Pediatric Bacterial Meningitis
Comprehensive evidence-based guide to diagnosis and management of bacterial meningitis in neonates, infants, and children - emergency recognition, age-specific pathogens, CSF interpretation, empiric antibiotics, and...
What matters first
Comprehensive evidence-based guide to diagnosis and management of bacterial meningitis in neonates, infants, and children - emergency recognition, age-specific pathogens, CSF interpretation, empiric antibiotics, and...
Petechial/purpuric rash
25 Apr 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Petechial/purpuric rash
- Shock
- Bulging fontanelle
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Pediatric Bacterial Meningitis
Quick Reference Card
Critical Time-Sensitive Actions
DOOR-TO-ANTIBIOTIC TIME: ≤60 MINUTES
- Do NOT delay antibiotics for LP or imaging
- Antibiotics remain effective for CSF culture up to 4 hours post-dose
- Mortality increases 12.6% per hour of delay 1
Emergency Recognition Triad
| Feature | Classic Presentation | Caveat |
|---|---|---|
| Fever | High-grade (> 38.5°C) | May be absent in neonates, immunocompromised |
| Altered mental status | Lethargy, confusion, irritability | Most reliable predictor of bacterial etiology |
| Meningismus | Neck stiffness, Kernig/Brudzinski positive | Absent in less than 12 months; sensitivity only 30% in children |
Age-Specific Pathogens: The Critical Three
| Age Group | Primary Pathogens | Coverage Required |
|---|---|---|
| less than 1 month | Group B Streptococcus (40-50%), E. coli K1 (20-30%), Listeria monocytogenes (5-10%) | Ampicillin + Cefotaxime/Gentamicin + Acyclovir |
| 1-3 months | GBS, E. coli, S. pneumoniae, H. influenzae type b, Listeria | Ampicillin + Ceftriaxone ± Vancomycin |
| > 3 months | S. pneumoniae (50-60%), N. meningitidis (25-30%), H. influenzae type b (less than 5% post-vaccine) | Ceftriaxone + Vancomycin |
CSF Interpretation at a Glance
| Parameter | Normal | Bacterial | Viral | TB/Fungal |
|---|---|---|---|---|
| Opening pressure | less than 20 cm H₂O | ↑↑ (> 30) | Normal/↑ | ↑↑ |
| WBC (cells/μL) | less than 5 | > 1000 (PMN) | 10-500 (lymph) | 50-500 (lymph) |
| Protein (mg/dL) | less than 45 | > 100 | 50-100 | > 100 |
| Glucose (mg/dL) | > 45 (> 50% serum) | less than 40 (less than 40% serum) | Normal | ↓↓ |
| Gram stain | Negative | Positive (60-90%) | Negative | AFB (20-40%) |
| Lactate (mmol/L) | less than 2.1 | > 4.0 | less than 3.5 | > 4.0 |
Empiric Antibiotic Dosing Quick Reference
| Age | Regimen | Doses |
|---|---|---|
| less than 1 month | Ampicillin + Cefotaxime + Acyclovir | Amp 75-100 mg/kg q6-8h; Cefo 50 mg/kg q6-8h; Acy 20 mg/kg q8h |
| 1-3 months | Ampicillin + Ceftriaxone ± Vancomycin | Amp 75-100 mg/kg q6h; CTX 50 mg/kg q12h; Vanc 15 mg/kg q6h |
| > 3 months | Ceftriaxone + Vancomycin | CTX 50-100 mg/kg/day (max 4g); Vanc 60 mg/kg/day divided q6h |
Definition and Overview
Core Definition
Bacterial meningitis is an acute purulent infection of the leptomeninges (pia mater and arachnoid) and subarachnoid space, representing a medical emergency with high morbidity and mortality in children. The condition is characterized by inflammation of the meninges caused by bacterial invasion, leading to cerebral edema, elevated intracranial pressure, and potential neurological sequelae 2.
Clinical Significance
Bacterial meningitis remains one of the top 10 causes of infection-related death worldwide and carries significant risk of permanent neurological disability in survivors. Despite advances in vaccination and antimicrobial therapy, mortality rates remain 5-15% in developed countries and up to 50% in resource-limited settings 3.
Classification System
By Etiology:
| Category | Characteristics | Common Organisms |
|---|---|---|
| Acute Bacterial | Rapid onset (less than 24-48h), purulent CSF | S. pneumoniae, N. meningitidis, H. influenzae, GBS, E. coli |
| Viral (Aseptic) | Subacute, lymphocytic CSF, benign course | Enteroviruses, HSV, VZV, arboviruses |
| Tuberculous | Chronic (weeks), basilar predominance | Mycobacterium tuberculosis |
| Fungal | Chronic, immunocompromised host | Cryptococcus, Candida, Aspergillus |
| Parasitic | Geographic exposure, eosinophilic CSF | Angiostrongylus, Naegleria fowleri |
By Age-Based Risk Stratification:
| Age Category | Risk Level | Unique Considerations |
|---|---|---|
| Neonates (less than 28 days) | Highest | Immature immune system, vertical transmission, blood-brain barrier vulnerability |
| Young infants (1-3 months) | Very High | Transition period; both neonatal and community pathogens |
| Infants (3-12 months) | High | Post-maternal antibody decline, pre-vaccine completion |
| Toddlers (1-5 years) | Moderate-High | Peak incidence for pneumococcal/meningococcal disease |
| School-age (> 5 years) | Moderate | More classic presentation, better prognosis |
Epidemiology
Global Burden
The World Health Organization estimates approximately 2.5 million cases of bacterial meningitis annually worldwide, with the highest burden in the African "meningitis belt" and in children under 5 years of age 4.
Incidence by Age and Pathogen
Pre-Vaccine Era vs. Post-Vaccine Era:
| Pathogen | Pre-vaccine Incidence | Post-vaccine Incidence | Reduction |
|---|---|---|---|
| H. influenzae type b | 40-50/100,000 less than 5 years | less than 0.5/100,000 | > 99% |
| S. pneumoniae | 15-25/100,000 less than 2 years | 5-8/100,000 | 60-75% |
| N. meningitidis | 1-3/100,000 | 0.3-0.5/100,000 | 70-85% |
| Group B Streptococcus | 0.5-1.0/1,000 neonates | 0.2-0.4/1,000 | 50-70% |
Risk Factors
Host Factors:
| Factor | Relative Risk | Mechanism |
|---|---|---|
| Age less than 2 years | 10-20× | Immature immune response, incomplete vaccination |
| Complement deficiency (C5-C9) | 5,000-10,000× | Impaired bactericidal activity against Neisseria |
| Asplenia (functional/anatomic) | 50-100× | Impaired opsonization of encapsulated organisms |
| HIV infection | 5-10× | Cell-mediated immune deficiency |
| CSF leak/skull fracture | 100-200× | Direct bacterial access to CNS |
| Cochlear implants | 30× | Biofilm formation, direct route |
| Recent neurosurgery | 50× | Blood-brain barrier disruption |
Environmental Factors:
| Factor | Association | Notes |
|---|---|---|
| Crowded living conditions | 2-4× risk | Day care, military barracks |
| Exposure to tobacco smoke | 2-3× risk | Impaired mucociliary clearance |
| Winter/spring months | Seasonal peak | Viral co-infection, indoor crowding |
| Endemic areas | Variable | Meningitis belt (Sub-Saharan Africa) |
Mortality and Morbidity Outcomes
Mortality by Pathogen (Treated Cases):
| Organism | Mortality Rate | Highest Risk Period |
|---|---|---|
| S. pneumoniae | 15-25% | First 48 hours |
| N. meningitidis | 5-10% | First 24 hours |
| H. influenzae type b | 3-6% | First 72 hours |
| Group B Streptococcus | 10-15% | Neonatal period |
| E. coli K1 | 15-30% | Neonatal period |
| Listeria monocytogenes | 20-30% | Extremes of age |
Long-term Sequelae (Survivors):
| Sequelae | Incidence | Risk Factors |
|---|---|---|
| Sensorineural hearing loss | 10-30% | Pneumococcal etiology, delayed treatment |
| Cognitive impairment | 10-20% | Young age, coma at presentation |
| Motor deficits | 5-10% | Vascular complications |
| Seizure disorder | 5-10% | Seizures during acute illness |
| Hydrocephalus | 3-5% | Gram-negative organisms, delayed treatment |
| Behavioral problems | 15-25% | Prolonged illness |
Pathophysiology
Mechanisms of CNS Invasion
The pathogenesis of bacterial meningitis involves a complex cascade of events that can be conceptualized in distinct phases:
Phase 1: Colonization and Mucosal Invasion
- Nasopharyngeal colonization (pneumococcus, meningococcus) or vertical transmission (GBS, E. coli)
- Bacterial adherence via pili, adhesins, and capsular polysaccharides
- Mucosal invasion through paracellular and transcellular routes
Phase 2: Bloodstream Survival and Invasion
- Encapsulated organisms resist complement-mediated killing
- High-grade bacteremia (> 10³ CFU/mL) increases CNS invasion risk
- Survival in bloodstream: 4-8 hours before CNS seeding
Phase 3: Blood-Brain Barrier Penetration
| Mechanism | Organisms | Target |
|---|---|---|
| Transcellular (receptor-mediated) | S. pneumoniae, GBS | Brain microvascular endothelium |
| Paracellular (tight junction disruption) | E. coli K1, N. meningitidis | Endothelial junctions |
| Choroid plexus invasion | Listeria, E. coli | Choroid epithelium |
| Trojan horse (monocyte carriage) | Listeria | Infected macrophages |
Phase 4: Subarachnoid Space Inflammation
The CSF is an "immunologically privileged" compartment with:
- Low complement levels
- Minimal immunoglobulin
- Absent resident phagocytes
- Limited opsonization capacity
This allows rapid bacterial proliferation and triggers intense inflammatory response.
