Lymphoma (Adult)
Lymphomas are a heterogeneous group of malignancies arising from lymphoid tissue, representing the fifth most common cancer in developed countries. They are broadly classified into Hodgkin lymphoma (HL) and...
What matters first
Lymphomas are a heterogeneous group of malignancies arising from lymphoid tissue, representing the fifth most common cancer in developed countries. They are broadly classified into Hodgkin lymphoma (HL) and...
Superior vena cava obstruction
5 Jan 2025
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Superior vena cava obstruction
- Spinal cord compression
- B symptoms (fever, weight loss less than 10%, night sweats)
- Tumour lysis syndrome
Linked comparisons
Differentials and adjacent topics worth opening next.
- Leukaemia
- Reactive Lymphadenopathy
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Lymphoma (Adult)
1. Overview
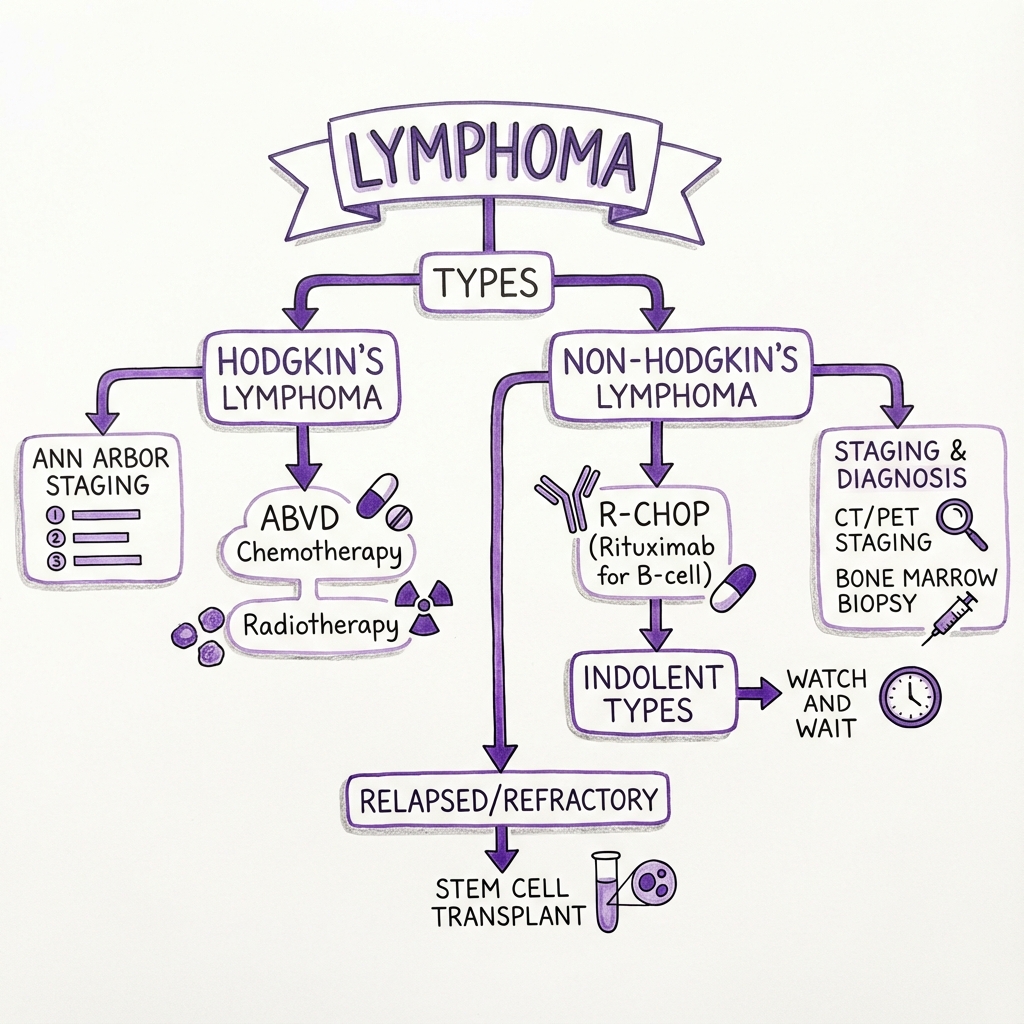
Lymphomas are a heterogeneous group of malignancies arising from lymphoid tissue, representing the fifth most common cancer in developed countries. [1] They are broadly classified into Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL), based on the presence or absence of Reed-Sternberg cells on histology. This fundamental distinction drives profoundly different biological behaviour, staging, treatment approaches, and prognosis. [2]
Hodgkin lymphoma accounts for approximately 10% of all lymphomas and is characterised by the presence of pathognomonic Reed-Sternberg cells within a reactive inflammatory background. It exhibits a bimodal age distribution (peaks at 20-30 years and > 55 years), contiguous lymph node spread, and excellent cure rates exceeding 80% with combination chemotherapy and radiotherapy. [3]
Non-Hodgkin lymphomas are more common, comprising over 90% of lymphomas, and represent a diverse collection of over 60 distinct subtypes with variable aggressiveness. The most common NHL subtype is diffuse large B-cell lymphoma (DLBCL), accounting for 30-40% of cases, followed by follicular lymphoma (20-25%). [4] NHL typically presents with non-contiguous spread and requires immunophenotyping for accurate classification and targeted therapy selection. The introduction of rituximab (anti-CD20 monoclonal antibody) has revolutionised outcomes for B-cell lymphomas. [5]
Early recognition of lymphoma, particularly oncological emergencies such as superior vena cava obstruction, spinal cord compression, and tumour lysis syndrome, is critical for optimal patient outcomes. Distinguishing benign reactive lymphadenopathy from malignant lymphoma and initiating timely biopsy is a core clinical skill across multiple specialties.
2. Epidemiology
Incidence and Prevalence
Lymphoma is the fifth most common cancer in the UK, with approximately 14,000 new cases diagnosed annually (comprising ~4,000 HL and ~10,000 NHL cases). [1] The age-standardised incidence rate is approximately 20 per 100,000 population for NHL and 3 per 100,000 for HL. [6]
| Type | Annual Incidence (UK) | Male:Female Ratio | 5-Year Survival |
|---|---|---|---|
| Hodgkin Lymphoma | ~4,000 cases | 1.3:1 | 85% |
| Non-Hodgkin Lymphoma | ~10,000 cases | 1.2:1 | 68% |
| DLBCL | ~4,500 cases | 1.2:1 | 63% |
| Follicular Lymphoma | ~2,500 cases | 1:1 | 90% (5-year) |
Age Distribution
Hodgkin Lymphoma: Exhibits classical bimodal age distribution with first peak in the third decade (20-30 years, predominantly nodular sclerosing subtype) and second peak after age 55 years (mixed cellularity subtype). [3]
Non-Hodgkin Lymphoma: Incidence increases steadily with age, with median age at diagnosis of 65-70 years for most subtypes. Notable exceptions include:
- Burkitt lymphoma: Children and young adults (median age 30 years)
- Primary mediastinal B-cell lymphoma: Young adults (median age 35 years)
- Mantle cell lymphoma: Older adults (median age 68 years) [4]
Risk Factors
Immunodeficiency States: HIV infection (200-fold increased risk of NHL), iatrogenic immunosuppression (post-transplant lymphoproliferative disorder), and primary immunodeficiencies. [7]
Infectious Agents:
- Epstein-Barr virus (EBV): Associated with endemic Burkitt lymphoma, post-transplant lymphoproliferative disorder, and HL
- Helicobacter pylori: MALT lymphoma of the stomach (70-80% association)
- Human T-lymphotropic virus-1 (HTLV-1): Adult T-cell leukaemia/lymphoma
- Hepatitis C virus: Associated with marginal zone lymphoma and DLBCL [8]
Autoimmune Disorders: Sjögren syndrome (44-fold increased risk of MALT lymphoma), rheumatoid arthritis, systemic lupus erythematosus, and coeliac disease (enteropathy-associated T-cell lymphoma). [9]
Occupational Exposures: Agricultural chemicals, pesticides, herbicides, and organic solvents have been implicated in epidemiological studies, though causative mechanisms remain unclear. [10]
Genetic Predisposition: First-degree relatives of lymphoma patients have 2-3-fold increased risk; specific genetic syndromes include ataxia-telangiectasia, Wiskott-Aldrich syndrome, and X-linked lymphoproliferative syndrome. [11]
Geographic Variation
Hodgkin lymphoma shows higher incidence in developed countries. Endemic Burkitt lymphoma demonstrates geographic clustering in equatorial Africa, linked to EBV infection and malaria co-endemicity. HTLV-1-associated adult T-cell leukaemia/lymphoma is endemic in southwestern Japan, the Caribbean, and parts of Africa. [8]
3. Aetiology & Pathophysiology
Hodgkin Lymphoma
Cellular Origin
Hodgkin lymphoma is characterised by rare malignant Reed-Sternberg cells and their mononuclear variants (Hodgkin cells), which typically represent less than 1% of the tumour mass. These cells are derived from germinal centre or post-germinal centre B cells that have undergone somatic hypermutation and class-switch recombination. [12]
Reed-Sternberg cells are large (> 20 μm), binucleated or multinucleated cells with prominent eosinophilic nucleoli, creating the pathognomonic "owl's eye" appearance. They are surrounded by a reactive inflammatory background comprising T lymphocytes, eosinophils, plasma cells, histiocytes, and fibroblasts. [3]
Immunophenotype
Classical Hodgkin lymphoma Reed-Sternberg cells express:
- CD30 (100%)
- CD15 (75-85%)
- PAX5 (weak/variable)
- Usually negative for CD20, CD45 (LCA)
Nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL) shows distinct features:
