Polycystic Ovary Syndrome (PCOS)
PCOS is not simply an ovarian disorder but a systemic metabolic condition with far-reaching health implications. Women with PCOS face substantially elevated risks of type 2 diabetes mellitus (4-fold increase),...
What matters first
PCOS is not simply an ovarian disorder but a systemic metabolic condition with far-reaching health implications. Women with PCOS face substantially elevated risks of type 2 diabetes mellitus (4-fold increase),...
Rapid Virilisation (Androgen Secreting Tumour)
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Rapid Virilisation (Androgen Secreting Tumour)
- Post-menopausal Bleeding (Endometrial Cancer)
- Severe Abdominal Pain (Ovarian Hyperstimulation/Torsion)
- Testosterone less than 5 nmol/L (Adrenal/Ovarian Tumour)
Linked comparisons
Differentials and adjacent topics worth opening next.
- Congenital Adrenal Hyperplasia
- Hypothalamic Amenorrhoea
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Polycystic Ovary Syndrome
1. Clinical Overview
Polycystic Ovary Syndrome (PCOS) is the most common endocrinological disorder affecting women of reproductive age, with a prevalence of 8-13% globally. [1] It represents a complex, heterogeneous syndrome characterised by reproductive, metabolic, and psychological manifestations. The syndrome is fundamentally driven by insulin resistance and hyperandrogenism, resulting in chronic anovulation, polycystic ovarian morphology, and a constellation of metabolic disturbances. [2]
PCOS is not simply an ovarian disorder but a systemic metabolic condition with far-reaching health implications. Women with PCOS face substantially elevated risks of type 2 diabetes mellitus (4-fold increase), cardiovascular disease, endometrial cancer, obstructive sleep apnoea, and mental health disorders. [3,4] The condition significantly impacts quality of life through fertility challenges, cosmetic concerns (hirsutism, acne, alopecia), and psychological burden.
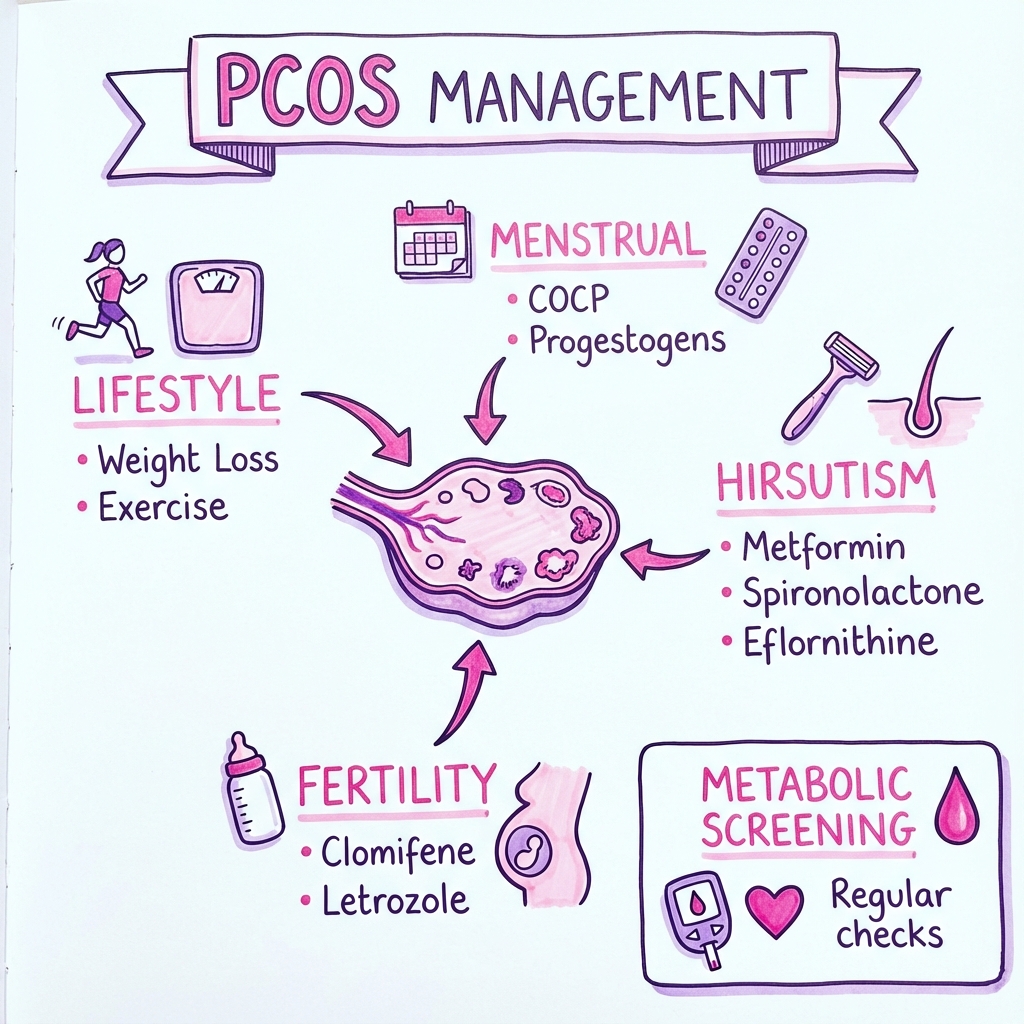
Management is individualised and symptom-directed, focusing on metabolic optimisation (weight loss, insulin sensitisation), menstrual regulation (endometrial protection), hyperandrogenism control, and fertility restoration when desired. [5] Lifestyle modification remains the cornerstone of first-line therapy across all presentations.
Clinical Pearls
The "String of Pearls" Sign: The classic transvaginal ultrasound appearance shows ≥20 small (2-9mm) antral follicles arranged peripherally around an echogenic, hyperplastic ovarian stroma. However, in the era of high-resolution ultrasound, up to 25% of normal ovulatory women may exhibit polycystic ovarian morphology (PCOM). PCOM alone does not equal PCOS syndrome. [6]
Insulin is the Master Regulator: Hyperinsulinaemia stimulates ovarian theca cells to produce excess androgens via upregulation of cytochrome P450c17α activity. Simultaneously, insulin suppresses hepatic production of sex hormone-binding globulin (SHBG), increasing the bioavailability of free testosterone. This dual mechanism explains why weight loss (reducing insulin levels) is the most effective treatment modality. [7]
Virilisation vs Hirsutism - A Critical Distinction:
- Hirsutism: Excessive terminal hair in male-pattern distribution (Ferriman-Gallwey score ≥8). Compatible with PCOS.