Inflammatory Cascade
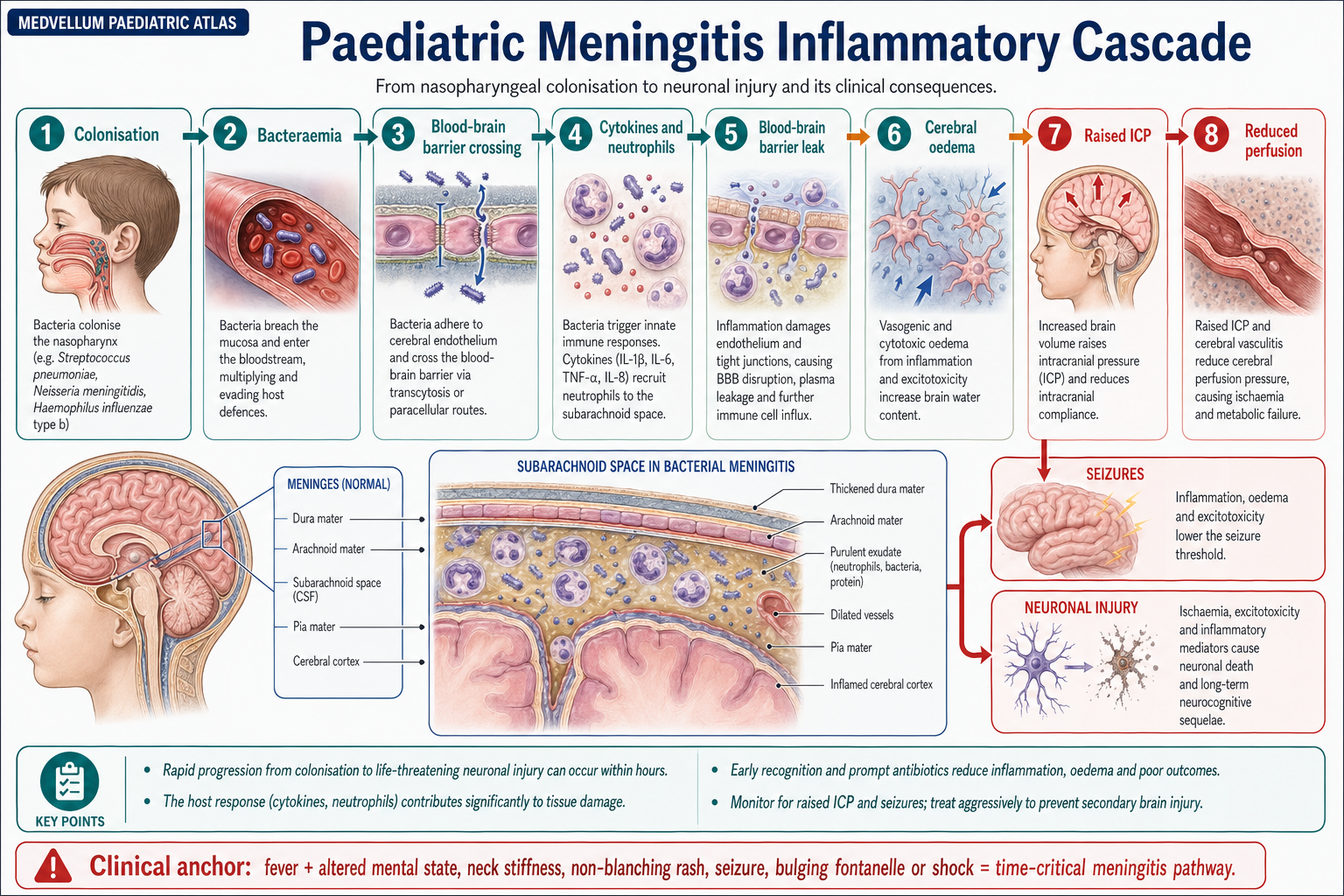
Key Inflammatory Mediators:
| Mediator | Source | Effect | Peak Level |
|---|---|---|---|
| TNF-α | Microglia, macrophages | BBB permeability, apoptosis | 2-6 hours |
| IL-1β | Microglia, astrocytes | Fever, inflammation | 4-8 hours |
| IL-6 | Endothelium, leukocytes | Acute phase response | 6-12 hours |
| IL-8 | Endothelium | Neutrophil chemotaxis | 4-12 hours |
| Matrix metalloproteinases | Neutrophils | BBB degradation | 12-24 hours |
| Nitric oxide | iNOS in macrophages | Vasodilation, oxidative damage | 6-24 hours |
Cerebral Complications
Intracranial Pressure Dynamics:
- Vasogenic edema: BBB breakdown → plasma protein extravasation
- Cytotoxic edema: Neuronal/glial cell swelling from energy failure
- Interstitial edema: Impaired CSF absorption
- Increased CSF production: Choroid plexus inflammation
Cerebrovascular Complications:
| Complication | Mechanism | Incidence |
|---|---|---|
| Vasculitis | Inflammation of meningeal vessels | 15-25% |
| Arterial infarction | Thrombosis, spasm | 5-15% |
| Venous thrombosis | Sagittal sinus, cortical veins | 3-5% |
| Subdural effusion | BBB dysfunction, inflammation | 10-30% |
| Subdural empyema | Secondary infection of effusion | 1-2% |
Pathogen-Specific Virulence Factors
| Organism | Key Virulence Factors | Clinical Implications |
|---|---|---|
| S. pneumoniae | Polysaccharide capsule, pneumolysin, autolysin | High mortality, hearing loss risk |
| N. meningitidis | LOS endotoxin, capsule, pili, Opa/Opc proteins | DIC, purpura fulminans, adrenal hemorrhage |
| H. influenzae type b | PRP capsule, IgA protease | Subdural effusions common |
| GBS | Capsular sialic acid, β-hemolysin | Neonatal focus, high relapse rate |
| E. coli K1 | K1 capsule (sialic acid), FimH adhesin | Neonatal meningitis, poor prognosis |
| Listeria | Listeriolysin O, ActA (intracellular spread) | Rhombencephalitis, immunocompromised |
Clinical Presentation
Age-Specific Presentations
Neonates (less than 28 Days)
Neonatal Meningitis: The Great Mimicker Classic meningeal signs are absent in > 90% of neonates. Maintain high index of suspicion in any unwell neonate.
Clinical Features:
| System | Early Signs | Late Signs |
|---|---|---|
| General | Temperature instability (hypo- or hyperthermia), poor feeding | Lethargy, weak cry, apnea |
| Neurological | Irritability, high-pitched cry | Bulging fontanelle, seizures, opisthotonus |
| Respiratory | Tachypnea, respiratory distress | Apnea, cyanosis |
| Cardiovascular | Tachycardia | Poor perfusion, hypotension |
| Gastrointestinal | Feeding intolerance, vomiting | Abdominal distension, ileus |
Early-Onset vs. Late-Onset Neonatal Meningitis:
| Feature | Early-Onset (less than 7 days) | Late-Onset (7-90 days) |
|---|---|---|
| Transmission | Vertical (maternal) | Horizontal (nosocomial/community) |
| Primary organisms | GBS, E. coli, Listeria | GBS, E. coli, S. aureus, CoNS |
| Associated features | Maternal risk factors (PROM, GBS+, fever) | Often no maternal factors |
| Mortality | 20-30% | 10-15% |
Infants (1-12 Months)
Clinical Features:
| Feature | Prevalence | Notes |
|---|---|---|
| Fever | 90-95% | May be only sign |
| Irritability | 80-90% | Inconsolable, paradoxical |
| Poor feeding/vomiting | 60-70% | Non-specific |
| Lethargy | 50-60% | Concerning sign |
| Bulging fontanelle | 25-35% | Specific but late sign |
| Seizures | 20-30% | Often focal |
| Neck stiffness | 15-25% | Unreliable under 12 months |
"Paradoxical Irritability": Infant cries more when held/comforted (due to meningeal irritation with movement)
Children (> 12 Months)
Classic Triad Prevalence:
| Sign | Children 1-5 years | Children > 5 years |
|---|---|---|
| Fever | 85-95% | 90-95% |
| Headache | 40-60% | 80-90% |
| Neck stiffness | 40-60% | 70-85% |
| All three | 20-30% | 40-50% |
Additional Features:
| Feature | Prevalence | Significance |
|---|---|---|
| Photophobia | 30-50% | Meningeal irritation |
| Nausea/vomiting | 60-80% | Raised ICP, direct irritation |
| Altered mental status | 30-50% | Poor prognostic sign |
| Seizures | 15-25% | Early: poor prognosis; Late: may indicate complication |
| Petechial/purpuric rash | 10-20% (50-80% in meningococcal) | Medical emergency |
| Focal deficits | 10-15% | Vascular complication, abscess |
Physical Examination Signs
Meningeal Signs
Kernig's Sign:
- Patient supine with hip flexed to 90°
- Attempt to extend knee
- Positive: Pain/resistance at > 135° flexion prevented
- Sensitivity: 5-10% (children); Specificity: 95%
Brudzinski's Sign:
- Patient supine
- Passive neck flexion
- Positive: Involuntary hip/knee flexion
- Sensitivity: 5-10% (children); Specificity: 95%
Nuchal Rigidity:
- Resistance to passive neck flexion
- Most reliable in children > 2 years
- Sensitivity: 30-70%; Specificity: 70-95%
Jolt Accentuation:
- Patient turns head horizontally 2-3×/second
- Positive: Worsening headache
- Sensitivity: 97%; Specificity: 60% (adults)
Assessment of Raised Intracranial Pressure
| Sign | Finding | Implication |
|---|---|---|
| Fontanelle (infants) | Bulging, tense, non-pulsatile | ↑ICP, needs urgent intervention |
| Consciousness | GCS decline, Cushing response | Impending herniation |
| Pupils | Unilateral dilation, sluggish | Uncal herniation |
| Posturing | Decorticate/decerebrate | Severe ↑ICP |
| Papilledema | Blurred disc margins, venous engorgement | Chronic ↑ICP (develops over hours-days) |
| VI nerve palsy | Lateral gaze limitation | False localizing sign of ↑ICP |
Meningococcal Disease: Special Considerations
MENINGOCOCCEMIA: TIME-CRITICAL EMERGENCY
- Median time from first symptom to death: 24 hours
- Petechiae can evolve to purpura fulminans within 2-4 hours
- Administer antibiotics IMMEDIATELY (do not wait for IV access - give IM)
Evolution of Meningococcal Rash:
| Stage | Time | Appearance | Distribution |
|---|---|---|---|
| 1. Macular | 0-4 hours | Blanching pink macules | Non-specific |
| 2. Petechial | 4-8 hours | Non-blanching less than 2mm lesions | Trunk, extremities, conjunctivae |
| 3. Purpuric | 8-12 hours | Larger non-blanching lesions | Spreading, coalescing |
| 4. Purpura fulminans | 12-24 hours | Extensive hemorrhagic necrosis | Diffuse, with DIC |
Glass Test (Tumbler Test):
- Press clear glass firmly against rash
- Non-blanching = concerning for meningococcemia
- Sensitivity 50-60% in early disease; higher specificity
Red Flags and Contraindications
Absolute Contraindications to Immediate LP
DO NOT PERFORM LP IF:
-
Signs of cerebral herniation:
- Abnormal posturing (decerebrate/decorticate)
- Unequal, dilated, or unreactive pupils
- Abnormal oculocephalic reflexes
- Cushing triad (bradycardia, hypertension, irregular respiration)
-
Cardiovascular instability:
- Shock requiring active resuscitation
- Respiratory failure
-
Local contraindications:
- Skin infection over LP site
- Suspected spinal epidural abscess
-
Coagulopathy:
- Platelets less than 50,000/μL
- INR > 1.5
- Active DIC (start antibiotics first)
ALWAYS START ANTIBIOTICS BEFORE CT/LP IF MENINGITIS SUSPECTED
Indications for CT Before LP
The IDSA recommends CT before LP in patients with 5:
| Clinical Feature | Rationale |
|---|---|
| Immunocompromised state | Higher risk of space-occupying lesion |
| History of CNS disease | Mass lesion, shunt malfunction |
| New-onset seizures | Focal lesion risk |
| Papilledema | Established raised ICP |
| Focal neurological deficit | Space-occupying lesion |
| Altered consciousness (GCS ≤12) | Higher herniation risk |
Important Caveat: A normal CT does NOT exclude raised ICP. Up to 5% of patients with normal CT may still herniate post-LP 6.
Life-Threatening Presentations Requiring Immediate Intervention
| Presentation | Immediate Action | Timeframe |
|---|---|---|
| Purpuric rash + fever | IV/IM antibiotics, aggressive fluid resuscitation | less than 5 minutes |
| Signs of herniation | Head elevation, hyperventilation (short-term), osmotherapy, neurosurgery consult | less than 10 minutes |
| Status epilepticus | Benzodiazepines, airway protection, antibiotics | less than 5 minutes |
| Septic shock | IV fluids (20 mL/kg bolus), vasopressors, antibiotics | less than 15 minutes |
| Respiratory failure | Intubation, mechanical ventilation, antibiotics | less than 10 minutes |
Differential Diagnosis
Primary Differential Considerations
| Diagnosis | Key Distinguishing Features | Investigations |
|---|---|---|
| Viral meningitis | Less toxic, lymphocytic CSF, normal glucose | CSF viral PCR (enterovirus, HSV) |
| Viral encephalitis | Altered mentation > meningismus, behavioral changes, seizures | CSF HSV PCR, MRI (temporal lobe changes) |
| Brain abscess | Focal deficits, headache, fever; may lack meningismus | CT/MRI with contrast |
| Subarachnoid hemorrhage | Thunderclap headache, sudden onset, xanthochromia | CT head, LP (xanthochromia) |
| Tuberculous meningitis | Subacute onset (weeks), cranial nerve palsies, basilar enhancement | CSF AFB, adenosine deaminase, PCR |
| Partially treated bacterial meningitis | Prior antibiotics, atypical CSF | Clinical history, PCR |
| Drug-induced aseptic meningitis | NSAID, IVIG, or antibiotic exposure | Temporal relationship to drug |
| Autoimmune meningitis | Recurrent episodes, systemic autoimmune features | Autoantibodies, biopsy |
Mimics in Specific Populations
Neonates:
| Condition | Features | Differentiation |
|---|---|---|
| Neonatal sepsis without meningitis | Similar presentation | LP negative |
| Inborn errors of metabolism | Poor feeding, lethargy, seizures | Metabolic workup, no fever |
| Non-accidental injury | Altered consciousness, bulging fontanelle | Retinal hemorrhages, imaging |
| Intracranial hemorrhage | Bulging fontanelle, seizures | CT/MRI, coagulation studies |
Infants and Children:
| Condition | Features | Differentiation |
|---|---|---|
| Complex febrile seizure | Postictal drowsiness, fever | LP often performed; CSF normal |
| Acute viral gastroenteritis with dehydration | Lethargy, fever | Hydration improves mental status |
| Sinusitis with extension | Headache, fever, focal signs | CT sinuses, MRI brain |
| Kawasaki disease | Irritability, fever | Other diagnostic criteria, CSF pleocytosis |
Diagnostic Approach
Clinical Decision Rules
Bacterial Meningitis Score (BMS)
The Bacterial Meningitis Score was developed and validated by Nigrovic et al. 7 to identify children with CSF pleocytosis at very low risk of bacterial meningitis:
Criteria (all must be absent for low risk):
| Criterion | Points | Definition |
|---|---|---|
| Positive CSF Gram stain | 1 | Any organisms seen |
| CSF ANC ≥1,000 cells/μL | 1 | Absolute neutrophil count |
| CSF protein ≥80 mg/dL | 1 | Elevated protein |
| Peripheral blood ANC ≥10,000/μL | 1 | Systemic inflammation |
| Seizure at or prior to presentation | 1 | Any seizure activity |
Interpretation:
- Score 0: Very low risk of bacterial meningitis (less than 0.1%)
- Score ≥1: Cannot rule out bacterial meningitis
Validation: Sensitivity 99.3%, Negative predictive value 99.9% 7
Limitations:
- Not validated in infants less than 29 days
- Not for use in immunocompromised patients
- Not for patients with prior antibiotics
UK-ChiMES Clinical Decision Models (2024)
A prospective multicenter UK study (Martin et al., 2024) 8 involving 3,002 children with suspected meningitis/encephalitis developed two novel clinical decision rules:
Pre-LP Model (before lumbar puncture):
- Sensitivity: 82%
- Specificity: 71%
- Used to assess bacterial meningitis risk before LP
Post-LP Model (after CSF results):
- Sensitivity: 84%
- Specificity: 93%
- Incorporates CSF parameters for definitive risk stratification
Key Findings:
- Bacterial meningitis comprised only 6% (180/3,002) of suspected cases
- Enterovirus most common in less than 6 months and 10-16 years
- N. meningitidis/S. pneumoniae most common at 6 months-9 years
- Bacterial Meningitis Score had NPV of 95.3% in this cohort
These models provide evidence-based tools to improve diagnostic accuracy and reduce unnecessary antibiotic use 8.
Lumbar Puncture
Technique and Safety
Positioning Options:
| Position | Advantages | Disadvantages |
|---|---|---|
| Lateral decubitus | More accurate opening pressure | May be difficult in obese/young infants |
| Sitting | Easier landmark identification | Cannot measure opening pressure |
Needle Selection:
| Age | Needle Gauge | Length |
|---|---|---|
| Neonate | 22G | 1.5 inch (3.8 cm) |
| Infant | 22G | 1.5 inch (3.8 cm) |
| Child | 22G | 2.5-3.5 inch (6.4-8.9 cm) |
| Adolescent | 20-22G | 3.5 inch (8.9 cm) |
Required CSF Volume:
| Test | Minimum Volume |
|---|---|
| Cell count + differential | 0.5-1 mL |
| Gram stain + culture | 1-2 mL |
| Glucose + protein | 0.5 mL |
| PCR panel | 0.2-0.5 mL |
| Total recommended | 3-4 mL |
CSF Interpretation
Normal CSF Values by Age:
| Parameter | Preterm | Term Neonate | 1-3 months | > 3 months |
|---|---|---|---|---|
| WBC (cells/μL) | less than 25 | less than 22 | less than 15 | less than 5 |
| % PMN | less than 60% | less than 60% | less than 5% | less than 5% |
| Protein (mg/dL) | less than 150 | less than 100 | less than 80 | less than 45 |
| Glucose (mg/dL) | 34-119 | 34-119 | 40-80 | 45-80 |
| CSF:serum glucose | > 50% | > 50% | > 50% | > 60% |
CSF Findings in Bacterial vs. Viral Meningitis:
| Parameter | Bacterial | Viral | Sensitivity | Specificity |
|---|---|---|---|---|
| WBC > 1000/μL | Common | Rare | 80% | 85% |
| PMN > 80% | Common | Early viral | 75% | 80% |
| Glucose less than 40 mg/dL | Common | Rare | 80% | 95% |
| CSF:serum glucose less than 0.4 | Common | Rare | 80% | 95% |
| Protein > 100 mg/dL | Common | Occasional | 70% | 75% |
| Lactate > 4 mmol/L | Common | Rare | 90% | 90% |
| Positive Gram stain | 60-90% | Negative | 60-90% | 99% |
Traumatic LP Interpretation
Correction Formulas:
-
WBC correction for bloody tap:
- Predicted WBC = Observed WBC - (WBC_blood × RBC_CSF / RBC_blood)
- Alternative: Subtract 1 WBC per 500-1000 RBCs
-
Protein correction:
- Subtract 1 mg/dL protein per 1000 RBCs
When to Repeat LP:
- Traumatic tap with clinical suspicion
- Unable to interpret results
- Not indicated routinely after 48-72 hours of treatment
Laboratory Investigations
Essential Blood Tests
| Test | Purpose | Expected Findings |
|---|---|---|
| CBC with differential | Assess WBC, platelet count | Leukocytosis (often left shift), thrombocytopenia in severe disease |
| Blood cultures (×2) | Identify pathogen | Positive in 50-80% of bacterial meningitis |
| CRP | Inflammatory marker | Elevated; helps distinguish bacterial from viral |
| Procalcitonin | Bacterial infection marker | ≥0.5 ng/mL suggests bacterial; \u003e 2 ng/mL highly specific 9 |
| Procalcitonin (prognostic) | Severity/outcome biomarker | Cutoff 19.6 ng/mL (admission) predicts short-term complications; 62.4 ng/mL (24h) predicts mortality 10 |
| Serum glucose | Calculate CSF:serum ratio | Obtain before/with LP |
| Coagulation studies | Pre-LP assessment | PT/INR, aPTT, platelets if concerns |
| BMP/electrolytes | Assess for SIADH, shock | Hyponatremia common (SIADH in 25-50%) |
| Lactate | Sepsis severity | Elevated in septic shock |
CSF Studies
Routine:
| Test | Tube | Priority |
|---|---|---|
| Cell count + differential | 1 and 4 | Essential |
| Gram stain and culture | 2 | Essential |
| Protein | 3 | Essential |
| Glucose | 3 | Essential |
Additional/Specialized:
| Test | Indication | Sensitivity/Specificity |
|---|---|---|
| CSF multiplex PCR | All suspected bacterial meningitis | 90-95% / 99% for covered organisms 11 |
| HSV PCR | Neonates, encephalitis features | 98% / 99% |
| Enterovirus PCR | Suspected viral meningitis | 95% / 99% |
| CSF lactate | Differentiate bacterial vs. viral | 90% / 90% (cutoff > 4 mmol/L) |
| Latex agglutination | Rapid antigen detection | Variable; largely replaced by PCR |
| CSF procalcitonin | Bacterial vs. viral | 90% / 85% |
Imaging
Indications for Neuroimaging
CT Head (Non-contrast first, then contrast):
| Indication | Rationale |
|---|---|
| Signs of raised ICP or herniation | Rule out mass effect before LP |
| Focal neurological signs | Mass lesion, abscess, infarct |
| Prolonged or new seizures | Focal lesion, complications |
| Immunocompromised host | Opportunistic infections, masses |
| Deteriorating clinical course | Complications (hydrocephalus, abscess, infarct) |
| Persistent fever > 48-72h on antibiotics | Secondary complications |
| Suspected mastoiditis/sinusitis extension | Source identification |
MRI Brain (With gadolinium):
| Indication | Findings |
|---|---|
| Suspected encephalitis | Temporal lobe involvement (HSV) |
| Subdural empyema | Rim-enhancing collection |
| Vascular complications | DWI for acute infarct |
| Venous sinus thrombosis | MRV abnormalities |
| Basilar meningitis | Meningeal enhancement, hydrocephalus |
| Poor response to therapy | Evaluate for secondary complications |
Treatment
Emergency Management Algorithm
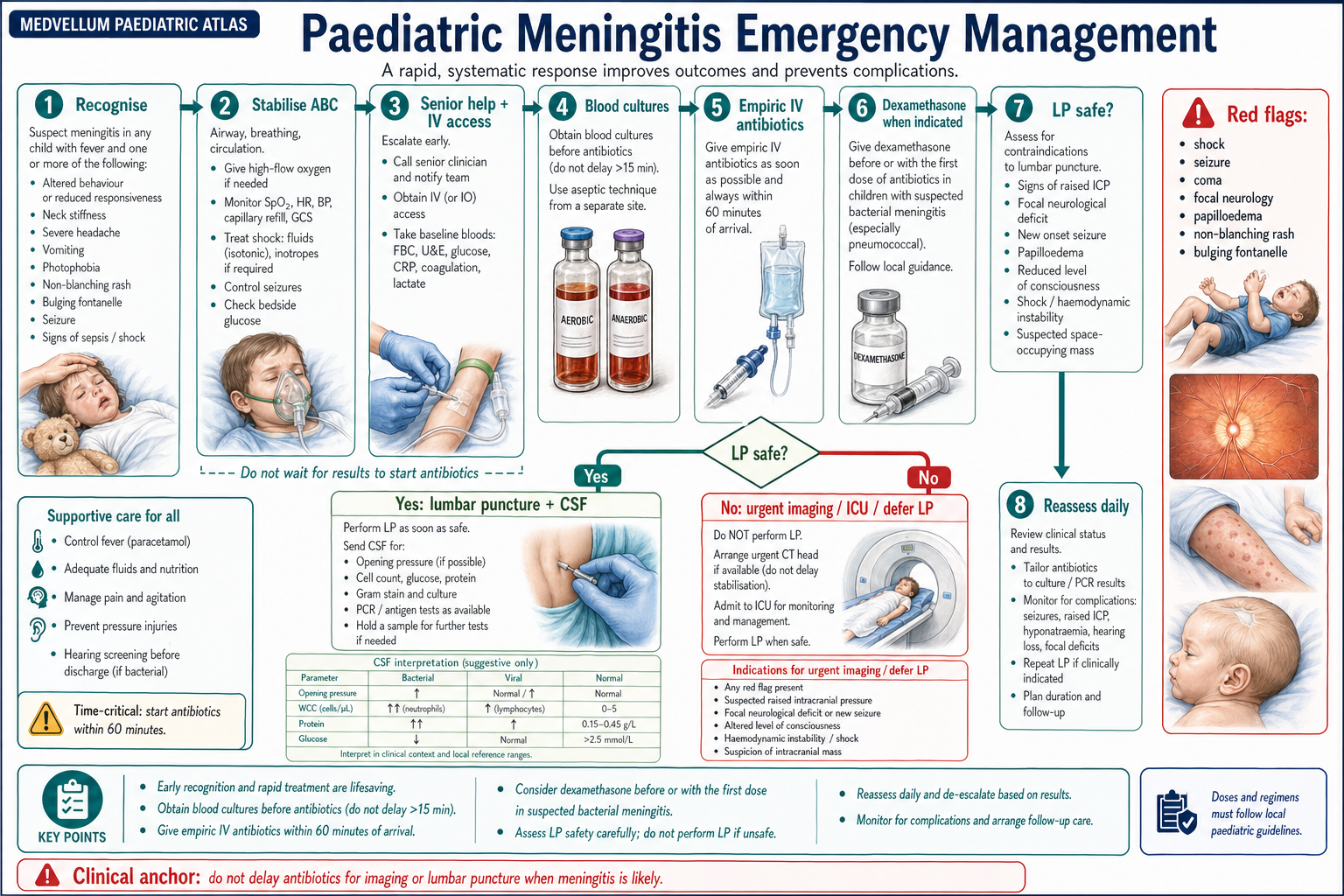
Antimicrobial Therapy
Empiric Treatment by Age
Neonates (less than 1 month):
| Drug | Dose | Interval | Coverage |
|---|---|---|---|
| Ampicillin | 75-100 mg/kg/dose | q6-8h (age-dependent) | GBS, Listeria, Enterococcus |
| Cefotaxime | 50 mg/kg/dose | q6-8h | E. coli, GNR, GBS |
| OR Gentamicin | 4-5 mg/kg/dose | q24-48h (if cefotaxime unavailable) | GNR synergy |
| Acyclovir | 20 mg/kg/dose | q8h | HSV (always add in neonates) |
Dosing by Postnatal and Gestational Age:
| Postmenstrual Age | Ampicillin | Cefotaxime | Gentamicin |
|---|---|---|---|
| ≤29 weeks PMA | q12h (0-28d), q8h (> 28d) | q12h (0-28d), q8h (> 28d) | q48h |
| 30-36 weeks PMA | q12h (0-14d), q8h (> 14d) | q12h (0-14d), q8h (> 14d) | q36h |
| 37-44 weeks PMA | q8h (0-7d), q6h (> 7d) | q8h (0-7d), q6h (> 7d) | q24h |
| ≥45 weeks PMA | q6h | q6h | q24h |
Infants 1-3 months:
| Drug | Dose | Interval | Coverage |
|---|---|---|---|
| Ampicillin | 75-100 mg/kg/dose | q6h | Listeria, Enterococcus |
| Ceftriaxone | 50 mg/kg/dose | q12h or 100 mg/kg q24h | Pneumococcus, H. flu, GNR, Meningococcus |
| ± Vancomycin | 15 mg/kg/dose | q6h | Resistant pneumococcus |
| ± Acyclovir | 20 mg/kg/dose | q8h | If HSV concern |
Children > 3 months:
| Drug | Dose | Interval | Maximum | Coverage |
|---|---|---|---|---|