- CD20+ (B-cell marker)
- CD30- and CD15-
- "Popcorn cells" rather than classical Reed-Sternberg cells [12]
Pathophysiological Mechanisms
Reed-Sternberg cells evade immune destruction through multiple mechanisms:
- Overexpression of PD-L1 and PD-L2 (immune checkpoint ligands)
- Secretion of cytokines (IL-5, IL-13, TGF-β) that recruit immunosuppressive cells
- Loss of MHC class I/II expression reducing antigen presentation
- Activation of NF-κB, JAK/STAT, and PI3K/AKT signalling pathways promoting survival [13]
Non-Hodgkin Lymphoma
Pathogenesis
NHL arises from malignant transformation of B cells (85%), T cells (12%), or NK cells (3%) at various stages of differentiation. Pathogenesis involves:
-
Chromosomal Translocations:
- t(14;18)(q32;q21): BCL2-IGH fusion in follicular lymphoma (85% cases) causing BCL2 overexpression and apoptosis resistance [14]
- t(11;14)(q13;q32): CCND1-IGH fusion in mantle cell lymphoma causing cyclin D1 overexpression and cell cycle dysregulation
- t(8;14)(q24;q32): MYC-IGH fusion in Burkitt lymphoma causing MYC overexpression and rapid proliferation
-
Oncogene Activation: MYC, BCL2, BCL6 activation through translocations or mutations
-
Tumour Suppressor Loss: TP53, CDKN2A/B deletion or mutation
-
Immune Evasion: Loss of MHC expression, PD-L1 overexpression, immune checkpoint activation
-
Microenvironment Interactions: Malignant cells interact with follicular dendritic cells, T cells, and stromal cells creating a supportive niche [15]
WHO Classification (2016, Revised 2022)
Mature B-Cell Neoplasms (most common):
- Diffuse large B-cell lymphoma (DLBCL), NOS (30-40%)
- Follicular lymphoma (20-25%)
- Marginal zone lymphoma (extranodal MALT, nodal, splenic) (8-10%)
- Mantle cell lymphoma (6%)
- Burkitt lymphoma (2%)
- Primary mediastinal large B-cell lymphoma (2-3%)
- Lymphoplasmacytic lymphoma (Waldenström macroglobulinaemia) (1%)
Mature T-Cell and NK-Cell Neoplasms (10-15%):
- Peripheral T-cell lymphoma, NOS (4%)
- Angioimmunoblastic T-cell lymphoma (2%)
- Anaplastic large cell lymphoma (ALK+ and ALK-) (2%)
- Extranodal NK/T-cell lymphoma, nasal type (1%)
- Adult T-cell leukaemia/lymphoma (endemic regions) [16]
Molecular Subtypes
DLBCL Cell-of-Origin Classification (gene expression profiling):
- Germinal centre B-cell-like (GCB): Better prognosis, 60% 5-year OS
- Activated B-cell-like (ABC): Worse prognosis, 35% 5-year OS
- Unclassified: Intermediate prognosis [17]
Hans algorithm (immunohistochemistry surrogate):
- CD10, BCL6, MUM1 expression patterns predict GCB vs ABC subtypes
Double-Hit and Triple-Hit Lymphomas: Lymphomas with MYC rearrangement plus BCL2 and/or BCL6 rearrangement (5-10% of DLBCL) have particularly aggressive behaviour and poor prognosis with standard R-CHOP, requiring intensified regimens. [18]
4. Clinical Presentation
Symptoms
Lymphadenopathy (75-80% of cases)
Hodgkin Lymphoma: Typically presents with painless, progressive enlargement of cervical or supraclavicular lymph nodes (60-70% of cases). Nodes are firm, rubbery, and mobile. Contiguous spread is characteristic—disease extends from one node group to adjacent groups in an orderly fashion. [3]
Non-Hodgkin Lymphoma: Presents with painless lymphadenopathy in varied distributions depending on subtype. Spread is non-contiguous. Common sites:
- Cervical/supraclavicular (50%)
- Axillary (20%)
- Inguinal (15%)
- Mediastinal (PMBCL, lymphoblastic lymphoma)
- Retroperitoneal (follicular lymphoma) [4]
Alcohol-Induced Pain: Rare (less than 5%) but highly specific symptom for Hodgkin lymphoma—patients report pain in affected lymph nodes within minutes of alcohol consumption. Mechanism unknown. [19]
B Symptoms (30-40% of cases)
Constitutionally defined symptoms indicating advanced disease and adverse prognosis, incorporated into Ann Arbor staging as "B" designation:
| B Symptom | Definition | Clinical Significance |
|---|---|---|
| Fever | Temperature > 38°C, unexplained, recurrent | May show Pel-Ebstein pattern (cyclic weeks of fever followed by afebrile periods) in HL |
| Weight Loss | Unintentional loss > 10% body weight in 6 months | Quantify and document carefully |
| Night Sweats | Drenching sweats requiring change of bedclothes | Must be severe; mild sweating insufficient |
B symptoms are more common in Hodgkin lymphoma (40%) than NHL (20%), and indicate worse prognosis (lower 5-year survival by ~10-15%). Presence of B symptoms upstages disease (e.g., IIA → IIB) and influences treatment intensity. [20]
Pruritus (15-30% in HL)
Generalised itching without rash, often severe and refractory to antihistamines. More common in Hodgkin lymphoma (particularly nodular sclerosis subtype). Not classified as a B symptom but may indicate active disease. Mechanism involves cytokine release (IL-31, IL-5). [21]
Extranodal Symptoms
Gastrointestinal Involvement (10-15% of NHL):
- Abdominal pain, nausea, early satiety (gastric MALT lymphoma)
- Change in bowel habit, bleeding (intestinal lymphoma)
- B symptoms less common in localised GI lymphomas
Bone Marrow Involvement (20-30% of NHL):
- Cytopenias: Anaemia (fatigue, dyspnoea), thrombocytopenia (bruising, bleeding), neutropenia (infections)
- More common in follicular lymphoma (40-50% have BM involvement at diagnosis)
CNS Involvement (2-5% of aggressive NHL):
- Headache, focal neurological deficits, cranial nerve palsies
- High risk in Burkitt lymphoma, lymphoblastic lymphoma, testicular DLBCL
- CSF analysis and MRI brain/spine essential in high-risk cases [22]
Skin Involvement:
- Mycosis fungoides/Sézary syndrome (cutaneous T-cell lymphomas)
- Secondary skin involvement in systemic lymphoma (5%)
Signs
Lymph Node Examination
Character: Firm, rubbery (lymphoma) vs hard (metastatic carcinoma) vs soft, tender (reactive)
Size: Pathological lymphadenopathy typically > 1 cm; > 2 cm warrants urgent investigation
Distribution: Document all involved sites systematically (cervical, supraclavicular, axillary, epitrochlear, inguinal, femoral)
Mobility: Mobile (early disease) vs fixed/matted (advanced disease or surrounding tissue infiltration)
Overlying Skin: Usually normal; erythema/warmth suggests infection; skin involvement indicates aggressive disease
Waldeyer's Ring Examination
Tonsillar enlargement (particularly unilateral) may indicate lymphoma involvement; oropharyngeal examination essential.