- Virilisation: Deep voice, clitoromegaly (> 1cm), male-pattern baldness, increased muscle mass, decreased breast size. This is NOT typical of PCOS and mandates urgent investigation for androgen-secreting tumour (ovarian or adrenal). Total testosterone > 5 nmol/L (> 150 ng/dL) is a critical red flag requiring imaging. [8]
2. Epidemiology
Prevalence and Incidence
| Statistic | Value | Population | Source |
|---|---|---|---|
| Overall prevalence | 8-13% | Reproductive-age women | [1] |
| Prevalence (Rotterdam criteria) | 10-18% | Varies by diagnostic criteria | [9] |
| Prevalence (adolescents) | 3-11% | Post-menarchal girls | [10] |
| Annual incidence | 5-10 per 1000 | Women aged 15-44 years | [11] |
Demographics and Risk Factors
Age Distribution:
- Peak presentation: 20-30 years (reproductive age)
- Symptoms may begin in adolescence (irregular menses, acne, hirsutism)
- Symptoms often improve post-menopause as androgen levels decline naturally [12]
Ethnic Variations:
- South Asian women: Higher prevalence (20-25%), more severe insulin resistance, earlier onset of metabolic complications [13]
- African-American women: Higher prevalence of hirsutism, obesity, and metabolic syndrome [14]
- Caucasian women: Classic presentation with moderate metabolic risk
- East Asian women: Lower rates of hirsutism despite similar androgen levels (lower 5α-reductase activity) [15]
Key Risk Factors:
- Genetic predisposition: 20-40% of first-degree relatives affected (sisters, mothers) [16]
- Obesity: Present in 40-80% of PCOS patients; exacerbates insulin resistance [17]
- Premature adrenarche: Early development of pubic/axillary hair (less than 8 years) [18]
- Intrauterine environment: Low birth weight or macrosomia; maternal hyperandrogenism [19]
- Lifestyle factors: Sedentary behaviour, high glycaemic index diet [20]
3. Aetiology and Pathophysiology
Primary Mechanisms
PCOS is a multifactorial disorder arising from complex interactions between genetic, metabolic, and environmental factors. The pathophysiology involves dysfunction across multiple systems:
1. Hypothalamic-Pituitary Axis Dysregulation
LH Hypersecretion:
- Increased gonadotropin-releasing hormone (GnRH) pulse frequency results in elevated luteinising hormone (LH) secretion
- LH:FSH ratio typically > 2:1 (though not diagnostic) [21]
- High LH drives excess androgen production by ovarian theca cells
- Reduced FSH impairs follicular maturation, causing follicular arrest at 5-10mm stage [22]
2. Ovarian Dysfunction
Hyperandrogenism:
- Theca cells overproduce androgens (androstenedione, testosterone) in response to LH stimulation
- Enhanced expression and activity of steroidogenic enzymes (CYP17A1, 3β-HSD)
- Granulosa cell dysfunction impairs conversion of androgens to oestrogens [23]
Follicular Arrest:
- High intra-ovarian androgen concentrations inhibit follicular development
- Accumulation of arrested antral follicles creates polycystic morphology
- Anovulation results from failure of dominant follicle selection [24]
3. Insulin Resistance and Hyperinsulinaemia
Present in 50-70% of women with PCOS, independent of obesity (though obesity exacerbates insulin resistance): [25]
Mechanisms:
- Post-receptor defect in insulin signalling pathway (serine phosphorylation of insulin receptor substrate-1)
- Reduced glucose transporter-4 (GLUT-4) translocation
- Peripheral insulin resistance with compensatory hyperinsulinaemia [26]
Metabolic Consequences:
- Ovarian: Insulin acts synergistically with LH to stimulate androgen production
- Hepatic: Suppressed SHBG synthesis → increased free androgen bioavailability
- Adipose: Impaired glucose uptake, increased lipolysis, dyslipidaemia
- Systemic: Increased risk of type 2 diabetes, metabolic syndrome, cardiovascular disease [27]
4. Androgen Excess
Sources:
- Ovarian (60%): Testosterone, androstenedione
- Adrenal (20%): Dehydroepiandrosterone sulphate (DHEAS)
- Peripheral (20%): Conversion of androgens to dihydrotestosterone (DHT) by 5α-reductase in skin [28]
Clinical Effects:
- Hirsutism (excessive terminal hair growth)
- Acne (sebaceous gland stimulation)
- Androgenic alopecia (frontal/vertex thinning)
- Disruption of normal follicular development [29]
5. Chronic Low-Grade Inflammation
Elevated markers of inflammation (CRP, IL-6, TNF-α) are consistently found in PCOS, contributing to:
- Insulin resistance
- Endothelial dysfunction
- Atherosclerosis progression
- Potentially contributing to anovulation [30]
Exam Detail: Molecular Pathophysiology - Viva Level:
The "two-hit" hypothesis proposes that PCOS arises from:
- Genetic susceptibility: Polymorphisms in genes regulating insulin action, steroid synthesis, and gonadotropin secretion
- Environmental trigger: Obesity, dietary factors, sedentary lifestyle
Key Genetic Loci Implicated (GWAS studies): [31]
- DENND1A (ovarian androgen biosynthesis)
- THADA (insulin secretion)
- INSR (insulin receptor function)
- FSH receptor polymorphisms
The Vicious Cycle:
Insulin Resistance → Hyperinsulinaemia
↓ ↓
↓ SHBG Ovarian Androgen ↑
↓ ↓
Free Androgens ↑ ← ← ← ← ┘
↓
Follicular Arrest → Anovulation
↓
Obesity (weight gain from anovulation)
↓
Worsened Insulin Resistance
Phenotypic Heterogeneity
PCOS manifests as four distinct phenotypes based on Rotterdam criteria combinations: [32]
| Phenotype | Features | Prevalence | Metabolic Risk |
|---|---|---|---|
| A (Classic) | Hyperandrogenism + Ovulatory dysfunction + PCOM | 50-60% | Highest |
| B (Classic) | Hyperandrogenism + Ovulatory dysfunction | 15-20% | High |
| C (Ovulatory) | Hyperandrogenism + PCOM | 15-20% | Moderate |
| D (Non-hyperandrogenic) | Ovulatory dysfunction + PCOM | 10-15% | Lower |
4. Diagnostic Criteria
Rotterdam Criteria (2003) - International Consensus
Diagnosis requires 2 out of 3 features (after exclusion of other causes): [33]
-
Oligo-ovulation or Anovulation:
- Menstrual cycles > 35 days apart OR
- less than 8 menses per year OR
- Amenorrhoea (absence of menses for ≥3 months)
-
Hyperandrogenism (Clinical or Biochemical):
- Clinical: Hirsutism (Ferriman-Gallwey score ≥8), acne, androgenic alopecia
- Biochemical: Elevated total testosterone, free testosterone, or Free Androgen Index (FAI)
-
Polycystic Ovarian Morphology (PCOM):
- ≥20 follicles (2-9mm diameter) per ovary OR
- Ovarian volume > 10cm³ (using prolate ellipsoid formula: 0.5 × L × W × D)
- Best assessed via transvaginal ultrasound (days 3-5 of cycle if menstruating)
Alternative Markers (2023 Update): [34]
- Anti-Müllerian Hormone (AMH) > 35 pmol/L can replace PCOM on ultrasound in adults
- AMH particularly useful when transvaginal ultrasound unavailable or inappropriate
Exclusion Criteria
Must exclude other causes of hyperandrogenism and ovulatory dysfunction:
- Thyroid dysfunction (TSH)
- Hyperprolactinaemia (Prolactin)
- Non-classic congenital adrenal hyperplasia (17-OH Progesterone)
- Androgen-secreting tumours (if testosterone > 5 nmol/L)
- Cushing's syndrome (if clinical features present)
- Acromegaly (if clinical features present) [35]
Special Diagnostic Considerations
Adolescents:
- Irregular menses are physiological for 2-3 years post-menarche
- PCOM is common in adolescence
- Diagnosis requires persistent oligomenorrhoea + hyperandrogenism
- Avoid labelling PCOS diagnosis less than 2 years post-menarche [36]
Post-menopausal Women:
- Hyperandrogenism typically declines
- Historical diagnosis based on reproductive-age features
- Metabolic complications persist and worsen [37]
5. Clinical Presentation
Cardinal Features
Menstrual Irregularity (70-80% of patients)
- Oligomenorrhoea: Cycles > 35 days apart
- Amenorrhoea: Absence of menses ≥3 months
- Dysfunctional uterine bleeding: Irregular heavy bleeding (unopposed oestrogen)
- May have regular cycles in 20-30% (ovulatory PCOS phenotype) [38]
Hyperandrogenism (60-80% of patients)
Hirsutism (most common):
- Excessive terminal hair in androgen-sensitive areas (face, chest, abdomen, thighs)
- Quantified by Ferriman-Gallwey Score (9 body areas, scored 0-4; total > 8 abnormal)
- Ethnicity-dependent: less common in East Asian women despite equivalent androgen levels [39]
Acne:
- Persistent acne beyond adolescence
- Predominantly affecting face, chest, upper back
- Often resistant to conventional topical therapy [40]
Androgenic Alopecia:
- Frontal and vertex thinning (Ludwig pattern I-III)
- Progressive hair loss in 20-40% of patients [41]
Infertility (40-75% seeking fertility treatment)
- Anovulation is the primary mechanism
- PCOS accounts for 70-80% of anovulatory infertility
- Conception rates approach normal with ovulation induction [42]
