Cushing's Syndrome
The pathophysiology involves systemic dysregulation of protein and fat metabolism, creating a catabolic state characterized by proximal muscle wasting, skin thinning, and paradoxical central obesity. Diagnostic...
What matters first
The pathophysiology involves systemic dysregulation of protein and fat metabolism, creating a catabolic state characterized by proximal muscle wasting, skin thinning, and paradoxical central obesity. Diagnostic...
Severe, refractory hypokalemia (suggests ectopic ACTH secretion)
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Severe, refractory hypokalemia (suggests ectopic ACTH secretion)
- Rapid onset of clinical features (less than 3-6 months)
- Acute proximal myopathy preventing standing or walking
- Opportunistic infections (e.g., Pneumocystis jirovecii) due to severe immunosuppression
Linked comparisons
Differentials and adjacent topics worth opening next.
- Pseudo-Cushing's Syndrome (Alcoholism/Depression)
- Polycystic Ovary Syndrome (PCOS)
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Cushing's Syndrome (Adult)
1. Clinical Overview
Summary
Cushing's Syndrome (CS) represents the clinical manifestation of chronic, inappropriate exposure to excessive levels of glucocorticoids. While the most common cause worldwide is exogenous iatrogenic administration of synthetic steroids, endogenous CS remains a diagnostic challenge associated with significant morbidity and mortality. Endogenous CS is categorized into ACTH-dependent (80%), primarily caused by an ACTH-secreting pituitary adenoma (termed Cushing's Disease), and ACTH-independent (20%), caused by primary adrenal pathology including adenomas, carcinomas, or bilateral adrenal hyperplasia. [1,2]
The pathophysiology involves systemic dysregulation of protein and fat metabolism, creating a catabolic state characterized by proximal muscle wasting, skin thinning, and paradoxical central obesity. Diagnostic protocols have evolved toward a multi-step verification process: confirming hypercortisolism via screening tests, followed by ACTH-driven localization, and ultimately anatomical and functional imaging. Modern management has transitioned from purely surgical approaches to a multimodal strategy incorporating potent steroidogenesis inhibitors like osilodrostat and levoketoconazole, targeted biologics such as pasireotide, and glucocorticoid receptor antagonists. [3,4]
The disease carries substantial morbidity even after cure, with persistent cardiovascular risk, metabolic dysfunction, and cognitive impairment. Early diagnosis and treatment are critical to prevent irreversible complications including vertebral fractures, cardiovascular disease, and opportunistic infections. [5]
Key Facts
- The Pituitary Prevalence: Cushing's Disease (pituitary source) accounts for approximately 70% of all endogenous cases, with a marked female predominance (female:male ratio 5-8:1). [1]
- Ectopic Alert: Ectopic ACTH syndrome (e.g., small cell lung cancer, bronchial carcinoid) often presents with severe hypokalemia, metabolic alkalosis, and hyperpigmentation due to high ACTH levels and POMC cross-reactivity with MSH receptors. [6]
- Screening Trifecta: 24-hour urinary free cortisol (UFC), late-night salivary cortisol (LNSC), and the 1-mg overnight dexamethasone suppression test (ONDST) are the validated screening tools, with at least two positive tests required for diagnosis. [2]
- Curative Aim: Surgical resection (transsphenoidal for pituitary, laparoscopic for adrenal) remains the first-line treatment for all resectable sources, with biochemical remission rates of 70-90% for microadenomas. [7]
- The "Cushingoid" Phenotype: While classic, features like buffalo humps and moon facies have lower specificity than proximal myopathy, wide purple striae, and facial plethora. [8]
- Subclinical Disease: Recent studies suggest that 1-3% of patients with poorly controlled type 2 diabetes and hypertension may have occult subclinical Cushing's syndrome, representing an underdiagnosed entity. [9]
- Mortality Impact: Untreated Cushing's syndrome carries a standardized mortality ratio (SMR) of 3.7-4.8, primarily due to cardiovascular and infectious complications. [10]
Clinical Pearls
The "Striae Distinction": Clinical striae in Cushing's are classically > 1 cm in width and violaceous (purple). These reflect dermal atrophy and fragility of capillaries due to protein catabolism. Narrow, pale, or silvery striae are common in simple obesity or rapid growth and lack the diagnostic specificity for hypercortisolism.