Hepatosplenomegaly
- Hepatomegaly (10-15% of cases): Suggests advanced disease (Stage IV)
- Splenomegaly (30-40% of HL, 10-20% of NHL): Palpable spleen suggests splenic involvement; imaging required for confirmation
- Massive splenomegaly: Characteristic of splenic marginal zone lymphoma [23]
Signs of Oncological Emergencies
Superior Vena Cava Obstruction: Facial/neck swelling, dilated chest wall veins, cyanosis, respiratory distress—requires urgent imaging and treatment
Spinal Cord Compression: Back pain, lower limb weakness, sensory level, sphincter dysfunction—MRI spine within 24 hours and urgent oncology/neurosurgery referral
Tumour Lysis Syndrome: Acute kidney injury, hyperkalaemia, hyperphosphataemia, hypocalcaemia—preventable with hydration and allopurinol/rasburicase prophylaxis
5. Differential Diagnosis
Lymphadenopathy has a broad differential diagnosis; systematic evaluation is essential to avoid missing malignancy or serious infection.
| Differential | Key Distinguishing Features |
|---|---|
| Reactive Lymphadenopathy | Tender, soft, mobile nodes; associated infection (URTI, mononucleosis); resolves within 4-6 weeks |
| Infectious Mononucleosis (EBV) | Bilateral cervical lymphadenopathy, fever, pharyngitis, atypical lymphocytes on blood film, positive Monospot test |
| Tuberculosis | Chronic, painless, matted cervical nodes; constitutional symptoms; positive interferon-gamma release assay (IGRA); caseating granulomas on biopsy |
| Metastatic Carcinoma | Hard, fixed nodes; primary malignancy evident (lung, breast, GI); older age group |
| Chronic Lymphocytic Leukaemia (CLL) | Lymphocytosis > 5×10⁹/L, generalised lymphadenopathy, older patients (median age 72), smudge cells on film |
| Acute Leukaemia | Cytopenias, circulating blasts, bone marrow failure symptoms (bleeding, infection, anaemia) |
| Sarcoidosis | Bilateral hilar lymphadenopathy (BHL), erythema nodosum, uveitis, hypercalcaemia; non-caseating granulomas |
| Kikuchi-Fujimoto Disease | Young women, cervical lymphadenopathy, fever; benign self-limiting; histiocytic necrotising lymphadenitis on biopsy |
| Castleman Disease | Unicentric (localised mass) or multicentric (systemic symptoms, HHV-8 association in HIV); characteristic histology |
Red Flags Warranting Urgent Investigation
- Lymph nodes > 2 cm persisting > 4-6 weeks
- Supraclavicular lymphadenopathy (Virchow's node)
- Hard, fixed, or rapidly enlarging nodes
- Associated B symptoms
- Unexplained splenomegaly
- Generalised lymphadenopathy with cytopenias
NICE Guidelines (NG12): Refer urgently (suspected cancer pathway, appointment within 2 weeks) for unexplained lymphadenopathy. [24]
6. Investigations
First-Line Investigations
Complete Blood Count (FBC)
- Anaemia: Normocytic normochromic anaemia of chronic disease (30-40%); autoimmune haemolytic anaemia (5% of HL, positive DAT)
- Thrombocytopenia: Bone marrow involvement or immune-mediated destruction
- Neutropenia: Bone marrow involvement or chemotherapy effect
- Lymphocytosis: May suggest CLL rather than lymphoma; circulating lymphoma cells rare except in leukaemic phase (mantle cell, Burkitt)
- Eosinophilia: Classic association with Hodgkin lymphoma (10-15% of cases) [25]
Biochemistry
- Lactate Dehydrogenase (LDH): Elevated in aggressive lymphomas (Burkitt, DLBCL); reflects tumour burden and cell turnover; independent adverse prognostic factor
- Albumin: Low albumin indicates advanced disease and poor prognosis
- Uric Acid: Elevated in high-grade lymphomas; risk factor for tumour lysis syndrome
- Renal Function: Baseline essential; may be impaired by tumour infiltration, hypercalcaemia, or urate nephropathy
- Liver Function Tests: Hepatic involvement (elevated ALP, GGT); hyperbilirubinaemia may indicate bile duct compression
- Calcium: Hypercalcaemia in aggressive NHL (Burkitt, DLBCL) or bony involvement
- Beta-2 Microglobulin: Prognostic marker; elevated in aggressive disease [26]
Inflammatory Markers
- ESR: Elevated in Hodgkin lymphoma (included in Hasenclever prognostic index)
- CRP: Elevated in aggressive disease; non-specific but useful for monitoring
Serology
- HIV Test: Mandatory in all lymphoma patients (consent required); HIV-associated lymphomas are aggressive and require modified treatment
- Hepatitis B and C Serology: Hepatitis B reactivation risk with rituximab and chemotherapy; prophylactic antivirals required if HBsAg or anti-HBc positive
- EBV Serology: May be useful in suspected EBV-associated lymphomas
Diagnostic Investigations
Lymph Node Biopsy (Gold Standard)
Excisional Biopsy (preferred):
- Entire lymph node removed (> 1 cm preferred)
- Preserves architecture essential for diagnosis
- Superior to core biopsy or FNA for initial diagnosis
- Allows comprehensive histology, immunohistochemistry, flow cytometry, and molecular studies [27]
Core Needle Biopsy:
- Acceptable alternative if excisional biopsy not feasible
- Multiple cores (4-6) required
- Useful for deep-seated nodes (retroperitoneal, mediastinal) under CT or ultrasound guidance
Fine Needle Aspiration (FNA):
- NOT recommended for initial lymphoma diagnosis—insufficient material for subtyping
- May be useful for confirming relapse in patients with known lymphoma
Histopathological Analysis
Hodgkin Lymphoma:
- Presence of Reed-Sternberg cells (large binucleated cells with prominent nucleoli)
- Reactive inflammatory background (T cells, eosinophils, histiocytes)
- Immunophenotype: CD30+, CD15+, PAX5 (weak), CD20- (usually), CD45-
- Subclassification: Nodular sclerosis (70%), mixed cellularity (20%), lymphocyte-rich (5%), lymphocyte-depleted (5%)
- NLPHL: "Popcorn cells," CD20+, CD15-, CD30- [12]
Non-Hodgkin Lymphoma:
- Architecture: Follicular vs diffuse growth pattern
- Cell morphology: Small vs large cell; cleaved vs non-cleaved nuclei
- Immunophenotyping (essential): CD20, CD3, CD5, CD10, BCL2, BCL6, MUM1, Ki-67
- Flow cytometry: Clonality (kappa/lambda light chain restriction)
- Molecular studies:
- "FISH for translocations: t(14;18) BCL2, t(11;14) CCND1, t(8;14) MYC"
- MYC/BCL2/BCL6 rearrangements (double/triple-hit status in DLBCL)
- IgH gene rearrangement (clonality confirmation) [16]
Staging Investigations
CT Chest, Abdomen, Pelvis (CT CAP)
- Contrast-Enhanced CT: Standard staging modality for all lymphomas
- Assesses:
- Lymph node involvement (cervical, mediastinal, axillary, abdominal, pelvic, inguinal)
- Extranodal disease (liver, spleen, lung, kidneys, bone)
- Complications (SVC obstruction, ureteric obstruction, bowel obstruction)
- Measurable disease documented for response assessment (RECIST criteria)
PET-CT (FDG-PET)
- Standard for Hodgkin Lymphoma and Aggressive NHL: Superior to CT alone for staging and response assessment
- Advantages:
- Detects metabolically active disease (higher sensitivity than CT)
- Differentiates active disease from residual masses post-treatment
- Interim PET after 2-4 cycles chemotherapy guides treatment adaptation (Deauville criteria)
- End-of-treatment PET determines complete metabolic response [28]
- Deauville 5-Point Scale (PET response assessment):
- "Score 1-3: Negative (complete metabolic response)"
- "Score 4: Probably positive"
- "Score 5: Positive (active disease)"
- Not routinely used: Indolent lymphomas (follicular, marginal zone) due to low FDG avidity
Bone Marrow Biopsy
Indications:
- Staging of NHL (particularly follicular, mantle cell, marginal zone)
- Unexplained cytopenias
- Not routinely required in Hodgkin lymphoma if PET-CT negative [29]
Technique:
- Posterior iliac crest aspiration and trephine biopsy
- Immunohistochemistry and flow cytometry if infiltration suspected
Lumbar Puncture (CSF Analysis)
Indications (CNS prophylaxis/staging):
- Burkitt lymphoma (mandatory)
- Lymphoblastic lymphoma (mandatory)
- DLBCL with high-risk features:
- Testicular involvement
- Paranasal sinus involvement
- Breast involvement
- Epidural involvement
-
1 extranodal site + elevated LDH
- High IPI score (4-5) [22]
CSF Studies:
- Cell count, cytology, flow cytometry, protein, glucose
- Positive if malignant cells identified or lymphocytosis with clonal population
Additional Investigations
Echocardiogram/MUGA Scan: Baseline cardiac function assessment before anthracycline chemotherapy (doxorubicin cardiotoxicity risk)
Pulmonary Function Tests: Baseline before bleomycin (pulmonary fibrosis risk)
Fertility Counselling: Sperm banking/oocyte preservation before gonadotoxic chemotherapy in patients of reproductive age
Virology Screening: HIV, hepatitis B/C (mandatory before rituximab or chemotherapy)
7. Staging (Ann Arbor Classification)
The Ann Arbor staging system (Cotswolds modification) is used for both Hodgkin and non-Hodgkin lymphomas. Stage is determined by anatomical extent of disease and presence/absence of B symptoms.
