arthritis Rheumatol. 2014;66(2):357-368. doi:10.1002/art.38239
-
Tsang ASMWP, Bultink IEM, Voskuyl AE. Systemic lupus erythematosus in Asians: a review. Lupus. 2011;20(3):257-269. doi:10.1177/0961203310388445
-
García MA, Marcos JC, Marcos AI, et al. Male gender is not a poor prognostic factor in lupus nephritis. Lupus. 2005;14(12):968-974. doi:10.1191/0961203305lu2241oa
-
Isenberg DA, Manson JJ, Ehrenstein MR, Rahman A. Fifty years of anti-ds DNA antibodies: are we approaching journey's end? Rheumatology (Oxford). 2007;46(7):1052-1056. doi:10.1093/rheumatology/kem112
-
Trouw LA, Groeneveld TW, Seelen MA, et al. Anti-C1q autoantibodies deposit in glomeruli but are only pathogenic in combination with glomerular C1q-containing immune complexes. J Clin Invest. 2004;114(5):679-688. doi:10.1172/JCI21075
-
Smyth A, Oliveira GH, Lahr BD, Bailey KR, Norby SM, Garovic VD. A systematic review and meta-analysis of pregnancy outcomes in patients with systemic lupus erythematosus and lupus nephritis. Clin J Am Soc Nephrol. 2010;5(11):2060-2068. doi:10.2215/CJN.00240110
Last Updated: 2026-01-05
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Systemic Lupus Erythematosus
- Glomerulonephritis Overview
Differentials
Competing diagnoses and look-alikes to compare.
- IgA Nephropathy
- Membranoproliferative Glomerulonephritis
- ANCA-Associated Vasculitis
Consequences
Complications and downstream problems to keep in mind.
- End-Stage Renal Disease
- Chronic Kidney Disease