| Ceftriaxone | 50-100 mg/kg/day | q12-24h | 4 g/day | S. pneumoniae, N. meningitidis, H. flu |
| Vancomycin | 60 mg/kg/day | q6h | 2 g/dose | Resistant S. pneumoniae (MIC > 1 μg/mL) |
Pathogen-Directed Therapy
Once Organism Identified:
| Organism | First-Line | Alternative | Duration |
|---|---|---|---|
| S. pneumoniae (PCN-S, MIC less than 0.1) | Penicillin G OR Ampicillin | Ceftriaxone | 10-14 days |
| S. pneumoniae (PCN-I, MIC 0.1-1) | Ceftriaxone | Cefotaxime | 10-14 days |
| S. pneumoniae (PCN-R, MIC > 1) | Ceftriaxone + Vancomycin | Add rifampin | 10-14 days |
| N. meningitidis | Penicillin G OR Ceftriaxone | Chloramphenicol | 7 days |
| H. influenzae (β-lactamase neg) | Ampicillin | Ceftriaxone | 7-10 days |
| H. influenzae (β-lactamase pos) | Ceftriaxone | Cefotaxime | 7-10 days |
| Group B Streptococcus | Penicillin G ± Gentamicin | Ampicillin | 14-21 days |
| E. coli | Ceftriaxone OR Cefotaxime | Meropenem (ESBL) | 21 days |
| Listeria monocytogenes | Ampicillin ± Gentamicin | TMP-SMX | 21 days |
Adjunctive Dexamethasone
Rationale: Dexamethasone reduces the inflammatory response to bacterial lysis induced by antibiotics, thereby decreasing meningeal inflammation, cerebral edema, and neurological sequelae 12.
Evidence: The Cochrane meta-analysis (2015) demonstrated that dexamethasone:
- Reduces hearing loss (RR 0.67; 95% CI 0.51-0.88)
- Reduces neurological sequelae in high-income countries
- Most beneficial for H. influenzae meningitis
- Probable benefit for pneumococcal meningitis in children 12
Dosing Protocol:
| Parameter | Recommendation |
|---|---|
| Dose | 0.15 mg/kg/dose IV |
| Frequency | Every 6 hours |
| Duration | 4 days (some protocols: 2 days) |
| Timing | Before or with first antibiotic dose (ideally 15-20 min before) |
When to Use:
| Population | Recommendation | Evidence Level |
|---|---|---|
| Children > 6 weeks with bacterial meningitis | Recommended | Strong |
| Hib meningitis | Strongly recommended | High quality |
| Pneumococcal meningitis | Recommended | Moderate quality |
| Neonates | NOT recommended | Insufficient evidence |
| Already received antibiotics | Consider if less than 4 hours since first dose | Weak |
When to Discontinue:
- Confirmed viral meningitis
- Gram stain and culture negative after 48-72 hours in well child
- TB meningitis (use separate steroid protocol)
Supportive Care
Fluid Management
SIADH Management:
- SIADH occurs in 25-50% of bacterial meningitis
- Monitor serum sodium q6-12h initially
- Avoid hypotonic fluids
- Fluid restriction only if symptomatic hyponatremia
| Clinical State | Fluid Strategy |
|---|---|
| Euvolemic, stable | Isotonic maintenance (D5NS or D5LR) |
| Suspected SIADH | Isotonic fluids, consider 75% maintenance if Na less than 130 |
| Septic shock | Aggressive resuscitation (20 mL/kg boluses, repeat PRN) |
| Raised ICP | Avoid hyponatremia, consider hypertonic saline |
Seizure Management
| Medication | Dose | Notes |
|---|---|---|
| Lorazepam | 0.1 mg/kg IV (max 4 mg) | First-line for acute seizure |
| Midazolam | 0.2 mg/kg IM/IN | If no IV access |
| Levetiracetam | 40-60 mg/kg IV (max 3g) | Second-line, maintenance |
| Phenobarbital | 20 mg/kg IV | Neonates, refractory seizures |
| Phenytoin/Fosphenytoin | 20 mg PE/kg IV | Alternative second-line |
Prophylactic Anticonvulsants: Not routinely recommended unless seizures occur
Management of Raised ICP
| Intervention | Mechanism | Notes |
|---|---|---|
| Head elevation 30° | Improve venous drainage | First-line, non-invasive |
| Avoid hyperventilation | Short-term PCO₂ reduction | Only brief temporizing measure |
| Hypertonic saline (3%) | Osmotic effect | 3-5 mL/kg bolus, maintain Na 145-155 |
| Mannitol | Osmotic diuresis | 0.5-1 g/kg; monitor osmolar gap |
| Neurosurgical consultation | EVD if refractory | For hydrocephalus, refractory ICP |
Pain Management
| Medication | Dose | Notes |
|---|---|---|
| Paracetamol | 15 mg/kg q4-6h (max 75 mg/kg/day) | First-line antipyretic/analgesic |
| Ibuprofen | 10 mg/kg q6-8h | Avoid if dehydrated or renal impairment |
| Morphine | 0.05-0.1 mg/kg IV q2-4h | For severe pain; monitor respirations |
Duration of Therapy
| Organism | Duration | Notes |
|---|---|---|
| N. meningitidis | 5-7 days | Shortest duration 13 |
| H. influenzae | 7-10 days | Standard; recent meta-analysis supports shorter courses 13 |
| S. pneumoniae | 7-14 days | Shorter courses (7 days) non-inferior in uncomplicated cases 13 |
| Group B Streptococcus | 14-21 days | Risk of relapse with shorter courses |
| Listeria monocytogenes | 21 days | High relapse risk |
| Gram-negative bacilli | 21 days or longer | Poor CSF penetration |
| Unknown organism | 10-14 days | Based on clinical response |
Recent Evidence on Duration:
A 2023 systematic review and meta-analysis by Sudo et al. 13 evaluated 6 RCTs (1,333 children) comparing shorter (≤7 days) versus longer (10-14 days) antibiotic courses for bacterial meningitis. Key findings:
- No significant differences in treatment failure, relapse, mortality, or neurological complications
- Shorter therapy supported for uncomplicated meningitis due to S. pneumoniae, H. influenzae, and N. meningitidis
- Important for antimicrobial stewardship and reducing adverse effects
- Caution advised for complicated cases or infections by other pathogens
Complications
Acute Complications
Syndrome of Inappropriate ADH Secretion (SIADH)
| Feature | Details |
|---|---|
| Incidence | 25-50% of bacterial meningitis |
| Mechanism | Hypothalamic-pituitary dysfunction, meningeal inflammation |
| Diagnosis | Hyponatremia + euvolemia + urine osm > 100 + urine Na > 40 |
| Management | Fluid restriction (unless shocked), isotonic fluids |
| Resolution | Usually within 48-72 hours |
Cerebral Edema and Raised ICP
| Feature | Details |
|---|---|
| Incidence | 10-30% |
| Risk factors | Young age, delayed treatment, pneumococcal etiology |
| Monitoring | Clinical signs, ICP monitoring in severe cases |
| Complications | Herniation, brainstem compression |
Subdural Collections
| Type | Incidence | Management |
|---|---|---|
| Subdural effusion (sterile) | 10-30% (especially Hib) | Observation if asymptomatic |
| Subdural empyema | 1-2% | Neurosurgical drainage + antibiotics |
Indications for drainage:
- Mass effect with midline shift