| Stage | Definition | Substage |
|---|---|---|
| I | Single lymph node region or single extranodal site (IE) | |
| II | ≥2 lymph node regions on same side of diaphragm | IIE: Contiguous extranodal involvement |
| III | Lymph node regions on both sides of diaphragm | III₁: Splenic, hilar, coeliac, portal nodes III₂: Para-aortic, iliac, mesenteric nodes |
| IV | Disseminated/multifocal extranodal involvement | Liver, bone marrow, lung (non-contiguous), CNS, bone |
Modifiers:
- A: Absence of B symptoms
- B: Presence of B symptoms (fever > 38°C, unintentional weight loss > 10% in 6 months, drenching night sweats)
- E: Contiguous extranodal extension from nodal site
- X: Bulky disease (> 10 cm mass or mediastinal mass >⅓ thoracic diameter on CXR)
- S: Splenic involvement
Examples:
- Stage IIA: Two lymph node regions same side of diaphragm, no B symptoms
- Stage IIIB: Lymph nodes both sides of diaphragm with B symptoms
- Stage IVB: Bone marrow involvement with B symptoms [30]
Prognostic Scores
International Prognostic Score (IPS) for Hodgkin Lymphoma
Seven adverse factors (1 point each):
- Albumin less than 40 g/L
- Haemoglobin less than 105 g/L
- Male sex
- Age ≥45 years
- Stage IV disease
- White cell count ≥15×10⁹/L
- Lymphocyte count less than 0.6×10⁹/L or less than 8% of WCC
Interpretation:
- Score 0-2: 5-year freedom from progression (FFP) ~80%
- Score 3-4: 5-year FFP ~60%
- Score ≥5: 5-year FFP ~40% [31]
International Prognostic Index (IPI) for Aggressive NHL (DLBCL)
Five adverse factors (1 point each):
- Age > 60 years
- Stage III or IV disease
- Elevated serum LDH (above upper limit of normal)
- ECOG performance status ≥2
-
1 extranodal site
Interpretation (in rituximab era, R-IPI):
- Low risk (0-1): 5-year OS ~90%
- Low-intermediate risk (2): 5-year OS ~80%
- High-intermediate risk (3): 5-year OS ~60%
- High risk (4-5): 5-year OS ~50% [32]
Follicular Lymphoma International Prognostic Index (FLIPI)
Five adverse factors (1 point each):
- Age > 60 years
- Stage III-IV
- Haemoglobin less than 120 g/L
- Elevated serum LDH
-
4 nodal areas involved
Interpretation:
- Low risk (0-1): 10-year OS ~70%
- Intermediate risk (2): 10-year OS ~50%
- High risk (≥3): 10-year OS ~35% [33]
8. Management
Hodgkin Lymphoma
Early-Stage Favourable (IA, IIA without risk factors)
Standard Treatment:
- ABVD × 2 cycles + Involved-Field Radiotherapy (IFRT) 20 Gy [34]
ABVD Regimen (administered every 14 days):
- Adriamycin (doxorubicin) 25 mg/m² IV day 1, 15
- Bleomycin 10 units/m² IV day 1, 15
- Vinblastine 6 mg/m² IV day 1, 15
- Dacarbazine 375 mg/m² IV day 1, 15
Risk Factors (determine "favourable" vs "unfavourable"):
- Bulky mediastinal mass (>⅓ thoracic diameter)
- Age ≥50 years
- Elevated ESR (≥50 without B symptoms, ≥30 with B symptoms)
- ≥3 nodal areas involved
Early-Stage Unfavourable (IA, IIA with risk factors, IIB)
Standard Treatment:
- ABVD × 4 cycles + IFRT 30 Gy [34]
Alternative (selected patients):
- BEACOPP (Bleomycin, Etoposide, Adriamycin, Cyclophosphamide, Oncovin, Procarbazine, Prednisolone): More intensive; used in some European centres for unfavourable early-stage disease
Advanced-Stage (IIB with bulky disease, III, IV)
Standard Treatment:
- ABVD × 6 cycles [35]
- PET-Guided Approach (RATHL trial): Interim PET after 2 cycles:
- "PET-negative (Deauville 1-3): Continue ABVD or AVD (omit bleomycin to reduce pulmonary toxicity)"
- "PET-positive (Deauville 4-5): Escalate to BEACOPP or BrECADD"
Escalated BEACOPP (for high-risk disease):
- More intensive regimen with higher cure rates but greater toxicity
- Used in young patients with high IPS scores or PET-positive interim scans
- 5-year FFP ~90% vs 85% with ABVD [36]
Radiotherapy Consolidation:
- Considered for PET-positive residual masses after chemotherapy
- IFRT 30-36 Gy to residual disease
Relapsed/Refractory Hodgkin Lymphoma
Second-Line Chemotherapy:
- ICE (Ifosfamide, Carboplatin, Etoposide)
- DHAP (Dexamethasone, High-dose Ara-C, Platinum)
- GDP (Gemcitabine, Dexamethasone, Platinum)
Autologous Stem Cell Transplantation (ASCT):
- Standard of care for relapsed/refractory HL with chemosensitive disease
- High-dose chemotherapy (BEAM: BCNU, Etoposide, Ara-C, Melphalan) followed by stem cell rescue
- 5-year OS ~60-70% [37]
Novel Agents:
- Brentuximab Vedotin (anti-CD30 antibody-drug conjugate): Response rate ~75% in relapsed/refractory HL; approved post-ASCT or in ASCT-ineligible patients [38]
- PD-1 Inhibitors (Nivolumab, Pembrolizumab): Response rate ~65-70%; particularly effective in classical HL due to PD-L1 amplification (9p24.1) [39]
Non-Hodgkin Lymphoma
Diffuse Large B-Cell Lymphoma (DLBCL)
Standard First-Line Treatment:
- R-CHOP × 6 cycles (administered every 21 days) [40]
R-CHOP Regimen:
- Rituximab 375 mg/m² IV day 1
- Cyclophosphamide 750 mg/m² IV day 1
- Hydroxydaunorubicin (Doxorubicin) 50 mg/m² IV day 1
- Oncovin (Vincristine) 1.4 mg/m² IV day 1 (max 2 mg)
- Prednisolone 100 mg PO days 1-5
CNS Prophylaxis (intrathecal methotrexate or high-dose systemic methotrexate):
- Testicular, paranasal sinus, breast, epidural DLBCL
-
1 extranodal site + elevated LDH
- High IPI score (4-5) [22]
Consolidation Radiotherapy:
- IFRT to initial bulky sites (> 10 cm) or PET-positive residual masses
- 30-36 Gy
Limited-Stage DLBCL (Stage I-II):
- R-CHOP × 3-4 cycles + IFRT 30-40 Gy (preferred)
- R-CHOP × 6 cycles alone (alternative if radiotherapy contraindicated)
Advanced-Stage DLBCL (Stage III-IV):
- R-CHOP × 6 cycles (standard)
- Interim PET after 3-4 cycles guides prognosis but not routinely treatment modification
Double-Hit/Triple-Hit Lymphomas:
- R-CHOP inadequate (5-year OS ~30%)
- Intensified regimens: DA-EPOCH-R (dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, rituximab) [18]
Relapsed/Refractory DLBCL
Second-Line Chemotherapy (salvage):
- R-ICE, R-DHAP, R-GDP (rituximab + platinum-based regimens)
Autologous Stem Cell Transplantation:
- Standard of care for chemosensitive relapsed DLBCL
- 3-year OS ~50% [41]
CAR-T Cell Therapy:
- Axicabtagene Ciloleucel (Axi-cel) and Tisagenlecleucel (Tisa-cel): Anti-CD19 CAR-T cell therapies
- Approved for relapsed/refractory DLBCL after ≥2 prior lines
- Response rate ~50-80%; durable remissions in 30-40%
- Toxicities: Cytokine release syndrome (CRS), immune effector cell-associated neurotoxicity syndrome (ICANS) [42]
Bispecific Antibodies:
- Glofitamab (CD20×CD3 bispecific): Response rate ~50% in heavily pre-treated DLBCL [43]
Follicular Lymphoma (Indolent NHL)
Management Strategy: Risk-adapted; observation vs treatment based on tumour burden and symptoms.
Watch-and-Wait (Asymptomatic, Low Tumour Burden):
- Observation without immediate treatment
- Median time to treatment ~3 years
- No survival disadvantage compared to early treatment [44]
Indications for Treatment (GELF/BNLI Criteria):
- Symptomatic disease (B symptoms, pruritus)
- Bulky disease (> 7 cm)
- Rapidly progressive disease
- Cytopenias (Hb less than 100 g/L, platelets less than 100×10⁹/L)
- Threatened end-organ function (ureteric obstruction, spinal cord compression)
First-Line Treatment (Limited Stage I-II):
- Involved-Site Radiotherapy (ISRT) 24-30 Gy: Potentially curative (20-30% long-term disease-free survival)
First-Line Treatment (Advanced Stage III-IV or Symptomatic):
- Bendamustine-Rituximab (BR) × 6 cycles: Preferred in elderly/frail patients; lower toxicity [45]
- R-CVPP (Rituximab, Cyclophosphamide, Vinblastine, Procarbazine, Prednisolone)
- R-Chlorambucil: Older, less fit patients
Maintenance Rituximab:
- Rituximab 375 mg/m² every 2-3 months for 2 years post-induction
- Prolongs progression-free survival (PFS) by ~4 years
- Recommended after chemotherapy induction [46]
Relapsed Follicular Lymphoma:
- Repeat rituximab-based regimen (if first remission > 2 years)
- Obinutuzumab (anti-CD20 glycoengineered antibody) + chemotherapy
- Lenalidomide-Rituximab (oral immunomodulatory agent)
- Radioimmunotherapy: Yttrium-90 or Iodine-131-labelled anti-CD20 antibodies (reserved for specific cases)
- Autologous SCT: Considered in young, fit patients with early relapse
Transformation to Aggressive Lymphoma (Richter transformation):
- Occurs in 2-3% per year (cumulative 30% at 10 years)
- Suspected if: Rapid lymph node enlargement, B symptoms, elevated LDH, PET-avid lesion (SUV > 10)
- Requires re-biopsy
- Treated as DLBCL (R-CHOP); poor prognosis (median OS ~1 year) [47]
Mantle Cell Lymphoma
First-Line Treatment (Fit Patients less than 65 years):
- Intensive induction: R-HyperCVAD (Rituximab, Cyclophosphamide, Vincristine, Doxorubicin, Dexamethasone alternating with Methotrexate, Cytarabine) or Nordic protocol
- Autologous SCT consolidation in first remission
- Rituximab maintenance for 3 years: Prolongs PFS [48]
First-Line Treatment (Older/Unfit Patients):
- Bendamustine-Rituximab (BR) × 6 cycles + rituximab maintenance
Novel Agents:
- Ibrutinib (Bruton tyrosine kinase inhibitor): Response rate ~65% in relapsed/refractory MCL; approved first-line in combination with rituximab [49]
- Acalabrutinib (second-generation BTK inhibitor): Better tolerability
- Venetoclax (BCL2 inhibitor): Active in relapsed MCL
Burkitt Lymphoma
Treatment: Intensive multi-agent chemotherapy with CNS prophylaxis
- CODOX-M/IVAC (Cyclophosphamide, Vincristine, Doxorubicin, Methotrexate alternating with Ifosfamide, Etoposide, Cytarabine)
- Hyper-CVAD + Rituximab
- Intrathecal chemotherapy (methotrexate, cytarabine) with every cycle
- 5-year OS ~70-80% in adults [50]
Tumour Lysis Syndrome Prophylaxis (mandatory):
- Aggressive hydration (3 L/m²/day)
- Allopurinol 300 mg daily (or rasburicase 0.2 mg/kg if high risk)
- Monitor electrolytes (K⁺, PO₄³⁻, Ca²⁺), uric acid, creatinine every 6-12 hours
Supportive Care
Infection Prophylaxis
- Pneumocystis jirovecii Prophylaxis: Co-trimoxazole 960 mg three times weekly during and for 6 months post-chemotherapy (particularly with fludarabine, bendamustine, or steroids)
- Antiviral Prophylaxis: Aciclovir 400 mg BD if herpes zoster risk or history
- Antifungal Prophylaxis: Fluconazole if prolonged neutropenia expected
- Hepatitis B Reactivation Prophylaxis: Lamivudine or entecavir if HBsAg+ or anti-HBc+ receiving rituximab [51]
Haemopoietic Growth Factors
- G-CSF (Filgrastim, Pegfilgrastim): Primary prophylaxis if febrile neutropenia risk > 20% (BEACOPP, dose-dense regimens)
Antiemetics
- 5-HT3 Antagonists (Ondansetron 8 mg TDS) + NK1 Antagonists (Aprepitant) + Dexamethasone for highly emetogenic chemotherapy (ABVD, R-CHOP)
Fertility Preservation
- Sperm Banking: Offered to all male patients pre-chemotherapy
- Oocyte/Embryo Cryopreservation: Offered to female patients if feasible (may delay treatment 2-3 weeks)
- GnRH Agonists: Limited evidence for ovarian protection
9. Complications
Treatment-Related Complications
| Complication | Causative Agent | Frequency | Prevention | Management |
|---|---|---|---|---|
| Febrile Neutropenia | Chemotherapy (all regimens) | 10-20% | G-CSF prophylaxis | Immediate broad-spectrum antibiotics (piperacillin-tazobactam) |
| Cardiotoxicity | Doxorubicin (anthracyclines) | 5-10% (cumulative) | Baseline echo; limit cumulative dose less than 450 mg/m² | ACE inhibitors, beta-blockers |
| Pulmonary Fibrosis | Bleomycin | 5-10% | Monitor PFTs; omit if PET-negative interim | Corticosteroids; avoid supplemental O₂ unless essential |
| Peripheral Neuropathy | Vincristine | 20-30% | Dose reduction if grade ≥2 | Duloxetine, gabapentin; vincristine cessation |
| Infusion Reactions | Rituximab | 30-40% (first infusion) | Premedication (paracetamol, antihistamine) | Slow/stop infusion; IV hydrocortisone, antihistamines |
| Tumour Lysis Syndrome | Burkitt, DLBCL, high tumour burden | 5-10% (high-risk) | Hydration, allopurinol/rasburicase | IV fluids, rasburicase, electrolyte correction, renal replacement therapy |
| Hepatitis B Reactivation | Rituximab, chemotherapy | 20-50% (if HBsAg+) | Antiviral prophylaxis (lamivudine) | Antiviral therapy, pause chemotherapy |
| Secondary Malignancies | Chemotherapy, radiotherapy | 5-15% (10-20 years) | Minimise radiotherapy fields | Age-appropriate cancer screening |
Disease-Related Complications
Superior Vena Cava Obstruction (2-3% of cases):
- Mediastinal lymphoma (PMBCL, lymphoblastic, DLBCL)
- Facial/neck swelling, dyspnoea, chest wall venous distension
- Urgent CT chest, tissue diagnosis (biopsy if not previously confirmed), chemotherapy or radiotherapy
Spinal Cord Compression (1-2% of cases):
- Back pain, lower limb weakness, sensory level, bowel/bladder dysfunction
- MRI spine within 24 hours
- High-dose dexamethasone 16 mg daily, urgent radiotherapy or surgical decompression [52]
Hyperviscosity Syndrome (rare in lymphoma):
- Waldenström macroglobulinaemia (lymphoplasmacytic lymphoma)
- Blurred vision, headache, bleeding, confusion
- Urgent plasmapheresis
Autoimmune Cytopenias (5-10%):
- Autoimmune haemolytic anaemia (AIHA): Positive DAT, spherocytes, reticulocytosis; treat with corticosteroids ± rituximab
- Immune thrombocytopenia (ITP): Isolated thrombocytopenia; treat underlying lymphoma
10. Prognosis
Hodgkin Lymphoma
Hodgkin lymphoma is one of the most curable cancers, with overall cure rates exceeding 80%. [3]
| Stage | 5-Year Overall Survival (OS) |
|---|---|
| Early-stage favourable (IA, IIA) | 95% |
| Early-stage unfavourable | 90% |
| Advanced-stage (III-IV) | 80-85% |
Adverse Prognostic Factors (International Prognostic Score):
- High IPS score (≥4) associated with 5-year FFP ~40-50%
- Bulky mediastinal disease
- B symptoms
- PET-positive interim scan (Deauville 4-5) after 2 cycles
Long-Term Survivorship Issues:
- Secondary malignancies (breast, lung, thyroid cancer) due to radiotherapy/chemotherapy exposure—10-15% risk at 20 years [53]
- Cardiovascular disease (coronary artery disease, heart failure) due to mediastinal radiotherapy and anthracyclines
- Infertility (particularly with BEACOPP)
- Hypothyroidism (neck radiotherapy)
Follow-Up:
- Clinical review every 3 months for 2 years, then 6-monthly to 5 years, then annually
- Annual thyroid function tests if neck radiotherapy
- Cardiovascular risk assessment and management
- Age-appropriate cancer screening (mammography if chest radiotherapy in women)
Non-Hodgkin Lymphoma
Prognosis varies widely based on histological subtype, stage, IPI score, and molecular features.
Diffuse Large B-Cell Lymphoma (DLBCL)
| IPI Score | Risk Category | 5-Year OS (R-CHOP Era) |
|---|---|---|
| 0-1 | Low | 90% |
| 2 | Low-Intermediate | 80% |
| 3 | High-Intermediate | 65% |
| 4-5 | High | 50% |
Cell-of-Origin Impact:
- GCB subtype: 5-year OS ~75%
- ABC subtype: 5-year OS ~55%
- Double/triple-hit lymphomas: 5-year OS ~30% with R-CHOP (improved with DA-EPOCH-R) [17,18]
Follicular Lymphoma
Follicular lymphoma is indolent but incurable. Median overall survival is 15-20 years with modern treatment (rituximab era). [44]
| FLIPI Score | Risk Category | 10-Year OS |
|---|---|---|
| 0-1 | Low | 70% |
| 2 | Intermediate | 50% |
| ≥3 | High | 35% |
Disease Course:
- Relapsing-remitting pattern; median time to first treatment ~3 years
- Median PFS after first-line rituximab-based therapy ~4-5 years
- Cumulative risk of transformation to aggressive lymphoma (DLBCL) ~30% at 10 years (poor prognosis) [47]
Mantle Cell Lymphoma
Aggressive yet partially chemosensitive; median OS ~5-7 years. [48]
- Intensive therapy + ASCT in young patients: Median OS ~10 years
- Older patients (BR + rituximab maintenance): Median PFS ~4 years
Burkitt Lymphoma
Highly aggressive but potentially curable with intensive chemotherapy.
- 5-year OS ~70-80% in adults with modern regimens (CODOX-M/IVAC, Hyper-CVAD-R)
- Prognosis worse in older patients (> 60 years) and HIV-associated disease [50]
11. Key Guidelines
-
NICE NG52 (2016): Non-Hodgkin's lymphoma: diagnosis and management. Recommendations on investigation, staging, and treatment pathways. [24]
-
ESMO Clinical Practice Guidelines:
- Hodgkin Lymphoma (Eichenauer et al., 2018): Evidence-based recommendations for diagnosis, staging, treatment, and follow-up. [34]
- Diffuse Large B-Cell Lymphoma (Tilly et al., 2015): First-line and relapsed/refractory DLBCL management. [40]
-
NCCN Guidelines: Comprehensive algorithms for all lymphoma subtypes, updated annually.
-
British Society for Haematology (BSH) Guidelines: UK-specific recommendations on lymphoma management.
-
Lugano Classification (2014): Updated staging and response criteria for lymphomas, incorporating PET-CT. [28]
12. Common Exam Questions
MRCP/FRACP Style Questions
-
"A 25-year-old presents with painless cervical lymphadenopathy and alcohol-induced pain in the neck. What is the most likely diagnosis?"
- Answer: Hodgkin lymphoma (alcohol-induced pain is rare but highly specific)
-
"What is the most common subtype of non-Hodgkin lymphoma in adults?"
- Answer: Diffuse large B-cell lymphoma (30-40% of NHL)
-
"A patient with DLBCL has testicular involvement. What additional investigation is mandatory?"
- Answer: Lumbar puncture (CSF analysis) for CNS staging—testicular DLBCL has high CNS relapse risk
-
"What is the immunophenotype of Reed-Sternberg cells in classical Hodgkin lymphoma?"
- Answer: CD30+, CD15+, CD20- (usually), CD45-
-
"Which chromosomal translocation is characteristic of follicular lymphoma?"
- Answer: t(14;18) BCL2-IGH translocation (85% of cases)
-
"What is the first-line treatment for advanced-stage Hodgkin lymphoma?"
- Answer: ABVD chemotherapy × 6 cycles
-
"A patient with Burkitt lymphoma is about to start chemotherapy. What is the most important preventive measure?"
- Answer: Tumour lysis syndrome prophylaxis (aggressive hydration, allopurinol/rasburicase, electrolyte monitoring)
-
"What is the role of rituximab in B-cell lymphomas?"
- Answer: Anti-CD20 monoclonal antibody; standard component of treatment for CD20+ B-cell lymphomas (DLBCL, follicular lymphoma, mantle cell lymphoma)
Viva Points
Opening Statement for Hodgkin Lymphoma: "Hodgkin lymphoma is a lymphoid malignancy characterised by the presence of Reed-Sternberg cells on histology, accounting for approximately 10% of lymphomas. It exhibits a bimodal age distribution with peaks at 20-30 years and > 55 years. The disease typically presents with painless, progressive cervical or supraclavicular lymphadenopathy and demonstrates contiguous spread through lymph node chains. The immunophenotype is CD30+, CD15+, with weak PAX5 expression. First-line treatment is combination chemotherapy with ABVD, with or without radiotherapy depending on stage. Hodgkin lymphoma is highly curable, with 5-year survival exceeding 80-85% across all stages."
Opening Statement for Non-Hodgkin Lymphoma: "Non-Hodgkin lymphomas are a heterogeneous group of lymphoid malignancies comprising over 60 distinct subtypes, classified based on cell of origin (B-cell, T-cell, NK-cell), morphology, immunophenotype, and molecular features. The most common subtype is diffuse large B-cell lymphoma, accounting for 30-40% of cases, which is aggressive but curable with R-CHOP chemotherapy (rituximab, cyclophosphamide, doxorubicin, vincristine, prednisolone). NHL demonstrates non-contiguous spread, in contrast to Hodgkin lymphoma. Prognosis varies widely by subtype—aggressive lymphomas like DLBCL are potentially curable, whereas indolent lymphomas like follicular lymphoma are incurable but have prolonged survival measured in decades."
Common Mistakes
❌ Using FNA for initial lymphoma diagnosis: Fine needle aspiration provides insufficient material for subtyping; excisional lymph node biopsy is required.
❌ Missing CNS prophylaxis in high-risk DLBCL: Testicular, paranasal sinus, breast, and epidural DLBCL require intrathecal chemotherapy or high-dose systemic methotrexate to prevent CNS relapse.
❌ Treating follicular lymphoma immediately without indication: Asymptomatic, low-burden follicular lymphoma should be observed ("watch-and-wait") with no survival disadvantage compared to early treatment.
❌ Forgetting hepatitis B screening before rituximab: Rituximab causes HBV reactivation in 20-50% of HBsAg+ patients; antiviral prophylaxis is mandatory.
❌ Missing tumour lysis syndrome prophylaxis in Burkitt lymphoma: Burkitt lymphoma has extremely high cell turnover; aggressive hydration and allopurinol/rasburicase are mandatory before chemotherapy.
❌ Assuming all lymphomas are treated the same: Lymphoma subtypes have vastly different biology and treatment—DLBCL requires intensive chemotherapy (R-CHOP), whereas follicular lymphoma may not require immediate treatment.
13. Patient/Layperson Explanation
What is Lymphoma?
Lymphoma is a type of cancer that starts in the lymphatic system—the network of glands (lymph nodes) and vessels that helps your body fight infections. White blood cells called lymphocytes become abnormal and grow out of control, forming lumps in lymph nodes or other organs.
Two Main Types
Hodgkin Lymphoma:
- Less common (about 10% of lymphomas)
- Usually very curable—more than 8 out of 10 people are cured
- More common in young adults and older people
- Diagnosed by finding specific abnormal cells (Reed-Sternberg cells) under a microscope
Non-Hodgkin Lymphoma:
- More common (about 90% of lymphomas)
- Many different subtypes with different behaviours
- Some types are aggressive but curable; others grow slowly and can be managed for many years
- Treatment depends on the specific type
Symptoms
The most common symptom is painless swelling of lymph nodes in the neck, armpit, or groin. These lumps don't go away and may slowly get bigger.
Other symptoms may include:
- Unexplained fevers
- Night sweats (soaking the bedsheets)
- Weight loss (losing more than 10% of your body weight without trying)
- Tiredness
- Itching all over the body
Diagnosis
To diagnose lymphoma, a doctor will:
- Remove a swollen lymph node (biopsy) for examination under a microscope
- CT or PET scans to see where the lymphoma has spread
- Blood tests to check your general health
Treatment
Hodgkin Lymphoma:
- Chemotherapy (usually a combination called ABVD)
- Sometimes radiotherapy (focused beams to kill cancer cells)
- Most people are cured with treatment
Non-Hodgkin Lymphoma:
- Depends on the type:
- "Aggressive types (like DLBCL): Chemotherapy with rituximab (R-CHOP regimen)—potentially curable"
- "Slow-growing types (like follicular lymphoma): May not need immediate treatment; can be watched closely and treated when symptoms develop"
Side Effects
Chemotherapy can cause:
- Hair loss (temporary—hair grows back after treatment)
- Nausea and vomiting (controlled with anti-sickness medicines)
- Increased infection risk (antibiotics given if needed)
- Tiredness
Outlook
- Hodgkin lymphoma: More than 80% of people are cured
- Aggressive non-Hodgkin lymphomas: 60-70% are cured with chemotherapy
- Slow-growing non-Hodgkin lymphomas: Not curable but people often live for 15-20 years or more with treatment
Support
- Lymphoma Action (UK charity): lymphoma-action.org.uk
- Macmillan Cancer Support: macmillan.org.uk
- Your hospital team will provide information and support throughout treatment
14. References
-
Cancer Research UK. Non-Hodgkin lymphoma statistics. Available at: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/non-hodgkin-lymphoma
-
Shankland KR, Armitage JO, Hancock BW. Non-Hodgkin lymphoma. Lancet. 2012;380(9844):848-857. doi:10.1016/S0140-6736(12)60605-9
-
Ansell SM. Hodgkin lymphoma: 2023 update on diagnosis, risk-stratification, and management. Am J Hematol. 2022;97(11):1478-1488. doi:10.1002/ajh.26717
-
Armitage JO, Gascoyne RD, Lunning MA, Cavalli F. Non-Hodgkin lymphoma. Lancet. 2017;390(10091):298-310. doi:10.1016/S0140-6736(16)32407-2
-
Coiffier B, Lepage E, Briere J, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med. 2002;346(4):235-242. doi:10.1056/NEJMoa011795
-
Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2018. National Cancer Institute. Bethesda, MD.
-
Shiels MS, Engels EA. Evolving epidemiology of HIV-associated malignancies. Curr Opin HIV AIDS. 2017;12(1):6-11. doi:10.1097/COH.0000000000000327
-
Shannon-Lowe C, Rickinson AB, Bell AI. Epstein-Barr virus-associated lymphomas. Philos Trans R Soc Lond B Biol Sci. 2017;372(1732):20160271. doi:10.1098/rstb.2016.0271
-
Zintzaras E, Voulgarelis M, Moutsopoulos HM. The risk of lymphoma development in autoimmune diseases: a meta-analysis. Arch Intern Med. 2005;165(20):2337-2344. doi:10.1001/archinte.165.20.2337
-
Alexander DD, Mink PJ, Adami HO, et al. The non-Hodgkin lymphomas: a review of the epidemiologic literature. Int J Cancer. 2007;120 Suppl 12:1-39. doi:10.1002/ijc.22719
-
Cerhan JR, Slager SL. Familial predisposition and genetic risk factors for lymphoma. Blood. 2015;126(20):2265-2273. doi:10.1182/blood-2015-04-537498
-
Küppers R, Rajewsky K, Zhao M, et al. Hodgkin disease: Hodgkin and Reed-Sternberg cells picked from histological sections show clonal immunoglobulin gene rearrangements and appear to be derived from B cells at various stages of development. Proc Natl Acad Sci U S A. 1994;91(23):10962-10966. doi:10.1073/pnas.91.23.10962
-
Steidl C, Connors JM, Gascoyne RD. Molecular pathogenesis of Hodgkin's lymphoma: increasing evidence of the importance of the microenvironment. J Clin Oncol. 2011;29(14):1812-1826. doi:10.1200/JCO.2010.32.8401
-
Tsujimoto Y, Gorham J, Cossman J, Jaffe E, Croce CM. The t(14;18) chromosome translocations involved in B-cell neoplasms result from mistakes in VDJ joining. Science. 1985;229(4720):1390-1393. doi:10.1126/science.3929382
-
Scott DW, Gascoyne RD. The tumour microenvironment in B cell lymphomas. Nat Rev Cancer. 2014;14(8):517-534. doi:10.1038/nrc3774
-
Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127(20):2375-2390. doi:10.1182/blood-2016-01-643569
-
Alizadeh AA, Eisen MB, Davis RE, et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature. 2000;403(6769):503-511. doi:10.1038/35000501
-
Howlett C, Snedecor SJ, Landsburg DJ, et al. Front-line, dose-escalated immunochemotherapy is associated with a significant progression-free survival advantage in patients with double-hit lymphomas: a systematic review and meta-analysis. Br J Haematol. 2015;170(4):504-514. doi:10.1111/bjh.13463
-
Sharma M, Rai H, Patil J. Alcohol-induced pain in Hodgkin lymphoma: a rare but important clinical sign. BMJ Case Rep. 2017;2017:bcr2017221594. doi:10.1136/bcr-2017-221594
-
Hasenclever D, Diehl V. A prognostic score for advanced Hodgkin's disease. N Engl J Med. 1998;339(21):1506-1514. doi:10.1056/NEJM199811193392104
-
Binder M, Léchenne B, Ummanni R, et al. Stereotypical chronic lymphocytic leukemia B-cell receptors recognize survival promoting antigens on stromal cells. PLoS One. 2010;5(12):e15992. doi:10.1371/journal.pone.0015992
-
Schmitz N, Zeynalova S, Nickelsen M, et al. CNS International Prognostic Index: a risk model for CNS relapse in patients with diffuse large B-cell lymphoma treated with R-CHOP. J Clin Oncol. 2016;34(26):3150-3156. doi:10.1200/JCO.2015.65.6520
-
Matutes E, Oscier D, Montalban C, et al. Splenic marginal zone lymphoma proposals for a revision of diagnostic, staging and therapeutic criteria. Leukemia. 2008;22(3):487-495. doi:10.1038/sj.leu.2405068
-
National Institute for Health and Care Excellence. Non-Hodgkin's lymphoma: diagnosis and management (NG52). Published July 2016. Available at: https://www.nice.org.uk/guidance/ng52
-
Von Wasielewski R, Mengel M, Fischer R, et al. Classical Hodgkin's disease. Clinical impact of the immunophenotype. Am J Pathol. 1997;151(4):1123-1130.