Pseudo-Cushing's Trap: Chronic alcoholism, severe depression, and extreme obesity can cause biochemically positive screening tests (Pseudo-Cushing's states). These conditions activate the HPA axis through stress pathways. Clinical features are often more reliable than borderline laboratory results in these populations, and the dexamethasone-CRH test can help differentiate. [11]
Cyclic Cushing's Challenge: Approximately 15% of Cushing's cases exhibit cyclic or periodic hypercortisolism, with alternating periods of normal and elevated cortisol production. This requires multiple serial measurements over weeks to months for diagnosis. [12]
IPSS Gradient Interpretation: In bilateral inferior petrosal sinus sampling (BIPSS), a central-to-peripheral ACTH ratio > 2 basally or > 3 post-CRH stimulation indicates pituitary source. Lateralization (interpetrosal ratio > 1.4) can guide surgical approach but has lower reliability. [13]
2. Epidemiology
Incidence and Prevalence
- Incidence: Endogenous CS is rare, with an incidence of 0.7–2.4 per million population per year in Europe and North America. [14]
- Prevalence: Estimated prevalence is 39-79 cases per million population. The true prevalence may be higher when including subclinical disease. [1]
Demographics
-
Gender Distribution:
- "Cushing's Disease shows a strong female predominance (5:1 to 8:1 ratio)"
- "Ectopic ACTH is more common in males (2:1) due to higher incidence of associated lung malignancies"
- "Adrenal adenomas show moderate female predominance (2:1)"
- Adrenal carcinomas have equal gender distribution
-
Age of Onset:
- "Cushing's Disease: Peaks in the 3rd to 4th decades (25-40 years)"
- "Adrenal Adenoma: Peak incidence 30-50 years"
- "Adrenal Carcinoma: Bimodal distribution (childhood less than 5 years and adults 40-50 years)"
- "Ectopic ACTH: Typically > 50 years, reflecting age distribution of causative tumors"
Special Populations
- Pediatric Cushing's: Rare, with different etiologic distribution—adrenal causes more common than in adults. Growth arrest is a key differentiating feature. [15]
- Pregnancy and Cushing's: Extremely rare (1 in 1 million pregnancies). Adrenal adenomas account for 40-50% of cases in pregnancy, higher than in non-pregnant populations. Associated with high maternal and fetal morbidity. [16]
- Subclinical Prevalence: Studies of adrenal incidentalomas reveal that 5-20% demonstrate autonomous cortisol secretion without overt Cushingoid features, termed "subclinical Cushing's syndrome" or "mild autonomous cortisol excess." [9,17]
3. Pathophysiology
The HPA Axis Disruption
In healthy physiology, the hypothalamus releases corticotropin-releasing hormone (CRH), which stimulates the anterior pituitary corticotroph cells to release adrenocorticotropic hormone (ACTH). ACTH then binds to melanocortin 2 receptors (MC2R) on the adrenal cortex (zona fasciculata), triggering cortisol synthesis and secretion. Cortisol provides negative feedback to both the hypothalamus and pituitary, maintaining homeostasis.
Cushing's syndrome represents a failure of this regulatory system through multiple potential mechanisms:
1. ACTH-Dependent Causes (80%)
A. Cushing's Disease (Pituitary ACTH Excess, 70% of endogenous cases)
-
Molecular Pathogenesis: A monoclonal expansion of corticotroph cells in the anterior pituitary forms a microadenoma (less than 10mm in 80-90% of cases) or macroadenoma (> 10mm). Recent genomic studies have identified somatic mutations in the USP8 (ubiquitin-specific protease 8) gene in approximately 35-40% of these adenomas. [18]
-
USP8 Mechanism: USP8 mutations (predominantly p.Pro720Arg and p.Ser718del) prevent degradation of epidermal growth factor receptor (EGFR), leading to sustained EGFR signaling, increased POMC transcription, and autonomous ACTH production. These mutations cluster in the 14-3-3 protein binding domain, disrupting USP8 auto-inhibition. [18]
-
Other Genetic Drivers: Additional mutations in genes including CDK5, BRAF, and rarely MEN1 have been identified. Germline mutations in MEN1, CDKN1B (MEN4), and PRKAR1A (Carney Complex) predispose to pituitary adenomas.
-
Pathophysiology: The adenoma produces ACTH autonomously, with partial preservation of negative feedback (explaining why high-dose dexamethasone can suppress cortisol in 70-80% of cases). Bilateral adrenal hyperplasia develops secondary to chronic ACTH stimulation.
B. Ectopic ACTH Syndrome (10-15% of endogenous cases)
-
Tumor Sources:
- "Small cell lung cancer (SCLC): 40-50% of ectopic ACTH"
- "Bronchial carcinoid tumors: 25-30%"
- "Pancreatic neuroendocrine tumors (NETs): 10%"
- "Thymic carcinoid: 5-10%"
- "Medullary thyroid carcinoma: 5%"
- "Pheochromocytoma/paraganglioma: Rare (less than 5%)"
-
Molecular Mechanism: These tumors ectopically express the POMC gene and its transcriptional machinery. They produce not only mature ACTH but also high levels of POMC precursors and related peptides (β-lipotropin, β-endorphin). The tumors bypass normal HPA regulation entirely.
-
Clinical Phenotype: Ectopic ACTH typically presents with rapid onset (less than 6 months), severe hypercortisolism with very high cortisol levels (often > 1000 nmol/L), severe hypokalemia (less than 2.5 mmol/L), metabolic alkalosis, peripheral edema, and hyperpigmentation. The classic cushingoid habitus may be absent due to rapid tempo. [6]
C. Ectopic CRH Syndrome (Rare, less than 1%)
- Extremely rare syndrome where tumors (typically bronchial carcinoids) produce CRH, leading to pituitary corticotroph hyperplasia and secondary ACTH excess. Biochemically mimics Cushing's disease.
2. ACTH-Independent Causes (20%)
A. Unilateral Adrenal Adenoma (10-15%)
-
Pathophysiology: Benign cortical adenoma autonomously secretes cortisol. The high cortisol levels suppress hypothalamic CRH and pituitary ACTH via negative feedback, leading to atrophy of the contralateral normal adrenal gland and the non-tumorous portion of the ipsilateral gland.
-
Genetic Drivers: Somatic activating mutations in PRKACA (protein kinase A catalytic subunit alpha) are found in 35-65% of cortisol-secreting adenomas. The most common mutation is PRKACA c.617A>C (p.Leu206Arg), which renders PKA constitutively active. [19]
-
Other Mutations: CTNNB1 (β-catenin), GNAS (Gs-alpha, associated with McCune-Albright syndrome), and chromosome 17q gains are also implicated.
B. Adrenocortical Carcinoma (5%)
-
Pathophysiology: Malignant tumor of the adrenal cortex with autonomous steroid production. Often produces multiple steroids (cortisol, androgens, aldosterone), leading to mixed clinical presentations.
-
Molecular Features: TP53 mutations (> 50%), IGF2 overexpression, alterations in CTNNB1, and chromatin remodeling genes. Germline mutations in TP53 (Li-Fraumeni syndrome) and Lynch syndrome genes predispose to ACC.
-
Clinical Features: Large tumors (typically > 6cm), often with local invasion or metastases at presentation. Poor prognosis with 5-year survival of 30-40%. [20]
C. Primary Bilateral Macronodular Adrenal Hyperplasia (BMAH)
-
Mechanism: Bilateral adrenal enlargement with multiple nodules (> 1cm) causing autonomous cortisol production. May involve aberrant expression of ectopic hormone receptors (GIP receptor, LH receptor, vasopressin receptors) that inappropriately stimulate cortisol secretion.
-
Genetics: ARMC5 (armadillo repeat containing 5) germline mutations found in 25-55% of cases. Loss of heterozygosity leads to bilateral nodule formation. [21]
D. Primary Pigmented Nodular Adrenocortical Disease (PPNAD)
-
Characteristics: Small pigmented micronodules (less than 1cm) in both adrenal glands. Adrenals may be normal-sized or small.
-
Association: Part of Carney Complex (autosomal dominant syndrome with cardiac myxomas, skin pigmentation, endocrine tumors). Caused by inactivating mutations in PRKAR1A gene, leading to unregulated PKA activity.
Systemic Effects of Hypercortisolism
Metabolic Effects
Protein Catabolism:
- Cortisol inhibits amino acid uptake in muscle and skin
- Suppresses collagen synthesis and promotes breakdown
- Results in thin skin, easy bruising, poor wound healing, proximal myopathy, and striae
- Contributes to growth arrest in children
Carbohydrate Metabolism:
- Stimulates hepatic gluconeogenesis
- Induces insulin resistance in peripheral tissues
- Reduces glucose uptake in muscle and adipose tissue
- Results in impaired glucose tolerance or frank diabetes (40-45% of patients)
Lipid Redistribution:
- Cortisol stimulates lipoprotein lipase activity in centripetal fat deposits (trunk, face, supraclavicular fossae)
- Promotes lipolysis in peripheral adipose tissue (extremities)
- Results in characteristic central obesity with peripheral wasting
- Visceral adiposity contributes to metabolic syndrome
Bone and Mineral Metabolism
- Osteoporosis: Cortisol inhibits osteoblast differentiation and function, stimulates osteoclast activity via RANKL upregulation, and decreases intestinal calcium absorption while increasing renal calcium excretion
- Suppresses sex hormone production, further impairing bone health
- Rapid bone loss: 10-20% per year in active disease
- Vertebral compression fractures occur in 30-67% of patients, often asymptomatic [22]
Cardiovascular Effects
-
Hypertension (present in 70-85%): Multiple mechanisms:
- At high concentrations, cortisol saturates the protective enzyme 11β-hydroxysteroid dehydrogenase type 2 (11β-HSD2), which normally converts cortisol to inactive cortisone in renal tubular cells
- Unconverted cortisol binds mineralocorticoid receptors (MR), causing sodium retention and potassium wasting
- Enhanced vascular reactivity to catecholamines
- Suppression of vasodilatory systems (nitric oxide, prostaglandins)
-
Cardiovascular Remodeling:
- Left ventricular hypertrophy
- Increased arterial stiffness
- Endothelial dysfunction
- Accelerated atherosclerosis
- Increased cardiovascular mortality (4-5 fold) [23]
Immune System
-
Immunosuppression:
- Inhibition of cytokine production (IL-1, IL-2, IL-6, TNF-α)
- Induction of T-cell apoptosis via mitochondrial pathway
- Impaired neutrophil migration and function
- Reduced eosinophils and lymphocytes
-
Infection Risk:
- "Opportunistic infections: Pneumocystis jirovecii pneumonia, cryptococcosis, aspergillosis, strongyloidiasis"
- Typical bacterial infections (skin, urinary, respiratory)
- Infection is a major cause of mortality in severe CS
Neuropsychiatric Effects
- Depression (50-70%), anxiety, emotional lability
- Cognitive impairment: memory deficits, attention problems
- Psychosis (5-10% of severe cases)
- May not fully reverse after cure, suggesting structural brain changes
- Hippocampal atrophy demonstrated on MRI
Reproductive Effects
- Women: Menstrual irregularities (amenorrhea, oligomenorrhea in 70-80%), reduced fertility, hirsutism from adrenal androgen excess
- Men: Hypogonadotropic hypogonadism, erectile dysfunction, reduced libido, infertility due to suppression of gonadotropins
4. Clinical Presentation
Discriminating Features
While many features of Cushing's syndrome overlap with common conditions like metabolic syndrome and obesity, certain clinical findings have higher specificity for pathologic hypercortisolism. Four features have been identified as most discriminating: [8]
-
Proximal Myopathy: Difficulty rising from a chair without using arms, difficulty climbing stairs, inability to squat. Results from muscle protein catabolism. Sensitivity 40-70%, specificity 90-95%.
-
Easy Bruising: Spontaneous ecchymoses with minimal or no trauma, particularly on extremities and dorsum of hands. Due to vascular fragility and dermal thinning. Sensitivity 55-70%, specificity 90%.
-
Purple Striae (> 1cm width): Widely spaced, violaceous, atrophic striae on abdomen, breasts, thighs, or axillae. Must be > 1cm width and purple/red (not pale). Result from dermal atrophy and weakened connective tissue. Sensitivity 40-60%, specificity 95-100%.
-
Facial Plethora: Persistent reddish-purple hue to face, independent of flushing or rosacea. Due to thinning of facial skin and telangiectasias. Sensitivity 85%, specificity 85%.
Systemic Manifestations
Constitutional
- Weight gain (particularly central): 90-95%
- Fatigue and weakness: 85-95%
- Decreased exercise tolerance: 80%
Skin and Soft Tissue
- Thin skin, parchment-like: 80%
- Easy bruising: 65%
- Purple striae (> 1cm): 55-70%
- Acne (facial and truncal): 40-50%
- Hirsutism (women): 65-80%
- Facial plethora: 85-90%
- Fungal skin infections: 40%
- Poor wound healing: 65%
- Hyperpigmentation: Rare, suggests ectopic ACTH
Metabolic and Endocrine
- Central obesity: 90-95%
- "Moon facies" (rounded, full face): 85%
- "Buffalo hump" (dorsocervical fat pad): 55%
- "Supraclavicular fat pads: 70%"
- "Truncal obesity with extremity wasting: 80%"
- Glucose intolerance/Diabetes mellitus: 40-45%
- Hypertension: 70-85%
- Dyslipidemia: 60-70%
- Menstrual irregularities (women): 70-80%
- Erectile dysfunction (men): 70-85%
Musculoskeletal
- Proximal muscle weakness: 40-70%
- Osteoporosis/osteopenia: 50-80%
- Vertebral compression fractures: 30-67%
- Avascular necrosis (femoral head): 5-10%
- Growth retardation (children): 90%
Neuropsychiatric
- Depression: 50-70%
- Emotional lability, irritability: 60-80%
- Insomnia: 60-75%
- Anxiety: 50-65%
- Cognitive impairment: 50-70%
- Psychosis: 5-10%
- Suicidal ideation: 5-15%
Cardiovascular
- Hypertension: 70-85%
- Left ventricular hypertrophy: 40-50%
- Congestive heart failure: 15-20%
- Thromboembolic disease: 5-10% (increased risk)
Immune/Infectious
- Recurrent infections: 30-40%
- Opportunistic infections: 5-10% (severe cases)
Presentation Patterns by Etiology
Cushing's Disease (Pituitary)
- Gradual onset over months to years
- Classic cushingoid habitus
- Moderate hypercortisolism
- Hyperpigmentation rare (unless Nelson's syndrome develops post-bilateral adrenalectomy)
Ectopic ACTH
- Rapid onset (less than 6 months, often less than 3 months)
- Severe hypercortisolism
- Weight loss more common than weight gain
- Severe hypokalemia (less than 2.5 mmol/L) and metabolic alkalosis
- Hyperpigmentation common
- Peripheral edema
- Glucose intolerance/diabetes
- Cushingoid habitus may be absent due to rapid tempo
- Older age group (typically > 50 years)
Adrenal Adenoma
- Gradual onset
- Classic cushingoid features
- Unilateral adrenal mass on imaging
- Suppressed ACTH
Adrenal Carcinoma
- May have rapid onset
- Mixed steroid excess (virilization in women, feminization in men)
- Large adrenal mass (> 6cm typically)
- Local symptoms (abdominal/flank pain)
- Constitutional symptoms (weight loss, fever)
- Metastases may be present
Special Clinical Scenarios
Cyclic Cushing's Syndrome
- Periodic hypercortisolism alternating with eucortisolism or hypocortisolism
- Occurs in 15% of cases (all etiologies)
- Cycle length: days to months
- Diagnosis requires serial measurements over weeks-months
- Patients may present during normocortisolic phase [12]
Subclinical Cushing's Syndrome
- Biochemical evidence of autonomous cortisol secretion without overt cushingoid phenotype
- Often discovered incidentally during evaluation of adrenal incidentalomas
- Subtle manifestations: hypertension, diabetes, obesity, osteoporosis
- Higher prevalence of cardiovascular and metabolic complications compared to non-functioning adenomas [9,17]
5. Differential Diagnosis
Pseudo-Cushing's States
Conditions that activate the HPA axis and can produce biochemical findings similar to Cushing's syndrome without autonomous cortisol production:
| Condition | Clinical Features | Distinguishing Features | Diagnostic Approach |
|---|---|---|---|
| Chronic Alcoholism | Cushingoid features, elevated UFC, abnormal ONDST | Cortisol normalizes within 1-2 weeks of abstinence | History, serial testing after abstinence |
| Severe Depression | Obesity, elevated UFC possible | LNSC may be normal; CRH-dex test helpful | Psychiatric evaluation, CRH-dex test |
| Extreme Obesity | Central adiposity, moon facies | Lacks catabolic features (thin skin, striae, myopathy) | Normal screening tests typically |
| Pregnancy | Weight gain, striae, elevated total cortisol | Free cortisol should be appropriately elevated for trimester | Gestational age correlation |
| Poorly Controlled Diabetes | Metabolic syndrome features | Normal screening tests | Glycemic control assessment |
Dexamethasone-CRH Test for Pseudo-Cushing's
- Patient receives dexamethasone 0.5mg every 6 hours for 48 hours (8 doses)
- Two hours after last dose, CRH (1 μg/kg) is given IV
- Plasma cortisol measured 15 minutes post-CRH
- Cushing's syndrome: Cortisol > 38 nmol/L (1.4 μg/dL) - sensitivity 95%, specificity 100%
- Pseudo-Cushing's: Cortisol suppressed less than 38 nmol/L [11]
Other Differential Diagnoses
| Condition | Overlapping Features | Differentiating Features | Diagnostic Tests |
|---|---|---|---|
| PCOS | Hirsutism, acne, obesity, oligomenorrhea | Young women, lacks myopathy/thin skin/striae; ovarian dysfunction | LH/FSH ratio, pelvic ultrasound, normal cortisol |
| Metabolic Syndrome | Central obesity, HTN, diabetes | Lacks catabolic features | Normal cortisol screening |
| Exogenous Glucocorticoid Use | Identical to endogenous CS | History of corticosteroid use (oral, inhaled, topical, injected) | Medication history, ACTH suppressed |
| Primary Aldosteronism | Hypertension, hypokalemia | No cushingoid features | Aldosterone-renin ratio, normal cortisol |
| Congenital Adrenal Hyperplasia | Variable presentation based on enzyme deficiency | Typically presents in childhood; specific enzyme deficiencies | 17-hydroxyprogesterone, ACTH stimulation test |
| Familial Partial Lipodystrophy | Central adiposity, insulin resistance | Fat loss in extremities, normal cortisol | Genetic testing |
6. Investigations
Diagnostic Pathway Overview
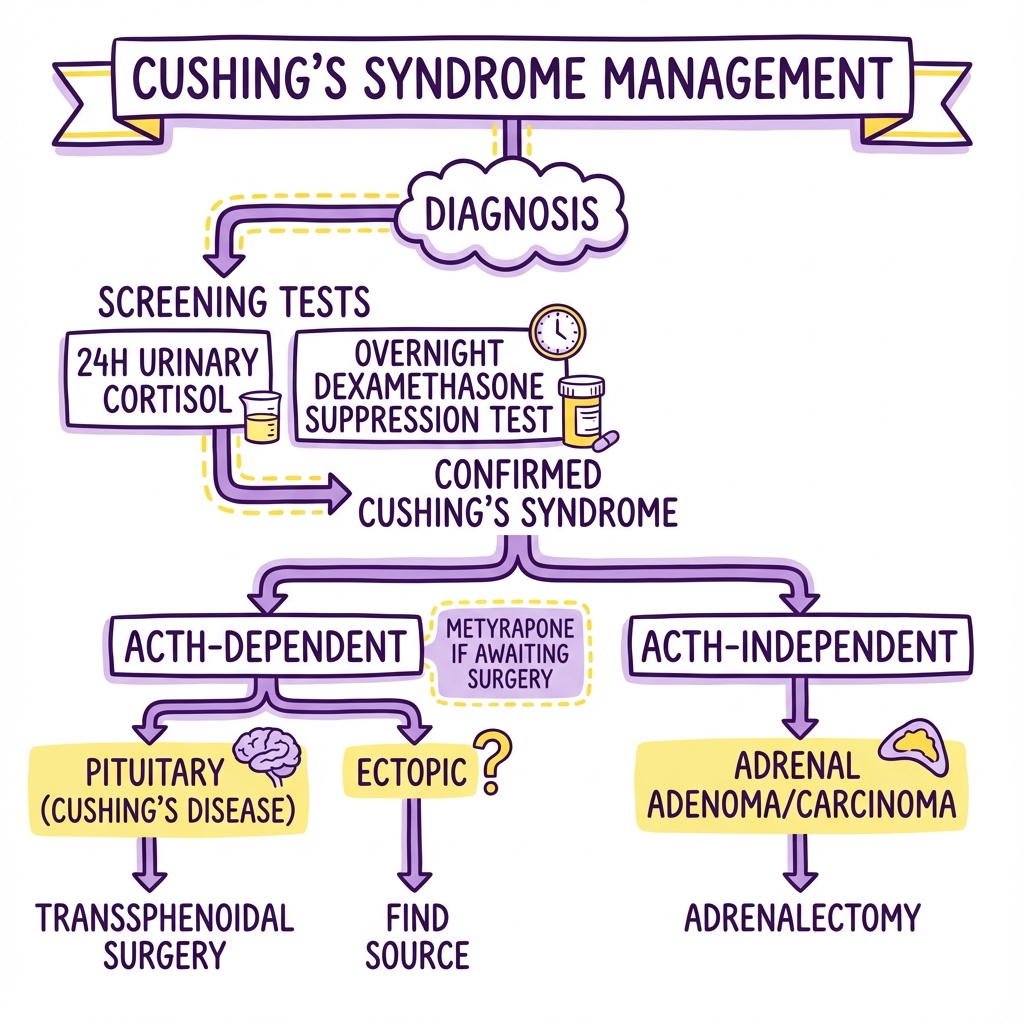
The diagnosis of Cushing's syndrome follows a structured three-step approach:
- Confirm hypercortisolism (screening tests)
- Determine ACTH-dependence (ACTH measurement)
- Localize the source (imaging and dynamic testing)
Step 1: Confirmation of Hypercortisolism
General Principle: At least two different positive screening tests are required to confirm Cushing's syndrome due to test limitations and possibility of Pseudo-Cushing's states. No single test has 100% sensitivity and specificity. [2]
Test 1: 24-Hour Urinary Free Cortisol (UFC)
Principle: Measures unbound (free) cortisol filtered by kidneys over 24 hours, reflecting integrated cortisol production.
Normal Values: less than 50-100 μg/24hr (depending on assay)
Interpretation:
- Normal: Excludes CS (if adequate collection)
- Elevated (> 3x ULN): Highly suggestive of CS - specificity approaches 100%
- Mildly elevated (1-3x ULN): Nonspecific, seen in Pseudo-Cushing's states
Performance:
- Sensitivity: 45-71% (single measurement)
- Specificity: 70-98% (variable by cutoff)
- Performing 2-3 collections increases sensitivity to 85-95%
Limitations:
- Inaccurate in chronic kidney disease (CrCl less than 60 ml/min)
- Affected by high fluid intake (dilution effect)
- Requires complete 24-hour collection (assess with creatinine)
- