Crohn's Disease
Crohn's disease is a chronic, relapsing-remitting inflammatory bowel disease (IBD) characterised by transmural inflammat... MRCP exam preparation.
What matters first
Crohn's disease is a chronic, relapsing-remitting inflammatory bowel disease (IBD) characterised by transmural inflammat... MRCP exam preparation.
Bowel obstruction
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Bowel obstruction
- Fistulae (enterocutaneous, enteroenteric, enterovesical)
- Intra-abdominal abscess
- Perforation
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Intestinal Tuberculosis
- Bowel Malignancy
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Crohn's Disease
1. Topic Overview
Summary
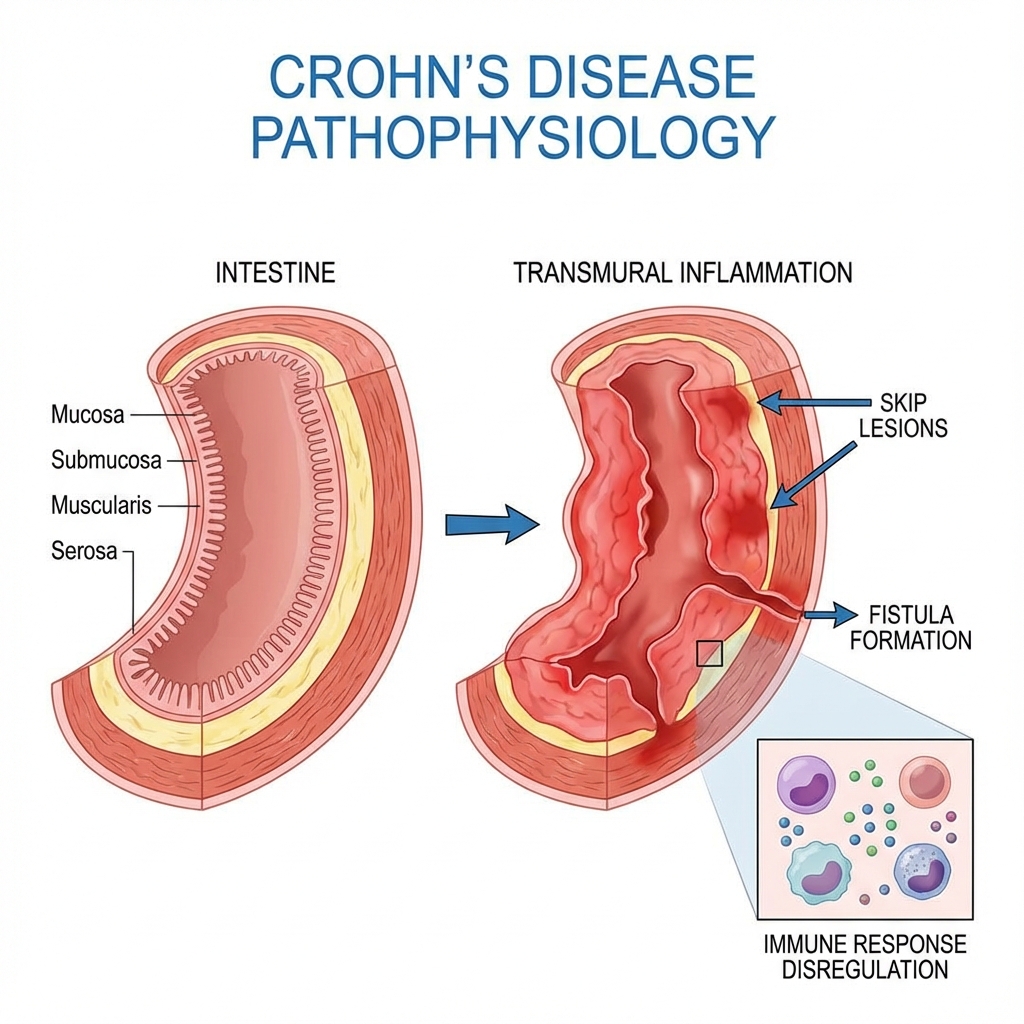
Crohn's disease is a chronic, relapsing-remitting inflammatory bowel disease (IBD) characterised by transmural inflammation that can affect any part of the gastrointestinal tract from mouth to anus. Unlike ulcerative colitis, Crohn's demonstrates discontinuous "skip lesions" with intervening normal mucosa, and the inflammation extends through all layers of the bowel wall, predisposing to strictures, fistulae, and abscesses. [1,2]
The terminal ileum is the most commonly affected site (30-40% of cases), followed by ileocolonic disease (40-50%), isolated colonic involvement (15-25%), and upper gastrointestinal disease (5%). [1] The transmural nature of inflammation is the hallmark feature distinguishing Crohn's from ulcerative colitis, leading to characteristic complications including fibrostenotic strictures, penetrating disease with fistula formation, and abscess development. [2,3]
The pathogenesis involves a complex interplay between genetic susceptibility (notably NOD2/CARD15, ATG16L1, IL23R, and IRGM variants), environmental triggers (smoking, diet, microbiome alterations, antibiotic exposure), intestinal barrier dysfunction, and aberrant immune responses characterised by excessive Th1 and Th17 cytokine production. [4,5] The disease affects approximately 3.1 million adults in Europe and North America, with incidence rates of 5-20 per 100,000 person-years in high-income countries and rising incidence in newly industrialised nations. [6]
Modern management has evolved from a purely symptomatic "step-up" approach to a proactive "treat-to-target" strategy, aiming for deep remission with endoscopic mucosal healing and transmural healing on cross-sectional imaging. [7] Biological therapies, including anti-TNF agents (infliximab, adalimumab), anti-integrin therapy (vedolizumab), anti-IL-12/23 agents (ustekinumab), and selective IL-23 inhibitors (risankizumab), have revolutionised treatment by achieving sustained remission, preventing complications, and reducing surgical intervention rates. [8,9,10]
Surgery remains non-curative with recurrence rates of 30-50% at 5 years post-resection, necessitating lifelong medical surveillance and prophylactic therapy. [11,12] A multidisciplinary approach involving gastroenterologists, colorectal surgeons, specialist nurses, dietitians, radiologists, and when needed, psychologists, is essential for optimal patient outcomes.
Key Facts at a Glance
| Feature | Details |
|---|---|
| Definition | Chronic transmural inflammatory bowel disease affecting any GI tract segment |
| Age of Onset | Bimodal: peak 15-30 years; second smaller peak 50-70 years |
| Most Common Location | Terminal ileum (30-40%), ileocolonic (40-50%) |
| Pattern | Skip lesions, transmural inflammation, cobblestone mucosa |
| Histology | Non-caseating granulomas (30-40% of cases), transmural lymphoid aggregates |
| Smoking Effect | Increases risk 2-fold, worsens disease activity (opposite to UC) |
| Surgery Rate | 50-70% require surgery within 10 years of diagnosis |
| Recurrence Post-Surgery | 30-50% at 5 years (endoscopic), 10-30% clinical recurrence |
| Inheritance | First-degree relative: 10-15% lifetime risk |
| Mortality | Standardised mortality ratio 1.4-1.5× general population |
Clinical Pearls for Examinations
"Terminal Ileum, Transmural, Skip Lesions" — The classic triad distinguishing Crohn's from UC.
"Perianal Disease = Think Crohn's" — Perianal fistulae, skin tags, and abscesses occur in 25-35% of Crohn's patients but are rare in UC, making them diagnostically useful.
"Granulomas are Pathognomonic but Not Sensitive" — Non-caseating granulomas confirm Crohn's when present, but their absence does not exclude it (found in only 30-40% of biopsies).
"Smoking Paradox" — Smoking doubles the risk of developing Crohn's and worsens outcomes (opposite effect to UC where smoking is protective).
"Top-Down vs Step-Up" — Early aggressive treatment with biologics in high-risk patients (young age, perianal disease, extensive disease, deep ulceration) may alter disease course and reduce complications.
"Treat-to-Target Strategy" — Aim for clinical remission plus objective evidence of mucosal healing (calprotectin less than 150-250 μg/g, endoscopic remission SES-CD ≤2), not just symptom control.
"Post-Operative Recurrence is the Rule" — 70% develop endoscopic recurrence at 1 year; prophylaxis with thiopurines, methotrexate, or anti-TNF reduces this significantly.
Why This Matters Clinically
Crohn's disease is a debilitating lifelong condition with substantial impact on quality of life, educational attainment, employment, and psychosocial wellbeing. Without adequate treatment, the natural history involves progressive bowel damage with stricture and fistula formation, repeated surgeries, and risk of short bowel syndrome.
The paradigm shift toward early intensive therapy and tight disease monitoring has demonstrated superior outcomes: reduced hospitalisations, fewer surgical interventions, and improved long-term remission rates. [7,13] Recognising high-risk features at diagnosis allows stratification for early biologic therapy, preventing irreversible complications.
Moreover, Crohn's increases colorectal cancer risk in patients with colonic involvement (cumulative risk 2.9% at 10 years, 5.6% at 20 years, 8.3% at 30 years with extensive colitis), necessitating surveillance colonoscopy. [14] Extra-intestinal manifestations, medication-related complications (immunosuppression, osteoporosis, malignancy), and psychosocial burden require holistic, multidisciplinary care.