-
A predictive model for aggressive non-Hodgkin's lymphoma. The International Non-Hodgkin's Lymphoma Prognostic Factors Project. N Engl J Med. 1993;329(14):987-994. doi:10.1056/NEJM199309303291402
-
Feldman AL, Arber DA, Pittaluga S, et al. Clonally related follicular lymphomas and histiocytic/dendritic cell sarcomas: evidence for transdifferentiation of the follicular lymphoma clone. Blood. 2008;111(12):5433-5439. doi:10.1182/blood-2007-11-124792
-
Cheson BD, Fisher RI, Barrington SF, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 2014;32(27):3059-3068. doi:10.1200/JCO.2013.54.8800
-
El-Galaly TC, d'Amore F, Mylam KJ, et al. Routine bone marrow biopsy has little or no therapeutic consequence for positron emission tomography/computed tomography-staged treatment-naive patients with Hodgkin lymphoma. J Clin Oncol. 2012;30(36):4508-4514. doi:10.1200/JCO.2012.42.4036
-
Lister TA, Crowther D, Sutcliffe SB, et al. Report of a committee convened to discuss the evaluation and staging of patients with Hodgkin's disease: Cotswolds meeting. J Clin Oncol. 1989;7(11):1630-1636. doi:10.1200/JCO.1989.7.11.1630
-
Hasenclever D, Diehl V. A prognostic score for advanced Hodgkin's disease. International Prognostic Factors Project on Advanced Hodgkin's Disease. N Engl J Med. 1998;339(21):1506-1514. doi:10.1056/NEJM199811193392104
-
Sehn LH, Berry B, Chhanabhai M, et al. The revised International Prognostic Index (R-IPI) is a better predictor of outcome than the standard IPI for patients with diffuse large B-cell lymphoma treated with R-CHOP. Blood. 2007;109(5):1857-1861. doi:10.1182/blood-2006-08-038257
-
Solal-Céligny P, Roy P, Colombat P, et al. Follicular lymphoma international prognostic index. Blood. 2004;104(5):1258-1265. doi:10.1182/blood-2003-12-4434
-
Eichenauer DA, Aleman BMP, André M, et al. Hodgkin lymphoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29(Suppl 4):iv19-iv29. doi:10.1093/annonc/mdy080
-
Canellos GP, Anderson JR, Propert KJ, et al. Chemotherapy of advanced Hodgkin's disease with MOPP, ABVD, or MOPP alternating with ABVD. N Engl J Med. 1992;327(21):1478-1484. doi:10.1056/NEJM199211193272102
-
Engert A, Diehl V, Franklin J, et al. Escalated-dose BEACOPP in the treatment of patients with advanced-stage Hodgkin's lymphoma: 10 years of follow-up of the GHSG HD9 study. J Clin Oncol. 2009;27(27):4548-4554. doi:10.1200/JCO.2008.19.8820
-
Linch DC, Winfield D, Goldstone AH, et al. Dose intensification with autologous bone-marrow transplantation in relapsed and resistant Hodgkin's disease: results of a BNLI randomised trial. Lancet. 1993;341(8852):1051-1054. doi:10.1016/0140-6736(93)92411-l
-
Younes A, Gopal AK, Smith SE, et al. Results of a pivotal phase II study of brentuximab vedotin for patients with relapsed or refractory Hodgkin's lymphoma. J Clin Oncol. 2012;30(18):2183-2189. doi:10.1200/JCO.2011.38.0410
-
Ansell SM, Lesokhin AM, Borrello I, et al. PD-1 blockade with nivolumab in relapsed or refractory Hodgkin's lymphoma. N Engl J Med. 2015;372(4):311-319. doi:10.1056/NEJMoa1411087
-
Tilly H, Gomes da Silva M, Vitolo U, et al. Diffuse large B-cell lymphoma (DLBCL): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2015;26 Suppl 5:v116-v125. doi:10.1093/annonc/mdv304
-
Gisselbrecht C, Glass B, Mounier N, et al. Salvage regimens with autologous transplantation for relapsed large B-cell lymphoma in the rituximab era. J Clin Oncol. 2010;28(27):4184-4190. doi:10.1200/JCO.2010.28.1618
-
Neelapu SS, Locke FL, Bartlett NL, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377(26):2531-2544. doi:10.1056/NEJMoa1707447
-
Hutchings M, Morschhauser F, Iacoboni G, et al. Glofitamab, a novel, bispecific CD20×CD3 antibody, induces durable complete remissions in relapsed or refractory B-cell lymphoma: a phase I trial. J Clin Oncol. 2021;39(18):1959-1970. doi:10.1200/JCO.20.03175
-
Ardeshna KM, Qian W, Smith P, et al. Rituximab versus a watch-and-wait approach in patients with advanced-stage, asymptomatic, non-bulky follicular lymphoma: an open-label randomised phase 3 trial. Lancet Oncol. 2014;15(4):424-435. doi:10.1016/S1470-2045(14)70027-0
-
Rummel MJ, Niederle N, Maschmeyer G, et al. Bendamustine plus rituximab versus CHOP plus rituximab as first-line treatment for patients with indolent and mantle-cell lymphomas: an open-label, multicentre, randomised, phase 3 non-inferiority trial. Lancet. 2013;381(9873):1203-1210. doi:10.1016/S0140-6736(12)61763-2
-
Salles G, Seymour JF, Offner F, et al. Rituximab maintenance for 2 years in patients with high tumour burden follicular lymphoma responding to rituximab plus chemotherapy (PRIMA): a phase 3, randomised controlled trial. Lancet. 2011;377(9759):42-51. doi:10.1016/S0140-6736(10)62175-7
-
Al-Tourah AJ, Gill KK, Chhanabhai M, et al. Population-based analysis of incidence and outcome of transformed non-Hodgkin's lymphoma. J Clin Oncol. 2008;26(32):5165-5169. doi:10.1200/JCO.2008.16.0283
-
Dreyling M, Campo E, Hermine O, et al. Newly diagnosed and relapsed mantle cell lymphoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28(suppl_4):iv62-iv71. doi:10.1093/annonc/mdx223
-
Wang ML, Rule S, Martin P, et al. Targeting BTK with ibrutinib in relapsed or refractory mantle-cell lymphoma. N Engl J Med. 2013;369(6):507-516. doi:10.1056/NEJMoa1306220
-
Dunleavy K, Pittaluga S, Shovlin M, et al. Low-intensity therapy in adults with Burkitt's lymphoma. N Engl J Med. 2013;369(20):1915-1925. doi:10.1056/NEJMoa1308392
-
Hwang JP, Somerfield MR, Alston-Johnson DE, et al. Hepatitis B virus screening for patients with cancer before therapy: American Society of Clinical Oncology provisional clinical opinion update. J Clin Oncol. 2020;38(31):3698-3715. doi:10.1200/JCO.20.01757
-
Huisman M, Paes FM, Schnater JM, Kroft LJ. Spinal epidural lymphoma: case report and review of the literature. Spine J. 2013;13(10):e45-e49. doi:10.1016/j.spinee.2013.03.002
-
Swerdlow AJ, Higgins CD, Smith P, et al. Second cancer risk after chemotherapy for Hodgkin's lymphoma: a collaborative British cohort study. J Clin Oncol. 2011;29(31):4096-4104. doi:10.1200/JCO.2011.34.8268
Medical Disclaimer: MedVellum content is for educational purposes only and does not constitute medical advice. Clinical decisions should be made in consultation with qualified healthcare professionals and with reference to current guidelines and patient-specific factors.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Lymphoid System Physiology
- Immunophenotyping
Differentials
Competing diagnoses and look-alikes to compare.
- Leukaemia
- Reactive Lymphadenopathy
Consequences
Complications and downstream problems to keep in mind.