Associated Features
Metabolic Manifestations
Obesity:
- Present in 40-80% (varies by ethnicity and geography)
- Predominantly central/abdominal distribution
- BMI > 30 kg/m² in 50-70% of affected women [43]
Acanthosis Nigricans:
- Hyperpigmented, velvety skin thickening
- Typically in neck folds, axillae, groin, under breasts
- Marker of severe insulin resistance [44]
Metabolic Syndrome (prevalence 30-47%):
- Central obesity (waist circumference > 80cm)
- Hypertension (> 130/85 mmHg)
- Dyslipidaemia (↓HDL, ↑triglycerides)
- Impaired fasting glucose or type 2 diabetes [45]
Psychological Impact
- Depression: 2-4 fold increased risk [46]
- Anxiety disorders: Significantly elevated prevalence
- Body image disturbance: Related to hirsutism, obesity, acne
- Reduced quality of life: Across multiple domains
- Eating disorders: Higher rates of binge eating disorder [47]
6. Differential Diagnosis
| Condition | LH | FSH | Testosterone | DHEAS | 17-OHP | Other Key Features |
|---|---|---|---|---|---|---|
| PCOS | ↑/N | N | ↑ (1.5-4 nmol/L) | N/↑ | N | PCOM, insulin resistance |
| Hypothalamic Amenorrhoea | ↓ | ↓ | N | N | N | Low BMI, stress, excessive exercise |
| Premature Ovarian Insufficiency | ↑↑ | ↑↑ | N | N | N | Age less than 40, menopausal symptoms |
| Hyperprolactinaemia | N | N | N | N | N | ↑Prolactin, galactorrhoea |
| Non-classic CAH | N | N | ↑ | ↑↑ | ↑↑ | 17-OHP > 6 nmol/L, salt-wasting rare |
| Androgen-secreting Tumour | N | N | ↑↑↑ (> 5 nmol/L) | ↑↑↑ | N | Rapid virilisation, pelvic/adrenal mass |
| Cushing's Syndrome | N | N | ↑ | ↑ | N | Moon facies, striae, hypertension |
| Thyroid Dysfunction | N | N | N | N | N | Abnormal TSH, specific thyroid symptoms |
Key Differentiating Points
Age of Onset:
- PCOS: Post-menarchal, reproductive age
- POI: less than 40 years with cessation of previously regular cycles
- Hypothalamic amenorrhoea: Association with recent stressor (weight loss, athletics)
Severity of Virilisation:
- PCOS: Hirsutism ± mild acne
- Tumour: Rapid progression, clitoromegaly, voice deepening, male-pattern baldness
Hormonal Clues:
- LH:FSH > 3:1 → Consider PCOS
- FSH > 25 IU/L → Consider POI (confirm with repeat in 4-6 weeks)
- Testosterone > 5 nmol/L → Urgent imaging for adrenal/ovarian tumour
- 17-OHP > 6 nmol/L → ACTH stimulation test for non-classic CAH [48]
7. Investigations
First-Line Investigations
Hormonal Assessment (Day 2-5 of menstrual cycle if present):
| Investigation | Purpose | Interpretation |
|---|---|---|
| FSH, LH | Assess ovarian function | LH often elevated; LH:FSH > 2:1 suggestive (not diagnostic) |
| Testosterone (total) | Assess hyperandrogenism | 1.5-4 nmol/L typical; > 5 nmol/L requires tumour exclusion |
| SHBG | Calculate Free Androgen Index | Usually low due to insulin suppression |
| Free Androgen Index | FAI = (Total T ÷ SHBG) × 100 | > 5 suggests hyperandrogenism |
| TSH | Exclude thyroid dysfunction | Rule out hypo/hyperthyroidism |
| Prolactin | Exclude hyperprolactinaemia | Mild elevation can occur with PCOS; > 1000 mIU/L suggests prolactinoma |
| 17-OH Progesterone | Exclude non-classic CAH | less than 6 nmol/L excludes; > 6 nmol/L requires ACTH stimulation test |
Metabolic Assessment:
| Investigation | Purpose | Frequency |
|---|---|---|
| HbA1c | Screen for diabetes/prediabetes | Baseline, then annually |
| Fasting glucose | Alternative to HbA1c | If HbA1c unavailable |
| Oral Glucose Tolerance Test | Assess glucose metabolism | If HbA1c 39-47 mmol/mol |
| Lipid profile | Assess cardiovascular risk | Baseline, then every 2-5 years |
| Blood pressure | Screen for hypertension | Every visit |
| BMI and waist circumference | Assess obesity and distribution | Every visit |
Second-Line Investigations
Imaging:
- Transvaginal ultrasound: ≥20 follicles per ovary OR ovarian volume > 10cm³
- Perform days 3-5 of cycle (if menstruating)
- Avoid if recent hormonal contraception use (less than 3 months)
- Transabdominal if transvaginal not appropriate (adolescents, no sexual activity) [49]
Advanced Hormonal Tests:
- AMH (Anti-Müllerian Hormone): > 35 pmol/L supports PCOS (correlates with PCOM)
- DHEAS: Elevated in 20-30%; if very high (> 19 μmol/L), consider adrenal imaging
- 24-hour urinary free cortisol: If clinical features of Cushing's syndrome [50]
Additional Metabolic Tests:
- Liver function tests: NAFLD is common (30-50% of PCOS patients)
- Sleep study: If symptoms of obstructive sleep apnoea (OSA)
Investigations to Avoid
❌ LH:FSH ratio: Not required for diagnosis; supportive but not diagnostic ❌ Routine insulin levels: Not standardised; insulin resistance clinically evident ❌ Routine pelvic MRI: Unnecessary unless adnexal mass on ultrasound
Exam Detail: Interpretation Pearls - Viva Level:
Borderline 17-OHP (3-6 nmol/L):
- Perform ACTH stimulation test
- Non-classic CAH diagnosed if 17-OHP > 30 nmol/L at 60 minutes post-Synacthen
Discordant Androgen Levels:
- High DHEAS + normal testosterone → Adrenal source (consider adrenal imaging)
- High testosterone + normal DHEAS → Ovarian source
- Both high → Mixed ovarian/adrenal contribution
AMH Utility:
- Correlates with antral follicle count
- Useful when ultrasound unavailable or contraindicated
- Predicts ovarian response in IVF
- May predict metabolic risk severity [51]
PCOM in Adolescents:
- Up to 50% of normal adolescents have PCOM
- PCOM alone insufficient for diagnosis
- Requires persistent oligomenorrhoea (> 1 year post-menarche) + hyperandrogenism
8. Management
Management is individualised and symptom-directed, targeting:
- Lifestyle optimisation and metabolic health
- Menstrual regulation (endometrial protection)
- Hyperandrogenism control (cosmetic concerns)
- Fertility restoration (if desired)
- Long-term complication prevention
Lifestyle Modification - First-Line for ALL Patients
Weight Loss:
- Target: 5-10% body weight reduction
- Evidence: Restores ovulation in 55-100% of anovulatory women; improves insulin sensitivity, lipid profile, and hirsutism [52]
- Approach: Caloric restriction (500-750 kcal deficit), low glycaemic index diet
Exercise:
- Target: 150 minutes moderate-intensity aerobic exercise weekly + resistance training
- Evidence: Improves insulin sensitivity independent of weight loss [53]
Dietary Modification:
- Low glycaemic index carbohydrates
- Increased fibre intake
- Reduced saturated fat
- Mediterranean diet patterns beneficial [54]
Management by Primary Concern
1. Menstrual Irregularity and Endometrial Protection
Problem: Chronic anovulation → unopposed oestrogen → endometrial hyperplasia/cancer risk
First-Line:
- Combined Oral Contraceptive Pill (COCP): [55]
- "Mechanism: Suppresses LH, increases SHBG, induces withdrawal bleeding"
- "Options: Standard formulations (e.g., Microgynon, Rigevidon)"
- "Benefit: Also treats hyperandrogenism"
- "Monitoring: BP, thrombosis risk assessment"
Alternatives:
-
Levonorgestrel Intrauterine System (LNG-IUS) (Mirena):
- "Mechanism: Local progestogenic endometrial suppression"
- "Benefit: Contraception + endometrial protection without systemic hormones"
- "Ideal for: Women with COCP contraindications [56]"
-
Cyclical Progestogens:
- "Regimen: Medroxyprogesterone acetate 10mg days 1-10 every 1-3 months"
- "Purpose: Induce withdrawal bleed, prevent hyperplasia"
- Less effective for hyperandrogenism than COCP [57]
Monitoring:
- If amenorrhoeic and declining all hormonal options: endometrial surveillance
- Transvaginal ultrasound (endometrial thickness)
- If > 10mm in amenorrhoeic patient, consider biopsy
2. Hyperandrogenism (Hirsutism and Acne)
First-Line - Combined Oral Contraceptive:
- Increases SHBG → reduces free androgens
- Anti-androgenic formulations preferred: [58]
- "Co-cyprindiol (Dianette): Cyproterone acetate 2mg + EE 35μg"
- "Drospirenone-containing pills (Yasmin): Anti-mineralocorticoid activity"
- Timeline: Improvement takes 6-12 months (hair growth cycle duration)
Topical Therapy - Hirsutism:
- Eflornithine 11.5% cream (Vaniqa):
- "Mechanism: Irreversible ornithine decarboxylase inhibitor (slows hair growth)"
- "Application: Facial hirsutism, twice daily"
- "Onset: 6-8 weeks"
- "Evidence: Reduces hair growth rate by 30-50% [59]"
Cosmetic Measures:
- Laser hair removal (alexandrite, diode, Nd:YAG lasers)
- Electrolysis
- Bleaching, waxing, threading (temporary)
Anti-Androgen Therapy (Specialist Endocrinology/Dermatology):
-
Spironolactone 50-200mg daily: [60]
- "Mechanism: Androgen receptor antagonist + 5α-reductase inhibitor"
- "Efficacy: Reduces hirsutism by 40-60%"
- "Contraindication: Pregnancy (feminisation of male fetus)"
- "Monitoring: Potassium, renal function"
- Must use reliable contraception
-
Finasteride 2.5-5mg daily:
- "Mechanism: 5α-reductase inhibitor (blocks DHT formation)"
- Category X in pregnancy (teratogenic)
- "Efficacy: Reduces hirsutism, improves androgenic alopecia"
Acne Management:
- COCP (first-line)
- Topical retinoids, benzoyl peroxide
- Oral antibiotics (doxycycline, lymecycline)
- Isotretinoin (severe, resistant cases - requires contraception) [61]
3. Anovulatory Infertility
Pre-conception Optimisation:
- Weight loss (if BMI > 30): Improves ovulation, pregnancy rates, reduces miscarriage risk
- Folic acid 5mg daily (higher dose due to obesity, metformin use)
- Smoking cessation, alcohol moderation
- Optimise glycaemic control (if impaired glucose tolerance/diabetes) [62]
First-Line - Ovulation Induction:
- Letrozole 2.5-7.5mg daily (days 2-6 of cycle): [63]
- "Mechanism: Aromatase inhibitor → reduced oestrogen → increased FSH"
- "Evidence: Superior to clomiphene (higher live birth rate, lower multiple pregnancy rate)"
- "2023 Guideline: First-line agent for ovulation induction in PCOS"
- "Ovulation rate: 70-85%"
- "Monitoring: Mid-luteal progesterone (confirm ovulation), pregnancy test"
Second-Line:
- Clomiphene Citrate 50-150mg daily (days 2-6 of cycle):
- "Mechanism: Selective oestrogen receptor modulator (SERM)"
- "Ovulation rate: 60-85%"
- "Risk: Multiple pregnancy (5-10%), ovarian hyperstimulation syndrome (OHSS) [64]"
Adjunctive Therapy:
- Metformin 500mg TDS or 850mg BD (slow titration to minimise GI side effects):
- "Mechanism: Improves insulin sensitivity"
- "Evidence: Modest benefit in ovulation induction; reduces OHSS risk in IVF [65]"
- "Use: Particularly in women with BMI > 30 or impaired glucose tolerance"
- Continue in pregnancy (reduces gestational diabetes risk in some studies)
Third-Line - Gonadotropin Therapy:
- Low-dose FSH protocol
- Requires specialist reproductive medicine input
- Risk: OHSS, multiple pregnancy
- Monitoring: Ultrasound follicle tracking [66]
Assisted Reproductive Technology (ART):
- In Vitro Fertilisation (IVF):
- "Indications: Failed ovulation induction, tubal factor, male factor"
- "PCOS-specific risks: OHSS (use antagonist protocol, metformin, trigger with GnRH agonist)"
- "Pregnancy rates: Equivalent to non-PCOS women [67]"
4. Insulin Sensitisers
Metformin:
- Indications in PCOS: [68]
- Impaired glucose tolerance or type 2 diabetes (standard diabetes indication)
- Adjunct to ovulation induction (especially if BMI > 30)
- Prevention of type 2 diabetes in high-risk patients
- Dose: 500mg OD → 500mg TDS or 850mg BD (titrate over 2-4 weeks)
- Evidence: Improves insulin sensitivity, modest weight loss (2-3kg), reduces androgen levels
- Not first-line for anovulation, hirsutism, or acne alone (lifestyle + COCP superior)
- Side effects: GI upset (diarrhoea, nausea), vitamin B12 deficiency (long-term)
Inositol Supplements (Myo-inositol, D-chiro-inositol):
- Emerging evidence for metabolic improvement, ovulation induction
- Dose: Myo-inositol 2-4g daily
- Generally well-tolerated; further research needed for definitive recommendation [69]
Management Algorithm
PCOS DIAGNOSED
↓
┌───────────┴───────────┐
↓ ↓
ALL PATIENTS IDENTIFY PRIMARY GOAL
Lifestyle Modification ↓
• Weight loss 5-10% ┌──────┼──────┬─────────┐
• Exercise 150 min/wk ↓ ↓ ↓ ↓
• Low GI diet FERTILITY MENSES HIRSUTISM METABOLIC
↓ ↓ ↓ ↓ ↓
Screen Metabolic: Letrozole COCP COCP + Metformin
• HbA1c annually 2.5-7.5mg OR Topical (if IGT/T2DM)
• Lipids q2-5y ↓ Mirena Eflornithine
• BP each visit Clomiphene + Screen:
(2nd line) Cyclical Laser/ • CVD risk
↓ Progestogen Electrolysis • OSA
Gonadotropins ↓ • NAFLD
↓ Spironolactone
IVF (Specialist)
Special Populations
Adolescents
- Diagnosis: Avoid labelling less than 2 years post-menarche (irregular cycles physiological)
- Require: Persistent oligomenorrhoea + clinical/biochemical hyperandrogenism
- Management: COCP for menstrual regulation, lifestyle intervention paramount [70]
Pregnancy
- Increased risks: Gestational diabetes (40%), pregnancy-induced hypertension (15-20%), preterm birth, macrosomia [71]
- Management: Early glucose screening (OGTT at 16-20 weeks + standard 24-28 weeks), low-dose aspirin if hypertension risk, fetal surveillance
Post-menopausal
- Hyperandrogenism symptoms decline
- Metabolic and cardiovascular risks persist and escalate
- Focus on diabetes screening, cardiovascular risk reduction, endometrial surveillance if prolonged amenorrhoea [72]
Exam Detail: Specific Treatment Protocols - Viva Level:
Letrozole Protocol:
- Start cycle day 2-3 (or after induced bleed with progestogen)
- Dose: 2.5mg daily × 5 days
- If no ovulation: Increase to 5mg, then 7.5mg in subsequent cycles
- Confirm ovulation: Day 21 progesterone > 30 nmol/L
- Max 6 cycles before reassessment
Clomiphene Protocol:
- Start day 2: 50mg daily × 5 days
- Increase to 100mg, then 150mg if no ovulation
- Monitor: Risk of multiple follicles (ultrasound if available)
- Max 6 ovulatory cycles (theoretical increased ovarian cancer risk with prolonged use - unproven)
COCP Selection:
- Anti-androgenic formulations preferred:
- "Co-cyprindiol (Dianette): Most potent anti-androgen; higher VTE risk"
- "Drospirenone 3mg/EE 30μg (Yasmin): Anti-mineralocorticoid"
- "Desogestrel, gestodene: Moderate anti-androgenic activity"
- Avoid: Levonorgestrel-containing pills (relatively androgenic)
Metformin in Pregnancy:
- Controversial; some RCTs show reduced gestational diabetes and miscarriage rates
- If used pre-pregnancy for ovulation induction or diabetes: Can continue first trimester
- Not standard practice; individualise decision [73]
9. Complications and Long-Term Health Risks
Metabolic Complications
| Complication | Risk Increase | Prevalence in PCOS | Prevention Strategy |
|---|---|---|---|
| Type 2 Diabetes | 4-fold | 30-40% by age 40 | Weight loss, metformin (if IGT), annual HbA1c screening |
| Impaired Glucose Tolerance | 3-5 fold | 30-35% | Lifestyle modification, low GI diet |
| Metabolic Syndrome | 2-3 fold | 30-47% | Weight management, exercise, BP/lipid control |
| Dyslipidaemia | 2-fold | 70% | Statin therapy if indicated by cardiovascular risk score |
| Non-Alcoholic Fatty Liver Disease | 2-3 fold | 40-55% | Weight loss, insulin sensitisation |
Type 2 Diabetes: [74]
- Risk begins in third decade
- Earlier onset than general population
- Screen with HbA1c or fasting glucose annually
- OGTT if HbA1c borderline (39-47 mmol/mol)
Metabolic Syndrome: [75]
- Defined by ≥3 of: Central obesity, hypertension, high triglycerides, low HDL, impaired fasting glucose
- Substantially increases cardiovascular disease risk
- Aggressive cardiovascular risk factor management required
Cardiovascular Disease
Evidence:
- Increased surrogate markers: Carotid intima-media thickness, coronary artery calcification, endothelial dysfunction [76]
- Conflicting evidence on hard cardiovascular endpoints (MI, stroke)
- Likely mediated by metabolic syndrome, obesity, diabetes
Risk Factors:
- Obesity, dyslipidaemia, hypertension, insulin resistance, chronic inflammation
Prevention:
- Calculate cardiovascular risk (QRISK3 or equivalent)
- Address modifiable risk factors: Smoking cessation, BP control, statin therapy (if indicated)
- Aspirin not routinely indicated (unless specific cardiovascular indication) [77]
Reproductive Complications
Endometrial Cancer:
- Risk: 2-6 fold increased (chronic unopposed oestrogen exposure) [78]
- Prevention: Ensure regular withdrawal bleeds (≥4 per year)
- COCP, LNG-IUS, or cyclical progestogen
- Surveillance: If prolonged amenorrhoea + endometrial thickness > 10mm → biopsy
Pregnancy Complications: [79]
- Gestational diabetes: 40% (3-fold increase)
- Pregnancy-induced hypertension/pre-eclampsia: 15-20% (2-fold increase)
- Miscarriage: Possibly increased (conflicting evidence)
- Preterm birth, macrosomia, NICU admission
Obstructive Sleep Apnoea (OSA)
- Prevalence: 30-40% of obese PCOS patients [80]
- Independent of obesity (though obesity worsens severity)
- Contributes to insulin resistance, hypertension, cardiovascular risk
- Screen: Epworth Sleepiness Scale; sleep study if suggestive symptoms
- Management: Weight loss, CPAP therapy
Psychological and Quality of Life
- Depression: Prevalence 30-40% (2-4 fold increased risk) [81]
- Anxiety: Significantly elevated
- Body image disturbance: Related to hirsutism, acne, obesity
- Sexual dysfunction: Reduced libido, satisfaction
- Eating disorders: Higher rates of binge eating
- Management: Psychological support, CBT, antidepressants if indicated, peer support groups
10. Prognosis and Long-Term Outcomes
Natural History
Reproductive Years:
- Chronic condition with fluctuating symptoms
- Symptoms worsen with weight gain, improve with weight loss
- Anovulation and hyperandrogenism persist without treatment [82]
Post-Menopause:
- Androgen levels decline → hirsutism and acne often improve
- Metabolic complications worsen: Higher rates of diabetes, hypertension, cardiovascular disease
- Vigilance required for endometrial cancer risk (if history of prolonged amenorrhoea)
Fertility Outcomes
- With treatment: Excellent prognosis
- Ovulation induction success: 70-85%
- Live birth rates approach those of non-PCOS women with appropriate intervention
- May require ART (IVF) in 20-25% if ovulation induction fails or other factors present [83]
Metabolic Outcomes
- Diabetes risk: 30-40% develop T2DM by age 40
- Cardiovascular disease: Increased risk mediated by metabolic syndrome
- Weight trajectory: Tendency toward weight gain; sustained lifestyle modification challenging
- Liver disease: NAFLD may progress to NASH and fibrosis in subset [84]
Factors Associated with Better Prognosis
- Normal BMI or successful weight loss
- Early diagnosis and proactive metabolic screening
- Good adherence to lifestyle modification
- Absence of metabolic syndrome
- Regular menstrual cycles (with treatment) [85]
11. Prevention and Screening
Primary Prevention
Not Applicable: No evidence-based strategies prevent PCOS onset (genetic and developmental origins)
Risk Reduction:
- Healthy BMI maintenance from adolescence
- Regular physical activity
- Avoidance of excessive weight gain
Secondary Prevention (Screening)
Who to Screen:
- All women with PCOS for metabolic complications
Screening Protocol: [86]
| Parameter | Test | Frequency |
|---|---|---|
| Diabetes | HbA1c or fasting glucose | Annually |
| Glucose intolerance | OGTT | If HbA1c 39-47 mmol/mol |
| Dyslipidaemia | Fasting lipid profile | Baseline, then q2-5 years |
| Hypertension | Blood pressure | Every visit |
| Obesity | BMI, waist circumference | Every visit |
| OSA | Epworth Scale, consider sleep study | If symptomatic |
| Depression/Anxiety | Validated screening tools | Baseline, as needed |
| Endometrial cancer | Withdrawal bleed ≥4/year; USS if amenorrhoeic | Ongoing |
Gestational Diabetes Screening:
- All pregnant women with PCOS: OGTT at 16-20 weeks + 24-28 weeks [87]
Tertiary Prevention (Complication Management)
- Treat diabetes, hypertension, dyslipidaemia per standard guidelines
- Cardiovascular risk assessment and management
- OSA treatment (CPAP)
- Psychological support services
12. Key Guidelines and Evidence
International Guidelines
| Guideline | Organisation | Year | Key Recommendations |
|---|---|---|---|
| International Evidence-Based Guideline | ESHRE/AE-PCOS/ASRM | 2023 | Letrozole 1st-line fertility; lifestyle 1st-line all patients; AMH alternative to PCOM [34] |
| Assessment and Management of PCOS | RCOG | 2014 | Rotterdam criteria; screen for metabolic complications [88] |
| PCOS Diagnosis and Management | Endocrine Society | 2013 | Exclude other causes; screen for diabetes, CVD risk [89] |
| Fertility in PCOS | NICE | 2013 | Letrozole or clomiphene for ovulation induction [90] |
Landmark Evidence
1. Rotterdam ESHRE/ASRM Consensus (2004): [33]
- Established widely-adopted Rotterdam diagnostic criteria
- Recognised phenotypic heterogeneity of PCOS
2. Legro RS, et al. NEJM (2014) - Letrozole vs Clomiphene: [63]
- RCT (750 women): Letrozole superior to clomiphene for live birth (27.5% vs 19.1%, p=0.007)
- Established letrozole as first-line ovulation induction agent
3. Teede HJ, et al. (2023) International Guideline Update: [34]
- Comprehensive evidence-based recommendations across diagnosis, screening, management
- Endorsed AMH as alternative to ultrasound PCOM
- Emphasised psychological screening and support
4. Moran LJ, et al. Cochrane Review (2011) - Lifestyle Intervention: [52]
- Weight loss improves reproductive outcomes, metabolic markers, ovulation
- 5-10% body weight reduction clinically meaningful
5. Dumesic DA, et al. Endocrine Reviews (2015) - Pathophysiology: [2]
- Comprehensive review of PCOS mechanisms: Genetics, insulin resistance, hyperandrogenism
- Framework for understanding heterogeneous phenotypes
13. Patient and Layperson Explanation
What is PCOS?
Polycystic Ovary Syndrome (PCOS) is a very common hormonal condition affecting about 1 in 10 women. It occurs when the ovaries don't release an egg regularly (ovulation). Instead, small fluid-filled sacs (follicles) develop and may remain in the ovaries, creating a "polycystic" appearance on ultrasound scans. The name is somewhat misleading—these are not true cysts but simply eggs that haven't matured and been released.
PCOS also causes slightly higher levels of male-type hormones (called androgens), which are normally present in small amounts in women. These hormones can lead to symptoms like excess facial or body hair, acne, and scalp hair thinning.
Why do I have PCOS?
The exact cause isn't fully understood, but PCOS runs in families—if your mother or sister has it, you're more likely to develop it too. The core problem involves how your body handles insulin, the hormone that controls blood sugar levels.
Many women with PCOS have "insulin resistance," meaning their body doesn't respond well to insulin. To compensate, the body produces more insulin. High insulin levels tell the ovaries to make extra male hormones (androgens), which interfere with normal egg release and cause symptoms like excess hair growth.
Weight can worsen insulin resistance, but PCOS also occurs in lean women.
What are the main symptoms?
- Irregular or absent periods: Cycles longer than 35 days or fewer than 8 periods per year
- Excess hair growth (hirsutism): Particularly on the face, chest, stomach, back
- Acne: Especially if it persists beyond teenage years
- Thinning scalp hair
- Difficulty getting pregnant: Due to irregular or absent ovulation
- Weight gain: Particularly around the abdomen
- Darkening of skin in body folds (acanthosis nigricans)
Can I still have children?
Yes, absolutely. While PCOS is a leading cause of difficulty conceiving, the vast majority of women with PCOS can become pregnant with appropriate treatment. Medications like letrozole or clomiphene can stimulate the ovaries to release eggs regularly. If those don't work, other options like injectable fertility hormones or IVF are available. With treatment, pregnancy rates are similar to women without PCOS.
Do I need treatment even if I don't want children right now?
Yes, for several important reasons:
-
Protect your womb lining: Without regular periods, the lining of the womb (endometrium) can become too thick because it's exposed to oestrogen without the balancing hormone, progesterone. Over many years, this increases the risk of womb (endometrial) cancer. Taking the contraceptive pill, having a hormonal coil (IUS), or using periodic hormones ensures you have a period at least 3-4 times per year, keeping the lining healthy.
-
Long-term health: PCOS increases the risk of developing type 2 diabetes, high blood pressure, and heart disease later in life. Managing your weight, staying active, and having regular health checks can reduce these risks significantly.
What treatments are available?
Treatment is tailored to your main concerns:
For everyone:
- Lifestyle changes: Weight loss of even 5-10% (if overweight) can restore regular periods and improve other symptoms dramatically.
- Healthy eating: A balanced diet with less processed food and sugar.
- Regular exercise: At least 150 minutes per week.
For irregular periods:
- Contraceptive pill: Regulates your cycle and protects the womb lining.
- Hormonal coil (Mirena): Thins the womb lining.
- Periodic hormones: Induce a period every few months.
For excess hair and acne:
- Contraceptive pill: Especially types with anti-androgen effects.
- Eflornithine cream: Slows facial hair growth.
- Laser hair removal or electrolysis: For longer-term reduction.
- Spironolactone: Anti-androgen medication (specialist prescription).
For fertility:
- Letrozole or clomiphene: Medications that stimulate ovulation.
- Metformin: Helps if you have insulin resistance or diabetes.
- IVF: If other treatments don't work or there are other fertility factors.
Will it go away?
PCOS is a lifelong condition, but symptoms often improve after menopause as hormone levels naturally decline. Hair growth and acne typically reduce, but the metabolic risks (diabetes, heart disease) persist, so ongoing health monitoring is important.
With appropriate management—especially maintaining a healthy weight and lifestyle—many symptoms can be well-controlled, and long-term complications can be minimised.
Where can I get support?
- Your GP or gynaecologist: For diagnosis, treatment, and screening.
- Specialist clinics: Reproductive medicine or endocrinology if needed.
- Support organisations: Verity (UK PCOS charity), PCOS Awareness Association.
- Mental health support: Don't hesitate to seek help for anxiety, depression, or body image concerns—these are common and treatable.
14. Examination Focus
High-Yield Exam Questions
MRCOG / MRCP Written Exams
-
What are the Rotterdam criteria for diagnosing PCOS?
- Answer: 2 out of 3: (1) Oligo/anovulation, (2) Hyperandrogenism (clinical or biochemical), (3) Polycystic ovaries on ultrasound (≥20 follicles or volume > 10cm³), after exclusion of other causes.
-
Why are women with PCOS at increased risk of endometrial cancer?
- Answer: Chronic anovulation leads to unopposed oestrogen (no progesterone from corpus luteum). Continuous oestrogen stimulation causes endometrial hyperplasia, which can progress to cancer. Regular withdrawal bleeds (via COCP, IUS, or cyclical progestogen) are protective.
-
What is the first-line medication for ovulation induction in PCOS?
- Answer: Letrozole (aromatase inhibitor). Evidence shows higher live birth rates and lower multiple pregnancy rates compared to clomiphene citrate (2023 international guideline).
-
What investigation would you perform if a woman with suspected PCOS has a testosterone level > 5 nmol/L?
- Answer: Urgent imaging (pelvic ultrasound and/or CT adrenals) to exclude androgen-secreting tumour. This level is beyond typical PCOS range and suggests neoplasm.
-
Name three long-term health complications of PCOS.
- Answer: Type 2 diabetes (4-fold risk), endometrial cancer (2-6 fold risk), cardiovascular disease (mediated by metabolic syndrome), obstructive sleep apnoea, depression/anxiety.
Viva Voce Scenarios
Opening Statement: "Polycystic Ovary Syndrome is the most common endocrine disorder in reproductive-age women, affecting approximately 10% of this population. It is characterised by ovulatory dysfunction, hyperandrogenism, and polycystic ovarian morphology, with significant metabolic and reproductive sequelae."
Key Viva Points:
-
Pathophysiology of Insulin Resistance in PCOS:
- "Insulin resistance in PCOS involves post-receptor defects in insulin signalling. Hyperinsulinaemia stimulates ovarian theca cells to produce excess androgens and suppresses hepatic SHBG production, increasing free androgen levels. This creates a self-perpetuating cycle of hyperandrogenism and anovulation."
-
Why Letrozole over Clomiphene?:
- "The PPCOS II trial by Legro demonstrated that letrozole resulted in higher ovulation rates, live birth rates (27.5% vs 19.1%), and lower multiple pregnancy rates compared to clomiphene. Letrozole has a shorter half-life, reducing anti-oestrogenic effects on the endometrium and cervical mucus."
-
Endometrial Protection Explanation:
- "In anovulatory cycles, the corpus luteum does not form, so no progesterone is produced. Without progesterone to oppose oestrogen, the endometrium undergoes continuous proliferation, increasing the risk of hyperplasia and eventual malignant transformation. Inducing regular withdrawal bleeds via COCP, progestogen, or LNG-IUS mitigates this risk."
-
AMH in PCOS:
- "Anti-Müllerian Hormone is secreted by granulosa cells of small antral follicles. In PCOS, the increased follicle number results in elevated AMH (typically > 35 pmol/L). The 2023 guideline supports using AMH as an alternative to ultrasound for diagnosing polycystic ovarian morphology, particularly when transvaginal ultrasound is inappropriate or unavailable."
-
Phenotypic Heterogeneity:
- "PCOS exhibits four phenotypes based on Rotterdam criteria combinations. Classic phenotypes (A and B, with hyperandrogenism and ovulatory dysfunction) carry the highest metabolic risk. The non-hyperandrogenic phenotype (D) has milder metabolic features. This heterogeneity necessitates individualised screening and management."
Common Mistakes (That Fail Candidates)
❌ Using LH:FSH ratio as diagnostic criterion: This is supportive but NOT part of Rotterdam criteria.
❌ Missing red flags for androgen-secreting tumour: Testosterone > 5 nmol/L, rapid virilisation, clitoromegaly demand urgent imaging.
❌ Prescribing anti-androgens without contraception: Spironolactone and finasteride are teratogenic; must ensure effective contraception.
❌ Ignoring metabolic screening: PCOS is not just a reproductive disorder. Failing to screen for diabetes, dyslipidaemia, and hypertension is substandard care.
❌ Stating clomiphene is first-line for ovulation induction: Outdated. Letrozole is now first-line (2023 guideline).
❌ Diagnosing PCOS in early adolescence: Irregular menses and PCOM are physiological less than 2 years post-menarche. Avoid premature labelling.
Model Answers
Q: A 28-year-old woman presents with oligomenorrhoea, hirsutism, and infertility. Describe your approach.
Model Answer: "This presentation is highly suggestive of PCOS, but I would first take a thorough history focusing on menstrual pattern since menarche, rate of hirsutism progression (rapid onset suggests tumour), weight changes, family history, and fertility goals. I would exclude red flags such as virilisation signs or symptoms of Cushing's syndrome.
On examination, I would assess BMI, waist circumference, Ferriman-Gallwey score for hirsutism, and look for acanthosis nigricans indicating severe insulin resistance.
My initial investigations would include:
- Hormonal profile (day 2-5): FSH, LH, testosterone, SHBG, prolactin, TSH, 17-OH progesterone
- Metabolic screen: HbA1c, lipid profile, blood pressure
- Pelvic ultrasound: Assess for polycystic ovarian morphology (≥20 follicles or volume > 10cm³)
If Rotterdam criteria are met (2 out of 3: anovulation, hyperandrogenism, PCOM) and other causes excluded, I would diagnose PCOS.
Management would address her fertility goal as priority:
- First-line: Lifestyle modification (weight loss 5-10% if BMI elevated)
- Ovulation induction with letrozole 2.5-7.5mg (first-line per 2023 guideline)
- Consider metformin adjunct if BMI > 30 or impaired glucose tolerance
- Pre-conception: Folic acid 5mg, optimise glycaemic control
I would also address long-term health:
- Annual HbA1c screening for diabetes
- Cardiovascular risk assessment
- Endometrial protection if she declines fertility treatment (COCP or cyclical progestogen to ensure ≥4 bleeds/year)
- Psychological support as needed
Follow-up would involve monitoring ovulation (day 21 progesterone), pregnancy testing, and escalation to specialist reproductive medicine if first-line treatment fails after 6 cycles."
15. References
-
Bozdag G, et al. The prevalence and phenotypic features of polycystic ovary syndrome: a systematic review and meta-analysis. Hum Reprod. 2016;31(12):2841-2855. doi:10.1093/humrep/dew218
-
Dumesic DA, et al. Scientific Statement on the Diagnostic Criteria, Epidemiology, Pathophysiology, and Molecular Genetics of Polycystic Ovary Syndrome. Endocr Rev. 2015;36(5):487-525. doi:10.1210/er.2015-1018
-
Moran LJ, et al. Impaired glucose tolerance, type 2 diabetes and metabolic syndrome in polycystic ovary syndrome: a systematic review and meta-analysis. Hum Reprod Update. 2010;16(4):347-63. doi:10.1093/humupd/dmq001
-
Barry JA, et al. Risk of endometrial, ovarian and breast cancer in women with polycystic ovary syndrome: a systematic review and meta-analysis. Hum Reprod Update. 2014;20(5):748-758. doi:10.1093/humupd/dmu012
-
Teede HJ, et al. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Fertil Steril. 2018;110(3):364-379. doi:10.1016/j.fertnstert.2018.05.004
-
Dewailly D, et al. Definition and significance of polycystic ovarian morphology: a task force report from the Androgen Excess and Polycystic Ovary Syndrome Society. Hum Reprod Update. 2014;20(3):334-352. doi:10.1093/humupd/dmt061
-
Diamanti-Kandarakis E, Dunaif A. Insulin resistance and the polycystic ovary syndrome revisited: an update on mechanisms and implications. Endocr Rev. 2012;33(6):981-1030. doi:10.1210/er.2011-1034
-
Carmina E, Lobo RA. Evaluation of hormonal status in women with hirsutism. UpToDate. 2022.
-
Lizneva D, et al. Criteria, prevalence, and phenotypes of polycystic ovary syndrome. Fertil Steril. 2016;106(1):6-15. doi:10.1016/j.fertnstert.2016.05.003
-
Rosenfield RL. The Diagnosis of Polycystic Ovary Syndrome in Adolescents. Pediatrics. 2015;136(6):1154-1165. doi:10.1542/peds.2015-1430
-
March WA, et al. The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Hum Reprod. 2010;25(2):544-551. doi:10.1093/humrep/dep399
-
Schmidt J, et al. Reproductive hormone levels and anthropometry in postmenopausal women with polycystic ovary syndrome (PCOS): a 21-year follow-up study. J Clin Endocrinol Metab. 2011;96(7):2178-2185. doi:10.1210/jc.2010-2959
-
Wijeyaratne CN, et al. Phenotype and metabolic profile of South Asian women with polycystic ovary syndrome (PCOS): results of a large database from a specialist Endocrine clinic. Hum Reprod. 2011;26(1):202-213. doi:10.1093/humrep/deq310
-
Ezeh U, et al. Racial and ethnic differences in metabolic syndrome in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2015;100(1):E142-E145. doi:10.1210/jc.2014-2990
-
Yildiz BO, et al. Prevalence, phenotype and cardiometabolic risk of polycystic ovary syndrome under different diagnostic criteria. Hum Reprod. 2012;27(10):3067-3073. doi:10.1093/humrep/des232
-
Kahsar-Miller MD, et al. Prevalence of polycystic ovary syndrome (PCOS) in first-degree relatives of patients with PCOS. Fertil Steril. 2001;75(1):53-58. doi:10.1016/s0015-0282(00)01662-9
-
Lim SS, et al. Overweight, obesity and central obesity in women with polycystic ovary syndrome: a systematic review and meta-analysis. Hum Reprod Update. 2012;18(6):618-637. doi:10.1093/humupd/dms030
-
Ibáñez L, et al. Premature adrenarche—normal variant or forerunner of adult disease? Endocr Rev. 2000;21(6):671-696. doi:10.1210/edrv.21.6.0416
-
Sir-Petermann T, et al. Maternal serum androgens in pregnant women with polycystic ovarian syndrome: possible implications in prenatal androgenization. Hum Reprod. 2002;17(10):2573-2579. doi:10.1093/humrep/17.10.2573
-
Moran LJ, et al. Dietary composition in the treatment of polycystic ovary syndrome: a systematic review to inform evidence-based guidelines. J Acad Nutr Diet. 2013;113(4):520-545. doi:10.1016/j.jand.2012.11.018
-
Blank SK, et al. The origins and sequelae of abnormal neuroendocrine function in polycystic ovary syndrome. Hum Reprod Update. 2006;12(4):351-361. doi:10.1093/humupd/dml017
-
Franks S, et al. Follicle dynamics and anovulation in polycystic ovary syndrome. Hum Reprod Update. 2008;14(4):367-378. doi:10.1093/humupd/dmn015
-
Nelson VL, et al. Augmented androgen production is a stable steroidogenic phenotype of propagated theca cells from polycystic ovaries. Mol Endocrinol. 1999;13(6):946-957. doi:10.1210/mend.13.6.0311
-
Jonard S, Dewailly D. The follicular excess in polycystic ovaries, due to intra-ovarian hyperandrogenism, may be the main culprit for the follicular arrest. Hum Reprod Update. 2004;10(2):107-117. doi:10.1093/humupd/dmh010
-
Dunaif A. Insulin resistance and the polycystic ovary syndrome: mechanism and implications for pathogenesis. Endocr Rev. 1997;18(6):774-800. doi:10.1210/edrv.18.6.0318
-
Corbould A, et al. Insulin resistance in skeletal muscle and adipose tissue in women with polycystic ovary syndrome involves intrinsic and acquired defects in insulin signaling. Am J Physiol Endocrinol Metab. 2005;288(5):E1047-E1054. doi:10.1152/ajpendo.00361.2004
-
Mather KJ, et al. Hyperinsulinemia in polycystic ovary syndrome correlates with increased cardiovascular risk independent of obesity. Fertil Steril. 2000;73(1):150-156. doi:10.1016/s0015-0282(99)00468-9
-
Rosenfield RL, Ehrmann DA. The Pathogenesis of Polycystic Ovary Syndrome (PCOS): The Hypothesis of PCOS as Functional Ovarian Hyperandrogenism Revisited. Endocr Rev. 2016;37(5):467-520. doi:10.1210/er.2015-1104
-
Azziz R, et al. Positions statement: criteria for defining polycystic ovary syndrome as a predominantly hyperandrogenic syndrome: an Androgen Excess Society guideline. J Clin Endocrinol Metab. 2006;91(11):4237-4245. doi:10.1210/jc.2006-0178
-
Escobar-Morreale HF, et al. The polycystic ovary syndrome associated with morbid obesity may resolve after weight loss induced by bariatric surgery. J Clin Endocrinol Metab. 2005;90(12):6364-6369. doi:10.1210/jc.2005-1490
-
Day F, et al. Large-scale genome-wide meta-analysis of polycystic ovary syndrome suggests shared genetic architecture for different diagnosis criteria. PLoS Genet. 2018;14(12):e1007813. doi:10.1371/journal.pgen.1007813
-
Welt CK, et al. Defining constant versus variable phenotypic features of women with polycystic ovary syndrome using different ethnic groups and populations. J Clin Endocrinol Metab. 2006;91(11):4361-4368. doi:10.1210/jc.2006-1191
-
Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril. 2004;81(1):19-25. doi:10.1016/j.fertnstert.2003.10.004
-
Teede HJ, et al. Recommendations from the 2023 international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Fertil Steril. 2023;120(4):767-793. doi:10.1016/j.fertnstert.2023.07.025
-
Azziz R, et al. The Androgen Excess and PCOS Society criteria for the polycystic ovary syndrome: the complete task force report. Fertil Steril. 2009;91(2):456-488. doi:10.1016/j.fertnstert.2008.06.035
-
Hickey M, et al. Clinical, ultrasound and biochemical features of polycystic ovary syndrome in adolescents: implications for diagnosis. Hum Reprod Update. 2011;17(1):77-87. doi:10.1093/humupd/dmq027
-
Hudecova M, et al. Long-term follow-up of patients with polycystic ovary syndrome: reproductive outcome and ovarian reserve. Hum Reprod. 2009;24(5):1176-1183. doi:10.1093/humrep/den482
-
Broekmans FJ, et al. PCOS according to the Rotterdam consensus criteria: Change in prevalence among WHO-II anovulation and association with metabolic factors. BJOG. 2006;113(10):1210-1217. doi:10.1111/j.1471-0528.2006.01008.x
-
Yildiz BO, et al. Assessment, diagnosis, and treatment of hirsutism. Fertil Steril. 2018;109(2):203-219. doi:10.1016/j.fertnstert.2017.11.019
-
Ekback MP, et al. Acne in polycystic ovary syndrome: Prevalence and associated factors. Am J Clin Dermatol. 2021;22(3):393-403. doi:10.1007/s40257-021-00591-3
-
Futterweit W, et al. Prevalence of hyperandrogenism in 109 consecutive female patients with diffuse alopecia. J Am Acad Dermatol. 1988;19(5 Pt 1):831-836. doi:10.1016/s0190-9622(88)70242-8
-
Fauser BC, et al. Consensus on women's health aspects of polycystic ovary syndrome (PCOS): the Amsterdam ESHRE/ASRM-Sponsored 3rd PCOS Consensus Workshop Group. Fertil Steril. 2012;97(1):28-38. doi:10.1016/j.fertnstert.2011.09.024
-
Yildiz BO, et al. Impact of obesity on the risk for polycystic ovary syndrome. J Clin Endocrinol Metab. 2008;93(1):162-168. doi:10.1210/jc.2007-1834
-
González F, et al. Adiposity and acanthosis nigricans in women with polycystic ovary syndrome: correlation with insulin secretion and insulin resistance. Fertil Steril. 2006;85(6):1441-1447. doi:10.1016/j.fertnstert.2005.11.035
-
Apridonidze T, et al. Prevalence and characteristics of the metabolic syndrome in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2005;90(4):1929-1935. doi:10.1210/jc.2004-1469
-
Dokras A, et al. Screening women with polycystic ovary syndrome for metabolic syndrome. Obstet Gynecol. 2005;106(1):131-137. doi:10.1097/01.AOG.0000167408.30893.6b
-
Dokras A, et al. Increased risk for abnormal depression scores in women with polycystic ovary syndrome: a systematic review and meta-analysis. Obstet Gynecol. 2011;117(1):145-152. doi:10.1097/AOG.0b013e318202b0a4
-
Escobar-Morreale HF, et al. Epidemiology, diagnosis and management of hirsutism: a consensus statement by the Androgen Excess and Polycystic Ovary Syndrome Society. Hum Reprod Update. 2012;18(2):146-170. doi:10.1093/humupd/dmr042
-
Lujan ME, et al. Updated ultrasound criteria for polycystic ovary syndrome: reliable thresholds for elevated follicle population and ovarian volume. Hum Reprod. 2013;28(5):1361-1368. doi:10.1093/humrep/det062
-
Dewailly D, et al. Interactions between androgens, FSH, anti-Müllerian hormone and estradiol during folliculogenesis in the human normal and polycystic ovary. Hum Reprod Update. 2016;22(6):709-724. doi:10.1093/humupd/dmw027
-
Iliodromiti S, et al. The predictive accuracy of anti-Müllerian hormone for live birth after assisted conception: a systematic review and meta-analysis of the literature. Hum Reprod Update. 2014;20(4):560-570. doi:10.1093/humupd/dmu003
-
Moran LJ, et al. Lifestyle changes in women with polycystic ovary syndrome. Cochrane Database Syst Rev. 2011;(7):CD007506. doi:10.1002/14651858.CD007506.pub3
-
Harrison CL, et al. Exercise therapy in polycystic ovary syndrome: a systematic review. Hum Reprod Update. 2011;17(2):171-183. doi:10.1093/humupd/dmq045
-
Barrea L, et al. Nutrition and polycystic ovary syndrome: a systematic review. Nutrients. 2021;13(6):1949. doi:10.3390/nu13061949
-
De Leo V, et al. Hormonal contraceptives: pharmacology tailored to women's health. Hum Reprod Update. 2016;22(5):634-646. doi:10.1093/humupd/dmw016
-
Varma R, et al. Levonorgestrel-releasing intrauterine system for heavy menstrual bleeding. Cochrane Database Syst Rev. 2005;(4):CD002126. doi:10.1002/14651858.CD002126.pub2
-
Leminen H, Härkki P. Management of menstrual dysfunction in adolescents with polycystic ovary syndrome. Best Pract Res Clin Obstet Gynaecol. 2018;48:90-98. doi:10.1016/j.bpobgyn.2017.10.003
-
Martin KA, et al. Evaluation and treatment of hirsutism in premenopausal women: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2018;103(4):1233-1257. doi:10.1210/jc.2018-00241
-
Balfour JA, McClellan K. Topical eflornithine. Am J Clin Dermatol. 2001;2(3):197-201. doi:10.2165/00128071-200102030-00009
-
Brown J, et al. Metformin for polycystic ovary syndrome. Cochrane Database Syst Rev. 2009;(2):CD003053. doi:10.1002/14651858.CD003053.pub3
-
Bhate K, Williams HC. Epidemiology of acne vulgaris. Br J Dermatol. 2013;168(3):474-485. doi:10.1111/bjd.12149
-
Best D, Bhattacharya S. Obesity and fertility. Horm Mol Biol Clin Investig. 2015;24(1):5-10. doi:10.1515/hmbci-2015-0023
-
Legro RS, et al. Letrozole versus clomiphene for infertility in the polycystic ovary syndrome. N Engl J Med. 2014;371(2):119-129. doi:10.1056/NEJMoa1313517
-
Homburg R. Clomiphene citrate—end of an era? A mini-review. Hum Reprod. 2005;20(8):2043-2051. doi:10.1093/humrep/dei042
-
Costello M, et al. Metformin versus oral contraceptive pill in polycystic ovary syndrome: a Cochrane review. Hum Reprod. 2007;22(5):1200-1209. doi:10.1093/humrep/dem005
-
Thessaloniki ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Consensus on infertility treatment related to polycystic ovary syndrome. Hum Reprod. 2008;23(3):462-477. doi:10.1093/humrep/dem426
-
Heijnen EM, et al. A meta-analysis of outcomes of conventional IVF in women with polycystic ovary syndrome. Hum Reprod Update. 2006;12(1):13-21. doi:10.1093/humupd/dmi036
-
Tang T, et al. Insulin-sensitising drugs (metformin, rosiglitazone, pioglitazone, D-chiro-inositol) for women with polycystic ovary syndrome, oligo amenorrhoea and subfertility. Cochrane Database Syst Rev. 2012;(5):CD003053. doi:10.1002/14651858.CD003053.pub5
-
Unfer V, et al. Myo-inositol effects in women with PCOS: a meta-analysis of randomized controlled trials. Endocr Connect. 2017;6(8):647-658. doi:10.1530/EC-17-0243
-
Witchel SF, et al. The diagnosis of polycystic ovary syndrome during adolescence. Horm Res Paediatr. 2015;83(6):376-389. doi:10.1159/000375530
-
Boomsma CM, et al. A meta-analysis of pregnancy outcomes in women with polycystic ovary syndrome. Hum Reprod Update. 2006;12(6):673-683. doi:10.1093/humupd/dml036
-
Tehrani FR, et al. Long-term cardiovascular outcomes in women with polycystic ovary syndrome: a systematic review and meta-analysis. Reprod Biomed Online. 2021;43(2):274-287. doi:10.1016/j.rbmo.2021.05.003
-
Khattab S, et al. Metformin reduces abortion in pregnant women with polycystic ovary syndrome. Gynecol Endocrinol. 2006;22(12):680-684. doi:10.1080/09513590601010508
-
Ehrmann DA, et al. Prevalence and predictors of the metabolic syndrome in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2006;91(1):48-53. doi:10.1210/jc.2005-1329
-
Vgontzas AN, et al. Polycystic ovary syndrome is associated with obstructive sleep apnea and daytime sleepiness: role of insulin resistance. J Clin Endocrinol Metab. 2001;86(2):517-520. doi:10.1210/jcem.86.2.7185
-
Paradisi G, et al. Cardiovascular risk factors in healthy women with polycystic ovary syndrome. Eur J Endocrinol. 2010;162(1):53-57. doi:10.1530/EJE-09-0611
-
Wild RA, et al. Cardiovascular disease and mortality in women with polycystic ovary syndrome: a systematic review and meta-analysis. Hum Reprod Update. 2011;17(4):495-500. doi:10.1093/humupd/dmr001
-
Haoula Z, et al. Evaluating the association between endometrial cancer and polycystic ovary syndrome. Hum Reprod. 2012;27(5):1327-1331. doi:10.1093/humrep/des042
-
Qin JZ, et al. Obstetric complications in women with polycystic ovary syndrome: a systematic review and meta-analysis. Reprod Biol Endocrinol. 2013;11:56. doi:10.1186/1477-7827-11-56
-
Kahal H, et al. The prevalence of obstructive sleep apnoea in women with polycystic ovary syndrome: a systematic review and meta-analysis. Sleep Breath. 2020;24(2):339-350. doi:10.1007/s11325-019-01835-1
-
Blay SL, et al. Polycystic ovary syndrome and mental disorders: a systematic review and exploratory meta-analysis. Neuropsychiatr Dis Treat. 2016;12:2895-2903. doi:10.2147/NDT.S91700
-
Norman RJ, et al. Polycystic ovary syndrome. Lancet. 2007;370(9588):685-697. doi:10.1016/S0140-6736(07)61345-2
-
Palomba S, et al. Pregnancy complications in women with polycystic ovary syndrome. Hum Reprod Update. 2015;21(5):575-592. doi:10.1093/humupd/dmv029
-
Jones H, et al. Polycystic ovary syndrome with hyperandrogenism is characterized by an increased risk of hepatic steatosis compared to nonhyperandrogenic PCOS phenotypes and healthy controls, independent of obesity and insulin resistance. J Clin Endocrinol Metab. 2012;97(10):3709-3716. doi:10.1210/jc.2012-1382
-
Fauser BC, et al. Health consequences of polycystic ovary syndrome. Semin Reprod Med. 2008;26(1):72-84. doi:10.1055/s-2007-992927
-
Wild RA, et al. Assessment of cardiovascular risk and prevention of cardiovascular disease in women with the polycystic ovary syndrome: a consensus statement by the Androgen Excess and Polycystic Ovary Syndrome (AE-PCOS) Society. J Clin Endocrinol Metab. 2010;95(5):2038-2049. doi:10.1210/jc.2009-2724
-
American Diabetes Association. Standards of Medical Care in Diabetes—2023. Diabetes Care. 2023;46(Suppl 1):S19-S40. doi:10.2337/dc23-S002
-
Royal College of Obstetricians and Gynaecologists. Long-term Consequences of Polycystic Ovary Syndrome. Green-top Guideline No. 33. London: RCOG; 2014.
-
Legro RS, et al. Diagnosis and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2013;98(12):4565-4592. doi:10.1210/jc.2013-2350
-
National Institute for Health and Care Excellence. Fertility problems: assessment and treatment. NICE Clinical Guideline CG156. London: NICE; 2013 (Updated 2017).
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate specialists and current local/national guidelines.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Ovarian Physiology
- Menstrual Cycle Disorders
Differentials
Competing diagnoses and look-alikes to compare.
- Congenital Adrenal Hyperplasia
- Hypothalamic Amenorrhoea
- Premature Ovarian Insufficiency
- Prolactinoma
Consequences
Complications and downstream problems to keep in mind.
- Type 2 Diabetes Mellitus
- Endometrial Hyperplasia and Cancer
- Metabolic Syndrome