- Increasing head circumference
- Persistent fever despite antibiotics
- Neurological deterioration
Vascular Complications
| Complication | Incidence | Mechanism |
|---|---|---|
| Arterial infarction | 5-15% | Vasculitis, thrombosis |
| Venous thrombosis | 3-5% | Dehydration, hypercoagulability |
| Hemorrhage | 1-3% | DIC, vascular injury |
Seizures
| Timing | Significance | Management |
|---|---|---|
| Early (≤72h) | Common (20-30%), not always prognostic | Acute treatment |
| Late (> 72h) | May indicate complication | Imaging, evaluate for abscess/infarct |
| Status epilepticus | Medical emergency | Aggressive treatment, ICU |
Long-term Sequelae
Hearing Loss
The most common long-term complication of bacterial meningitis in children.
| Pathogen | Incidence | Mechanism |
|---|---|---|
| S. pneumoniae | 20-35% | Labyrinthitis, cochlear damage |
| H. influenzae | 10-15% | Labyrinthitis |
| N. meningitidis | 5-10% | Less common |
| GBS (neonatal) | 10-20% | Cochlear damage |
Screening Protocol:
- All survivors: Audiology evaluation before hospital discharge
- Repeat at 1 month and 6 months
- Annual follow-up if abnormal
- Cochlear implant evaluation for severe bilateral loss
Role of Dexamethasone: Reduces hearing loss, particularly in Hib meningitis (NNT = 10-15) 12
Neurological Sequelae
| Sequela | Incidence | Risk Factors |
|---|---|---|
| Cognitive impairment | 10-20% | Young age, coma, seizures during illness |
| Motor deficits (hemiparesis, quadriparesis) | 5-10% | Vascular complications |
| Epilepsy | 5-10% | Seizures during acute illness, cortical damage |
| Behavioral/attention problems | 15-25% | School-age at diagnosis |
| Hydrocephalus (requiring shunt) | 3-5% | Gram-negative, delayed treatment |
Neonatal-Specific Predictors of Poor Outcome
A 2024 systematic review by Liu et al. 14 analyzed 20 studies (neonates less than 90 days) and identified key prognostic factors:
Most Consistently Associated with Poor Outcome (by multivariate analysis):
- Preterm birth/low birth weight: 2.14-26.27-fold increased risk of death (risk increases with degree of prematurity)
- Coma: 11.14-31.85-fold increased risk of death
- Elevated CSF protein: Variable cutoffs (1.88-5.0 g/L); associated with poor outcomes
- Seizures: Especially persistent seizures \u003e72h; associated with moderate/severe disability
Pathogen-Specific Outcomes:
- GBS: Mortality 7-14%; moderate/severe disability in 34% of survivors
- E. coli/Gram-negative: Higher mortality (28.6% vs 10.7%); higher sequelae rate (58% vs 35% for GBS)
- S. pneumoniae: Associated with serious CNS complications (OR 4.83) and death (OR 4.62)
Neuroimaging Predictors:
- Abnormal cerebral ultrasound: Associated with adverse motor outcome (OR 5.3)
- Extensive MRI lesions: Predict adverse cognitive (OR 7.0) and motor outcomes (OR 10.7-12.6)
Neurodevelopmental Follow-up
Recommended Surveillance Schedule:
| Age at Meningitis | Follow-up | Assessments |
|---|---|---|
| Neonatal | q3 months × 2 years, then annually | Developmental milestones, hearing, vision |
| Infant/Toddler | q6 months × 2 years, then annually | Developmental, hearing, behavior |
| School-age | Annually × 5 years | Academic performance, hearing, psychosocial |
Prevention
Vaccination
Conjugate Vaccines
| Vaccine | Target | Schedule (US/UK) | Effectiveness |
|---|---|---|---|
| Hib conjugate | H. influenzae type b | 2, 4, 6 months + booster | > 99% reduction |
| PCV13/PCV15/PCV20 | 13/15/20 pneumococcal serotypes | 2, 4, 6 months + booster | 80-90% against vaccine serotypes |
| MenACWY | N. meningitidis A, C, W, Y | 11-12 years + booster | 85-100% |
| MenB | N. meningitidis serogroup B | 10+ years (varies by country) | 80-95% |
Catch-up and High-Risk Schedules
| Risk Group | Additional Vaccination |
|---|---|
| Asplenia (functional/anatomic) | Complete all meningococcal vaccines, PCV, Hib |
| Complement deficiency | MenACWY + MenB, boosters q5 years |
| HIV infection | Additional PCV doses |
| Cochlear implant recipients | Additional PCV doses |
| Travel to endemic areas | MenACWY before travel |
Chemoprophylaxis
Meningococcal Disease Contacts
Close contacts of meningococcal disease require prophylaxis within 24 hours of case identification
Definition of Close Contact:
- Household members
- Day care/school contacts (close proximity)
- Anyone with direct exposure to oral secretions (kissing, sharing utensils)
- Healthcare workers with unprotected exposure to respiratory secretions
- Airplane passengers sitting directly next to case for > 8 hours
Prophylaxis Regimens:
| Agent | Dose | Duration | Notes |
|---|---|---|---|
| Rifampin | 10 mg/kg q12h (max 600 mg) | 2 days | First-line for children |
| Ceftriaxone | 125 mg IM (≤15 years); 250 mg IM (> 15 years) | Single dose | Preferred for pregnancy |
| Ciprofloxacin | 20 mg/kg (max 500 mg) PO | Single dose | > 1 month age |
| Azithromycin | 10 mg/kg (max 500 mg) PO | Single dose | Alternative |
Hib Disease Contacts
Indications for Prophylaxis:
- Household with at least one contact less than 4 years who is unimmunized/under-immunized
- Household with immunocompromised individual
- Day care/nursery with two or more cases within 60 days
Regimen:
- Rifampin 20 mg/kg/day (max 600 mg) PO once daily × 4 days
Intrapartum Prophylaxis for GBS
| Risk Factor | Indication for IAP |
|---|---|
| Previous infant with invasive GBS disease | Always |
| GBS bacteriuria during current pregnancy | Always |
| Positive GBS screening (35-37 weeks) | Always |
| Unknown GBS status + any of: | Consider |
| - Delivery less than 37 weeks | |
| - Rupture of membranes > 18 hours | |
| - Intrapartum temperature ≥38°C |
IAP Regimen:
- Penicillin G 5 million units IV, then 2.5-3 million units q4h until delivery
- Alternative: Ampicillin 2g IV, then 1g q4h
Special Populations
Neonates
Unique Considerations:
| Factor | Implication |
|---|---|
| Immature blood-brain barrier | Easier pathogen entry |
| Deficient complement | Poor opsonization |
| Low immunoglobulin levels | Reduced humoral immunity |
| Decreased neutrophil function | Impaired bacterial killing |
| Non-specific presentation | Low threshold for workup |
HSV Meningitis/Meningoencephalitis:
Always consider HSV in neonates with meningitis, especially:
- Maternal history of genital herpes
- Vesicular rash
- Seizures
- CSF pleocytosis with negative bacterial cultures
- Hepatitis, coagulopathy
Management:
- Acyclovir 20 mg/kg IV q8h empirically for all neonates with suspected meningitis
- Continue for 21 days if HSV CNS disease confirmed
- Repeat LP at end of treatment to confirm negative HSV PCR
Immunocompromised Children
| Condition | Additional Pathogens | Modified Empiric Therapy |
|---|---|---|
| HIV/AIDS | Cryptococcus, TB, CMV, toxoplasmosis | Add amphotericin B, consider TB coverage |
| Post-transplant | Listeria, fungi, CMV | Add ampicillin, consider antifungals |
| Neutropenia | Pseudomonas, Aspergillus | Anti-pseudomonal coverage |
| Primary immunodeficiency | Cryptococcus, Mycobacteria | Individualized approach |
| Post-splenectomy | Encapsulated organisms | Aggressive coverage, even for "minor" fevers |
VP Shunt-Associated Meningitis
Common Organisms:
- Coagulase-negative staphylococci (50%)
- S. aureus (25%)
- Gram-negative bacilli (15-20%)
- Propionibacterium acnes (5%)
Management:
- Empiric: Vancomycin + Ceftazidime or Meropenem
- Shunt externalization or removal often required
- CSF obtained from shunt tap (by neurosurgery)
- Intraventricular antibiotics may be needed
Post-Neurosurgical Meningitis
Organisms:
- Gram-negative bacilli (Pseudomonas, Acinetobacter)
- S. aureus, coagulase-negative staphylococci
- Resistant organisms more common
Empiric Therapy:
- Vancomycin + Meropenem or Cefepime
- Consider intrathecal/intraventricular therapy for resistant organisms
Monitoring and Follow-up
Inpatient Monitoring
Clinical Parameters
| Parameter | Frequency | Target |
|---|---|---|
| Vital signs | q2-4h initially | Temperature normalizing, stable hemodynamics |
| Neurological assessment | q4-6h | GCS improving, no new deficits |
| Head circumference (infants) | Daily | No rapid increase |
| Fluid balance | q8-12h | Adequate output, no overload |
Laboratory Monitoring
| Test | Frequency | Purpose |
|---|---|---|
| Serum sodium | q6-12h initially, then daily | SIADH detection |
| CRP | Day 0, 2, and before discharge | Response to treatment |
| Blood cultures | Day 0; repeat if febrile > 48h on antibiotics | Clearance confirmation |
| CSF (repeat LP) | Not routine; see indications below |
Indications for Repeat LP:
| Indication | Timing |
|---|---|
| Poor clinical response at 48-72 hours | As needed |
| GNR meningitis (to confirm sterilization) | 48-72 hours |
| Persistent fever | Case-by-case |
| Neonatal meningitis | Consider at end of treatment |
| Concern for secondary complication | As indicated |
Discharge Criteria
| Criterion | Requirement |
|---|---|
| Clinical stability | Afebrile ≥24-48h, improving mental status |
| Oral intake | Tolerating oral fluids/feeds |
| Antibiotics | Completion plan established (may include outpatient IV) |
| Hearing evaluation | Audiology assessment completed or scheduled |
| Follow-up | Appointments arranged (pediatrics, audiology, neurology if indicated) |
| Parent education | Red flags reviewed, medication instructions clear |
Outpatient Follow-up Schedule
| Visit | Timing | Assessments |
|---|---|---|
| Post-discharge | 1-2 weeks | Clinical recovery, medication completion |
| Audiology | Before discharge, repeat 1 month, 6 months | Hearing evaluation |
| Neurology | 4-6 weeks if neurological complications | Developmental assessment |
| Developmental | 3-6 months | Milestone review |
| School-age | Annually for 5 years | Academic/behavioral screening |
Disposition
Admission Criteria
All suspected bacterial meningitis requires hospital admission.
ICU Admission Criteria
| Indication | Rationale |
|---|---|
| Altered consciousness (GCS ≤12) | Airway protection, ICP monitoring |
| Hemodynamic instability/shock | Vasopressor support |
| Respiratory failure | Mechanical ventilation |
| Status epilepticus | Aggressive seizure management |
| Signs of raised ICP/herniation | ICP management, neurosurgery |
| Rapidly evolving purpura/DIC | Critical care support |
| Neonates with meningitis | Higher monitoring needs |
General Ward Admission
| Indication | Criteria |
|---|---|
| Confirmed/suspected bacterial meningitis | GCS > 12, stable hemodynamics |
| Viral meningitis requiring IV therapy | Significant symptoms, unable to tolerate oral |
Outpatient Management
Viral meningitis may be managed as outpatient if:
- Child > 1 year of age
- Well-appearing, tolerating oral intake
- No toxic appearance or altered consciousness
- Confirmed enterovirus or other benign viral etiology
- Reliable caregiver with good access to healthcare
- Clear return precautions provided
Patient and Family Education
Explaining the Diagnosis
For Bacterial Meningitis:
"Your child has an infection called meningitis - this means bacteria have gotten into the fluid surrounding the brain and spinal cord. This is a very serious infection that needs strong antibiotics given through an IV. We're starting treatment right away because quick treatment gives your child the best chance of recovery and reduces the risk of complications."
For Viral Meningitis:
"Your child has viral meningitis. While this sounds scary, viral meningitis is usually much milder than bacterial meningitis. The symptoms - headache, fever, stiff neck - should improve over the next week or two. We may not need to give antibiotics since this is caused by a virus, but we'll watch closely to make sure they're recovering well."
Red Flags for Parents
Return immediately if:
- Worsening headache or confusion
- New seizures
- Increasing drowsiness or difficulty waking
- New rash that doesn't blanch with pressure
- Weakness in arms or legs
- Stiff neck getting worse
- High fever returning after improvement
- Poor feeding, vomiting everything
Long-term Outlook Counseling
Hearing:
- "We will test your child's hearing before leaving the hospital and again in the coming months. Some children who have meningitis develop hearing problems, and early detection helps us provide the right support."
Development:
- "Most children recover fully, but we recommend follow-up appointments to make sure your child is meeting developmental milestones. Let us know if you notice any concerns about their learning, behavior, or movement."
Infection Control Education
For Meningococcal Disease:
- Close contacts need preventive antibiotics
- Public health will be notified and assist with contact tracing
- Isolation precautions in hospital for first 24 hours of antibiotic treatment
Quality Metrics and Documentation
Key Performance Indicators
| Metric | Target | Rationale |
|---|---|---|
| Door-to-antibiotic time | ≤60 minutes | Mortality reduction |
| Blood cultures before antibiotics | > 90% | Pathogen identification |
| Dexamethasone given with/before first antibiotic dose | > 80% | Hearing loss prevention |
| LP performed (if no contraindication) | > 95% | Diagnostic accuracy |
| Audiology referral before discharge | 100% | Hearing loss detection |
| Follow-up appointment scheduled | 100% | Continuity of care |
Documentation Checklist
| Element | Required Documentation |
|---|---|
| Initial assessment | Time of presentation, initial GCS, vital signs, meningeal signs |
| Antibiotic timing | Exact time of first antibiotic dose |
| Dexamethasone | Dose, timing relative to antibiotics |
| LP details | Time, opening pressure, complications, appearance |
| CSF results | Cell count, differential, protein, glucose, Gram stain |
| Clinical response | Daily neurological assessment, fever curve |
| Complications | Seizures, SIADH, new focal signs |
| Hearing screening | Audiology results and follow-up plan |
| Discharge planning | Duration of antibiotics, follow-up appointments, return precautions |
Clinical Pearls
Diagnostic Pearls
-
"Treat first, diagnose second": Never delay antibiotics for diagnostic tests in suspected bacterial meningitis
-
Classic triad is unreliable in young children: Fever may be the only sign in infants; maintain high index of suspicion
-
LP interpretation requires serum glucose: Always send concurrent serum glucose for CSF:serum ratio
-
Negative Gram stain does not exclude bacterial meningitis: Sensitivity only 60-90%; prior antibiotics further reduce yield
-
CSF multiplex PCR is a game-changer: Rapid pathogen identification even after antibiotic administration 11
-
Procalcitonin > 0.5 ng/mL strongly suggests bacterial infection: Helps distinguish from viral, but don't delay treatment for result 9
Treatment Pearls
-
Dexamethasone timing is crucial: Give before or with first antibiotic dose for maximum benefit; little benefit after first dose 12
-
Vancomycin is not routine in all ages: Only needed > 3 months for resistant pneumococcus coverage
-
Ampicillin required until 3 months: Listeria coverage essential in young infants
-
Never forget HSV in neonates: Acyclovir should be part of empiric therapy for all neonates with suspected meningitis
-
SIADH is common: Monitor sodium closely; avoid hypotonic fluids
Disposition Pearls
-
Hearing test before discharge is mandatory: Sensorineural hearing loss is the most common permanent sequela
-
Meningococcal contacts need prophylaxis within 24 hours: Public health must be notified immediately
-
Viral meningitis in older children can be managed outpatient: If well-appearing, tolerating oral, with reliable follow-up
VIVA/OSCE Scenarios
Scenario 1: Neonatal Meningitis
Stem: A 14-day-old male infant presents with fever (38.5°C), irritability, and poor feeding for 12 hours. He was born at term via SVD with no maternal complications. On examination, he is lethargic with a bulging fontanelle.
Key Points for Discussion:
- Immediate actions: Stabilize, IV access, blood cultures, LP (if no contraindications), empiric antibiotics
- Empiric therapy: Ampicillin + Cefotaxime + Acyclovir (always cover HSV in neonates)
- Age-specific pathogens: GBS, E. coli K1, Listeria, HSV
- Neonatal CSF norms differ from older children (higher WBC, protein acceptable)
- Duration of therapy: 14-21 days for GBS, 21 days for GNR
Scenario 2: Meningococcal Septicemia
Stem: A 4-year-old previously healthy girl presents to ED with 6-hour history of fever and progressive non-blanching rash. She is drowsy with HR 160, BP 70/40, and spreading purpura.
Key Points for Discussion:
- Immediate IM/IV antibiotics (ceftriaxone) - do not delay for IV access
- Fluid resuscitation: 20 mL/kg boluses, prepare for vasopressors
- This is meningococcemia until proven otherwise - life-threatening emergency
- Contraindication to LP: hemodynamic instability
- Contact prophylaxis within 24 hours
- Complications: DIC, purpura fulminans, adrenal hemorrhage (Waterhouse-Friderichsen)
Scenario 3: LP Interpretation
Stem: A 2-year-old with 2 days of fever and vomiting has LP showing: WBC 850 cells/μL (85% PMN), protein 95 mg/dL, glucose 35 mg/dL (serum glucose 100 mg/dL), Gram stain showing Gram-positive diplococci.
Key Points for Discussion:
- CSF findings consistent with bacterial meningitis
- Likely organism: Streptococcus pneumoniae (Gram-positive diplococci)
- CSF:serum glucose ratio = 0.35 (less than 0.4 suggests bacterial)
- Treatment: Ceftriaxone + Vancomycin (until sensitivities known) + Dexamethasone
- Hearing screening essential (highest risk with pneumococcal disease)
Scenario 4: Fever Without Source in Infant
Stem: A 6-week-old infant presents with fever (38.2°C) for 4 hours. Mother reports no other symptoms. Baby is feeding well. On examination, the infant appears well but has no focus of infection.
Key Points for Discussion:
- "Well-appearing" doesn't exclude serious bacterial infection in young infants
- Rochester/Philadelphia/Boston criteria for low-risk fever without source
- Full sepsis workup indicated in this age group (blood culture, urine, LP)
- Empiric antibiotics after cultures if admitted
- This age group at risk for both "neonatal" and "community" pathogens
References
Related Topics
- Neonatal Sepsis
- Febrile Seizures in Children
- Pediatric Sepsis and Septic Shock
- Viral Encephalitis
- Fever Without Source in Infants
- Lumbar Puncture Technique
Footnotes
-
Proulx N, Fréchette D, Toye B, et al. Delays in the administration of antibiotics are associated with mortality from adult acute bacterial meningitis. QJM. 2005;98(4):291-298. doi:10.1093/qjmed/hci047 ↩
-
van de Beek D, Cabellos C, Dzupova O, et al. ESCMID guideline: diagnosis and treatment of acute bacterial meningitis. Clin Microbiol Infect. 2016;22 Suppl 3:S37-S62. doi:10.1016/j.cmi.2016.01.007 ↩
-
Thigpen MC, Whitney CG, Messonnier NE, et al. Bacterial meningitis in the United States, 1998-2007. N Engl J Med. 2011;364(21):2016-2025. doi:10.1056/NEJMoa1005384 ↩
-
Collaborators GBDMeningitis. Global, regional, and national burden of meningitis, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17(12):1061-1082. doi:10.1016/S1474-4422(18)30387-9 ↩
-
Tunkel AR, Hartman BJ, Kaplan SL, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis. 2004;39(9):1267-1284. doi:10.1086/425368 ↩
-
Rennick G, Shann F, de Campo J. Cerebral herniation during bacterial meningitis in children. BMJ. 1993;306(6883):953-955. doi:10.1136/bmj.306.6883.953 ↩
-
Nigrovic LE, Kuppermann N, Macias CG, et al. Clinical prediction rule for identifying children with cerebrospinal fluid pleocytosis at very low risk of bacterial meningitis. JAMA. 2007;297(1):52-60. doi:10.1001/jama.297.1.52 ↩ ↩2
-
Martin NG, Defres S, Willis L, et al. Paediatric meningitis in the conjugate vaccine era and a novel clinical decision model to predict bacterial aetiology. J Infect. 2024;88(5):106145. doi:10.1016/j.jinf.2024.106145 ↩ ↩2
-
Dubos F, Korczowski B, Aygun DA, et al. Serum procalcitonin level and other biological markers to distinguish between bacterial and aseptic meningitis in children: a European multicenter case cohort study. Arch Pediatr Adolesc Med. 2008;162(12):1157-1163. doi:10.1001/archpedi.162.12.1157 ↩ ↩2
-
Castagno E, Aguzzi S, Rossi L, et al. Clinical predictors and biomarkers in children with sepsis and bacterial meningitis. Pediatr Emerg Care. 2023;39(5):311-317. doi:10.1097/PEC.0000000000002865 ↩
-
Leber AL, Everhart K, Balada-Llasat JM, et al. Multicenter Evaluation of BioFire FilmArray Meningitis/Encephalitis Panel for Detection of Bacteria, Viruses, and Yeast in Cerebrospinal Fluid Specimens. J Clin Microbiol. 2016;54(9):2251-2261. doi:10.1128/JCM.00730-16 ↩ ↩2
-
Brouwer MC, McIntyre P, Prasad K, van de Beek D. Corticosteroids for acute bacterial meningitis. Cochrane Database Syst Rev. 2015;2015(9):CD004405. doi:10.1002/14651858.CD004405.pub5 ↩ ↩2 ↩3 ↩4
-
Sudo RYU, Câmara MCC, Kieling SV, et al. Shorter versus longer duration of antibiotic treatment in children with bacterial meningitis: a systematic review and meta-analysis. Eur J Pediatr. 2024;183(1):61-71. doi:10.1007/s00431-023-05275-8 ↩ ↩2 ↩3 ↩4
-
Liu Y, Feng Y, Guo Y, et al. Clinical predictors of poor outcome of bacterial meningitis in infants less than 90 days: a systematic review. Front Pediatr. 2024;12:1414778. doi:10.3389/fped.2024.1414778 ↩