2. Epidemiology
Incidence and Prevalence
Global Distribution:
- Highest incidence: Northern Europe, United Kingdom, North America (10-20 per 100,000 person-years) [6]
- Intermediate: Southern Europe, Australia, New Zealand (5-10 per 100,000)
- Rapidly increasing: Asia, South America, Africa (previously rare, now 1-5 per 100,000) [6]
- Prevalence in Western countries: 100-322 per 100,000 population [6]
Temporal Trends:
- Incidence stabilised or decreasing in historically high-incidence regions (Western Europe, North America)
- Sharply increasing in newly industrialised countries (South Korea, China, India, Brazil) — "westernisation" effect
- Overall prevalence continues to rise due to decreasing mortality and chronic nature
Age and Sex Distribution
| Factor | Characteristics |
|---|---|
| Age at Diagnosis | Bimodal: primary peak 15-30 years (60-70%), secondary peak 50-70 years (10-15%) |
| Sex Ratio | Slight female predominance (1.1-1.3:1), but varies by population |
| Paediatric Onset | 20-30% diagnosed before age 20; more extensive disease, higher genetic burden |
| Elderly Onset | 10-15% diagnosed after age 60; more colonic disease, fewer strictures/fistulae |
Ethnicity and Geography
- Highest risk: Ashkenazi Jewish populations (2-4× higher than general population)
- Caucasians more affected than other ethnic groups in historically high-prevalence areas
- Urban > Rural: Urban dwellers have 1.5-2× higher risk
- North-South gradient: Higher incidence at northern latitudes (potential Vitamin D/sunlight association)
- Migrant studies: Risk increases in migrants from low to high-incidence regions within one generation, supporting environmental factors [6]
Risk Factors
Genetic Factors
| Gene | Function | Effect Size | Notes |
|---|---|---|---|
| NOD2/CARD15 | Bacterial pattern recognition | OR 2-4 (heterozygous), 20-40 (homozygous) | First identified susceptibility gene; 3 main mutations; predicts ileal disease, early onset, stricturing [4] |
| ATG16L1 | Autophagy pathway | OR 1.2-1.5 | Impaired bacterial handling, altered Paneth cell function [4] |
| IL23R | IL-23 receptor signaling | OR 1.3-1.4 | Protective variants exist; validates IL-23 as therapeutic target |
| IRGM | Autophagy, immunity | OR 1.2-1.3 | Bacterial clearance defect |
| FUT2 | Fucosyltransferase | OR 1.1-1.2 | Alters gut microbiome composition |
- Family history: First-degree relative with Crohn's confers 10-15% lifetime risk (vs 0.5% general population)
- Twin concordance: Monozygotic twins 50-60%, dizygotic 10% — supports polygenic inheritance
- Over 240 susceptibility loci identified by GWAS — collectively explain ~25% of disease heritability [4,5]
Environmental Factors
| Factor | Effect | Evidence Level |
|---|---|---|
| Smoking | Increases risk 2×; worsens disease severity and recurrence | Strong (Level 1) [15] |
| Appendicectomy | Possible modest protective effect (OR 0.7-0.8) — mechanism unclear | Moderate |
| Antibiotics in childhood | Repeated courses increase risk (OR 1.5-2.5) | Moderate [6] |
| Diet | Western diet (high processed foods, low fiber) associated with increased risk | Moderate |
| Breastfeeding | Protective effect (OR 0.6-0.8 if breastfed > 6 months) | Moderate |
| NSAIDs | May trigger flares in susceptible individuals | Moderate |
| Oral contraceptives | Weak association with increased risk (OR 1.3-1.5) | Weak |
| Stress/Psychological factors | May trigger flares but not causative of disease | Weak |
Microbiome Alterations
- Dysbiosis: Reduced diversity, decreased Firmicutes (especially Faecalibacterium prausnitzii), increased Enterobacteriaceae and adherent-invasive E. coli (AIEC) [16]
- Mycobacterium avium paratuberculosis (MAP): Controversial — detected in some Crohn's tissue but causative role unproven
3. Aetiology and Pathophysiology
The Multi-Hit Hypothesis
Crohn's disease arises from the convergence of four key pathogenic factors: [4,5]
- Genetic susceptibility — polygenic inheritance conferring impaired bacterial sensing, autophagy defects, and aberrant immune responses
- Environmental triggers — smoking, dysbiosis, infections, diet, antibiotics
- Barrier dysfunction — increased intestinal permeability ("leaky gut"), impaired mucus layer
- Dysregulated immune response — loss of tolerance to commensal bacteria, excessive Th1/Th17 activation, inadequate regulatory T cell function
Detailed Pathophysiological Mechanism
Step 1: Genetic Predisposition and Barrier Dysfunction
NOD2/CARD15 Mutations (most important genetic factor):
- NOD2 is an intracellular pattern recognition receptor that detects bacterial peptidoglycan (muramyl dipeptide)
- Mutations impair bacterial sensing → defective autophagy → inadequate intracellular bacterial clearance → persistent inflammation [4]
- Predominantly affects ileal disease; patients with two mutant alleles have 20-40× increased risk
Autophagy Gene Defects (ATG16L1, IRGM):
- Autophagy is essential for clearing intracellular bacteria and regulating inflammatory responses
- Defects lead to impaired Paneth cell function (reduced antimicrobial peptide secretion), persistent bacterial antigens, and prolonged inflammation [4]
Barrier Dysfunction:
- Tight junction disruption (altered claudins, occludin, zonula occludens) increases intestinal permeability
- Allows bacterial translocation into submucosa and lamina propria
- Reduced mucus production (goblet cell dysfunction) facilitates bacterial-epithelial contact
Step 2: Dysbiosis and Environmental Triggers
Microbial Dysbiosis: [16]
- Reduced alpha diversity (fewer species)
- Loss of protective species (e.g., F. prausnitzii produces butyrate — anti-inflammatory short-chain fatty acid)
- Expansion of pathobionts (adherent-invasive E. coli — AIEC) that adhere to and invade epithelial cells, survive in macrophages, induce TNF-α
Smoking:
- Alters mucus composition, increases intestinal permeability, modifies microbiome
- Affects immune function: increases pro-inflammatory cytokines
- Smokers have 2× risk, worse prognosis, higher recurrence post-surgery [15]
Step 3: Innate Immune Activation
Dendritic Cells and Macrophages:
- Encounter translocated bacteria and damage-associated molecular patterns (DAMPs)
- Activate via Toll-like receptors (TLRs) and NOD2 → produce IL-12, IL-23, TNF-α
- Present antigens to naïve T cells in mesenteric lymph nodes
Neutrophil Infiltration:
- Early hallmark; neutrophils accumulate in crypts (crypt abscesses less prominent than UC)
- Release reactive oxygen species, proteases → tissue damage
Step 4: Adaptive Immune Dysregulation (Th1/Th17 Dominance)
Th1 Pathway:
- Driven by IL-12 from dendritic cells/macrophages
- Th1 cells produce IFN-γ, TNF-α → activate macrophages → perpetuate inflammation
- Characteristic of Crohn's (vs Th2 in UC)
Th17 Pathway:
- Driven by IL-23 (heterodimer of IL-23p19 and IL-12p40)
- Th17 cells produce IL-17A, IL-17F, IL-22 → neutrophil recruitment, epithelial barrier disruption
- IL-23/IL-17 axis is central to Crohn's pathogenesis — validated by efficacy of IL-23 inhibitors [9,10]
Regulatory T Cell (Treg) Dysfunction:
- Reduced number or impaired suppressive function of CD4+CD25+FoxP3+ Tregs
- Failure to downregulate effector T cell responses → uncontrolled inflammation
TNF-α as Master Cytokine:
- Produced by macrophages, T cells, fibroblasts
- Promotes leukocyte recruitment, activates endothelium, induces apoptosis, stimulates matrix metalloproteinases
- Neutralising TNF-α (infliximab, adalimumab) is highly effective therapeutically [8]
Step 5: Transmural Inflammation and Complications
Transmural Extension:
- Inflammation extends from mucosa → submucosa → muscularis propria → serosa
- Lymphoid aggregates form throughout bowel wall (transmural lymphoid aggregation)
- Granulomas (compact collections of epithelioid histiocytes) develop in 30-40% — non-caseating, pathognomonic when present [1,2]
Fibrosis and Stricture Formation:
- Chronic inflammation → transforming growth factor-β (TGF-β) activation → fibroblast proliferation → collagen deposition
- Leads to fibrostenotic strictures (30-40% of patients develop stricturing over time)
- Strictures cause obstructive symptoms (cramping pain, vomiting, bloating)
Fistula and Abscess Formation (Penetrating Disease):
- Transmural inflammation erodes through serosa → sinus tracts → fistulae to adjacent structures
- Types: Enteroenteric (bowel-to-bowel), enterocutaneous (bowel-to-skin), enterovesical (bowel-to-bladder), enterovaginal, perianal
- Bacterial seeding of sinus tracts → abscess formation (require drainage + antibiotics)
- 20-40% develop penetrating complications over disease course [3]
Skip Lesions:
- Patchy inflammation with areas of normal intervening mucosa
- Results from focal bacterial translocation and localized immune activation
- Contrasts with continuous inflammation in ulcerative colitis
Macroscopic and Microscopic Features
Macroscopic (Endoscopy/Surgery):
- Aphthous ulcers (early): small superficial erosions on otherwise normal mucosa
- Deep longitudinal and transverse ulcers → "cobblestone" appearance (pathognomonic)
- Skip lesions: inflamed segments separated by normal bowel
- Thickened, rigid bowel wall (transmural inflammation, fibrosis)
- Mesenteric fat wrapping ("creeping fat") — adipose tissue extends over serosal surface
- Strictures (narrowed lumen), fistulae, abscesses
Microscopic (Histology):
- Transmural inflammation with lymphoid aggregates in all bowel wall layers
- Non-caseating granulomas (30-40%): well-formed collections of epithelioid histiocytes without central necrosis — pathognomonic but not required for diagnosis [1,2]
- Focal cryptitis (patchy, less severe than UC)
- Preserved goblet cells (unlike UC where depleted)
- Pyloric gland metaplasia in severe cases
- Fissuring ulcers extending deep into bowel wall
- Neural hyperplasia, vascular proliferation
4. Classification Systems
Montreal Classification (2005)
The Montreal Classification categorises Crohn's disease by age at diagnosis, location, and behaviour — essential for prognostication and treatment planning. [17]
Age at Diagnosis (A)
| Category | Age Range | Notes |
|---|---|---|
| A1 | Below 16 years | Paediatric; more extensive disease, higher genetic load |
| A2 | 17-40 years | Peak incidence group |
| A3 | Above 40 years | Often less aggressive; higher proportion of colonic disease |
Location (L)
| Category | Location | Frequency | Notes |
|---|---|---|---|
| L1 | Ileal (terminal ileum ± limited cecal involvement) | 30-40% | Classic presentation; highest stricture risk |
| L2 | Colonic (colon ± rectum, no upper GI or ileal) | 15-25% | Resembles UC; lower fistula risk |
| L3 | Ileocolonic (ileum + any colonic segment) | 40-50% | Most common; highest complication rate |
| L4 modifier | Upper GI (proximal to terminal ileum) | 5-10% | Esophagus, stomach, duodenum, jejunum; add to L1-L3 |
Behaviour (B)
| Category | Behaviour | Frequency at Diagnosis | Notes |
|---|---|---|---|
| B1 | Non-stricturing, non-penetrating (inflammatory) | 70-80% | Initial phenotype; may progress to B2/B3 |
| B2 | Stricturing (stenosis, obstruction) | 10-15% at diagnosis, 30-40% over time | Fibrotic; may need endoscopic dilation or surgery |
| B3 | Penetrating (fistulae, abscess, perforation) | 5-10% at diagnosis, 20-40% over time | Higher morbidity; often requires surgery |
| p modifier | Perianal disease modifier | 25-35% overall | Add "p" to B1/B2/B3 (e.g., B2p = stricturing + perianal) |
Clinical Significance:
- Disease phenotype evolves over time: inflammatory (B1) → stricturing (B2) or penetrating (B3) in 30-50% within 10 years
- L1+B3 (ileal penetrating) has high risk of abscess
- B3p (penetrating + perianal) requires aggressive medical therapy (anti-TNF first-line) and often surgical intervention
- L3 (ileocolonic) has highest overall complication rate
Disease Activity Indices
Crohn's Disease Activity Index (CDAI)
The CDAI is a research tool (rarely used clinically) scoring:
- Stool frequency, abdominal pain, general wellbeing (each scored 0-3 daily for 7 days)
- Extra-intestinal manifestations, anti-diarrhoeal use, abdominal mass, haematocrit, body weight
Scoring:
- less than 150: Remission
- 150-220: Mild disease
- 220-450: Moderate disease
-
450: Severe disease
Clinical response in trials: decrease ≥70 points or ≥100 points (depending on study)
Harvey-Bradshaw Index (HBI) — Simplified CDAI
More practical for clinical use (single-day assessment):
| Component | Score |
|---|---|
| General wellbeing | 0 (very well) – 4 (terrible) |
| Abdominal pain | 0 (none) – 3 (severe) |
| Number of liquid stools per day | 1 point per stool |
| Abdominal mass | 0 (none), 1 (dubious), 2 (definite), 3 (tender) |
| Complications | 1 point each: arthralgia, uveitis, erythema nodosum, aphthous ulcers, pyoderma, anal fissure, fistula, abscess |
Interpretation:
- less than 5: Remission
- 5-7: Mild disease
- 8-16: Moderate disease
-
16: Severe disease
Endoscopic Scores
Simple Endoscopic Score for Crohn's Disease (SES-CD):
- Assesses 5 colonic segments: ileum, right colon, transverse, left colon, rectum
- Scores ulcer size (0-3), ulcerated surface (0-3), affected surface (0-3), stenosis (0-3) per segment
- Total 0-60; ≤2 = mucosal healing (treatment target) [7]
Rutgeerts Score (Post-Operative Recurrence): Evaluates neo-terminal ileum after ileocolic resection:
- i0: No lesions
- i1: ≤5 aphthous lesions
- i2: > 5 aphthous lesions, skip areas, or lesions confined to anastomosis
- i3: Diffuse aphthous ileitis
- i4: Diffuse inflammation with ulcers, nodules, stenosis
i2-i4 predict clinical recurrence and warrant treatment escalation. [11,12]
5. Clinical Presentation
Cardinal Symptoms
Gastrointestinal:
- Chronic diarrhoea (typically 3-6 stools/day): usually non-bloody (ileal disease) or mixed blood/mucus (colonic disease)
- Abdominal pain: Cramping, colicky, often right lower quadrant (terminal ileum); aggravated by eating (fear of food if stricturing)
- Weight loss: Due to malabsorption, anorexia, fear of eating, catabolism from chronic inflammation
- Rectal bleeding: Less prominent than UC; suggests colonic involvement
- Perianal symptoms: Pain, discharge, swelling (fistulae, abscesses, fissures) — 25-35% of patients [3]
Constitutional:
- Fatigue: Multifactorial (anaemia, inflammation, poor sleep, malnutrition)
- Fever: Low-grade in active disease; high-grade suggests abscess or severe inflammation
- Anorexia, malaise
- Growth failure in children/adolescents (delayed puberty, short stature)
Oral Manifestations:
- Aphthous ulcers (5-10%): small painful oral ulcers; correlate with disease activity
- Cobblestoning of buccal mucosa (rare)
- Angular cheilitis (associated with nutritional deficiency)
Clinical Examination Findings
General Inspection
| Finding | Significance |
|---|---|
| Cachexia, muscle wasting | Chronic malnutrition, malabsorption |
| Pallor | Anaemia (iron deficiency, anaemia of chronic disease, B12 deficiency) |
| Clubbing | Rare; suggests chronic inflammation, associated with liver disease |
| Fever | Active inflammation, abscess, perforation |
| Growth retardation | Paediatric patients: stunted height, delayed puberty |
Abdominal Examination
Inspection:
- Distension (obstruction, ascites if hypoalbuminaemia)
- Surgical scars (previous resections)
- Visible stomas (ileostomy, colostomy)
- Fistula openings (enterocutaneous)
Palpation:
- Right iliac fossa tenderness (terminal ileitis) — most common
- Palpable mass (phlegmon, abscess, thickened bowel loop, matted loops)
- Hepatomegaly (primary sclerosing cholangitis, fatty liver, hepatic abscess — rare)
Percussion:
- Tympanic if dilated loops (obstruction)
- Ascites (protein-losing enteropathy, portal hypertension)
Auscultation:
- High-pitched, hyperactive bowel sounds (partial obstruction)
- Absent bowel sounds (complete obstruction, peritonitis)
Perianal Examination (Essential — Do Not Omit)
Inspection:
- Skin tags (edematous, non-thrombosed) — 75% of perianal Crohn's
- Fistula openings — may see purulent discharge
- Fissures — often lateral or multiple (unlike idiopathic which is posterior midline)
- Erythema, induration (active inflammation, abscess)
Digital Rectal Examination:
- Assess sphincter tone
- Palpable fistula tracts, induration
- Masses, strictures
- Caution: May be too painful if active inflammation/abscess; defer if severe tenderness
Key Point: Always examine the perianal region — findings help distinguish Crohn's from UC and guide treatment (anti-TNF is first-line for perianal fistulising disease).
