Pneumothorax in Adults
Pneumothorax is defined as air in the pleural space, resulting in partial or complete lung collapse. It represents a com... MRCP, FRACP exam preparation.
What matters first
Pneumothorax is defined as air in the pleural space, resulting in partial or complete lung collapse. It represents a com... MRCP, FRACP exam preparation.
Tension pneumothorax: hypotension, tracheal deviation, distended neck veins - immediate needle decompression without CXR
8 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Tension pneumothorax: hypotension, tracheal deviation, distended neck veins - immediate needle decompression without CXR
- Bilateral pneumothorax: severe dyspnea, bilateral reduced breath sounds - life-threatening
- Haemopneumothorax: penetrating trauma with pneumothorax - requires large-bore chest drain
- Persistent air leak less than 5-7 days: referral to thoracic surgery for VATS
Exam focus
Current exam surfaces linked to this topic.
- MRCP, FRACP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Pulmonary Embolism
- Acute Coronary Syndrome
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
1. Clinical Overview
Definition and Classification
Pneumothorax is defined as air in the pleural space, resulting in partial or complete lung collapse. It represents a common respiratory emergency with diverse etiologies and varying degrees of clinical severity. [1]
Classification by Etiology:
-
Primary Spontaneous Pneumothorax (PSP): Occurs without preceding trauma or obvious underlying lung disease, typically due to rupture of apical subpleural blebs. [2]
-
Secondary Spontaneous Pneumothorax (SSP): Occurs in patients with established underlying pulmonary disease (COPD, asthma, interstitial lung disease, infections). [2]
-
Traumatic Pneumothorax: Results from penetrating or blunt chest trauma, or iatrogenic injury (central venous catheterization, thoracentesis, mechanical ventilation). [3]
-
Tension Pneumothorax: Life-threatening condition with progressive air accumulation and one-way valve mechanism, leading to mediastinal shift, vascular compromise, and cardiovascular collapse. [4]
Clinical Significance
Epidemiological Impact:
- PSP incidence: 18-28 per 100,000 per year in males, 1.2-6 per 100,000 in females. [5]
- SSP incidence: 6.3 per 100,000 per year, predominantly in patients aged > 55 years with chronic lung disease. [5]
- Male-to-female ratio: 3-6:1 for PSP. [2]
- Recurrence represents major clinical burden: 30% after first episode, 60% after second episode without surgical intervention. [6]
Healthcare Burden:
- Annual hospital admissions in UK: approximately 8,000 cases. [1]
- Mean hospital stay: 4-7 days for chest drain management. [7]
- Surgical intervention required in 15-20% of cases due to recurrence or persistent air leak. [8]
Key Clinical Associations
Primary Spontaneous Pneumothorax:
- Tall, thin body habitus: Increased apical pleural porosity and mechanical stress. [9]
- Young males (20-30 years): Peak incidence during rapid growth phase. [2]
- Smoking: Dose-dependent risk increase (relative risk 20-fold for heavy smokers). [10]
- Marfan syndrome: Fibrillin-1 deficiency leading to apical bullae formation. [11]
- Familial pneumothorax: FLCN gene mutations (Birt-Hogg-Dubé syndrome). [12]
Secondary Spontaneous Pneumothorax:
- COPD (most common cause of SSP): Bullae rupture in emphysematous lungs. [13]
- Asthma: Particularly during acute exacerbations with alveolar overdistension. [14]
- Infections: Tuberculosis (cavitary disease), Pneumocystis jirovecii pneumonia (PCP) in HIV, necrotizing pneumonia. [15]
- Interstitial lung diseases: Lymphangioleiomyomatosis (LAM), pulmonary Langerhans cell histiocytosis. [16]
- Cystic fibrosis: Advanced disease with bullae formation. [17]
- Malignancy: Sarcoma, pulmonary metastases (especially osteosarcoma). [18]
"Do Not Miss" Red Flag Presentations
1. Tension Pneumothorax
Clinical Features (Diagnosis is Clinical - Do NOT Wait for Imaging):
- Progressive severe dyspnea and chest pain
- Hypotension (SBP less than 90 mmHg) or rapid deterioration in previously stable patient
- Tachycardia (> 120 bpm)
- Tracheal deviation away from affected side (late sign)
- Distended neck veins (raised JVP from SVC compression)
- Absent breath sounds on affected side with hyperresonant percussion
- Cyanosis, altered consciousness
- Pulseless electrical activity (PEA) arrest if untreated [4]
Immediate Management:
- Needle decompression without waiting for imaging confirmation
- Site 1: 2nd intercostal space, midclavicular line (above 3rd rib to avoid neurovascular bundle)
- Site 2 (alternative, potentially more effective): 4th-5th intercostal space, mid-axillary line (triangle of safety) [19]
- Technique: Large-bore cannula (14-16G), insert perpendicular to chest wall until audible hiss of escaping air
- Confirmation: Clinical improvement (BP stabilization, reduced respiratory distress)
- Definitive treatment: Chest drain insertion after initial decompression [20]
2. Bilateral Pneumothorax
Clinical Features:
- Severe dyspnea out of proportion to unilateral findings
- Bilateral reduced breath sounds
- Profound hypoxia (SpO2 less than 85% on room air)
- May occur iatrogenically (bilateral subclavian line attempts) or in diseases with bilateral bullae (Marfan, LAM) [21]
Management:
- Bilateral chest drain insertion
- High-flow oxygen
- Consider ICU admission for respiratory support
3. Catamenial Pneumothorax
Clinical Features:
- Women of reproductive age (25-35 years typically)
- Recurrent pneumothorax within 72 hours of menstruation onset (90% right-sided)
- History of endometriosis or pelvic pain
- Pathophysiology: Thoracic endometriosis with diaphragmatic fenestrations or ectopic endometrial tissue in pleura [22]
Management:
- Hormonal suppression (GnRH agonists, danazol)
- VATS with diaphragmatic repair and pleurodesis
- Recurrence common (50%) without combined surgical and hormonal therapy [22]
4. Haemopneumothorax
Clinical Features:
- Pneumothorax following penetrating trauma
- Haemodynamic instability
- Decreased breath sounds with dullness to percussion (fluid level)
- Substantial blood loss into pleural space [3]
Management:
- Large-bore chest drain (28-32F) for blood evacuation
- Massive haemothorax (> 1500 mL immediate drainage or > 200 mL/hour): thoracic surgery for thoracotomy
- Blood transfusion and coagulation correction
5. Iatrogenic Pneumothorax Post-Procedure
High-Risk Procedures:
- Central venous catheterization (especially subclavian): 1-6% incidence [23]
- Thoracentesis: 5-30% incidence (higher with inexperienced operators)
- Transbronchial biopsy: 2-5%
- Percutaneous lung biopsy: 15-25%
- Mechanical ventilation with high peak pressures: Barotrauma risk [3]
Management:
- Small asymptomatic iatrogenic pneumothorax: Observation with serial CXR
- Symptomatic or enlarging: Aspiration or chest drain
- Prevention: Ultrasound guidance for procedures, lung-protective ventilation strategies
2. Epidemiology and Risk Factors
Incidence and Prevalence
Primary Spontaneous Pneumothorax (PSP):
- Male incidence: 18-28 per 100,000 population per year [5]
- Female incidence: 1.2-6 per 100,000 population per year [5]
- Peak age: 20-30 years (males), 30-40 years (females)
- Second smaller peak: > 60 years (overlapping with SSP) [2]
Secondary Spontaneous Pneumothorax (SSP):
- Overall incidence: 6.3 per 100,000 population per year [5]
- Predominantly affects patients > 55 years with chronic lung disease
- COPD patients: Lifetime risk approximately 26% [13]
- AIDS patients with PCP: 35% develop pneumothorax during infection [15]
Traumatic Pneumothorax:
- Occurs in 15-50% of major chest trauma cases [3]
- Iatrogenic: Variable depending on procedure (see above)
Geographical and Seasonal Variation:
- Higher incidence in temperate climates [24]
- Seasonal variation: Peaks in winter and spring (possibly related to atmospheric pressure changes) [24]
Risk Factors and Relative Risk
| Risk Factor | Relative Risk (RR) | Absolute Risk Increase | Key Mechanism | Reference |
|---|---|---|---|---|
| Smoking (current) | 22.0 (males), 9.0 (females) | Dose-dependent: 1% per pack-year | Airway inflammation → emphysema-like changes → apical bleb formation, impaired healing | [10] |
| Tall stature (> 185 cm) | 2.0-3.5 | 4% increase per 10 cm height | Increased negative apical pleural pressure from greater vertical lung height; apical pleural porosity | [9] |
| Low BMI (less than 18.5 kg/m²) | 1.8-2.4 | — | Ectomorphic body habitus; reduced intrathoracic adipose tissue protective effect | [9] |
| Family history (1st degree) | 11.5 | — | FLCN gene mutations (Birt-Hogg-Dubé), folliculin deficiency | [12] |
| Marfan syndrome | 4.5-8.0 | 4-10% lifetime risk | Fibrillin-1 deficiency → apical bullae; aortic root dilation does NOT correlate with pneumothorax risk | [11] |
| COPD (moderate-severe) | 10.0-25.0 | Lifetime risk ~26% | Emphysematous bullae rupture; increased work of breathing → alveolar stress | [13] |
| Previous pneumothorax | Ipsilateral recurrence: 30% (1st), 60% (2nd) | — | Pleural inflammation → weakened visceral pleura; incomplete healing | [6] |
| Cannabis smoking | 2.0-3.5 | — | Bullous lung disease from deep inhalation and Valsalva during smoking | [10] |
| Thoracic endometriosis | — | Accounts for 3-6% of spontaneous pneumothorax in women of reproductive age | Diaphragmatic fenestrations; ectopic endometrial tissue in pleura with cyclical necrosis | [22] |
Protective Factors
- Female sex: Lower baseline risk (hormonal factors may provide pleural protection) [5]
- Smoking cessation: Risk reduction within 2-3 years to near-baseline [10]
- Obesity (BMI > 30): Paradoxically protective (intrathoracic adipose tissue cushioning effect) [9]
3. Pathophysiology
Normal Pleural Physiology Recap
- Pleural space: Potential space between visceral pleura (covers lungs) and parietal pleura (lines chest wall)
- Pleural pressure: Negative pressure (-5 to -8 cmH2O), maintaining lung inflation
- Pleural fluid: 10-20 mL, allows frictionless lung movement
- Reabsorption balance: Produced by parietal pleura, reabsorbed by visceral pleural lymphatics [25]
Mechanisms of Pneumothorax Formation
Primary Spontaneous Pneumothorax (PSP)
Bleb-Bullae Hypothesis:
- Apical blebs: Small (less than 2 cm) subpleural air-filled cysts form at lung apices
- Histology: Thin-walled (less than 1 mm) cysts lined by flattened mesothelial cells
- Location: Apex of upper lobes (most common), superior segments of lower lobes
- Mechanical stress: Upright posture creates greatest negative pleural pressure at apex (gravity effect)
- Pressure gradient: Apex experiences -8 to -10 cmH₂O vs base -2 to -4 cmH₂O
- Alveolar distension: Greater transpulmonary pressure at apex → chronic alveolar overdistension
- Growth spurt theory: Rapid vertical growth in tall young males → increased apical stress → bleb formation [9]
- Critical period: Adolescent growth spurt (12-18 years) creates maximal apical stress
- Ischemic hypothesis: Rapid lung growth outpaces vascular supply → apical ischemia → airspace formation
- Molecular mechanisms:
- Matrix metalloproteinase (MMP) dysregulation: Increased MMP-2 and MMP-9 → collagen degradation → weakened pleura
- Elastin fiber abnormalities: Defective elastic fiber assembly in visceral pleura
- Oxidative stress: Smoking-induced reactive oxygen species → extracellular matrix damage
- Rupture: Spontaneous bleb rupture (often during rest, not exertion) → air enters pleural space
- Trigger mechanisms: Sudden atmospheric pressure changes, coughing, stretching
- Ruptured bleb rarely seen at surgery: Suggests healing occurs rapidly after initial air leak [2]
- Rapid sealing: Fibrin deposition and mesothelial proliferation seal defect within 24-48 hours
Distal Airway Inflammation Hypothesis:
- Even "primary" pneumothorax shows histological evidence of airway inflammation in 80% of cases [26]
- Smoking-induced small airway inflammation → air trapping → alveolar distension → subpleural bleb formation [10]
- Supports overlap between PSP and early emphysema spectrum
Atmospheric Pressure Changes:
- Sudden atmospheric pressure drops (weather changes, high altitude) may precipitate rupture [24]
- Explains increased incidence during air travel or mountain climbing in susceptible individuals
Secondary Spontaneous Pneumothorax (SSP)
COPD/Emphysema:
- Bullae formation: Destruction of alveolar septa → large air spaces (> 1 cm)
- Rupture mechanism: Increased work of breathing → dynamic hyperinflation → excessive transpulmonary pressure → bullae rupture
- Air leak persistence: Diseased lung tissue has poor healing capacity → prolonged air leaks common [13]
Asthma:
- Mucus plugging: Complete airway obstruction → distal air trapping → alveolar overdistension
- Check-valve effect: Air enters on inspiration but cannot exit → progressive distension → rupture
- Typically during acute exacerbation: High peak airway pressures [14]
Infections:
- Tuberculosis: Cavitation → subpleural cavity erosion into pleural space; bronchopleural fistula formation [15]
- PCP (Pneumocystis jirovecii): Necrotizing pneumonitis → thin-walled cyst formation → rupture (35% of AIDS patients with PCP develop pneumothorax) [15]
- Necrotizing bacterial pneumonia (Staph aureus, Klebsiella): Lung abscess → pleural rupture
Interstitial Lung Diseases:
- LAM (Lymphangioleiomyomatosis): Cystic lung destruction from abnormal smooth muscle proliferation → cyst rupture [16]
- Pulmonary Langerhans Cell Histiocytosis: Upper lobe cystic changes → pneumothorax in 25% of patients [16]
Cystic Fibrosis:
- Advanced disease: Bullae from recurrent infections and bronchiectasis
- Complicates 3-4% of CF patients; associated with severe disease and poor prognosis [17]
Traumatic Pneumothorax
Penetrating Trauma:
- Direct violation of pleural integrity (stab wound, gunshot)
- Air enters from: (a) External environment through chest wall defect, (b) Lung parenchyma injury [3]
Blunt Trauma:
- Rib fracture → sharp bone edge lacerates lung
- Sudden thoracic compression → alveolar rupture without rib fracture [3]
Iatrogenic:
- Central line insertion: Needle transgresses pleura during subclavian/internal jugular approach; lung apical position increases risk [23]
- Thoracentesis: Direct lung puncture if technique inadequate; higher risk without ultrasound guidance [23]
- Mechanical ventilation (barotrauma): High peak pressures (> 35 cmH2O) → alveolar rupture → interstitial air → tracks along bronchovascular bundles to mediastinum (pneumomediastinum) → ruptures into pleural space [3]
- Transbronchial biopsy: Peripheral lung biopsy through bronchoscope → direct pleural violation [3]
Pathophysiology of Tension Pneumothorax
Critical One-Way Valve Mechanism:
-
Air entry: Tissue flap acts as one-way valve
- Inspiration: Negative intrathoracic pressure → flap opens → air enters pleural space
- Expiration: Positive pressure → flap closes → air trapped in pleural space [4]
-
Progressive accumulation: Each respiratory cycle adds more air → increasing intrapleural pressure
-
Intrapleural pressure becomes positive: Normally -5 cmH2O → becomes +5 to +15 cmH2O [4]
-
Mediastinal shift:
- Increasing positive pressure on affected side → pushes mediastinum to contralateral side
- Heart, great vessels, trachea displaced
- Tracheal deviation: Late sign, indicates severe tension [20]
-
Vascular compression and cardiac compromise:
- SVC/IVC kinking: Reduced venous return → decreased preload
- Frank-Starling mechanism fails: Inadequate ventricular filling → reduced stroke volume
- Cardiac output drops: SBP falls despite compensatory tachycardia
- Coronary perfusion compromised: Hypotension → reduced diastolic coronary flow → myocardial ischemia [4]
-
Respiratory compromise:
- Ipsilateral lung completely collapsed
- Contralateral lung compressed by mediastinal shift
- Profound hypoxia and hypercapnia
-
Cardiovascular collapse:
- Progressive hypotension → shock → PEA arrest
- Death within minutes if untreated [4]
Why Tension Develops:
- Positive pressure ventilation: Most common setting (ICU patients, trauma with mechanical ventilation) [20]
- Large parenchymal injury: Large air leak overwhelms pleural reabsorption
- Delayed recognition: Small pneumothorax initially → progressive enlargement → sudden decompensation
Physiological Consequences of Pneumothorax
Respiratory Effects:
- Reduced lung compliance: Lung elastic recoil pulls lung away from chest wall → collapse
- V/Q mismatch: Collapsed lung receives minimal ventilation but continues perfusion → shunt physiology → hypoxemia
- Hypoxemia severity:
- "PSP: Minimal hypoxia (PaO2 typically > 70 mmHg) due to normal contralateral lung compensation [25]"
- "SSP: Severe hypoxia (PaO2 may be less than 60 mmHg) due to poor baseline lung function [13]"
Pleural Pressure Changes:
- Intrapleural pressure becomes less negative or atmospheric (0 cmH2O)
- Loss of negative pressure → lung collapse toward hilum
Spontaneous Reabsorption:
- Pleural air reabsorbed at rate of 1.25-2.2% per day on room air [25]
- Mechanism: Nitrogen gradient between pleural air (79% N2) and venous blood (low N2) → diffusion into capillaries
- High-flow oxygen accelerates reabsorption 4-fold: Oxygen washout of nitrogen → increased gradient → faster reabsorption (key therapeutic principle) [27]
4. Clinical Presentation
Symptom Profile
Primary Spontaneous Pneumothorax (PSP)
Timing and Onset:
- Typically at rest: 90% occur during sedentary activities or sleep (NOT during exertion) [2]
- Sudden onset: Immediate symptom development at moment of rupture
Symptoms:
-
Pleuritic chest pain (90% of patients):
- Sharp, unilateral (ipsilateral to pneumothorax)
- Sudden onset
- Worse on inspiration, coughing, movement
- Intensity variable: May be mild or severe
- Location: Chest wall, may radiate to shoulder (phrenic nerve irritation) [2]
-
Dyspnea (80% of patients):
- Sudden-onset breathlessness
- Variable severity depending on size
- Small pneumothorax (less than 20% lung volume): Mild dyspnea or asymptomatic
- Large pneumothorax (> 50% lung volume): Moderate to severe dyspnea [2]
-
Asymptomatic presentation (10-15% of cases):
- Incidental finding on CXR performed for other indication
- More common with small pneumothoraces (less than 15% lung volume) [2]
Symptom Duration:
- Chest pain: Typically resolves within 24-48 hours even without treatment (pleural inflammation settles)
- Dyspnea: Persists until lung re-expansion occurs
Secondary Spontaneous Pneumothorax (SSP)
More Severe Presentation (due to reduced respiratory reserve):
Symptoms:
-
Dyspnea (95% of patients):
- Severe even with small pneumothorax
- Baseline breathlessness worsens acutely
- May progress to respiratory failure rapidly [13]
-
Pleuritic chest pain (70-80%):
- Similar character to PSP
- May be masked by chronic chest discomfort from underlying disease
-
Cough: Dry, non-productive, worsened by pain
-
Cyanosis: If large pneumothorax or severe underlying disease
Symptoms of Underlying Disease:
- COPD: Chronic productive cough, wheeze
- Asthma: Wheeze, use of accessory muscles
- PCP: Fever, dry cough, weight loss in HIV patient [15]
Tension Pneumothorax
Rapidly Progressive Severe Symptoms:
- Severe dyspnea: Immediate, progressively worsening
- Chest pain: Severe, unilateral
- Anxiety, agitation: Sense of impending doom from hypoxia
- Syncope or pre-syncope: Reduced cardiac output
- Confusion, altered consciousness: Severe hypoxia and hypotension [4]
Physical Examination Findings
Inspection
General Appearance:
- PSP: Often minimal distress with small-moderate pneumothorax
- SSP: Evident respiratory distress even with small pneumothorax
- Tension: Severe distress, cyanosis, altered consciousness
Respiratory Pattern:
- Tachypnea (RR > 20 breaths/min in PSP; > 25 in SSP; > 30 in tension)
- Reduced chest expansion on affected side (best seen from foot of bed, comparing both hemithoraces)
- Use of accessory muscles (sternocleidomastoid, scalenes): Indicates respiratory distress [25]
Chest Wall:
- Surgical emphysema: Crepitus on palpation of chest wall/neck (indicates air tracking through tissue planes from pneumothorax or pneumomediastinum) [25]
- Trauma signs: Bruising, wounds, rib tenderness/crepitus
Tracheal Position:
- Normal/central: Simple pneumothorax (no mediastinal shift) [25]
- Deviation away from affected side: Tension pneumothorax (LATE SIGN - do not wait for this before treating) [20]
- Assessment: Palpate trachea in suprasternal notch
Percussion
Ipsilateral to Pneumothorax:
- Hyperresonant (increased resonance): Classic finding; air in pleural space produces enhanced resonance compared to normal lung
- Sensitivity: 60-70% (may be difficult to appreciate, especially in noisy emergency department) [25]
Technique: Percuss systematically comparing both hemithoraces at multiple levels (apices, mid-zones, bases)
Auscultation
Ipsilateral to Pneumothorax:
- Reduced or absent breath sounds: Most reliable clinical sign (sensitivity 85-90%) [25]
- Complete absence: Large pneumothorax
- Diminished: Small-moderate pneumothorax
- Normal breath sounds: Very small pneumothorax (less than 10%)
Technique:
- Auscultate systematically, comparing both sides
- Include apices (patient sitting forward, auscultate from posterior)
Other Auscultatory Findings:
- Hamman's sign (Hamman's crunch): Crunching or clicking sound synchronous with heartbeat, heard over precordium. Indicates pneumomediastinum (air in mediastinum), which may be associated with pneumothorax. [25]
- Wheeze: May indicate underlying asthma/COPD in SSP
Cardiovascular Examination
Simple Pneumothorax:
- Heart rate: Mild tachycardia (90-110 bpm)
- Blood pressure: Normal
- JVP: Normal
- Heart sounds: Normal (apex beat may be difficult to locate if large left-sided pneumothorax)
Tension Pneumothorax:
- Hypotension: SBP less than 90 mmHg or drop > 40 mmHg from baseline [4]
- Severe tachycardia: > 120 bpm
- Distended neck veins (raised JVP): SVC compression by positive intrapleural pressure; paradoxically elevated despite hypotension (differentiates from hypovolemic shock) [20]
- Pulsus paradoxus: Exaggerated drop in BP on inspiration (> 10 mmHg)
- Weak/thready pulses: Reduced cardiac output
Summary of Key Examination Findings by Type
| Sign/Examination | Simple PSP | Simple SSP | Tension Pneumothorax |
|---|---|---|---|
| Respiratory Rate | 18-24 | 24-30 | > 30 |
| Tracheal Position | Central | Central | Deviated away (late) |
| Chest Expansion | Reduced ipsilateral | Reduced ipsilateral | Reduced ipsilateral |
| Percussion | Hyperresonant | Hyperresonant | Hyperresonant |