Extra-Intestinal Manifestations (EIMs)
EIMs occur in 25-40% of Crohn's patients; some correlate with disease activity, others are independent. [18]
Musculoskeletal (Most Common)
| Manifestation | Prevalence | Features | Correlation with IBD Activity |
|---|---|---|---|
| Peripheral arthritis | 10-20% | Oligoarticular, asymmetric, large joints (knees, ankles, hips, wrists); non-erosive | Yes — improves with IBD treatment |
| Axial arthropathy | 3-10% | Ankylosing spondylitis, sacroiliitis; HLA-B27 associated; progressive | No — independent course |
| Enthesitis | 5-10% | Achilles tendinitis, plantar fasciitis | Variable |
Dermatological
| Manifestation | Prevalence | Features | Correlation |
|---|---|---|---|
| Erythema nodosum | 4-15% | Tender red nodules on shins; associated with active colonic disease | Yes |
| Pyoderma gangrenosum | 1-2% | Painful ulcerating skin lesions (often lower limbs); colonic disease | Variable |
| Sweet's syndrome | Rare | Painful erythematous papules, fever, neutrophilia | Yes |
Ophthalmological
| Manifestation | Prevalence | Features | Urgency |
|---|---|---|---|
| Episcleritis | 3-10% | Red, painful eye; superficial inflammation | Moderate; ophthalmology review |
| Uveitis/Iritis | 0.5-3% | Painful red eye, photophobia, blurred vision | Urgent — risk of blindness |
| Scleritis | Rare | Severe pain, vision loss | Urgent |
Clinical Pearl: Any red, painful eye in IBD patient → same-day ophthalmology assessment (uveitis can cause permanent vision loss).
Hepatobiliary
| Manifestation | Prevalence | Features | Notes |
|---|---|---|---|
| Primary sclerosing cholangitis (PSC) | 1-4% in Crohn's (less than UC where 5-8%) | Progressive bile duct fibrosis → cirrhosis, cholangiocarcinoma risk | Monitor LFTs, MRCP; may need transplant |
| Cholelithiasis | 2-3× general population | Due to bile acid malabsorption (ileal disease) | Risk factor for gallstones |
| Fatty liver | 10-40% | Non-alcoholic fatty liver disease; associated with malnutrition, steroids | Usually benign |
Renal and Urological
- Nephrolithiasis (kidney stones): 10-20% — calcium oxalate stones due to ileal malabsorption (fat binds calcium → free oxalate absorption)
- Ureteric obstruction: Rarely from retroperitoneal fibrosis, fistulae
- Fistulae: Enterovesical (bowel-bladder) → pneumaturia, faecaluria, recurrent UTI
Vascular
- Venous thromboembolism (VTE): 1.5-3.5× increased risk [19]
- Deep vein thrombosis (DVT), pulmonary embolism (PE)
- Due to chronic inflammation (elevated Factor VIII, fibrinogen), immobility during flares, surgery
- Consider thromboprophylaxis during hospitalisation and perioperatively
Nutritional and Metabolic
| Deficiency | Cause | Prevalence | Clinical Features |
|---|---|---|---|
| Iron deficiency | Chronic bleeding, malabsorption | 60-80% | Microcytic anaemia, fatigue |
| Vitamin B12 deficiency | Terminal ileal disease/resection | 20-40% (higher post-resection) | Macrocytic anaemia, neuropathy, glossitis |
| Folate deficiency | Malabsorption, methotrexate use | 20-30% | Macrocytic anaemia |
| Vitamin D deficiency | Malabsorption (fat-soluble), reduced sun exposure | 50-70% | Osteomalacia, secondary hyperparathyroidism |
| Osteoporosis/Osteopenia | Corticosteroid use, vitamin D deficiency, chronic inflammation | 40-50% | Fracture risk |
| Zinc, selenium, magnesium | Diarrhoea, malabsorption | Common | Impaired immunity, hair loss, fatigue |
6. Differential Diagnosis
Key Differentials to Consider
Inflammatory Bowel Disease vs Other Inflammatory Conditions
| Condition | Distinguishing Features |
|---|---|
| Ulcerative colitis | Continuous inflammation from rectum, mucosal only, rectal bleeding prominent, cured by colectomy |
| Intestinal tuberculosis | Endemic areas, weight loss, night sweats, ascites, ileocecal region, caseating granulomas, positive AFB/culture [20] |
| Intestinal Behçet's disease | Oral + genital ulcers, uveitis, pathergy, deep punched-out ulcers at ileocecal valve |
| Yersinia enterocolitis | Acute onset, self-limiting, mesenteric adenitis mimics appendicitis, positive serology/culture |
| Actinomycosis | Rare, sulphur granules, chronic abdominal mass, fistulae |
Ischaemic and Vascular
| Condition | Distinguishing Features |
|---|---|
| Ischaemic colitis | Older age, cardiovascular risk factors, watershed areas (splenic flexure), acute onset, thumbprinting on imaging |
| Radiation enteritis | History of pelvic radiotherapy, affects radiated field, telangiectasia, fibrosis |
Infectious
| Pathogen | Key Features |
|---|---|
| Clostridioides difficile | Recent antibiotics, toxin assay positive, pseudomembranes |
| Campylobacter, Salmonella, Shigella | Acute diarrhoea, positive stool culture |
| Cytomegalovirus (CMV) | Immunocompromised (on immunosuppression), deep "punched out" ulcers, CMV PCR/immunostaining positive |
| Intestinal parasites (Giardia, Entamoeba) | Travel history, microscopy/PCR positive |
Malignancy
| Condition | Distinguishing Features |
|---|---|
| Colorectal carcinoma | Older age (> 50), change in bowel habit, iron deficiency anaemia, mass on imaging, biopsy diagnostic |
| Lymphoma | Systemic symptoms (fever, night sweats, weight loss), lymphadenopathy, extranodal involvement |
| Small bowel adenocarcinoma | Rare, chronic Crohn's increases risk 2-3×, stricture, bleeding |
Functional and Other
| Condition | Distinguishing Features |
|---|---|
| Irritable bowel syndrome (IBS) | No red flags, normal inflammatory markers, normal endoscopy/imaging, symptom-based diagnosis |
| Coeliac disease | Diarrhoea, malabsorption, positive anti-tissue transglutaminase, villous atrophy on duodenal biopsy |
| Appendicitis | Acute RLQ pain, fever, elevated WCC, imaging confirms inflamed appendix |
| Diverticulitis | Left-sided pain (sigmoid), older age, diverticula on CT, inflammatory mass |
Crohn's vs Ulcerative Colitis: Key Differences
| Feature | Crohn's Disease | Ulcerative Colitis |
|---|---|---|
| Distribution | Mouth to anus (skip lesions) | Rectum to proximal colon (continuous) |
| Rectal involvement | Spared in 50% | Always involved |
| Most common site | Terminal ileum, ileocolonic | Rectum, left colon |
| Depth of inflammation | Transmural | Mucosal/submucosal |
| Granulomas | 30-40% (non-caseating) | Absent |
| Fistulae | Common (20-40%) | Rare |
| Strictures | Common | Rare (may indicate cancer or Crohn's overlap) |
| Perianal disease | 25-35% | Rare (less than 5%) |
| Smoking | Worsens disease (OR 2.0) | Protective (paradox) |
| Bloody diarrhoea | Less common (unless colonic) | Hallmark feature |
| Surgery | Not curative; 50-70% need surgery | Curative (total colectomy) |
| Malignancy risk | Increased (especially with colonic involvement) | Higher (8-10% at 30 years extensive colitis) |
Indeterminate Colitis (IBD-U): 10-15% of IBD cases cannot be classified as Crohn's or UC based on clinical, endoscopic, and histological features. May declare as one or the other over time.
7. Investigations
Initial Assessment: First-Line Tests
Blood Tests
| Test | Purpose | Typical Findings in Active Crohn's |
|---|---|---|
| Full blood count (FBC) | Anaemia, infection, thrombocytosis | Hb ↓ (microcytic or normocytic), MCV ↓ (iron deficiency) or ↑ (B12/folate deficiency), platelets ↑ (inflammation), WCC ↑ if sepsis/abscess |
| Inflammatory markers | Disease activity | CRP ↑ (> 5 mg/L), ESR ↑ (> 20 mm/hr) — correlate with active inflammation but can be normal in 20-30% |
| Albumin | Nutritional status, protein loss | ↓ (less than 35 g/L) in severe disease (malnutrition, protein-losing enteropathy) |
| Liver function tests (LFTs) | Hepatobiliary disease, drug monitoring | May be abnormal if PSC, fatty liver, medication hepatotoxicity |
| Renal function (U&E) | Dehydration, medication monitoring | Urea/creatinine may be elevated if dehydrated or on nephrotoxic drugs |
| Iron studies | Iron deficiency anaemia | Ferritin ↓ (if low iron), or ↑ (acute phase reactant despite iron deficiency), transferrin saturation ↓, TIBC ↑ |
| Vitamin B12, folate | Malabsorption | B12 ↓ (ileal disease/resection), folate ↓ (small bowel disease, methotrexate) |
| Vitamin D (25-OH vitamin D) | Malabsorption, bone health | ↓ in 50-70%; target > 75 nmol/L |
Additional tests if indicated:
- Anti-Saccharomyces cerevisiae antibodies (ASCA): Positive in 50-60% of Crohn's (vs 10-15% UC) — not routinely used diagnostically
- Anti-neutrophil cytoplasmic antibodies (p-ANCA): Usually negative in Crohn's (positive in 60-70% UC)
- Thiopurine methyltransferase (TPMT): Test before starting azathioprine/mercaptopurine (low activity → toxicity risk)
Stool Tests
| Test | Purpose | Key Findings |
|---|---|---|
| Faecal calprotectin | Distinguish IBD from IBS; monitor disease activity | > 250 μg/g suggests IBD (sensitivity 95%, specificity 90%); less than 50 μg/g unlikely active IBD; trend more useful than single value [21] |
| Stool microscopy, culture, sensitivity (MCS) | Exclude infection | Negative in Crohn's; positive for Salmonella, Campylobacter, Shigella, E. coli if infective colitis |
| Clostridioides difficile toxin | Exclude C. diff (especially if recent antibiotics or hospitalisation) | Positive toxin A/B; treat appropriately |
| Ova, cysts, parasites | If travel history or immunocompromised | Detect Giardia, Entamoeba, Cryptosporidium |
| Faecal lactoferrin | Alternative inflammatory marker | Similar to calprotectin but less widely used |
Endoscopy
Ileocolonoscopy with Biopsies (Gold Standard for Diagnosis): [1,2]
Indications:
- Confirm diagnosis in suspected Crohn's
- Assess disease extent and severity
- Differentiate from UC
- Surveillance for dysplasia (long-standing colonic Crohn's)
Endoscopic Findings:
- Early disease: Aphthous ulcers (small 2-5 mm erosions on normal mucosa)
- Active disease: Deep longitudinal and transverse serpentine ulcers, cobblestone appearance (intersecting ulcers with intervening edematous mucosa)
- Skip lesions: Inflamed areas interspersed with normal mucosa
- Strictures: Luminal narrowing (scope may not pass)
- Fistula openings (less commonly visualised endoscopically)
Biopsy Histology:
- Transmural inflammation (inferred if muscularis mucosae involvement seen)
- Non-caseating granulomas (30-40%): diagnostic when present, but absence does not exclude Crohn's [1,2]
- Focal chronic inflammation (patchy, discontinuous)
- Preserved goblet cells (unlike UC)
- Pyloric gland metaplasia, fissuring ulcers
Capsule Endoscopy:
- Visualise small bowel mucosa (jejunum, ileum) inaccessible to standard endoscopy
- Indications: Suspected small bowel Crohn's with negative ileocolonoscopy and MRI
- Contraindications: Known or suspected stricture (risk of capsule retention — perform patency capsule first)
- Detects aphthous ulcers, erosions, strictures
Upper GI Endoscopy (OGD):
- If upper GI symptoms (dysphagia, nausea, epigastric pain)
- Detects oesophageal, gastric, duodenal Crohn's (5-10%)
- Biopsy gastric antrum and duodenum even if macroscopically normal
Cross-Sectional Imaging
MRI Enterography (Preferred for Small Bowel Assessment)
Advantages:
- No ionising radiation (important in young patients needing repeated imaging)
- Excellent soft tissue resolution
- Assesses bowel wall thickness, enhancement, strictures, fistulae, abscesses, mesenteric changes
Findings in Crohn's:
- Mural thickening: > 3 mm small bowel, > 4 mm colon
- Mural hyperenhancement (active inflammation)
- T2 hyperintensity (oedema)
- Strictures: Luminal narrowing with proximal dilatation (pre-stenotic dilation)
- Fistulae and sinus tracts
- Abscesses: Fluid collections with rim enhancement
- "Comb sign": Engorged vasa recta (mesenteric vessels perpendicular to bowel)
- Mesenteric lymphadenopathy
- Creeping fat: Mesenteric fat wrapping around bowel
MR Enterography vs MR Enteroclysis:
- Enterography: Oral contrast (e.g., polyethylene glycol) — better tolerated, widely used
- Enteroclysis: Nasojejunal tube contrast instillation — superior distension, less commonly performed
CT Enterography
Indications:
- Acute presentation (suspected abscess, perforation, obstruction) — faster than MRI
- Contraindication to MRI (pacemaker, claustrophobia)
Disadvantages:
- Ionising radiation (cumulative risk in young patients with lifelong disease)
Findings: Similar to MRI — bowel wall thickening, enhancement, complications
MRI Pelvis (For Perianal Disease)
Gold standard for perianal fistula assessment: [3]
Findings:
- Fistula tracts: classified by Parks classification (intersphincteric, transsphincteric, suprasphincteric, extrasphincteric)
- Abscesses: fluid collections
- Involvement of sphincter complex (determines surgical approach)
- Guides seton placement and monitors response to medical therapy
Ultrasound (Point-of-Care)
Intestinal Ultrasound:
- Increasingly used; operator-dependent
- Assess bowel wall thickness, vascularity (colour Doppler), strictures
- Useful for monitoring disease activity non-invasively
- Detect abscesses, collections
Abdominal Ultrasound:
- Initial imaging for suspected abscess in unstable patients
- Hepatobiliary assessment
Barium Studies (Largely Replaced by MRI/CT)
- Small bowel follow-through, barium enema: Historical; still occasionally used if MRI/CT unavailable
- Show mucosal irregularity, strictures ("string sign" of Kantor — narrow ileal segment), fistulae
8. Management
Overview: Treat-to-Target Strategy
Modern Crohn's management has shifted from symptomatic control ("step-up" approach) to proactive "treat-to-target" strategy aiming for: [7,13]
- Clinical remission: Symptom resolution (HBI less than 5, no abdominal pain, normal stool frequency)
- Biochemical remission: CRP less than 5 mg/L, faecal calprotectin less than 150-250 μg/g
- Endoscopic remission: Mucosal healing (SES-CD ≤2, absence of ulceration)
- Transmural healing: Resolution of inflammation on MRI (normalisation of bowel wall thickness and enhancement)
Rationale: Achieving deep remission (mucosal/transmural healing) reduces hospitalisation, surgery, disability, and may alter disease course long-term. [7,13]
Monitoring:
- Every 3-6 months: Clinical assessment (symptoms, weight), CRP, faecal calprotectin
- If targets not met: Escalate therapy (optimise dose, switch biologic, add immunomodulator)
- Endoscopy: 6-12 months after treatment initiation, then as clinically indicated
Induction of Remission (Acute Flare Management)
Mild-to-Moderate Disease (HBI 5-7, CRP less than 10 mg/L)
First-Line:
-
Budesonide 9 mg once daily for 8 weeks, then taper (ileocecal/right-sided disease)
- Locally acting steroid; reduced systemic side effects vs prednisolone
- Efficacy: 60% remission vs 20% placebo [22]
- Not effective for colonic or upper GI disease
-
Prednisolone 40 mg once daily (if extensive, left-sided, or upper GI disease)
- Taper over 8-10 weeks (reduce 5 mg weekly)
- Efficacy: 70-80% remission in mild-moderate disease
Adjunctive:
- Nutritional support (oral supplements if malnourished)
- Anti-diarrhoeals (loperamide) if no obstruction/toxic megacolon — use cautiously
- Analgesia: paracetamol (avoid NSAIDs — may exacerbate)
Follow-Up:
- Assess response at 2-4 weeks
- If inadequate response: escalate to moderate-severe protocol
Moderate-to-Severe Disease (HBI 8-16, CRP 10-40 mg/L)
First-Line:
-
Systemic corticosteroids:
- Prednisolone 40-60 mg once daily or IV hydrocortisone 100 mg QDS (if unable to tolerate oral)
- Taper prednisolone over 8-10 weeks once remission achieved
-
Early biologic therapy (consider if high-risk features):
- High-risk features: Young age (less than 40), extensive disease (L3), deep ulceration, perianal disease, prior hospitalisation/surgery, elevated CRP
- Infliximab 5 mg/kg IV at weeks 0, 2, 6 (induction regimen)
- Adalimumab 160 mg SC week 0, 80 mg week 2, then 40 mg every 2 weeks
Concomitant Immunomodulator:
- Azathioprine (2-2.5 mg/kg) or mercaptopurine (1-1.5 mg/kg) started alongside or shortly after induction
- Rationale: SONIC trial showed infliximab + azathioprine superior to either monotherapy (remission 57% vs 44% infliximab alone vs 30% azathioprine alone) [23]
- Check TPMT genotype/activity before starting (low activity → increased toxicity risk)
Severe Acute Disease (HBI > 16, CRP > 40 mg/L, Systemic Toxicity)
Hospitalisation required:
-
Resuscitation:
- IV fluids (correct dehydration, electrolyte disturbances)
- Nil by mouth if obstruction/pre-operative
- VTE prophylaxis (LMWH — high thrombosis risk)
- Nutritional assessment (consider enteral or parenteral nutrition if severely malnourished)
-
IV corticosteroids:
- Hydrocortisone 100 mg QDS or methylprednisolone 40-60 mg OD
-
Exclude complications:
- CT abdomen/pelvis (abscess, perforation, obstruction)
- Stool for C. difficile, CMV (especially if on immunosuppression)
-
If abscess: Percutaneous drainage (IR-guided) or surgical drainage + antibiotics (metronidazole + ciprofloxacin or piperacillin-tazobactam)
-
If no response to IV steroids within 3-5 days → Rescue therapy:
- Infliximab 5 mg/kg IV (single dose, then standard induction if responds)
- Surgical consultation if perforation, free air, peritonitis, or refractory disease
Maintenance of Remission
Once remission achieved, lifelong maintenance therapy required to prevent relapse.
Conventional Immunomodulators
| Drug | Dose | Mechanism | Efficacy | Monitoring | Adverse Effects |
|---|---|---|---|---|---|
| Azathioprine | 2-2.5 mg/kg OD | Purine analogue; inhibits T cell proliferation | Maintains remission in 60-70% | FBC, LFTs every 3 months; TPMT before starting | Nausea, myelosuppression, hepatotoxicity, pancreatitis (3-5%), lymphoma risk (small but increased), hypersensitivity |
| Mercaptopurine | 1-1.5 mg/kg OD | Active metabolite of azathioprine | Similar to azathioprine | As above | As above |
| Methotrexate | 25 mg SC/IM weekly | Folate antagonist | Maintains remission in 60-65% | FBC, LFTs, renal function every 3 months; CXR baseline | Nausea, stomatitis, myelosuppression, hepatotoxicity, pulmonary fibrosis (rare), teratogenic (contraception essential) |
Notes:
- Azathioprine/mercaptopurine: First-line immunomodulator; slow onset (3-6 months to full effect)
- Methotrexate: Second-line or if thiopurine-intolerant; also used post-operatively to prevent recurrence
- Co-prescribe folic acid 5 mg weekly (different day from methotrexate) to reduce side effects
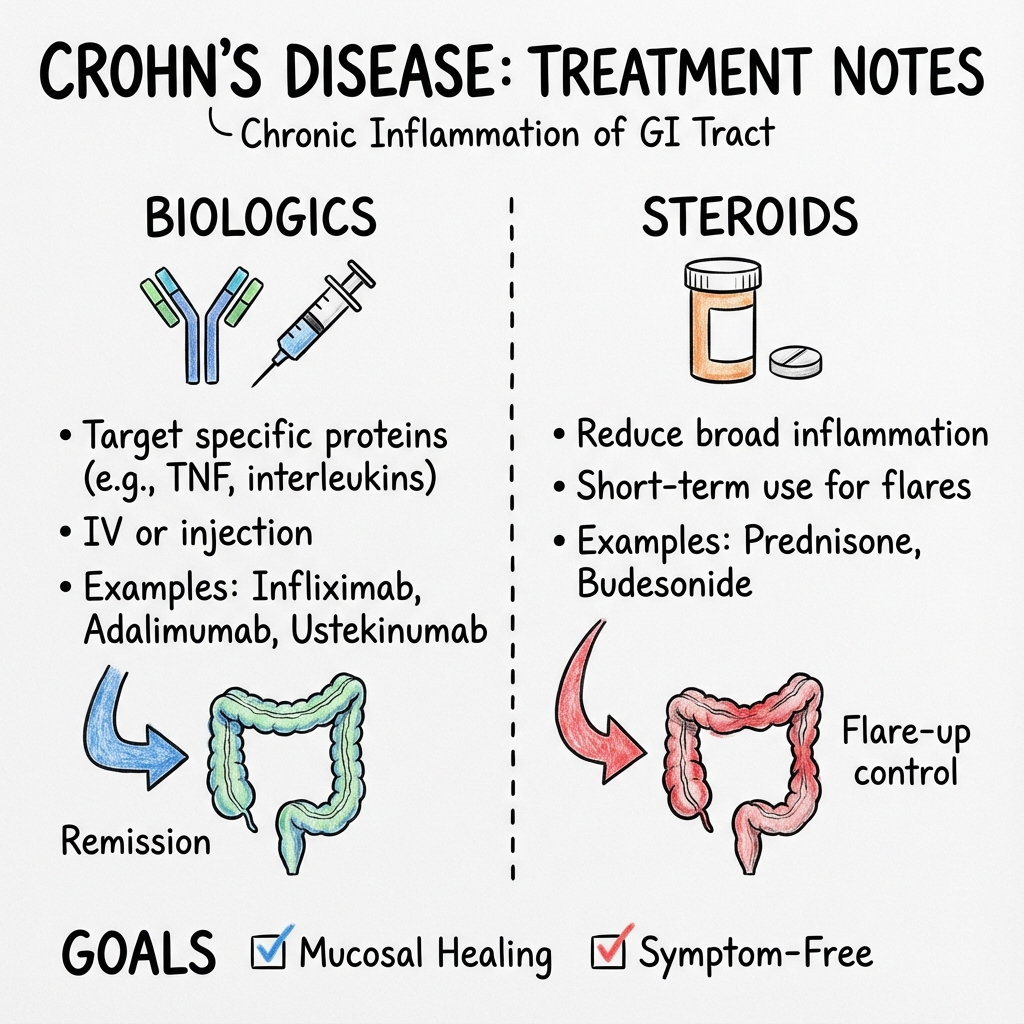
Biologic Therapies
Anti-TNF Agents
Indications:
- Moderate-severe disease failing conventional therapy
- Steroid-dependent or steroid-refractory disease
- Perianal fistulising disease (first-line)
- Post-operative recurrence prevention (selected patients)
| Drug | Mechanism | Induction Regimen | Maintenance | Efficacy | Notes |
|---|---|---|---|---|---|
| Infliximab | Chimeric (mouse-human) monoclonal antibody against TNF-α | 5 mg/kg IV at weeks 0, 2, 6 | 5 mg/kg IV every 8 weeks | Clinical remission 60-70%; mucosal healing 40-50% [8] | Infusion reactions, immunogenicity (combine with immunomodulator to reduce); check TB and Hep B before starting |
| Adalimumab | Fully human monoclonal antibody against TNF-α | 160 mg SC week 0, 80 mg week 2 | 40 mg SC every 2 weeks | Clinical remission 55-60%; mucosal healing 30-40% [8] | Self-administered; lower immunogenicity; injection site reactions |
Pre-Treatment Screening:
- TB: Quantiferon/T-SPOT, CXR (latent TB reactivation risk — treat if positive before starting anti-TNF)
- Hepatitis B: HBsAg, anti-HBc (reactivation risk)
- Vaccinations: Update (pneumococcal, influenza, Hep B) — avoid live vaccines once on therapy
Adverse Effects:
- Serious infections (TB, sepsis, fungal) — 5-10%
- Infusion/injection reactions
- Demyelination (rare; caution in MS family history)
- Heart failure exacerbation (avoid if NYHA III-IV)
- Lymphoma (controversial — small increased risk)
- Anti-drug antibodies (especially infliximab) → loss of response
Loss of Response:
- Primary non-response: Never achieve remission (~20-30%)
- Secondary loss of response: Initial response then lose efficacy (30-40% over time)
- Management: Check drug levels and anti-drug antibodies; dose escalation or switch to alternative biologic
Anti-Integrin Therapy
| Drug | Mechanism | Induction | Maintenance | Efficacy | Notes |
|---|---|---|---|---|---|
| Vedolizumab | Humanised monoclonal antibody against α4β7 integrin (gut-selective) | 300 mg IV weeks 0, 2, 6 | 300 mg IV every 8 weeks | Clinical remission 40-50%; slower onset (10-14 weeks) [24] | Gut-selective (reduced systemic immunosuppression); effective in anti-TNF failures; slower onset; PML risk negligible (vs natalizumab) |
Indications:
- Anti-TNF failures or contraindications
- Patients preferring gut-selective agent (lower systemic infection risk)
- Post-operative recurrence prevention (REPREVIO trial showed efficacy) [12]
Anti-IL-12/23 Therapy
| Drug | Mechanism | Induction | Maintenance | Efficacy | Notes |
|---|---|---|---|---|---|
| Ustekinumab | Human monoclonal antibody against IL-12/IL-23 p40 subunit | Weight-based IV (~6 mg/kg): ≤55 kg: 260 mg, 55-85 kg: 390 mg, > 85 kg: 520 mg | 90 mg SC every 8-12 weeks | Clinical remission 50-55%; effective in anti-TNF failures [9] | Effective post-anti-TNF failure; well-tolerated; SC maintenance convenient |
Indications:
- Anti-TNF failures or intolerance
- Multiple biologic failures
Selective IL-23 Inhibitors (Newest Class)
| Drug | Mechanism | Induction | Maintenance | Efficacy | Notes |
|---|---|---|---|---|---|
| Risankizumab | Humanised monoclonal antibody against IL-23 p19 subunit | 600 mg IV weeks 0, 4, 8 | 360 mg SC every 8 weeks | Clinical remission 55-60%; endoscopic response 40-45% [10] | High efficacy including biologic-experienced; selective IL-23 inhibition (spares IL-12 → potentially less infection risk) |
Indications:
- Anti-TNF failures, anti-integrin failures
- Patients requiring highly efficacious therapy
JAK Inhibitors (Emerging)
| Drug | Mechanism | Dose | Efficacy | Notes |
|---|---|---|---|---|
| Upadacitinib | Selective JAK1 inhibitor | Induction: 45 mg OD; Maintenance: 15-30 mg OD | Clinical remission 50-55%; endoscopic response 40% | Oral agent; effective in biologic failures; concerns: VTE, malignancy (FDA black box warning) — careful patient selection; not yet widely approved for Crohn's |
Combination Therapy vs Monotherapy
SONIC Trial (2010): [23]
- Infliximab + azathioprine vs infliximab alone vs azathioprine alone
- Results: Combination superior (57% vs 44% vs 30% steroid-free remission at 6 months)
- Implication: Combination therapy preferred for anti-TNF-naïve patients, especially moderate-severe disease
However:
- Increased infection risk with combination (monitor closely)
- Many patients on biologic monotherapy due to thiopurine intolerance
- Optimised biologic monotherapy (therapeutic drug monitoring, dose escalation) may be sufficient
Management of Perianal Fistulising Disease
Occurs in 25-35% of Crohn's patients; associated with significant morbidity. [3]
Assessment:
- Clinical examination: Inspect perineum, gentle digital rectal exam (DRE)
- MRI pelvis: Define fistula anatomy (Parks classification), abscesses, sphincter involvement
Medical Management:
-
Antibiotics (initial therapy):
- Metronidazole 400 mg TDS + ciprofloxacin 500 mg BD for 6-8 weeks
- Symptomatic improvement but high relapse rate on cessation
-
Anti-TNF (first-line definitive therapy):
- Infliximab or Adalimumab — most effective medical therapy
- Achieves fistula closure in 30-50% at 1 year [8]
- Continue long-term to maintain closure
-
Immunomodulators:
- Azathioprine as adjunctive therapy with anti-TNF
Surgical Management:
-
EUA (Examination Under Anaesthesia) + Seton insertion:
- Indicated if abscess or complex fistula
- "Seton: Loose non-cutting seton (e.g., Silastic vessel loop) placed through fistula tract"
- "Purpose: Promotes drainage, prevents abscess re-formation, allows medical therapy to work"
- Seton can remain long-term (months to years)
-
Fistulotomy/Fistulectomy: Only for low, simple fistulae (risk of incontinence if complex)
-
Advancement flap, LIFT procedure: More complex procedures for refractory cases
-
Defunctioning stoma: Rarely needed if severe, refractory disease (diverts faecal stream)
Algorithm:
- Abscess → Drainage (percutaneous or surgical) + Antibiotics
- No abscess, simple fistula → Antibiotics then Anti-TNF
- Complex fistula/recurrent abscess → EUA + Seton + Anti-TNF
- Refractory → Surgical options (flap, LIFT) or defunctioning stoma
Management of Strictures
Strictures occur in 30-40% over time due to chronic inflammation and fibrosis.
Clinical Presentation:
- Cramping abdominal pain (especially post-prandial)
- Bloating, distension
- Nausea, vomiting
- Change in bowel habit
Investigation:
- MRI/CT enterography: Identify stricture length, degree of narrowing, pre-stenotic dilatation, active inflammation vs pure fibrosis
Predominantly Inflammatory Stricture (mural enhancement, oedema on MRI):
- Medical therapy: Optimise biologics ± corticosteroids to reduce inflammation
- May respond to anti-inflammatory treatment
Predominantly Fibrotic Stricture (no enhancement, fixed narrowing):
-
Endoscopic Balloon Dilatation (EBD):
- Indications: Short (less than 4 cm), single or limited strictures, accessible by colonoscopy, no fistula/abscess
- Technique: Balloon inflated to 15-20 mm diameter under endoscopic guidance
- Success rate: 70-80% short-term improvement; 50% remain symptom-free at 1 year [25]
- Complications: Perforation (2-5%), bleeding (rare)
- May need repeat dilations
-
Surgical Resection:
- Indications: Failed EBD, multiple strictures, long strictures, associated fistula/abscess, dysplasia/cancer concern
- Technique: Stricturoplasty (preserve bowel length) or limited resection with primary anastomosis
- Goal: Minimal resection (preserve bowel to avoid short bowel syndrome)
Post-operative Management: See below.
Surgical Management
Key Principle: Surgery is not curative in Crohn's; recurrence is the rule.
Indications for Surgery:
- Failed medical therapy (refractory disease)
- Strictures causing obstruction
- Fistulae/abscesses not responding to medical/conservative management
- Perforation, haemorrhage
- Dysplasia or malignancy
- Growth failure in children despite medical therapy
Common Surgical Procedures
| Procedure | Indication | Details |
|---|---|---|
| Ileocecal resection | Terminal ileal disease, stricture, fistula | Most common surgery; resect terminal ileum + cecum, anastomose ileum to ascending colon |
| Segmental small bowel resection | Localised small bowel disease, stricture | Limited resection of affected segment, primary anastomosis |
| Stricturoplasty | Multiple strictures, preserve bowel length | Heineke-Mikulicz (short strictures) or Finney (longer) — widen lumen without resection |
| Colectomy (subtotal or total) | Extensive colonic disease, failed medical therapy | Subtotal colectomy + ileorectal anastomosis, or total colectomy + end ileostomy (if rectal involvement) |
| Perianal surgery | Perianal fistula, abscess | EUA, seton insertion, drainage |
Recurrence Rates:
- Endoscopic recurrence: 70-80% at 1 year (Rutgeerts i2-i4)
- Clinical recurrence: 30-50% at 5 years, 50-70% at 10 years [11]
Post-Operative Recurrence Prevention
Risk Stratification (Assess within 6-12 Months Post-Op):
High-Risk Features:
- Smoking
- Penetrating disease (B3)
- Prior resection (recurrent surgery)
- Extensive resection (> 50 cm)
- Perianal disease
Low-Risk:
- First resection, limited disease, non-smoker
Post-Operative Endoscopy (6-12 Months):
- Ileocolonoscopy to assess neo-terminal ileum
- Rutgeerts score: i2-i4 → escalate treatment
Medical Prophylaxis: [11,12]
| Risk Level | Recommended Prophylaxis |
|---|---|
| Low-risk | No prophylaxis, or 5-ASA (mesalazine) — weak evidence |
| Moderate-risk | Thiopurine (azathioprine 2-2.5 mg/kg) or methotrexate (25 mg weekly SC) |
| High-risk | Anti-TNF (infliximab, adalimumab) or vedolizumab (REPREVIO trial: vedolizumab superior to placebo, 63% vs 48% endoscopic remission) [12] |
Smoking Cessation: Strongly encourage — most important modifiable risk factor.
Algorithm:
- Assess risk factors
- Start prophylaxis (thiopurine for moderate-risk, anti-TNF/vedolizumab for high-risk)
- Ileocolonoscopy at 6-12 months → Rutgeerts score
- If i2-i4 (recurrence) → Escalate therapy
- If i0-i1 (no recurrence) → Continue current prophylaxis, repeat endoscopy 1-2 yearly
Nutritional Therapy
Exclusive Enteral Nutrition (EEN):
- Paediatric Crohn's: First-line therapy for induction (8-12 weeks elemental or polymeric formula); 60-80% achieve remission
- Adults: Less effective (compliance issues, palatability), but can be used if steroid contraindication
- Mechanism: Reduces inflammation, alters microbiome, bowel rest
Supplementary Enteral Nutrition:
- Adjunct to medical therapy if malnourished
- Oral nutritional supplements (high-protein, high-calorie)
Parenteral Nutrition:
- Short-term if severe malnutrition, bowel rest needed (pre-op), or intestinal failure
Dietary Advice:
- No specific "Crohn's diet" proven universally effective
- Avoid known triggers if identified (some patients report dairy, high-fiber, spicy foods worsen symptoms)
- If strictures: low-residue diet to reduce obstruction risk
Micronutrient Supplementation:
- Iron: Oral (ferrous sulfate/fumarate) if mild deficiency and tolerating; IV iron (ferric carboxymaltose, iron isomaltoside) if severe, malabsorption, or oral intolerance
- Vitamin B12: 1000 μg IM every 3 months if ileal resection > 20 cm or terminal ileal disease
- Vitamin D: 800-2000 IU daily (or higher if deficiency); monitor levels
- Calcium: 1000-1500 mg daily (especially if on corticosteroids)
- Folic acid: 5 mg weekly if on methotrexate; daily if deficiency
9. Complications
Intestinal Complications
Strictures
- Incidence: 30-40% over disease course
- Types: Inflammatory (reversible with medical therapy) vs fibrotic (require dilation/surgery)
- Complications of strictures: Obstruction, perforation proximal to stricture
Fistulae
Types:
- Enteroenteric (bowel-to-bowel): May cause malabsorption, bacterial overgrowth, bypass of nutrients
- Enterocutaneous (bowel-to-skin): Discharge, infection, fluid/electrolyte loss
- Enterovesical (bowel-to-bladder): Recurrent UTI, pneumaturia (air in urine), faecaluria
- Enterovaginal (bowel-to-vagina): Vaginal discharge (faeculent), distressing
- Perianal: See above
Management: Medical (anti-TNF) ± surgical drainage/repair
Abscess
- Intra-abdominal or pelvic: Fever, pain, mass, sepsis
- Diagnosis: CT/MRI
- Management: Antibiotics + percutaneous drainage (IR-guided) or surgical drainage; anti-TNF therapy once sepsis controlled
Perforation
- Free perforation: Peritonitis, acute abdomen — surgical emergency
- Sealed perforation: May present as abscess
Malignancy
Small Bowel Adenocarcinoma:
- 2-3× increased risk in Crohn's (rare overall — less than 1% lifetime risk)
- Associated with long-standing small bowel disease, chronic strictures
Colorectal Cancer:
- Risk similar to UC if extensive colonic involvement
- Cumulative risk with extensive colitis: 2.9% at 10 years, 5.6% at 20 years, 8.3% at 30 years [14]
- Surveillance: Colonoscopy every 1-5 years depending on risk factors (extent, duration, dysplasia history, PSC, family history)
Lymphoma:
- Controversial association with thiopurines (especially in young males)
- Hepatosplenic T-cell lymphoma (rare, fatal) — mostly in young males on combination thiopurine + anti-TNF
- Monitor clinically; weigh benefits vs risks
Extra-Intestinal Complications
See "Extra-Intestinal Manifestations" section above.
Nutritional and Metabolic Complications
- Malnutrition: Weight loss, sarcopenia, failure to thrive (children)
- Anaemia: Iron, B12, folate deficiency; anaemia of chronic disease
- Osteoporosis/Osteopenia: 40-50% prevalence — screen with DEXA scan (especially if prolonged steroids, postmenopausal women, > 50 years)
- Vitamin D deficiency: 50-70%
- Short bowel syndrome: If extensive resections (> 200 cm small bowel or > 100 cm with loss of ileocecal valve) → malabsorption, diarrhoea, dependence on parenteral nutrition
Treatment-Related Complications
Corticosteroids
- Short-term: Hyperglycaemia, mood changes, insomnia, increased appetite, acne
- Long-term (> 3 months): Osteoporosis, hypertension, diabetes, cataracts, glaucoma, adrenal suppression, increased infection risk
- Mitigation: Calcium + Vitamin D supplementation, bisphosphonate if prolonged use, taper as soon as possible
Thiopurines (Azathioprine, Mercaptopurine)
- Myelosuppression (1-5%): Monitor FBC every 3 months; dose-reduce or stop if WCC less than 3.0 or neutrophils less than 1.0
- Hepatotoxicity (5-10%): Monitor LFTs; dose-reduce if ALT > 2× ULN
- Pancreatitis (3-5%): Acute onset abdominal pain; stop drug immediately (do not rechallenge)
- Lymphoma risk: Small but increased (4-6 per 10,000 patient-years) — counsel patients
- Hypersensitivity (rare): Fever, rash, arthralgia — stop drug
Methotrexate
- Nausea (common): Take folic acid 5 mg weekly (different day)
- Myelosuppression, hepatotoxicity: Monitor FBC, LFTs every 3 months
- Pulmonary fibrosis (rare less than 1%): Baseline CXR, warn patients (dyspnoea)
- Teratogenic: Strict contraception; stop 3-6 months before conception
Anti-TNF
- Infections (5-10%): TB reactivation, sepsis, opportunistic infections (fungal, viral) — screen for TB, Hep B before starting; vaccinate; monitor
- Infusion/injection reactions: Manage with premedication (antihistamines, paracetamol)
- Demyelination (rare): Avoid if MS or family history of MS
- Heart failure exacerbation: Avoid if NYHA III-IV
- Malignancy risk: Possible small increase in lymphoma, skin cancers (controversial) — annual skin checks
- Paradoxical psoriasis (rare): New-onset or worsening psoriasis
Psychosocial Complications
- Depression and anxiety: 20-30% prevalence — screen regularly, refer for psychological support
- Quality of life impairment: Fatigue, pain, fear of incontinence, body image (stomas, perianal disease)
- Social isolation, impact on education/employment
- Sexual dysfunction, fertility concerns
Management: Multidisciplinary support (psychologist, IBD nurse specialist, patient support groups)
10. Prognosis and Outcomes
Natural History
Without Treatment:
- Chronic relapsing-remitting course with progressive bowel damage
- Majority develop complications (strictures, fistulae) over time
With Modern Treatment:
- Most patients achieve and maintain clinical remission
- Quality of life can approach that of general population with optimised therapy
- Shift from "step-up" to "top-down" early aggressive therapy improves long-term outcomes [7,13]
Surgical Outcomes
- Cumulative surgery rate: 50-70% at 10 years (decreasing with biologic use)
- Recurrence post-resection: Endoscopic 70-80% at 1 year, clinical 30-50% at 5 years [11]
- Multiple surgeries: 30-40% require ≥2 surgeries over lifetime
- Prophylactic medical therapy reduces recurrence [12]
Mortality
- Standardised mortality ratio (SMR): 1.4-1.5× general population [26]
- Causes of death: Complications of disease (sepsis, thromboembolism, malignancy), surgical complications, medication-related (infections)
- Mortality has decreased over past 2 decades with improved therapies
Prognostic Factors
Poor Prognostic Factors (Higher Risk of Complications, Surgery)
| Factor | Impact |
|---|---|
| Perianal disease | Higher complication rate, more aggressive disease |
| Young age at diagnosis (less than 40) | Longer disease duration, higher cumulative damage |
| Smoking | 2× risk of progression, recurrence post-surgery [15] |
| Stricturing (B2) or penetrating (B3) behaviour | Already complicated disease phenotype |
| Extensive disease (L3) | More bowel involvement |
| Deep ulceration on endoscopy | Marker of severe inflammation |
| Need for corticosteroids at diagnosis | Suggests moderate-severe disease |
| NOD2 mutations (homozygous) | Ileal disease, stricturing, earlier surgery |
| Elevated CRP/calprotectin despite therapy | Ongoing inflammation → damage |
Favourable Prognostic Factors
| Factor | Impact |
|---|---|
| Non-smoking | Better outcomes, lower recurrence |
| Colonic-only disease (L2) | Less aggressive than ileocolonic |
| Inflammatory phenotype (B1) | Less complicated (but may progress) |
| Early mucosal healing with therapy | Reduces hospitalisation, surgery [7] |
| Adherence to therapy | Critical for long-term remission |
Disease Modification
Mucosal Healing as Outcome:
- Achieving mucosal healing (absence of ulceration) associated with:
- Reduced hospitalisation (HR 0.5)
- Reduced surgery (HR 0.3)
- Sustained remission [7]
Top-Down (Early Aggressive) vs Step-Up Approach:
- SONIC, REACT trials: Early combination therapy (anti-TNF + immunomodulator) in high-risk patients improves outcomes [23]
- Treat-to-target: Tight monitoring and therapy adjustment to achieve remission superior to symptom-based management [13]
11. Evidence Base and Guidelines
Key International Guidelines
ECCO Guidelines on Therapeutics in Crohn's Disease (2020) [1]
Key Recommendations:
- Induction: Corticosteroids for mild-moderate; early biologics for moderate-severe or high-risk
- Maintenance: Thiopurines, methotrexate, or biologics (anti-TNF, vedolizumab, ustekinumab)
- Perianal fistulae: Anti-TNF first-line
- Post-operative prophylaxis: Thiopurines or anti-TNF based on risk
- Monitoring: CRP, calprotectin, endoscopy to assess mucosal healing
NICE Guideline NG129: Crohn's Disease (2019)
Key Recommendations:
- Offer information and support at diagnosis
- Conventional therapy (steroids, thiopurines) first-line for most
- Biologics (anti-TNF, vedolizumab, ustekinumab) if conventional therapy fails or high-risk
- Nutritional support for all patients
- Smoking cessation critical
British Society of Gastroenterology (BSG) IBD Guidelines (2019)
- Aligned with ECCO and NICE
- Emphasise multidisciplinary care, patient education, psychological support
Landmark Trials and Evidence
1. SONIC Trial (2010) — Combination Therapy [23]
Design: RCT comparing infliximab + azathioprine vs infliximab alone vs azathioprine alone in moderate-severe Crohn's
Results:
- Corticosteroid-free remission at 6 months: Combination 57%, infliximab 44%, azathioprine 30%
- Mucosal healing: Combination 44%, infliximab 30%, azathioprine 17%
Impact: Established combination therapy (anti-TNF + immunomodulator) as superior for achieving and maintaining remission.
2. EXTEND Trial (2012) — Mucosal Healing [27]
Design: RCT of adalimumab vs placebo in moderate-severe Crohn's; assessed mucosal healing
Results: Mucosal healing at week 12: 27% adalimumab vs 13% placebo; associated with better long-term outcomes
Impact: Validated mucosal healing as important treatment endpoint.
3. UNITI Trials (2016) — Ustekinumab [9]
Design: Phase 3 RCTs (UNITI-1, UNITI-2) in moderate-severe Crohn's, including anti-TNF failures
Results: Clinical response at week 6: 34-55% ustekinumab vs 20% placebo; effective in anti-TNF-experienced patients
Impact: Established ustekinumab as effective alternative mechanism after anti-TNF failure.
4. ADVANCE, MOTIVATE Trials (2022-2023) — Risankizumab [10]
Design: Phase 3 RCTs in moderate-severe Crohn's (biologic-naïve and biologic-experienced)
Results: Clinical remission week 12: 45-52% risankizumab vs 29-34% placebo; endoscopic response 35-40% vs 12-14%
Impact: Established risankizumab as highly efficacious agent including in biologic-experienced patients; selective IL-23 inhibition effective.
5. REPREVIO Trial (2024) — Vedolizumab Post-Operative Prophylaxis [12]
Design: Multicentre RCT of vedolizumab vs placebo for post-operative recurrence prevention
Results: Endoscopic remission (Rutgeerts i0-i1) at week 26: 63% vedolizumab vs 48% placebo
Impact: First biologic proven effective for post-operative prophylaxis; vedolizumab option for high-risk patients.
6. CALM Trial (2018) — Treat-to-Target [13]
Design: RCT comparing tight control (treat-to-target with escalation based on symptoms + biomarkers) vs clinical symptoms-based management
Results: Mucosal healing at 48 weeks: 46% tight control vs 30% symptoms-based; less bowel damage
Impact: Validated treat-to-target strategy; proactive monitoring and escalation superior to reactive symptom-based care.
Evidence Summary Table
| Intervention | Evidence Level | Key Studies | Efficacy (NNT or %) |
|---|---|---|---|
| Corticosteroids (induction) | 1a | Multiple RCTs | 70-80% remission (mild-moderate) |
| Budesonide (ileal disease) | 1a | RCTs, meta-analyses [22] | 60% remission vs 20% placebo (NNT 2-3) |
| Thiopurines (maintenance) | 1a | Meta-analyses | 60-70% maintain remission |
| Methotrexate (maintenance) | 1a | RCTs | 60-65% maintain remission |
| Anti-TNF (infliximab, adalimumab) | 1a | ACCENT, SONIC, CLASSIC [8,23] | 60-70% clinical remission; 40-50% mucosal healing |
| Vedolizumab | 1a | GEMINI 2 [24] | 40-50% remission; effective in anti-TNF failure |
| Ustekinumab | 1a | UNITI, IM-UNITI [9] | 50-55% remission; effective in anti-TNF failure |
| Risankizumab | 1a | ADVANCE, MOTIVATE [10] | 55-60% remission; high efficacy in biologic-experienced |
| Endoscopic balloon dilation (strictures) | 2b | Case series, observational | 70-80% short-term success [25] |
| Post-op prophylaxis (anti-TNF, vedolizumab) | 1a | Meta-analyses, REPREVIO [11,12] | Reduces endoscopic recurrence by 30-50% |
12. Patient/Layperson Explanation
What is Crohn's Disease?
Crohn's disease is a long-term condition that causes inflammation in your digestive system (gut). Unlike some gut problems that only affect the surface lining, Crohn's affects the full thickness of the bowel wall. It can happen anywhere from your mouth to your bottom, but most commonly affects the last part of the small intestine (terminal ileum) and the colon (large bowel).
The inflammation comes and goes — sometimes you'll have flare-ups (active disease) with symptoms, and other times you'll feel well (remission). The goal of treatment is to keep you in remission as much as possible and prevent complications.
What Causes Crohn's Disease?
We don't know the exact cause, but it's likely a combination of:
- Your genes: If a close family member has Crohn's, you're at higher risk (but it's not directly inherited like some conditions)
- Your immune system: It overreacts to normal gut bacteria, causing inflammation
- Environmental factors: Smoking, diet, antibiotics, and gut bacteria changes can trigger or worsen it
Important: Crohn's is NOT caused by stress or what you eat, though these can make symptoms worse.
What Are the Symptoms?
Common symptoms include:
- Diarrhoea (often without blood) — usually 3-6 times a day
- Tummy pain (cramping, especially on the right side)
- Weight loss and tiredness
- Mouth ulcers
- Problems around the bottom (pain, discharge, swelling) — this is a telltale sign of Crohn's
Some people also get:
- Joint pain or swelling
- Skin rashes (painful lumps on legs)
- Eye inflammation (red, painful eyes — needs urgent attention)
How is Crohn's Diagnosed?
Your doctor will:
- Take blood tests to check for inflammation and anaemia
- Test your poo (stool sample) to look for inflammation markers and rule out infections
- Do a camera test (colonoscopy) to look inside your bowel and take small samples (biopsies)
- Arrange scans (MRI or CT) to see the small bowel and check for complications
How is Crohn's Treated?
Crohn's can't be cured, but it can be well-controlled with treatment. The aim is to:
- Stop flare-ups (get you into remission)
- Keep you well (maintain remission)
- Prevent complications (strictures, fistulae)
Treatments include:
-
Steroids (e.g., prednisolone, budesonide):
- Used to calm down flare-ups quickly
- Not used long-term due to side effects
-
Immunosuppressants (e.g., azathioprine, methotrexate):
- Tablets or injections taken long-term to keep you in remission
- Regular blood tests needed to monitor
-
Biologic medicines (e.g., infliximab, adalimumab):
- Injections or infusions (drips) that target specific parts of your immune system
- Very effective for moderate or severe Crohn's
- Common options: infliximab (IV drip every 8 weeks), adalimumab (injection every 2 weeks)
-
Surgery:
- Sometimes needed if medicines don't work or if you develop complications (blockages, abscesses)
- Usually involves removing the diseased part of bowel
- Important: Surgery doesn't cure Crohn's — it often comes back, so you'll need ongoing treatment
-
Diet and nutrition:
- No special "Crohn's diet," but some people find certain foods trigger symptoms
- You may need supplements (iron, vitamin B12, vitamin D) if you're not absorbing nutrients well
- Children may use liquid nutrition (enteral feeds) as a treatment
What Complications Can Happen?
- Strictures (narrowing): Scar tissue narrows the bowel, causing blockages
- Fistulae: Abnormal connections between bowel and skin, bladder, or other organs
- Abscesses: Pockets of infection that need draining
- Malnutrition: Poor absorption of nutrients, especially if extensive bowel disease
- Increased bowel cancer risk: Small increased risk if you have long-standing colonic Crohn's (needs surveillance colonoscopies)
Living with Crohn's Disease
What to Expect:
- Crohn's is a lifelong condition, but most people lead full, active lives with proper treatment
- You'll have regular check-ups with your gastroenterology team (doctors, nurses, dietitians)
- Blood and stool tests every few months to monitor disease activity
- Camera tests (colonoscopy) periodically to check healing
Tips for Managing:
- Take your medicines as prescribed — even when you feel well (stopping can cause flares)
- Stop smoking — smoking doubles your risk of flares and complications
- Know your triggers — stress, certain foods, infections can worsen symptoms
- Stay active — exercise helps with fatigue, mood, and bone health
- Ask for support — IBD nurses, support groups (Crohn's & Colitis UK), counselling if needed
When to Seek Urgent Help
Go to A&E or call 999 if you have:
- Severe abdominal pain that won't go away
- Vomiting and can't keep fluids down
- Very high fever (> 38.5°C) with shaking
- Passing blood and feeling faint/dizzy
- Swelling of your tummy with no bowel movements
Contact your IBD team urgently if:
- Symptoms suddenly get much worse
- New lumps or discharge around your bottom
- Painful red eye (can damage vision)
- Signs of infection while on immunosuppressants (fever, feeling unwell)
Questions You Might Have
Will I need a stoma (bag)?
- Most people don't. Only a small number need a stoma, usually temporarily while the bowel heals, or if complications can't be managed otherwise.
Can I have children?
- Yes. Crohn's doesn't usually affect fertility. Most medicines are safe in pregnancy (discuss with your doctor). It's best to conceive when your disease is in remission.
Will I be able to work/study?
- Yes. With good disease control, most people work, study, and travel normally. You may need time off during flares, and you're entitled to workplace adjustments if needed.
Is there a cure?
- Not yet, but research is ongoing. Current treatments are very effective at controlling disease and preventing complications.
Support and Resources
- Crohn's & Colitis UK: crohnsandcolitis.org.uk (information, support groups, helpline)
- Your IBD team: Gastroenterology consultant, IBD nurse specialist, dietitian
- Online communities: Connect with others living with Crohn's
13. Viva/Exam Preparation
Opening Statement (Structured Answer)
Examiner: "Tell me about Crohn's disease."
Model Answer: "Crohn's disease is a chronic, relapsing inflammatory bowel disease characterised by transmural inflammation affecting any part of the gastrointestinal tract from mouth to anus, though most commonly the terminal ileum. It demonstrates discontinuous 'skip lesions' and is associated with complications including strictures, fistulae, and abscesses. The aetiology is multifactorial involving genetic susceptibility, notably NOD2 mutations, environmental triggers such as smoking, and dysregulated Th1 and Th17 immune responses. Management has evolved from symptomatic step-up approaches to proactive treat-to-target strategies aiming for mucosal and transmural healing using biologics such as anti-TNF agents, anti-integrin therapy, and IL-12/23 or IL-23 inhibitors. Surgery is non-curative with high recurrence rates, necessitating lifelong medical prophylaxis."
High-Yield Exam Points
Distinguish Crohn's from UC (Always Asked)
Mnemonic: "CROHN'S"
- Cobblestone mucosa
- Regional / Skip lesions
- Oral lesions (aphthous ulcers)
- Hallmark: transmural inflammation
- Non-caseating granulomas
- 'S** — Small bowel (terminal ileum), Strictures, Smoking worsens, Surgery non-curative
vs UC: "CLOSE"
- Continuous inflammation
- Limited to colon (rectum to proximal)
- Only mucosal depth
- Surgery curative (colectomy)
- Erythema, friability, bloody diarrhoea hallmark
Classifications You Must Know
-
Montreal Classification:
- Age: A1 (less than 16), A2 (17-40), A3 (> 40)
- Location: L1 (ileal), L2 (colonic), L3 (ileocolonic), +L4 (upper GI)
- Behaviour: B1 (inflammatory), B2 (stricturing), B3 (penetrating), +p (perianal)
-
Rutgeerts Score (Post-Op Recurrence):
- i0: No lesions
- i1: ≤5 aphthous lesions
- i2: > 5 aphthous lesions or anastomotic lesions
- i3: Diffuse ileitis
- i4: Ulcers, nodules, stenosis
- i2-i4 = escalate therapy
Management Algorithm (Step-by-Step for Viva)
Mild-Moderate:
- Budesonide 9 mg (if ileal/right colon) or Prednisolone 40 mg
- If inadequate response → escalate
Moderate-Severe:
- Prednisolone 40-60 mg OR early biologic if high-risk
- Add immunomodulator (azathioprine or methotrexate)
- Aim for mucosal healing
Severe Acute:
- Admit, IV fluids, VTE prophylaxis
- IV hydrocortisone 100 mg QDS
- CT to exclude abscess/perforation
- If no response 3-5 days → rescue with infliximab OR surgery
Maintenance:
- Thiopurines (azathioprine 2-2.5 mg/kg) OR methotrexate 25 mg weekly
- Biologics: Anti-TNF (infliximab, adalimumab), vedolizumab, ustekinumab, risankizumab
- Monitor: CRP, calprotectin every 3-6 months; endoscopy 6-12 months
Perianal Fistulae:
- MRI pelvis
- Antibiotics (metronidazole + ciprofloxacin)
- Anti-TNF (first-line definitive therapy)
- EUA + seton if abscess/complex
Post-Operative:
- Risk-stratify
- High-risk → anti-TNF or vedolizumab prophylaxis
- Ileocolonoscopy 6-12 months → Rutgeerts score → adjust therapy
Biologic Choice: When to Use What?
| Clinical Scenario | First Choice | Alternatives |
|---|---|---|
| Moderate-severe, biologic-naïve | Anti-TNF (infliximab, adalimumab) | Vedolizumab, ustekinumab |
| Perianal fistulising disease | Anti-TNF (infliximab or adalimumab) | — |
| Anti-TNF failure | Vedolizumab, ustekinumab, risankizumab | Switch anti-TNF (e.g., infliximab → adalimumab) |
| Post-operative prophylaxis (high-risk) | Anti-TNF, vedolizumab | — |
| Preference for gut-selective | Vedolizumab | — |
| Multiple biologic failures | Risankizumab, ustekinumab | Clinical trial |
Pre-Treatment Screening (Anti-TNF)
Always screen for:
- TB: Quantiferon or T-SPOT + CXR (treat latent TB before starting)
- Hepatitis B: HBsAg, anti-HBc (risk of reactivation)
- Vaccinations: Pneumococcal, influenza, Hep B (avoid live vaccines once on therapy)
- Baseline bloods: FBC, LFTs, renal function
Complications: What Gets You Marks
Intestinal:
- Strictures (30-40%) → obstruction, EBD or surgery
- Fistulae (20-40%): enteroenteric, enterocutaneous, enterovesical, perianal
- Abscess → drainage + antibiotics
- Perforation → surgery
- Small bowel adenocarcinoma (2-3×), colorectal cancer (if extensive colitis)
Extra-Intestinal:
- Joints (10-20%): peripheral arthritis (disease-activity linked), axial arthropathy (independent)
- Skin: erythema nodosum (yes, disease-linked), pyoderma gangrenosum (variable)
- Eyes: uveitis (urgent — risk blindness), episcleritis
- Hepatobiliary: PSC (1-4%), gallstones (ileal disease → bile acid malabsorption)
- VTE (1.5-3.5× risk — thromboprophylaxis during admission)
Treatment-Related:
- Steroids: osteoporosis, diabetes, infection
- Thiopurines: myelosuppression, hepatotoxicity, pancreatitis (3-5%), lymphoma (small risk)
- Anti-TNF: TB reactivation, serious infections, demyelination (rare)
Evidence and Trials
| Trial | Key Finding | Clinical Impact |
|---|---|---|
| SONIC (2010) [23] | Infliximab + azathioprine > monotherapy (57% vs 44% vs 30% remission) | Combination therapy superior |
| UNITI (2016) [9] | Ustekinumab effective in anti-TNF failures | IL-12/23 alternative mechanism |
| CALM (2018) [13] | Treat-to-target (tight control) > symptom-based (46% vs 30% mucosal healing) | Validates proactive monitoring |
| ADVANCE/MOTIVATE (2022-23) [10] | Risankizumab 55-60% remission (biologic-experienced) | Selective IL-23 highly efficacious |
| REPREVIO (2024) [12] | Vedolizumab reduces post-op recurrence (63% vs 48%) | First biologic proven for post-op prophylaxis |
Common Viva Questions and Model Answers
Q1: How do you differentiate Crohn's from ulcerative colitis?
A: "Crohn's disease differs from UC in several key aspects. Firstly, distribution: Crohn's can affect any part of the GI tract with skip lesions, whereas UC is continuous from rectum proximally within the colon only. Secondly, depth of inflammation: Crohn's is transmural leading to fistulae and strictures, whilst UC is mucosal. Histologically, Crohn's may show non-caseating granulomas in 30-40%, which are absent in UC. Clinically, perianal disease is common in Crohn's but rare in UC. Finally, smoking worsens Crohn's but is paradoxically protective in UC, and whilst surgery is curative in UC via total colectomy, it is non-curative in Crohn's with high recurrence rates."
Q2: A patient has failed anti-TNF therapy. What are your next options?
A: "Firstly, I'd assess the reason for failure: primary non-response versus secondary loss of response. If secondary loss of response, I'd check drug levels and anti-drug antibodies — low levels may warrant dose escalation or switching to another anti-TNF. If primary non-response or confirmed anti-TNF failure, I'd switch to an alternative mechanism: vedolizumab, an anti-integrin agent with gut-selective action; ustekinumab, targeting IL-12/23; or risankizumab, a selective IL-23 inhibitor, all of which have demonstrated efficacy in anti-TNF-experienced patients in trials such as UNITI and ADVANCE/MOTIVATE."
Q3: How would you manage perianal fistulising Crohn's disease?
A: "Perianal fistulae require combined medical and surgical approach. Initial assessment includes clinical examination and MRI pelvis to delineate fistula anatomy using Parks classification and identify abscesses. Medical management consists of antibiotics initially — metronidazole plus ciprofloxacin for symptom control — followed by anti-TNF therapy, either infliximab or adalimumab, which is first-line definitive treatment achieving fistula closure in 30-50% at one year. Concomitant immunomodulators such as azathioprine may be added. Surgically, if abscess present or complex fistula, I'd arrange EUA with seton placement to promote drainage whilst medical therapy takes effect. Definitive surgical procedures like fistulotomy or advancement flaps are reserved for refractory cases."
Q4: Describe your approach to preventing post-operative recurrence.
A: "Post-operative recurrence is common with 70-80% developing endoscopic recurrence at one year. I'd firstly stratify the patient's risk: high-risk features include smoking, penetrating disease, prior resections, extensive resection, and perianal disease. For high-risk patients, I'd commence prophylaxis with anti-TNF such as infliximab or adalimumab, or vedolizumab based on REPREVIO trial evidence. For moderate-risk, thiopurines or methotrexate are appropriate. Smoking cessation is paramount. I'd perform ileocolonoscopy at 6-12 months to assess the neo-terminal ileum using Rutgeerts score: i0-i1 indicates no recurrence and I'd continue current therapy, whilst i2-i4 indicates recurrence requiring treatment escalation."
14. References
-
Torres J, Bonovas S, Doherty G, et al. ECCO Guidelines on Therapeutics in Crohn's Disease: Medical Treatment. J Crohns Colitis. 2020;14(1):4-22. doi:10.1093/ecco-jcc/jjz180
-
Baumgart DC, Sandborn WJ. Crohn's disease. Lancet. 2012;380(9853):1590-1605. doi:10.1016/S0140-6736(12)60026-9
-
Gecse KB, Bemelman W, Kamm MA, et al. A global consensus on the classification, diagnosis and multidisciplinary treatment of perianal fistulising Crohn's disease. Gut. 2014;63(9):1381-1392. doi:10.1136/gutjnl-2013-306709
-
Liu JZ, van Sommeren S, Huang H, et al. Association analyses identify 38 susceptibility loci for inflammatory bowel disease and highlight shared genetic risk across populations. Nat Genet. 2015;47(9):979-986. doi:10.1038/ng.3359
-
Ramos GP, Papadakis KA. Mechanisms of Disease: Inflammatory Bowel Diseases. Mayo Clin Proc. 2019;94(1):155-165. doi:10.1016/j.mayocp.2018.09.013
-
Ng SC, Shi HY, Hamidi N, et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet. 2017;390(10114):2769-2778. doi:10.1016/S0140-6736(17)32448-0
-
Peyrin-Biroulet L, Sandborn W, Sands BE, et al. Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE): Determining Therapeutic Goals for Treat-to-Target. Am J Gastroenterol. 2015;110(9):1324-1338. doi:10.1038/ajg.2015.233
-
Hanauer SB, Feagan BG, Lichtenstein GR, et al. Maintenance infliximab for Crohn's disease: the ACCENT I randomised trial. Lancet. 2002;359(9317):1541-1549. doi:10.1016/S0140-6736(02)08512-4
-
Feagan BG, Sandborn WJ, Gasink C, et al. Ustekinumab as Induction and Maintenance Therapy for Crohn's Disease. N Engl J Med. 2016;375(20):1946-1960. doi:10.1056/NEJMoa1602773
-
Ferrante M, Panaccione R, Baert F, et al. Risankizumab as maintenance therapy for moderately to severely active Crohn's disease: results from the multicentre, randomised, double-blind, placebo-controlled, withdrawal phase 3 FORTIFY maintenance trial. Lancet. 2022;399(10340):2031-2046. doi:10.1016/S0140-6736(22)00466-4
-
De Cruz P, Kamm MA, Hamilton AL, et al. Crohn's disease management after intestinal resection: a randomised trial. Lancet. 2015;385(9976):1406-1417. doi:10.1016/S0140-6736(14)61908-5
-
D'Haens G, Taxonera C, Lopez-Sanroman A, et al. Vedolizumab to prevent postoperative recurrence of Crohn's disease (REPREVIO): a multicentre, double-blind, randomised, placebo-controlled trial. Lancet Gastroenterol Hepatol. 2025;10(1):26-33. doi:10.1016/S2468-1253(24)00317-0
-
Colombel JF, Panaccione R, Bossuyt P, et al. Effect of tight control management on Crohn's disease (CALM): a multicentre, randomised, controlled phase 3 trial. Lancet. 2018;390(10114):2779-2789. doi:10.1016/S0140-6736(17)32641-7
-
Lutgens MW, van Oijen MG, van der Heijden GJ, et al. Declining risk of colorectal cancer in inflammatory bowel disease: an updated meta-analysis of population-based cohort studies. Inflamm Bowel Dis. 2013;19(4):789-799. doi:10.1097/MIB.0b013e31828029c0
-
Parkes GC, Whelan K, Lindsay JO. Smoking in inflammatory bowel disease: impact on disease course and insights into the aetiology of its effect. J Crohns Colitis. 2014;8(8):717-725. doi:10.1016/j.crohns.2014.02.002
-
Caparrós E, Wiest R, Scharl M, et al. Dysbiotic microbiota interactions in Crohn's disease. Gut Microbes. 2021;13(1):1949096. doi:10.1080/19490976.2021.1949096
-
Silverberg MS, Satsangi J, Ahmad T, et al. Toward an integrated clinical, molecular and serological classification of inflammatory bowel disease: report of a Working Party of the 2005 Montreal World Congress of Gastroenterology. Can J Gastroenterol. 2005;19 Suppl A:5A-36A. doi:10.1155/2005/269076
-
Vavricka SR, Schoepfer A, Scharl M, et al. Extraintestinal Manifestations of Inflammatory Bowel Disease. Inflamm Bowel Dis. 2015;21(8):1982-1992. doi:10.1097/MIB.0000000000000392
-
Yuhara H, Steinmaus C, Corley D, et al. Meta-analysis: the risk of venous thromboembolism in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2013;37(10):953-962. doi:10.1111/apt.12294
-
Debi U, Ravisankar V, Prasad KK, et al. Abdominal tuberculosis of the gastrointestinal tract: revisited. World J Gastroenterol. 2014;20(40):14831-14840. doi:10.3748/wjg.v20.i40.14831
-
van Rheenen PF, Van de Vijver E, Fidler V. Faecal calprotectin for screening of patients with suspected inflammatory bowel disease: diagnostic meta-analysis. BMJ. 2010;341:c3369. doi:10.1136/bmj.c3369
-
Rezaie A, Kuenzig ME, Benchimol EI, et al. Budesonide for induction of remission in Crohn's disease. Cochrane Database Syst Rev. 2015;(6):CD000296. doi:10.1002/14651858.CD000296.pub4
-
Colombel JF, Sandborn WJ, Reinisch W, et al. Infliximab, azathioprine, or combination therapy for Crohn's disease. N Engl J Med. 2010;362(15):1383-1395. doi:10.1056/NEJMoa0904492
-
Sandborn WJ, Feagan BG, Rutgeerts P, et al. Vedolizumab as induction and maintenance therapy for Crohn's disease. N Engl J Med. 2013;369(8):711-721. doi:10.1056/NEJMoa1215739
-
Bettenworth D, Gustavsson A, Atreja A, et al. A Pooled Analysis of Efficacy, Safety, and Long-term Outcome of Endoscopic Balloon Dilation Therapy for Patients with Stricturing Crohn's Disease. Inflamm Bowel Dis. 2017;23(1):133-142. doi:10.1097/MIB.0000000000000988
-
Jess T, Frisch M, Simonsen J. Trends in overall and cause-specific mortality among patients with inflammatory bowel disease from 1982 to 2010. Clin Gastroenterol Hepatol. 2013;11(1):43-48. doi:10.1016/j.cgh.2012.09.026
-
Rutgeerts P, Van Assche G, Sandborn WJ, et al. Adalimumab induces and maintains mucosal healing in patients with Crohn's disease: data from the EXTEND trial. Gastroenterology. 2012;142(5):1102-1111.e2. doi:10.1053/j.gastro.2012.01.035
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate guidelines and specialists for patient care.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Differentials
Competing diagnoses and look-alikes to compare.
- Intestinal Tuberculosis
- Bowel Malignancy
Consequences
Complications and downstream problems to keep in mind.
- Short Bowel Syndrome