| Breath Sounds | Reduced/absent | Reduced/absent | Absent |
| Heart Rate | 90-110 | 100-120 | > 120 |
| Blood Pressure | Normal | Normal or low | Hypotensive (less than 90 mmHg) |
| JVP | Normal | Normal | Elevated |
| Clinical Urgency | Stable | Moderate | LIFE-THREATENING |
Differential Diagnosis
| Condition | Clinical Features Distinguishing from Pneumothorax | Key Diagnostic Test |
|---|---|---|
| Pulmonary Embolism (PE) | Bilateral chest signs (pneumothorax is unilateral); risk factors (surgery, malignancy, immobility); pleuritic pain may be similar but dyspnea often more gradual onset; ECG may show S1Q3T3 pattern or tachycardia | CTPA (gold standard); D-dimer (if low probability); Leg ultrasound (DVT) |
| Acute Myocardial Infarction (MI) | Central/crushing chest pain (not pleuritic); radiation to jaw/left arm; associated sweating, nausea; cardiac risk factors; bilateral equal breath sounds | ECG (ST elevation/depression, T-wave inversion); Troponin (elevated) |
| Acute Aortic Dissection | Tearing pain radiating to back; blood pressure differential between arms (> 20 mmHg); pulse deficit; aortic regurgitation murmur; may have neurological symptoms if carotid involvement | CT aorta with contrast (intimal flap, false lumen); Transoesophageal echo |
| Pleural Effusion | Stony dull percussion (not hyperresonant); gradual onset dyspnea; may have underlying cause (heart failure, malignancy); no sudden pleuritic pain | CXR (blunted costophrenic angle, meniscus); Ultrasound (fluid); Diagnostic tap |
| Pneumonia | Productive cough, fever, purulent sputum; dull percussion over consolidation (not hyperresonant); bronchial breathing, crackles | CXR (consolidation); Sputum culture; Blood cultures if septic |
| Rib Fracture (without pneumothorax) | Focal point tenderness over fracture site; history of trauma; pain on palpation and rib springing; breath sounds normal bilaterally | CXR (may show fracture, but often missed); CT chest if high suspicion |
| Musculoskeletal Chest Pain | Chest wall tenderness on palpation; pain reproduced by movement; no respiratory symptoms; normal examination | Clinical diagnosis; imaging if concern for underlying pathology |
| Pericarditis | Central chest pain, worse lying flat, better sitting forward; pericardial friction rub; ECG shows widespread ST elevation; may have viral prodrome | ECG (widespread concave ST elevation, PR depression); Echocardiogram (pericardial effusion) |
| Oesophageal Rupture (Boerhaave's) | Severe retrosternal pain after vomiting; surgical emphysema; Hamman's sign; history of forceful vomiting; rapidly unwell with sepsis | CT chest with oral contrast (oesophageal leak); CXR (pneumomediastinum, left pleural effusion) |
5. Investigations
Initial Assessment (ABCDE Approach)
Tension Pneumothorax: Clinical diagnosis - treat immediately with needle decompression BEFORE any investigations. [20]
For Non-Tension Presentations: Systematic investigation approach
Bedside Investigations
Pulse Oximetry (SpO2)
Expected Findings:
- PSP (small-moderate): SpO2 94-98% on room air (minimal hypoxia due to normal contralateral lung compensation) [25]
- PSP (large): SpO2 88-94% on room air
- SSP: SpO2 frequently less than 90% on room air even with small pneumothorax (poor baseline lung function) [13]
- Tension: SpO2 less than 85%, progressive desaturation
Interpretation: Hypoxia severity guides urgency of intervention
Arterial Blood Gas (ABG)
Indications:
- SSP (all cases)
- PSP with significant hypoxia (SpO2 less than 92%)
- Consideration for discharge (assess baseline respiratory function)
Expected Findings:
PSP:
- pH: Normal (7.35-7.45)
- PaO2: Mild reduction (9-11 kPa / 68-83 mmHg on room air)
- PaCO2: Normal or slightly reduced (compensatory tachypnea causing hyperventilation)
- Type 1 respiratory failure pattern (hypoxia without hypercapnia) [25]
SSP:
- pH: May be reduced if severe (less than 7.30 in severe cases with respiratory failure)
- PaO2: Moderate to severe reduction (less than 8 kPa / 60 mmHg common)
- PaCO2: Variable
- "COPD: May be chronically elevated (6-8 kPa), acute rise indicates decompensation"
- "Asthma: Initially low (hyperventilation), rising PaCO2 indicates exhaustion/respiratory failure"
- Type 2 respiratory failure possible in severe SSP (hypoxia + hypercapnia) [13]
Electrocardiogram (ECG)
Indications: All pneumothorax cases (exclude cardiac cause of chest pain)
Possible Findings:
- Most commonly normal or sinus tachycardia
- Large left-sided pneumothorax:
- "Reduced R-wave amplitude in left precordial leads (V4-V6): Air interface attenuates signal"
- Rightward QRS axis shift (if massive left-sided collapse)
- Tension pneumothorax:
- Electrical alternans (QRS amplitude variation beat-to-beat from mediastinal swinging)
- ST-segment depression (myocardial ischemia from hypotension)
- Right-sided pneumothorax: May mimic acute MI with T-wave inversion in right precordial leads [25]
Importance: Exclude concomitant acute coronary syndrome in older patients with chest pain
Imaging
Chest X-Ray (CXR) - Gold Standard for Diagnosis
Technique:
- Erect PA (posteroanterior): Mandatory (patient standing, X-ray from back to front)
- Inspiration film: Standard (full inspiration)
- Expiration films NO LONGER RECOMMENDED: Historical practice; does not increase diagnostic sensitivity and delays treatment [1,2]
Diagnostic Features:
-
Visceral Pleural Line (Pathognomonic):
- Thin white line representing visceral pleura, separated from chest wall
- Peripheral to line: Absent lung markings (air in pleural space)
- Central to line: Lung markings visible (collapsed lung)
- Best seen at apex (air rises to highest point in erect patient) [25]
-
Absence of Lung Markings Peripherally:
- No vascular or bronchial markings in pneumothorax space
- Beware: Confusing with bulla (bulla has thin walls but is within lung, not separating lung from chest wall)
-
Small Pneumothorax:
- May only be visible at apex
- Apical cap appearance
- Subtle visceral pleural line
-
Large Pneumothorax:
- Complete lung collapse toward hilum
- Entire hemithorax radiolucent (black)
-
Tension Pneumothorax (imaging should NOT delay treatment):
- Complete lung collapse
- Mediastinal shift: Trachea, heart shadow displaced to contralateral side
- Contralateral lung compression
- Flattened hemidiaphragm (depressed by positive intrapleural pressure)
- Widened intercostal spaces on affected side [20]
Pitfalls and Mimics:
- Skin folds: Can mimic pleural line, but skin fold extends beyond chest wall; no absent lung markings
- Large bullae: Thin-walled, may mimic pneumothorax but wall is within lung parenchyma, not peripheral to lung edge
- Pneumomediastinum: Air outlining mediastinal structures; may be associated with pneumothorax
Quantification of Size (British Thoracic Society Guidelines): [1]
Method: Measure horizontal distance between lung edge and chest wall at level of hilum (mid-point of collapsed lung)
Classification:
- Small pneumothorax: Rim of air less than 2 cm at hilum level
- Large pneumothorax: Rim of air ≥2 cm at hilum level
Clinical Significance:
- Guides management decisions (aspiration vs chest drain)
- Size does NOT directly correlate with symptoms (small pneumothorax in SSP may be highly symptomatic)
Historical Note:
- Light Index (older method, less used): Estimates pneumothorax volume as % of hemithorax
- "Formula: Volume % = 100 - (lung diameter / hemithorax diameter)³ × 100"
- Large if > 50%, but cumbersome calculation; BTS rim measurement preferred [1]
Computed Tomography (CT) Chest
Indications:
- Diagnostic uncertainty: CXR equivocal; differentiate pneumothorax from bullae
- Underlying lung disease: Identify bullae, emphysema, ILD, malignancy in SSP
- Pre-surgical planning: Assess lung parenchyma before VATS pleurodesis
- Occult pneumothorax: Suspected on clinical grounds but CXR normal (especially in trauma) [3]
- Catamenial pneumothorax: Identify diaphragmatic defects, thoracic endometriosis [22]
Advantages:
- Higher sensitivity: Detects pneumothoraces as small as 5 mL (vs 50 mL for CXR)
- Characterizes underlying disease: Bullae, ILD patterns, malignancy
- Assessment in supine trauma patients: CT superior to supine CXR (air collects anteriorly/medially when supine) [3]
Findings:
- Air in pleural space with precise anatomical localization
- Identification of blebs/bullae (PSP: apical blebs less than 2 cm; SSP: bullae > 1 cm)
- Underlying parenchymal disease
- Loculated pneumothorax
Limitation: Radiation exposure; cost; not first-line
Ultrasound (Point-of-Care Ultrasound / POCUS)
Indications:
- Trauma settings (FAST scan protocol): Rapid bedside assessment for pneumothorax in unstable patients
- ICU monitoring: Serial assessment without radiation exposure
- Procedural guidance: Confirming diagnosis pre-chest drain insertion [28]
Technique:
- High-frequency linear probe (5-10 MHz)
- Patient supine or semi-recumbent
- Probe placed in intercostal spaces, longitudinally aligned with ribs
- Scan anterior chest wall (2nd-4th ICS, midclavicular to mid-axillary line) [28]
Normal Lung Ultrasound Findings (Pneumothorax ABSENT):
- Lung sliding: Visceral pleura slides against parietal pleura with respiration; visible as shimmering movement at pleural line (also called "gliding sign")
- B-lines (comet-tail artifacts): Vertical hyperechoic lines extending from pleural line to bottom of screen; arise from normal aerated lung
- Lung pulse: Transmitted cardiac pulsations visible at pleural line [28]
Pneumothorax Ultrasound Findings:
- Absent lung sliding: No movement at pleural line (air in pleural space prevents visceral-parietal contact)
- Sensitivity: 86-98% [28]
- Specificity: 97-100% (but also absent in: adhesions, pleurodesis, apnea, main bronchus intubation)
- Absent B-lines: No comet-tail artifacts
- Lung point sign (Specific for pneumothorax):
- Transition point where pneumothorax edge meets normally aerated lung
- Intermittent lung sliding visible (sliding present, then absent, alternating with respiration)
- Specificity: 100%, Sensitivity: 66% (only detected if pneumothorax edge within scanning field) [28]
- A-lines only: Horizontal reverberation artifacts (present in both normal lung and pneumothorax; non-specific)
Advantages:
- Rapid (2-3 minutes)
- No radiation
- Higher sensitivity than CXR for small pneumothorax (especially in supine patients)
- Bedside availability [28]
Limitations:
- Operator-dependent
- Cannot quantify size accurately
- Subcutaneous emphysema impairs image quality
- Cannot replace CXR for documentation and medico-legal purposes
Laboratory Investigations
Routine Blood Tests (to identify underlying cause in SSP or assess fitness for intervention):
-
Full Blood Count (FBC):
- Leukocytosis: Infection (pneumonia, TB)
- Lymphopenia: HIV/PCP [15]
-
C-Reactive Protein (CRP):
- Elevated: Infection
-
HIV Test (if PCP suspected):
- Risk factors: Known HIV, immunosuppression, CD4 less than 200
-
Coagulation Screen (pre-chest drain insertion):
- INR, APTT: Assess bleeding risk (especially if on anticoagulation)
-
Alpha-1 Antitrypsin Level (young patient with emphysema/SSP):
- Deficiency causes early-onset emphysema [13]
Specialist Investigations (Selected Cases)
-
CT Thorax with Contrast (if malignancy suspected):
- Lung masses, lymphadenopathy
-
VATS Diagnostic Biopsy (at time of pleurodesis):
- Histological diagnosis of underlying ILD, malignancy
-
Hormonal Studies (catamenial pneumothorax):
- Performed in context of endometriosis workup [22]
6. Management
Overarching Principles
- Tension Pneumothorax = Clinical Diagnosis → Immediate Needle Decompression (Do NOT wait for imaging) [20]
- All SSP patients require admission and chest drain (poor respiratory reserve means even small pneumothorax poorly tolerated) [1]
- PSP management stratified by size and symptoms (observation vs aspiration vs chest drain) [1]
- High-flow oxygen accelerates pleural air reabsorption 4-fold (nitrogen washout mechanism) [27]
- Recurrence after 2nd pneumothorax mandates surgical pleurodesis (recurrence rate 60% without surgery) [6]
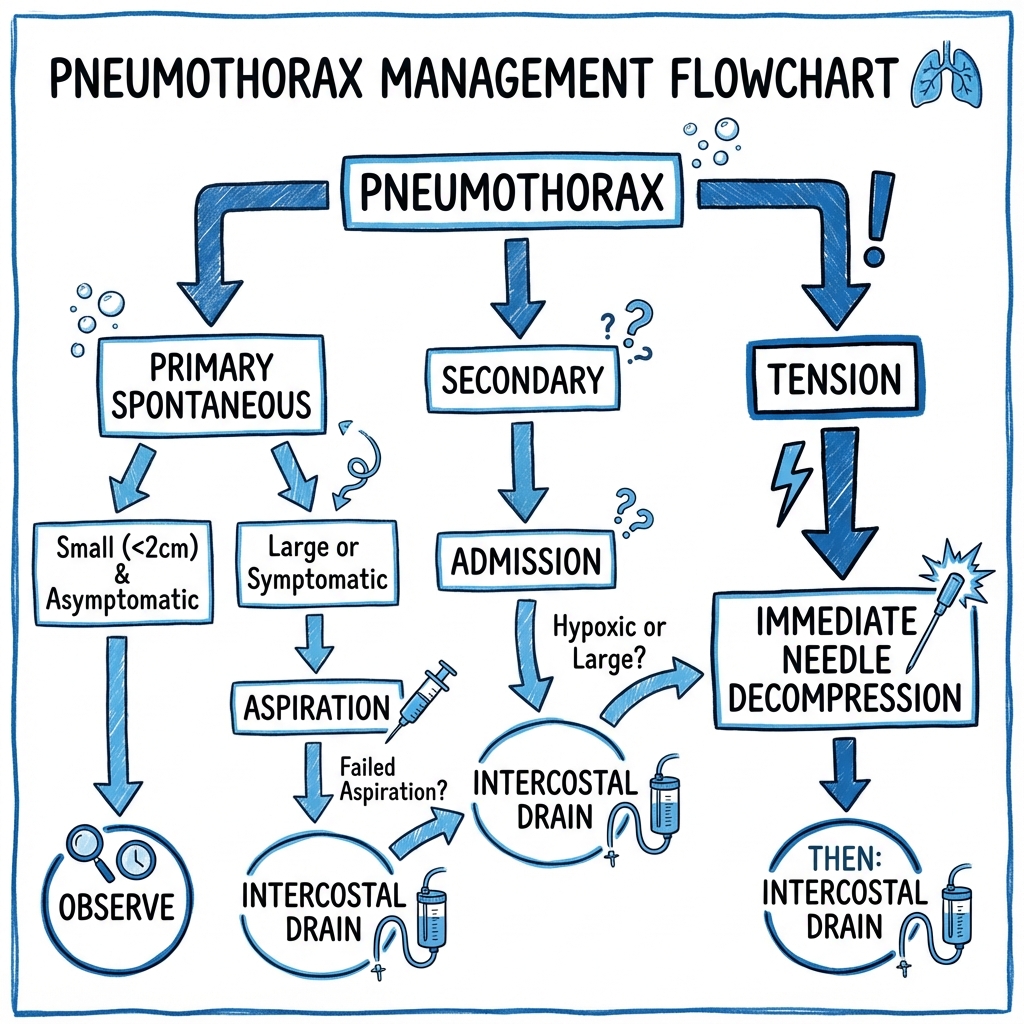
Management Algorithm (British Thoracic Society 2010 Guidelines) [1]
┌─────────────────────────────────────────┐
│ PNEUMOTHORAX SUSPECTED │
│ (Dyspnea + pleuritic pain ± │
│ unilateral reduced breath sounds) │
└──────────────────┬──────────────────────┘
↓
┌────────────────────┐
│ Tension features? │
│ (Clinical diagnosis)│
└─────┬──────────┬───┘
│ │
YES NO
│ │
↓ ↓
┌────────────────┐ │
│ IMMEDIATE │ │
│ NEEDLE │ │
│ DECOMPRESSION │ │
│ │ │
│ • 2nd ICS MCL │ │
│ OR │ │
│ • 4th-5th ICS │ │
│ MAL (triangle│ │
│ of safety) │ │
│ • 14-16G needle│ │
└────────┬───────┘ │
↓ │
Insert chest drain │
(definitive after │
decompression) │
│ │
└────────────┘
↓
┌────────────────┐
│ CXR confirm │
│ diagnosis │
└────────┬───────┘
↓
┌───────────────────────┐
│ Primary (PSP) or │
│ Secondary (SSP)? │
└─────┬───────────┬─────┘
│ │
PSP SSP
│ │
↓ ↓
┌─────────────┐ ┌──────────────────┐
│ Measure size│ │ ALL SSP PATIENTS:│
│ (rim at │ │ • ADMIT │
│ hilum) │ │ • CHEST DRAIN │
└──────┬──────┘ │ (12-14F) │
│ │ • High-flow O2 │
┌─────┴─────┐ │ • Treat │
less than 2cm ≥2cm │ underlying │
(Small) (Large)│ disease │
│ │ └──────────────────┘
↓ ↓
┌──────────┐ ┌──────────────┐
│ Symptoms?│ │ 1. ASPIRATION│
└────┬─────┘ │ (16-18G) │
│ │ Max 2.5L │
┌──┴──┐ └──────┬───────┘
YES NO ↓
│ │ Repeat CXR
│ │ (in 4h)
│ │ │
↓ ↓ ┌─────┴─────┐
┌──────────┐ Success? Failure
│ ASPIRATE │ (less than 2cm) (≥2cm)
│ OR │ │ │
│ DRAIN │ ↓ ↓
└──────────┘ ┌──────┐ ┌──────────┐
│OBSERVE│ │CHEST DRAIN│
│+O2 │ │ (12-14F) │
│ │ └──────────┘
│Follow-│
│up 2-4w│
└───────┘
Immediate Management of Tension Pneumothorax
Clinical Diagnosis Criteria (DO NOT WAIT FOR CXR - Imaging Delays Treatment and Increases Mortality): [4,20]
- Respiratory distress (severe dyspnea, tachypnea > 30/min)
- Hypotension (SBP less than 90 mmHg) or haemodynamic instability
- Tachycardia (> 120 bpm)
- Reduced/absent breath sounds unilaterally
- ± Tracheal deviation (LATE sign, often absent)
- ± Distended neck veins (raised JVP)
Immediate Needle Decompression:
Indication: Clinical suspicion of tension pneumothorax (do not wait for imaging confirmation)
Sites (Evidence supports BOTH locations; local protocols vary): [19,20]
-
Traditional Site: 2nd Intercostal Space, Midclavicular Line (2nd ICS MCL):
- Landmarks:
- Identify sternal angle (angle of Louis)
- 2nd rib articulates at sternal angle
- 2nd ICS is space below 2nd rib
- Midclavicular line (vertical line through midpoint of clavicle)
- Insertion: Insert needle ABOVE 3rd rib (neurovascular bundle runs along inferior border of rib)
- Advantages: Anterior location; easy landmark identification
- Disadvantages:
- 30% failure rate (especially obese patients; inadequate needle length; thick chest wall)
- Risk of internal mammary artery injury
- Smaller safe zone [19]
- Landmarks:
-
Alternative Site: 4th-5th Intercostal Space, Mid-Axillary Line (Lateral approach):
- Landmarks: "Triangle of Safety"
- Apex: Axilla (armpit)
- Anterior border: Lateral edge of pectoralis major
- Posterior border: Lateral edge of latissimus dorsi
- Base: 5th intercostal space (level of nipple in males)
- Insertion: 4th or 5th ICS in mid-axillary line within triangle of safety
- Advantages:
- Higher success rate (90% vs 70% for anterior approach)
- Thinner chest wall laterally
- Larger safe zone; fewer vital structures
- Easier conversion to chest drain (same site) [19]
- Disadvantages: May be difficult in supine patient with limited access to lateral chest
- Landmarks: "Triangle of Safety"
Technique:
-
Equipment:
- 14-16 Gauge (large-bore) cannula
- Length ≥5 cm (longer needles needed in obese patients; studies show 5 cm needles fail in 30% due to chest wall thickness) [19]
- Syringe (10-20 mL)
-
Procedure:
- DO NOT prep skin or delay (life-threatening emergency; immediate decompression required)
- Identify insertion site (see above)
- Insert needle perpendicular to chest wall
- Advance while aspirating syringe
- Audible hiss of escaping air: Confirms tension pneumothorax
- Advance cannula over needle into pleural space
- Remove needle, leave cannula in situ (secure with tape)
- Air continues to escape through cannula
-
Confirmation:
- Clinical improvement: Reduced respiratory distress, BP stabilization, improved SpO2
- Ongoing air escape from cannula
-
Definitive Management:
- Needle decompression is TEMPORIZING measure only
- Must insert chest drain after initial decompression (typically at same lateral site) [20]
Post-Decompression:
- Continuous monitoring (HR, BP, SpO2, RR)
- Chest drain insertion (see below)
- CXR to confirm diagnosis and assess lung re-expansion
Management of Primary Spontaneous Pneumothorax (PSP)
Small PSP (less than 2 cm rim at hilum) + Asymptomatic/Minimal Symptoms
Conservative Management (Observation):
Indications: [1]
- Pneumothorax less than 2 cm (BTS criteria)
- No significant breathlessness (mild pleuritic pain acceptable)
- Patient able to return if deterioration
- Lives close to hospital (less than 1 hour)
Intervention:
-
High-flow oxygen (if patient hypoxic):
- 10-15 L/min via non-rebreather mask
- Mechanism: Creates nitrogen gradient → accelerates pleural air reabsorption from 1.25% per day to 4-6% per day [27]
- Continue until normoxic on room air
-
Analgesia:
- Paracetamol 1g QDS regular
- ± NSAIDs (e.g. ibuprofen 400mg TDS) if no contraindications
- ± Opioids (e.g. codeine 30-60mg QDS) for severe pain
-
Observation (options):
- Outpatient management (BTS-supported approach):
- Suitable if asymptomatic/minimally symptomatic
- Safety netting advice (see below)
- Follow-up CXR in 2-4 weeks (outpatient)
- Resolves spontaneously in 3-8 weeks [1]
- Inpatient observation (24h):
- If patient anxious or lives far from hospital
- Repeat CXR at 24 h: If stable or improving → discharge with outpatient follow-up
- Outpatient management (BTS-supported approach):
Safety Netting / Discharge Advice:
- Return immediately if increasing breathlessness, chest pain, dizziness/syncope
- Avoid strenuous activity for 2 weeks
- No flying for 6 weeks post-resolution on CXR (or 1 week post-successful surgical pleurodesis) [1]
- Permanent diving ban unless bilateral surgical pleurodesis (recurrence underwater is fatal due to inability to surface) [1]
Large PSP (≥2 cm) or Symptomatic Small PSP
Active Intervention Required
First-Line: Needle Aspiration (BTS Guidelines Recommend Aspiration Before Chest Drain for PSP): [1]
Rationale:
- Success rate 50-70% for first pneumothorax
- Avoids chest drain insertion in > 50% of patients
- Shorter hospital stay (same-day discharge if successful)
- Lower complication rate than chest drain [1]
Indications:
- Large PSP (≥2 cm)
- OR symptomatic small PSP
Contraindications:
- Haemodynamic instability
- Bilateral pneumothorax
- Previous failed aspiration
- Secondary pneumothorax (SSP) - these need chest drain [1]
Equipment:
- 16-18 Gauge cannula
- 50 mL syringe
- Three-way tap
- Local anaesthetic (1% lidocaine 5-10 mL)
Procedure:
-
Patient position: Sitting upright (if able) or semi-recumbent 45°
-
Site selection (EITHER):
- 2nd ICS, midclavicular line (traditional anterior approach)
- 4th-5th ICS in triangle of safety (lateral approach; increasingly preferred as easier to convert to chest drain if needed)
-
Technique:
- Aseptic technique (sterile gloves, skin prep with chlorhexidine)
- Local anaesthetic infiltration to skin, subcutaneous tissue, pleura
- Insert 16-18G cannula above rib border (avoid neurovascular bundle)
- Confirm air aspiration (feel "give" as pleura punctured; aspirate air easily)
- Advance cannula into pleural space; remove needle
- Connect to 50 mL syringe via three-way tap
- Aspirate up to 2.5 litres total (to prevent re-expansion pulmonary oedema)
- Aspirate 50 mL aliquots, expelling air through three-way tap
- Continue until: (a) Resistance felt (lung re-expanded against catheter), OR (b) Patient coughs excessively (lung touching catheter), OR (c) 2.5L aspirated
- Remove cannula once aspiration complete [1]
-
Post-procedure:
- CXR at 4 hours post-aspiration: Assess lung re-expansion [1]
Success Criteria:
- Pneumothorax reduced to less than 2 cm on 4-hour CXR
- Clinical improvement (reduced dyspnea)
If Successful (less than 2 cm on CXR):
- Discharge same day with safety netting
- Outpatient CXR in 2-4 weeks
- Advise avoid flying for 6 weeks, diving indefinitely [1]
If Failed (≥2 cm on CXR or increasing symptoms):
- Proceed to chest drain insertion [1]
Second-Line: Chest Drain Insertion (if aspiration fails or contraindicated)
Indications:
- Failed aspiration (pneumothorax remains ≥2 cm post-aspiration)
- Haemodynamic instability
- Bilateral pneumothorax
- Secondary spontaneous pneumothorax (SSP) - ALL require chest drain [1]
Chest Drain Size:
- Small-bore (12-14 French): Preferred for PSP (Seldinger technique; less painful; lower complication rate)
- Large-bore (24-28 French): If haemopneumothorax or thick fluid [1]
Insertion Site:
- Triangle of Safety (ALWAYS use this site; minimizes risk of injury): [1]
- "Apex: Axilla"
- "Anterior: Lateral border of pectoralis major"
- "Posterior: Lateral border of latissimus dorsi"
- "Base: 5th intercostal space (nipple level in males)"
- Typical position: 5th ICS, mid-axillary line
Procedure (Seldinger Technique for Small-Bore Drains):
-
Patient position:
- Supine or semi-recumbent
- Ipsilateral arm abducted and hand behind head (opens intercostal spaces)
-
Equipment:
- Seldinger chest drain kit (guidewire, dilators, 12-14F drain)
- Underwater seal drainage system
- Local anaesthetic (1% lidocaine 10-20 mL)
- Sterile gown, gloves, drapes
-
Technique:
- Full aseptic technique
- Skin preparation (chlorhexidine)
- Identify insertion site (5th ICS, mid-axillary line, within triangle of safety)
- Local anaesthetic: Infiltrate skin, subcutaneous tissue, muscle, periosteum of rib, pleura (aspirate to confirm pleural space entered; air aspirated confirms position)
- Small skin incision (1-1.5 cm)
- Insert needle above rib border (avoid neurovascular bundle on inferior aspect)
- Confirm air aspiration
- Pass guidewire through needle into pleural space (advance 10-15 cm)
- Remove needle, leaving guidewire
- Pass dilators over guidewire sequentially (dilate tract)
- Pass chest drain over guidewire into pleural space (advance 10-15 cm; ensure all holes within pleural cavity)
- Remove guidewire
- Connect to underwater seal drainage system (bubbling confirms correct placement)
- Secure drain with suture (1-0 silk, secure to skin)
- Occlusive dressing
- CXR to confirm position: Drain tip should be apical for pneumothorax [1]
-
Post-insertion management:
- Underwater seal drainage: Bubbling indicates ongoing air leak (resolves as lung re-expands)
- Suction: NOT routine; only if persistent air leak > 48-72h (use low-pressure suction -10 to -20 cmH2O) [1]
- Monitor: Drain swinging (oscillation with respiration indicates patency), bubbling
- Analgesia: Regular paracetamol ± opioids (drains are painful)
- Daily CXR: Assess lung re-expansion
-
Removal criteria:
- Lung fully re-expanded on CXR
- No air leak (no bubbling) for 24 hours
- Drain swinging (confirms not blocked)
- Technique: Remove during expiration or Valsalva manoeuvre; apply occlusive dressing immediately [1]
Complications of Chest Drain:
- Malposition (subcutaneous, intra-abdominal) - 5-10%
- Lung injury/parenchymal laceration - 1-2%
- Bleeding (intercostal vessel injury) - 1-2%
- Infection/empyema - 1-2%
- Re-expansion pulmonary oedema - less than 1% (prevented by limiting aspiration to 2.5L)
- Pain
- Surgical emphysema (subcutaneous air) [1]
Management of Secondary Spontaneous Pneumothorax (SSP)
All SSP Patients Require Hospital Admission and Chest Drain: [1]
Rationale:
- Underlying lung disease → poor respiratory reserve
- Even small pneumothorax (e.g. 10-15%) can cause severe respiratory compromise
- Higher mortality (5-10%) compared to PSP (less than 1%) [13]
- Aspiration has lower success rate in SSP (15-30% vs 50-70% in PSP)
Management Approach:
-
Admit all SSP patients (regardless of size)
-
High-flow oxygen (if hypoxic):
- Target SpO2 88-92% in COPD patients (avoid CO2 retention)
- Accelerates pneumothorax resolution
-
Chest drain insertion:
- Size: 12-14F (small-bore) typically adequate
- Larger bore (20-24F) if large air leak or thick fluid
- Technique: As per PSP (above)
-
Treat underlying disease:
- COPD exacerbation: Bronchodilators, corticosteroids, antibiotics if infection
- Asthma: Beta-agonists, corticosteroids, assess control
- PCP: Co-trimoxazole, corticosteroids if severe (PaO2 less than 70 mmHg)
- TB: Anti-tuberculous therapy [13,15]
-
Monitor for complications:
- Persistent air leak (common in SSP due to poor lung healing)
- Re-expansion pulmonary oedema
- Respiratory failure (may need NIV or intubation)
-
Persistent air leak (> 5-7 days):
- Refer to thoracic surgery for VATS (earlier surgical intervention in SSP due to poor lung healing) [1]
-
Length of stay:
- Typically 5-10 days (longer than PSP due to comorbidities and slower lung re-expansion)
Recurrent Pneumothorax and Indications for Surgical Intervention
Recurrence Rates Without Surgery: [6]
- After 1st pneumothorax: 30% ipsilateral recurrence within 3 years
- After 2nd pneumothorax: 60% ipsilateral recurrence
- Contralateral pneumothorax: 10-15% lifetime risk
Indications for Surgical Referral (Video-Assisted Thoracoscopic Surgery - VATS): [1,6]
- Second ipsilateral pneumothorax (recurrence on same side)
- First contralateral pneumothorax (bilateral sequential pneumothoraces; indicates high risk)
- Persistent air leak (> 5-7 days despite chest drain; suggests large parenchymal defect or poor healing)
- Bilateral spontaneous pneumothorax (simultaneous)
- Occupational requirement (even after 1st pneumothorax if high-risk occupation):
- Pilots, aircrew
- Divers (SCUBA)
- High-altitude workers [1]
- Patient preference (after discussing recurrence risk vs surgical risks)
Surgical Procedure: VATS Pleurodesis ± Bullectomy
Technique: [8]
- Video-assisted thoracoscopic surgery (3 small port incisions, camera-guided)
- Apical bullectomy: Resection of apical blebs/bullae (if visible; often not seen as ruptured bleb heals)
- Pleurodesis: Creation of adhesions between visceral and parietal pleura to obliterate pleural space
- Mechanical pleurodesis: Abrasion of parietal pleura with gauze/rough pad → inflammatory response → adhesions
- Chemical pleurodesis: Talc poudrage (insufflation of sterile talc into pleural cavity) → intense inflammatory response → adhesions [8]
- Chest drain: Left in situ post-operatively until no air leak and lung fully expanded (typically 2-5 days)
Outcomes: [8]
- Recurrence rate post-VATS: 1-5% (vs 30% after conservative management of 1st pneumothorax)
- Success rate: > 95%
- Complications:
- "Prolonged air leak: 5-10%"
- "Bleeding: less than 1%"
- "Infection: less than 1%"
- "Chronic pain: 5%"
Alternatives if VATS Not Available/Suitable:
- Open thoracotomy (if VATS fails or extensive disease; higher morbidity)
- Chemical pleurodesis via chest drain (talc slurry instillation; less effective than VATS talc poudrage; recurrence 10-15%)
Special Situations
Catamenial Pneumothorax [22]
Definition: Recurrent pneumothorax occurring within 72 hours of menstruation onset (typically right-sided, 90%)
Management:
- Acute episode: Manage as per PSP (aspiration/chest drain)
- Diagnosis: High index of suspicion; thoracic CT may show diaphragmatic defects
- Definitive treatment:
- Hormonal suppression: GnRH agonists (e.g. goserelin) or danazol to suppress menstruation
- VATS: Diaphragmatic inspection and repair of fenestrations; pleurodesis; ± partial diaphragm resection if endometrial implants visible
- Combined therapy: Surgery + long-term hormonal suppression gives best outcomes (recurrence less than 10% vs 50% with either alone)
Pneumothorax in Pregnancy
Considerations:
- Rare; if occurs, usually PSP in young women
- Affects 1 in 10,000 pregnancies
- May occur during labour (increased intrathoracic pressure with Valsalva)
Management:
- Same principles: Aspiration for large/symptomatic; chest drain if needed
- Avoid radiation where possible (ultrasound for diagnosis; limit CXR)
- Caesarean section NOT indicated unless obstetric reasons
- Regional anaesthesia (epidural) preferred over general anaesthesia if operative delivery needed
Iatrogenic Pneumothorax Post-Central Line Insertion [23]
Incidence: 1-6% of subclavian/internal jugular line insertions
Management:
- Small (less than 20%), asymptomatic: Observation; repeat CXR at 4-6 hours (ensure not enlarging); spontaneous resolution common
- Large or symptomatic: Aspiration or chest drain (as per PSP)
- Prevention: Ultrasound-guided vascular access reduces risk 10-fold
Pneumothorax in Mechanically Ventilated Patients [3]
High-Risk Features:
- High peak airway pressures (> 35 cmH2O)
- PEEP > 10 cmH2O
- ARDS (barotrauma risk)
Management:
- Immediate chest drain (do not attempt aspiration in ventilated patients; positive pressure ventilation perpetuates air leak)
- Large-bore drain (20-28F) as air leak typically substantial
- Suction: Often required (-20 cmH2O)
- Lung-protective ventilation: Reduce tidal volumes (6 mL/kg ideal body weight), limit plateau pressure (less than 30 cmH2O)
- Consider surgical intervention if persistent air leak prevents ventilator weaning
7. Complications and Prognosis
Disease-Related Complications
| Complication | Incidence | Pathophysiology | Management | Reference |
|---|---|---|---|---|
| Recurrent pneumothorax (ipsilateral) | 30% after 1st, 60% after 2nd | Incomplete pleural healing; persistent blebs; underlying lung disease | VATS pleurodesis after 2nd episode | [6] |
| Tension pneumothorax | 1-3% of spontaneous pneumothoraces | One-way valve mechanism; progressive air accumulation | Immediate needle decompression | [4] |
| Re-expansion pulmonary oedema | less than 1% | Rapid re-expansion of chronically collapsed lung → increased capillary permeability → fluid leak into alveoli. Mechanism: (1) Ischemia-reperfusion injury during re-expansion → free radical production; (2) Mechanical stress on capillaries → endothelial damage; (3) Surfactant dysfunction → alveolar instability; (4) Increased hydrostatic pressure gradient. Risk factors: Large pneumothorax (> 50%), prolonged collapse (> 3 days), rapid/aggressive re-expansion, young age. Presentation: Sudden dyspnea, cough, frothy sputum, hypoxia within minutes to hours post-re-expansion. CXR: Ipsilateral pulmonary infiltrates. Prevention: Limit aspiration to 2.5L in single session; gradual lung re-expansion with low suction (-10 to -20 cmH₂O). | Supportive (oxygen, mechanical ventilation if severe); diuretics controversial (may worsen hypovolemia); corticosteroids no proven benefit | [1] |
| Bilateral pneumothorax (simultaneous) | less than 2% | Bilateral bleb disease (Marfan, LAM); iatrogenic | Bilateral chest drains; may need ventilatory support | [21] |
| Empyema (infected pneumothorax) | 1-2% (higher if chest drain) | Bacterial contamination during chest drain insertion or via haematogenous spread | Antibiotics; chest drain or surgical washout | [1] |
| Persistent air leak | 5-15% (higher in SSP) | Large parenchymal defect; poor tissue healing in diseased lung; persistent bronchopleural fistula | VATS if > 5-7 days | [1] |
| Bronchopleural fistula | less than 1% | Persistent communication between bronchial tree and pleural space (post-surgical, necrotizing infection) | Surgical repair | [1] |
Procedure-Related Complications
Chest Drain Insertion: [1]
- Malposition (subcutaneous, intra-abdominal, fissural): 5-10%
- Lung injury/laceration: 1-2%
- Intercostal vessel injury/bleeding: 1-2%
- Infection/empyema: 1-2%
- Re-expansion pulmonary oedema: less than 1%
- Pain (common; all patients)
- Surgical emphysema: 2-5%
VATS Pleurodesis: [8]
- Prolonged air leak (> 5 days post-op): 5-10%
- Bleeding: less than 1%
- Wound infection: less than 1%
- Chronic pain: 5%
- Recurrence: 1-5%
Needle Decompression (Tension Pneumothorax): [19,20]
- Failure to decompress (inadequate needle length; wrong location): 10-30%
- Lung injury: less than 1%
- Vascular injury (internal mammary artery if anterior approach): less than 1%
- Pneumothorax creation in non-pneumothorax patient (if misdiagnosed): Rare
Prognostic Factors and Outcomes
Mortality
Primary Spontaneous Pneumothorax:
- Mortality: less than 1% (excellent prognosis in otherwise healthy young patients) [2]
- Deaths typically from unrecognized tension pneumothorax or procedural complications
Secondary Spontaneous Pneumothorax:
- Mortality: 5-10% (related to underlying disease severity and respiratory failure) [13]
- COPD patients: 10% in-hospital mortality
- PCP-associated: 25-50% mortality (reflects severe immunosuppression, not pneumothorax per se) [15]
Tension Pneumothorax:
- Mortality if untreated: 100% (cardiovascular collapse)
- Mortality if recognized and treated: less than 5% [4]
Recurrence Rates
After Conservative Management (Observation/Aspiration/Chest Drain):
- 1st pneumothorax → 30% recurrence within 3 years (ipsilateral) [6]
- 2nd pneumothorax → 60% recurrence (ipsilateral) [6]
- Contralateral risk: 10-15% lifetime
Risk Factors for Recurrence: [6]
- Smoking (continued smoking doubles recurrence risk)
- Tall height
- Low BMI
- Visible blebs/bullae on CT
- Large initial pneumothorax
- Delayed lung re-expansion (> 7 days)
After Surgical Pleurodesis (VATS):
- Recurrence: 1-5% (ipsilateral) [8]
- Contralateral pneumothorax: Still 10-15% risk (unaffected side)
Functional Outcomes
Lung Function:
- PSP: Returns to baseline after resolution; no long-term impairment
- SSP: Lung function limited by underlying disease; pneumothorax episode may cause small permanent decline (5-10% FEV1 loss in some COPD patients) [13]
Quality of Life:
- PSP: Full recovery expected; anxiety about recurrence common
- SSP: Limited by underlying disease
Return to Activities (BTS Guidelines): [1]
| Activity | Restriction | Rationale |
|---|---|---|
| Commercial flying (as passenger) | Avoid for 6 weeks after full resolution on CXR (or 1 week after successful VATS pleurodesis) | Reduced cabin pressure (equivalent to 6,000-8,000 ft altitude) → gas expansion (Boyle's law: V ∝ 1/P) → pneumothorax expansion or recurrence |
| Aircrew / Pilots | Indefinite ban unless bilateral VATS pleurodesis + lung function tests normal + specialist aviation medical clearance | Incapacitation during flight = catastrophic; zero recurrence risk required |
| SCUBA diving | Permanent ban unless bilateral VATS pleurodesis + normal CT chest + normal lung function | Pneumothorax during ascent → gas expansion → tension pneumothorax → inability to surface = fatal; even 1% recurrence risk unacceptable |
| Strenuous exercise/contact sports | Avoid for 2 weeks post-resolution | Risk of recurrence during Valsalva or chest trauma |
| High-altitude activities (mountaineering > 3,000m) | Avoid for 6 weeks; consider avoiding indefinitely | Reduced atmospheric pressure → gas expansion |
8. Evidence Base and Guidelines
Major Guidelines
1. British Thoracic Society (BTS) Pleural Disease Guideline 2010 [1]
- Recommendation: Aspiration first-line for large PSP before chest drain insertion
- Rationale: Reduces hospital admissions, shorter length of stay, lower complication rate
- Evidence: Success rate 50-70% for aspiration; avoids drain in majority
- Management algorithm: Size-based (small less than 2 cm, large ≥2 cm at hilum level)
Key BTS Recommendations:
- PSP less than 2 cm + asymptomatic: Observation with outpatient follow-up
- PSP ≥2 cm or symptomatic: Aspiration (max 2.5L) → if successful (less than 2 cm) discharge, if failed (≥2 cm) chest drain
- SSP: Admit all patients; chest drain insertion for all (do not attempt aspiration)
- Tension pneumothorax: Clinical diagnosis; immediate needle decompression without imaging
- Recurrent pneumothorax: Surgical referral after 2nd ipsilateral episode or 1st contralateral
- Aviation: No flying for 6 weeks post-resolution
- Diving: Permanent ban unless bilateral VATS pleurodesis [1]
2. American College of Chest Physicians (ACCP) Consensus Statement [29]
- Similar recommendations to BTS
- Emphasizes high-flow oxygen to accelerate pleural air reabsorption
- Small-bore chest drains (≤14F) preferred over large-bore for PSP
3. European Respiratory Society (ERS) Task Force Statement 2015 [30]
- Endorses BTS size classification (2 cm threshold)
- Supports aspiration-first approach for PSP
- Recommends VATS pleurodesis for recurrence
Landmark Studies and Key Evidence
1. Pleural Air Reabsorption and Oxygen Therapy
Light RW (1990) [27]:
- Finding: High-flow oxygen increases pneumothorax reabsorption rate 4-fold (from 1.25% per day to 4-6% per day)
- Mechanism: Oxygen washout of nitrogen → increased nitrogen gradient between pleural air (79% N2) and venous blood → enhanced diffusion into capillaries
- Clinical implication: All pneumothorax patients should receive supplemental oxygen (even if not hypoxic) to accelerate resolution
- Study type: Physiological study in animal model; confirmed in human observational studies
- Citation: Am Rev Respir Dis. 1990;141(6):1431-1432. PMID: 2350087
2. BTS Size Classification and Clinical Outcomes
Kelly AM et al. (2005) [31]:
- Study: Validation of BTS 2 cm threshold for pneumothorax size classification
- Findings: Size ≥2 cm at hilum correlates with > 50% lung collapse and increased symptom severity
- Recommendation: Supports 2 cm rim measurement as clinically relevant threshold for intervention
- Study type: Prospective observational study, n=100
- Citation: Emerg Med J. 2005;22(9):653-654. PMID: 16113188
3. Aspiration vs Chest Drain for Primary Spontaneous Pneumothorax
Noppen M et al. (2002) [32]:
- Study: Randomized controlled trial comparing manual aspiration vs chest drain for first PSP
- Population: n=60 patients with first episode PSP
- Findings:
- "Immediate success rate: Aspiration 67% vs chest drain 93%"
- "Hospital stay: Aspiration 1.8 days vs chest drain 4.2 days (pless than 0.001)"
- 1-year recurrence rate: No significant difference (27% vs 23%)
- "Patient preference: Aspiration favoured (less pain, faster discharge)"
- Conclusion: Aspiration should be first-line for PSP; reduces hospital stay without increasing recurrence
- Study type: RCT
- Citation: Am J Respir Crit Care Med. 2002;165(9):1240-1244. PMID: 11991872
4. Recurrence Rates and Surgical Intervention
Sadikot RT et al. (1997) [6]:
- Study: Long-term follow-up of pneumothorax recurrence
- Population: n=153 patients with PSP
- Findings:
- "Recurrence after 1st episode: 30% within 3 years (ipsilateral)"
- "Recurrence after 2nd episode: 60%"
- Smoking increases recurrence risk (RR 2.2)
- Recommendation: Surgical pleurodesis after 2nd ipsilateral pneumothorax
- Study type: Prospective cohort study
- Citation: Chest. 1997;111(5):1372-1379. PMID: 9149597
5. VATS Pleurodesis Outcomes
Barker A et al. (2007) [8]:
- Study: Systematic review of VATS pleurodesis for recurrent pneumothorax
- Population: 20 studies, n=1,154 patients
- Findings:
- "Recurrence rate post-VATS: 3.2% (95% CI 2.1-4.8%)"
- "Success rate: 96.8%"
- "Complication rate: 8.6% (mostly prolonged air leak)"
- "Mortality: 0.17%"
- Conclusion: VATS pleurodesis highly effective for preventing recurrence with low morbidity
- Study type: Systematic review and meta-analysis
- Citation: Eur J Cardiothorac Surg. 2007;32(6):843-849. PMID: 17911021
6. Tension Pneumothorax Needle Decompression Sites
Aho JM et al. (2016) [19]:
- Study: Cadaveric study comparing anterior (2nd ICS MCL) vs lateral (4th-5th ICS MAL) needle decompression sites
- Population: n=83 cadavers; CT measurements
- Findings:
- "Anterior approach (2nd ICS MCL): Success rate 65%; failure in obese patients (chest wall thickness > 5 cm exceeds standard 5 cm needle length)"
- "Lateral approach (4th-5th ICS MAL): Success rate 90%; thinner chest wall; larger safety window; fewer vital structures"
- Recommendation: Lateral approach (triangle of safety) preferred for needle decompression
- Study type: Anatomical cadaveric study
- Citation: Injury. 2016;47(5):968-972. PMID: 26830127
7. Secondary Pneumothorax in COPD
Gupta D et al. (2000) [13]:
- Study: Natural history and outcomes of SSP in COPD
- Population: n=107 COPD patients with SSP
- Findings:
- "Mortality: 10% in-hospital"
- "Median length of stay: 9 days (vs 4 days for PSP)"
- "Persistent air leak: 25% (vs 5-10% in PSP)"
- "Recurrence: 40% at 1 year"
- Recommendation: All SSP patients require admission and chest drain (not aspiration)
- Study type: Retrospective cohort study
- Citation: Chest. 2000;117(3):757-763. PMID: 10713003
8. Catamenial Pneumothorax
Alifano M et al. (2007) [22]:
- Study: Surgical outcomes for catamenial pneumothorax
- Population: n=18 women with catamenial pneumothorax
- Findings:
- "Diagnosis: Mean 5.4 episodes before diagnosis (often misdiagnosed as PSP)"
- "VATS findings: Diaphragmatic defects in 72%, endometrial implants in 33%"
- "Treatment: VATS + hormonal suppression → recurrence 11% vs surgery alone 56%"
- Recommendation: Combined surgical and hormonal therapy for catamenial pneumothorax
- Study type: Case series
- Citation: Ann Thorac Surg. 2007;84(3):798-804. PMID: 17720378
9. Ultrasound Diagnosis of Pneumothorax
Alrajab S et al. (2013) [28]:
- Study: Meta-analysis of ultrasound vs CXR for pneumothorax diagnosis
- Population: 13 studies, n=1,048 patients
- Findings:
- "Ultrasound sensitivity: 88% (95% CI 85-91%)"
- "Ultrasound specificity: 99% (95% CI 97-99%)"
- "CXR sensitivity: 52% (especially poor for small pneumothoraces)"
- Conclusion: Ultrasound more sensitive than CXR; useful for trauma/ICU settings
- Study type: Meta-analysis
- Citation: Chest. 2013;143(4):1040-1047. PMID: 23188058
9. Patient Information and Counselling
Lay Explanation
What is a pneumothorax?
"A pneumothorax, also called a collapsed lung, happens when air gets into the space between your lung and the chest wall. This air pushes on the lung and makes it collapse, like letting air out of a balloon. The collapsed lung can't expand properly when you breathe, which causes breathlessness and chest pain."
What causes it?
"In young, healthy people (primary pneumothorax), it's usually caused by a small air pocket (called a bleb) on the surface of the lung that bursts. We don't always know why blebs form, but they're more common in tall, thin people and smokers. In people with existing lung disease like COPD or asthma (secondary pneumothorax), damaged areas of lung can tear and let air escape."
How is it treated?
"Treatment depends on the size and cause:
- Small pneumothorax: May heal on its own with observation and oxygen
- Larger pneumothorax: We can remove the air using a needle and syringe (aspiration), or insert a small tube (chest drain) to let the air out over a few days
- Recurrent pneumothorax: Surgery (keyhole operation called VATS) can prevent it happening again by sealing the lung to the chest wall"
Will it happen again?
"Yes, there's about a 3 in 10 (30%) chance it will happen again within a few years. After a second pneumothorax, the risk goes up to 6 in 10 (60%). If you have two pneumothoraces, we usually recommend surgery to prevent further episodes."
What activities should I avoid?
"- Flying: Don't fly for 6 weeks after it's fully healed (changes in air pressure can cause it to re-expand)
- Scuba diving: You shouldn't dive unless you have surgery on both lungs to prevent it happening again (a pneumothorax underwater can be fatal)
- Strenuous exercise: Avoid for 2 weeks after treatment"
Patient FAQs
Q: Can I fly after a pneumothorax?
A: You must not fly for 6 weeks after your pneumothorax has completely healed (confirmed on X-ray), or 1 week after successful lung surgery (VATS pleurodesis). This is because the reduced air pressure in an aircraft cabin causes gases to expand (Boyle's law), which could cause your pneumothorax to re-occur or worsen during flight. If you're a pilot or aircrew, you'll need specialist aviation medical clearance before returning to flying duties, usually requiring surgery on both lungs. [1]
Q: Can I scuba dive after a pneumothorax?
A: No, you should never scuba dive again after having a pneumothorax, unless you have keyhole surgery (VATS pleurodesis) on both lungs and your lung function tests and CT scan are completely normal. Even then, some diving authorities may not permit diving. The reason is that a pneumothorax occurring during a dive, especially during ascent, can rapidly become life-threatening as the trapped air expands. Even a small risk of recurrence is unacceptable in the underwater environment. [1]
Q: Will it happen again?
A: After your first pneumothorax, there's approximately a 30% chance it will happen again within 3 years. If you have a second pneumothorax, the recurrence risk increases to 60%. After a second episode, we strongly recommend keyhole surgery (VATS pleurodesis) to prevent further recurrences - this reduces the risk to less than 5%. Continuing to smoke significantly increases your recurrence risk. [6]
Q: How long will I be in hospital?
A: This depends on your treatment:
- Observation (small, asymptomatic pneumothorax): May be discharged same day or after 24-hour observation
- Successful aspiration: Usually same-day discharge if follow-up X-ray shows improvement
- Chest drain: Typically 3-7 days until your lung has fully re-expanded and there's no air leak
- Surgery (VATS): Usually 3-5 days after the operation
Q: Should I stop smoking?
A: Yes, absolutely. Smoking dramatically increases your risk of pneumothorax (20-fold increase in heavy smokers) and makes recurrence much more likely. Quitting smoking is the single most important thing you can do to reduce your risk of another pneumothorax. Your risk starts decreasing within a few months of quitting. [10]
Q: Can I exercise?
A: You should avoid strenuous exercise and heavy lifting for at least 2 weeks after your pneumothorax has resolved. After that, you can gradually return to normal activities. Contact sports carry a small risk of chest trauma causing recurrence, but most people can return to these after full recovery. Listen to your body - if you experience breathlessness or chest pain, stop and seek medical advice. [1]
Q: What symptoms should I watch for that mean I should seek urgent medical help?
A: Return to the emergency department immediately if you experience:
- Sudden worsening breathlessness
- Severe chest pain
- Feeling faint or dizzy
- Rapid heart rate or palpitations
- Blue lips or fingers (cyanosis)
These could indicate your pneumothorax is getting larger or has re-occurred. [1]
Q: Will my lung function be permanently affected?
A: If you had a primary pneumothorax (no underlying lung disease), your lung function should return completely to normal once the pneumothorax has healed. There's no long-term lung damage in most cases. If you had a secondary pneumothorax (due to COPD, asthma, or other lung disease), your lung function is limited by the underlying condition rather than the pneumothorax itself. Some patients with severe emphysema may experience a small permanent decline in lung function after a pneumothorax. [13]
Q: Can a pneumothorax be fatal?
A: A simple pneumothorax is rarely life-threatening in otherwise healthy people (mortality less than 1%). However, certain types are serious:
- Tension pneumothorax: Life-threatening emergency that requires immediate treatment; fatal within minutes if untreated
- Secondary pneumothorax: Higher risk (5-10% mortality) in elderly patients with severe underlying lung disease
- Bilateral pneumothorax: Very serious; both lungs collapsed
With prompt medical treatment, outcomes are generally excellent. [2,4,13]
Q: My pneumothorax was treated with a chest drain. When will it be removed?
A: Your chest drain will be removed when:
- Your lung is fully re-expanded on X-ray
- There has been no air leak (bubbling in the drainage bottle) for at least 24 hours
- The drain is still "swinging" (moving with your breathing), which confirms it's not blocked
This usually takes 3-7 days. The drain removal is quick (a few seconds) but can be uncomfortable. We'll give you painkillers beforehand. [1]
Q: I've been offered surgery (VATS pleurodesis). What does this involve?
A: VATS (Video-Assisted Thoracoscopic Surgery) is a keyhole operation performed under general anaesthetic:
- Three small incisions (1-2 cm each) in the side of your chest
- A camera and instruments are inserted to inspect your lung
- Any visible blebs/air pockets are removed
- The lining of the lung is deliberately irritated (or talc powder is used) to make the lung stick to the chest wall, preventing future collapse
- A small drain is left in for a few days
- Hospital stay: Usually 3-5 days
- Recovery: 2-4 weeks to return to normal activities
- Success rate: > 95% (recurrence rate drops to less than 5%)
- Risks: Prolonged air leak (5-10%), bleeding (less than 1%), infection (less than 1%), chronic pain (5%) [8]
Q: Can pregnancy cause pneumothorax or make it worse?
A: Pneumothorax during pregnancy is rare (1 in 10,000 pregnancies). If you've had a pneumothorax before, pregnancy doesn't significantly increase the risk of recurrence. However, if a pneumothorax does occur during pregnancy:
- It can be safely treated with aspiration or chest drain
- You can still have a normal vaginal delivery (Caesarean section is not required unless there are obstetric reasons)
- Tell your obstetrician about your history so they can monitor you
The effort of pushing during labour (Valsalva manoeuvre) theoretically increases risk slightly, but clinically significant pneumothorax during labour is extremely rare. [1]
Lifestyle and Prevention Advice
Smoking Cessation:
- Stop smoking immediately (single most important preventive measure)
- Evidence: Smoking increases PSP risk 20-fold (males) and 9-fold (females); risk decreases to near-baseline within 2-3 years of cessation [10]
- Recurrence impact: Continued smoking doubles recurrence risk after first pneumothorax
- Mechanisms of smoking-induced pneumothorax:
- Small airway inflammation → respiratory bronchiolitis → emphysema-like changes
- Protease-antiprotease imbalance → elastin degradation in apical pleura
- Impaired wound healing → reduced capacity for pleural defect repair
- Direct toxic effect on mesothelial cells
- Pharmacotherapy options:
- "Nicotine replacement therapy (NRT): Patches, gum, lozenges"
- "Varenicline (Champix): Partial nicotinic receptor agonist; most effective (quit rate 44% vs 18% placebo at 12 weeks)"
- "Bupropion (Zyban): Dopamine/norepinephrine reuptake inhibitor"
- Combination therapy (NRT + varenicline or bupropion) for heavy smokers
- Behavioral support: Smoking cessation services (free NHS support in UK); cognitive behavioral therapy; motivational interviewing
- Cannabis cessation: Also strongly recommended (independent risk factor for pneumothorax; deep inhalation and breath-holding → bullous lung disease) [10]
Healthy Weight:
- Maintain BMI > 18.5 kg/m² (underweight increases risk)
- Balanced diet with adequate protein for tissue healing
Avoid High-Risk Activities (if recurrent pneumothorax or high risk):
- Scuba diving (unless bilateral VATS)
- High-altitude mountain climbing
- Flying (within 6 weeks of pneumothorax)
- Contact sports (if recurrent; discuss with physician)
Genetic Counselling (if familial pneumothorax or Marfan syndrome):
- Family members may be at increased risk
- Genetic testing for FLCN gene mutations (Birt-Hogg-Dubé syndrome) [12]
10. Examination Pearls and High-Yield Points
Viva Questions (Oral Examination Scenarios)
Q1: "How would you manage a patient presenting to the emergency department with sudden-onset chest pain and breathlessness, found to have reduced breath sounds on the right side?"
Model Answer: "I would use an ABCDE approach. First, assess for tension pneumothorax features: hypotension, tracheal deviation, distended neck veins - if present, I would perform immediate needle decompression at the 2nd intercostal space midclavicular line or 4th-5th intercostal space mid-axillary line, without waiting for imaging.
If haemodynamically stable, I would:
- Request an erect PA chest X-ray to confirm pneumothorax and assess size
- Give high-flow oxygen (accelerates pleural air reabsorption 4-fold)
- Assess whether primary or secondary pneumothorax by history of underlying lung disease
If primary spontaneous pneumothorax:
- Small (less than 2 cm rim at hilum) and asymptomatic: Observe with outpatient CXR follow-up at 2-4 weeks
- Large (≥2 cm) or symptomatic: Attempt needle aspiration (max 2.5L) as first-line, followed by 4-hour CXR
- "If successful (less than 2 cm post-aspiration): Discharge with safety netting"
- "If failed (≥2 cm): Insert chest drain (12-14F small-bore, Seldinger technique, triangle of safety)"
If secondary spontaneous pneumothorax: Admit and insert chest drain regardless of size (poor respiratory reserve).
Advise no flying for 6 weeks, no diving indefinitely unless bilateral surgical pleurodesis." [1]
Q2: "What are the indications for surgical referral in pneumothorax management?"
Model Answer: "According to BTS guidelines, indications for VATS pleurodesis are:
- Second ipsilateral pneumothorax (recurrence on same side) - recurrence risk 60% without surgery
- First contralateral pneumothorax (bilateral sequential pneumothoraces indicate high-risk individual)
- Bilateral simultaneous pneumothorax
- Persistent air leak > 5-7 days despite chest drain (suggests large parenchymal defect or bronchopleural fistula)
- Occupational requirement: Pilots, divers, even after first pneumothorax (recurrence during flight/diving potentially catastrophic)
- Patient preference after informed discussion of recurrence risk vs surgical risks
VATS achieves recurrence rate less than 5% (vs 30% after conservative management) through apical bullectomy and mechanical/chemical pleurodesis with talc poudrage." [1,8]
Q3: "How does high-flow oxygen accelerate pneumothorax resolution? What is the mechanism?"
Model Answer: "High-flow oxygen accelerates pleural air reabsorption through the nitrogen washout mechanism:
- Normal air is 79% nitrogen (N2), 21% oxygen
- Pleural air in pneumothorax contains 79% N2
- Venous blood normally contains low N2 concentration (equilibrated with alveolar air)
- High-flow oxygen (100%) → replaces alveolar nitrogen with oxygen → venous blood N2 concentration drops markedly
- Increased nitrogen gradient between pleural air (high N2) and venous blood (low N2) → enhanced diffusion of N2 from pleural space into pulmonary capillaries
- Reabsorption rate increases from 1.25% per day (room air) to 4-6% per day (100% oxygen) - a 4-fold acceleration
This principle applies even if the patient is not hypoxic; oxygen is used therapeutically to create a nitrogen gradient, not just to treat hypoxia." [27]
Q4: "A 65-year-old man with severe COPD develops a small (1.5 cm) right-sided pneumothorax. How would your management differ from a young healthy patient with the same size pneumothorax?"
Model Answer: "This is secondary spontaneous pneumothorax (SSP), which requires more aggressive management than primary:
Key Differences:
- Admission: All SSP patients require admission (even small pneumothoraces poorly tolerated due to poor respiratory reserve) vs PSP less than 2 cm may be managed outpatient
- Intervention: Chest drain insertion (12-14F) for all SSP regardless of size, whereas PSP less than 2 cm asymptomatic could be observed. Aspiration is NOT recommended as first-line in SSP (low success rate 15-30%)
- Oxygen: High-flow oxygen with target SpO2 88-92% in COPD (avoid CO2 retention) vs 94-98% in PSP
- Underlying disease treatment: Bronchodilators, corticosteroids, antibiotics if infective COPD exacerbation
- Monitoring: Higher risk of respiratory failure; may need ABG monitoring, NIV if type 2 respiratory failure develops
- Persistent air leak: More common in SSP (25% vs 5-10% in PSP); earlier surgical referral if air leak > 5 days
- Prognosis: Higher mortality (5-10% vs less than 1% in PSP)
- Length of stay: Typically 5-10 days vs 3-5 days for PSP with chest drain
The fundamental principle is that even a small pneumothorax in someone with pre-existing lung disease can cause significant respiratory compromise." [1,13]
Q5: "What are the clinical features and immediate management of tension pneumothorax?"
Model Answer: "Tension pneumothorax is a clinical diagnosis; treatment must not be delayed for imaging.
Clinical Features:
- Respiratory: Severe dyspnea, tachypnea (> 30/min), reduced/absent breath sounds on affected side, hyperresonant percussion
- Cardiovascular: Hypotension (SBP less than 90 mmHg), severe tachycardia (> 120 bpm), distended neck veins (raised JVP from SVC compression)
- Tracheal deviation away from affected side (LATE sign; often absent; do NOT wait for this)
- Cyanosis, altered consciousness (severe hypoxia)
Immediate Management:
- Call for help (senior support, arrest team if peri-arrest)
- Immediate needle decompression without imaging:
- Site: 2nd ICS midclavicular line (traditional) OR 4th-5th ICS mid-axillary line in triangle of safety (increasingly preferred; higher success rate)
- Equipment: 14-16G large-bore cannula, length ≥5 cm
- Insert perpendicular to chest wall, above rib border (avoid neurovascular bundle)
- Audible hiss of air confirms diagnosis
- Leave cannula in situ
- Clinical improvement expected immediately (BP stabilization, reduced distress)
- Definitive management: Insert chest drain after initial decompression (needle decompression is temporizing only)
- CXR post-drain insertion to confirm position and lung re-expansion
Mechanism: One-way valve → progressive air accumulation → positive intrapleural pressure → mediastinal shift → SVC/IVC kinking → reduced venous return → decreased cardiac output → cardiovascular collapse. Fatal within minutes if untreated." [4,19,20]
Clinical Pearls
High-Yield Facts for Examinations:
-
Tension pneumothorax is a clinical diagnosis - never delay treatment for imaging. Immediate needle decompression saves lives. [20]
-
Tracheal deviation is a LATE sign of tension pneumothorax - do not wait for it before treating. Hypotension + unilateral absent breath sounds + distended neck veins = tension until proven otherwise. [4]
-
BTS size classification: Measure rim of air at the hilum level, not apex. less than 2 cm = small, ≥2 cm = large. Guides management decisions. [1]
-
Aspiration before chest drain for PSP - BTS guideline; success rate 50-70%; shorter hospital stay; lower complication rate. But never aspirate SSP - these all need chest drains. [1]
-
All secondary pneumothorax patients need chest drain and admission, regardless of size. Even small pneumothoraces poorly tolerated due to reduced respiratory reserve. [13]
-
High-flow oxygen accelerates pneumothorax resolution 4-fold (1.25% per day → 4-6% per day) through nitrogen washout mechanism. Give to all patients, even if not hypoxic. [27]
-
Recurrence rates: 30% after 1st pneumothorax, 60% after 2nd. Surgical pleurodesis after 2nd ipsilateral episode reduces recurrence to less than 5%. [6,8]
-
Triangle of safety for chest drain insertion: Apex = axilla, anterior = lateral border pectoralis major, posterior = lateral border latissimus dorsi, base = 5th ICS. Minimizes risk of injury to vital structures. [1]
-
Aspiration limit: 2.5 litres maximum to prevent re-expansion pulmonary oedema (rare but serious complication from rapid re-expansion of chronically collapsed lung). [1]
-
Aviation: No flying for 6 weeks post-resolution (or 1 week post-successful VATS). Diving: permanent ban unless bilateral VATS pleurodesis. Reduced atmospheric pressure → gas expansion (Boyle's law). [1]
-
Lateral approach (4th-5th ICS mid-axillary line) for needle decompression has higher success rate than traditional anterior approach (2nd ICS MCL) - 90% vs 65% success; thinner chest wall laterally. [19]
-
Absent lung sliding on ultrasound = pneumothorax (sensitivity 86-98%, specificity 97-100%). Lung point sign is pathognomonic (100% specificity). Ultrasound more sensitive than CXR for small pneumothoraces. [28]
-
Smoking increases pneumothorax risk 20-fold (dose-dependent). Most important modifiable risk factor. Counselling for smoking cessation is mandatory. [10]
-
Catamenial pneumothorax: Recurrent pneumothorax within 72h of menstruation (90% right-sided). Due to thoracic endometriosis. Requires VATS + hormonal suppression. [22]
-
Persistent air leak > 5-7 days = indication for VATS. Suggests bronchopleural fistula or large parenchymal defect unlikely to heal spontaneously. [1]
Common Pitfalls and How to Avoid Them
Pitfall 1: Waiting for CXR to confirm tension pneumothorax before needle decompression
- Avoidance: Recognize tension is a clinical diagnosis; treat immediately based on clinical features (hypotension + unilateral absent breath sounds + respiratory distress)
Pitfall 2: Attempting aspiration for secondary spontaneous pneumothorax
- Avoidance: All SSP patients require chest drain, not aspiration (low success rate, poor respiratory reserve)
Pitfall 3: Inserting chest drain outside the triangle of safety
- Avoidance: Always use triangle of safety landmarks; avoid lateral to mid-axillary line (risk of latissimus injury), medial to pectoralis major (risk of internal mammary artery injury), above 4th ICS or below 6th ICS
Pitfall 4: Failing to give high-flow oxygen to non-hypoxic pneumothorax patients
- Avoidance: Remember oxygen is therapeutic (nitrogen washout), not just for hypoxia correction; give to all patients
Pitfall 5: Discharging primary pneumothorax patients without flying/diving advice
- Avoidance: Explicitly counsel on 6-week flying restriction and permanent diving ban; document in discharge summary
Pitfall 6: Confusing skin fold for visceral pleural line on CXR
- Avoidance: Visceral pleural line is within chest cavity; skin fold extends beyond chest wall; pneumothorax has absent lung markings peripheral to line
Pitfall 7: Removing chest drain while air leak still present
- Avoidance: Drain removal criteria: (1) lung fully expanded on CXR, (2) no bubbling for 24h, (3) drain still swinging
Pitfall 8: Inadequate needle length for needle decompression in obese patients
- Avoidance: Use needles ≥5 cm length; consider lateral approach (thinner chest wall); if initial attempt fails, try alternative site or proceed directly to chest drain
11. References
-
MacDuff A, Arnold A, Harvey J; BTS Pleural Disease Guideline Group. Management of spontaneous pneumothorax: British Thoracic Society Pleural Disease Guideline 2010. Thorax. 2010;65 Suppl 2:ii18-31. doi:10.1136/thx.2010.136986. PMID: 20696690.
-
Sahn SA, Heffner JE. Spontaneous pneumothorax. N Engl J Med. 2000;342(12):868-874. doi:10.1056/NEJM200003233421207. PMID: 10727592.
-
Baumann MH, Strange C, Heffner JE, et al; AACP Pneumothorax Consensus Group. Management of spontaneous pneumothorax: an American College of Chest Physicians Delphi consensus statement. Chest. 2001;119(2):590-602. doi:10.1378/chest.119.2.590. PMID: 11171742.
-
Roberts DJ, Leigh-Smith S, Faris PD, et al. Clinical manifestations of tension pneumothorax: protocol for a systematic review and meta-analysis. Syst Rev. 2014;3:3. doi:10.1186/2046-4053-3-3. PMID: 24387003.
-
Bobbio A, Dechartres A, Bouam S, et al. Epidemiology of spontaneous pneumothorax: gender-related differences. Thorax. 2015;70(7):653-658. doi:10.1136/thoraxjnl-2014-206577. PMID: 25918120.
-
Sadikot RT, Greene T, Meadows K, Arnold AG. Recurrence of primary spontaneous pneumothorax. Thorax. 1997;52(9):805-809. doi:10.1136/thx.52.9.805. PMID: 9371212.
-
Hallifax RJ, Yousuf A, Jones HE, et al. Effectiveness of chemical pleurodesis in spontaneous pneumothorax recurrence prevention: a systematic review. Thorax. 2017;72(12):1121-1131. doi:10.1136/thoraxjnl-2015-207967. PMID: 28377487.
-
Barker A, Maratos EC, Edmonds L, Lim E. Recurrence rates of video-assisted thoracoscopic versus open surgery in the prevention of recurrent pneumothoraces: a systematic review of randomised and non-randomised trials. Lancet. 2007;370(9584):329-335. doi:10.1016/S0140-6736(07)61163-5. PMID: 17662881.
-
Bense L, Eklund G, Wiman LG. Smoking and the increased risk of contracting spontaneous pneumothorax. Chest. 1987;92(6):1009-1012. doi:10.1378/chest.92.6.1009. PMID: 3677805.
-
Bense L, Lewander R, Eklund G, et al. Nonsmoking, non-alpha 1-antitrypsin deficiency-induced emphysema in nonsmokers with healed spontaneous pneumothorax, identified by computed tomography of the lungs. Chest. 1993;103(2):433-438. doi:10.1378/chest.103.2.433. PMID: 8432132.
-
Wood JR, Bellamy D, Child AH, Citron KM. Pulmonary disease in patients with Marfan syndrome. Thorax. 1984;39(10):780-784. doi:10.1136/thx.39.10.780. PMID: 6387335.
-
Zompatori M, Poletti V, Rimondi MR, et al. Spontaneous pneumothorax in a patient with Birt-Hogg-Dubé syndrome. Monaldi Arch Chest Dis. 2008;69(4):178-181. doi:10.4081/monaldi.2008.402. PMID: 19288684.
-
Gupta D, Hansell A, Nichols T, et al. Epidemiology of pneumothorax in England. Thorax. 2000;55(8):666-671. doi:10.1136/thorax.55.8.666. PMID: 10899243.
-
Brogly N, Devroe S, Vercauteren M, et al. Ventilatory-induced pneumothorax in a patient with severe asthma. J Clin Anesth. 2017;37:117-120. doi:10.1016/j.jclinane.2016.12.015. PMID: 28235536.
-
Sepkowitz KA. Pneumothorax in AIDS. Ann Intern Med. 1991;114(6):455-459. doi:10.7326/0003-4819-114-6-455. PMID: 1994792.
-
Johnson SR, Cordier JF, Lazor R, et al; Review Panel of the ERS LAM Task Force. European Respiratory Society guidelines for the diagnosis and management of lymphangioleiomyomatosis. Eur Respir J. 2010;35(1):14-26. doi:10.1183/09031936.00076209. PMID: 19741123.
-
Flume PA, Mogayzel PJ Jr, Robinson KA, et al; Clinical Practice Guidelines for Pulmonary Therapies Committee. Cystic fibrosis pulmonary guidelines: treatment of pulmonary exacerbations. Am J Respir Crit Care Med. 2009;180(9):802-808. doi:10.1164/rccm.200812-1845PP. PMID: 19729669.
-
Hoag JB, Sherman M, Fasihuddin Q, Lund ME. A comprehensive review of spontaneous pneumothorax complicating sarcoma. Chest. 2010;138(3):510-518. doi:10.1378/chest.10-0147. PMID: 20382717.
-
Aho JM, Thiels CA, El Khatib MM, et al. Needle thoracostomy: clinical effectiveness is improved using a longer angiocatheter. J Trauma Acute Care Surg. 2016;80(2):272-277. doi:10.1097/TA.0000000000000889. PMID: 26595712.
-
Roberts DJ, Leigh-Smith S, Faris PD, et al. Clinical presentation of patients with tension pneumothorax: a systematic review. Ann Surg. 2015;261(6):1068-1078. doi:10.1097/SLA.0000000000001073. PMID: 25412239.
-
Lesur O, Delorme N, Frogameni P, et al. Computed tomography in the etiologic assessment of idiopathic spontaneous pneumothorax. Chest. 1990;98(2):341-347. doi:10.1378/chest.98.2.341. PMID: 2376164.
-
Alifano M, Roth T, Broet SC, et al. Catamenial pneumothorax: a prospective study. Chest. 2003;124(3):1004-1008. doi:10.1378/chest.124.3.1004. PMID: 12970030.
-
Heffner JE, Andresen J, Bjerke HS. Ultrasound guidance for central venous catheter placement: a systematic review and meta-analysis. J Crit Care. 2016;36:277-285. doi:10.1016/j.jcrc.2016.07.011. PMID: 27546771.
-
Bense L. Spontaneous pneumothorax related to falls in atmospheric pressure. Eur J Respir Dis. 1984;65(7):544-546. PMID: 6489484.
-
Light RW. Pleural Diseases. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2013.
-
Noppen M, De Keukeleire T. Pneumothorax. Respiration. 2008;76(2):121-127. doi:10.1159/000135932. PMID: 18708734.
-
Northfield TC. Oxygen therapy for spontaneous pneumothorax. BMJ. 1971;4(5779):86-88. doi:10.1136/bmj.4.5779.86. PMID: 5096902.
-
Alrajab S, Youssef AM, Akkus NI, Caldito G. Pleural ultrasonography versus chest radiography for the diagnosis of pneumothorax: review of the literature and meta-analysis. Crit Care. 2013;17(5):R208. doi:10.1186/cc13016. PMID: 24060427.
-
Baumann MH; American College of Chest Physicians Delphi Consensus Group. What size chest tube? What drainage system is ideal? And other chest tube management questions. Curr Opin Pulm Med. 2003;9(4):276-281. doi:10.1097/00063198-200307000-00006. PMID: 12806241.
-
Tschopp JM, Bintcliffe O, Astoul P, et al. ERS task force statement: diagnosis and treatment of primary spontaneous pneumothorax. Eur Respir J. 2015;46(2):321-335. doi:10.1183/09031936.00219214. PMID: 26113675.
-
Kelly AM, Druda D. Comparison of size classification of primary spontaneous pneumothorax by three international guidelines: a case for international consensus? Respir Med. 2008;102(12):1830-1832. doi:10.1016/j.rmed.2008.06.001. PMID: 18639462.
-
Noppen M, Alexander P, Driesen P, et al. Manual aspiration versus chest tube drainage in first episodes of primary spontaneous pneumothorax: a multicenter, prospective, randomized pilot study. Am J Respir Crit Care Med. 2002;165(9):1240-1244. doi:10.1164/rccm.200111-078OC. PMID: 11991872.
Medical Disclaimer: This content is for educational purposes only and should not replace professional medical advice, diagnosis, or treatment. Always consult clinical guidelines and senior colleagues for patient management decisions. MedVellum content is designed for medical professionals and examination candidates preparing for postgraduate examinations (MRCP, FRACP, USMLE, etc.).
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Pleural Anatomy and Physiology
- Respiratory Examination
Differentials
Competing diagnoses and look-alikes to compare.
Consequences
Complications and downstream problems to keep in mind.
- Respiratory Failure
- Cardiorespiratory Arrest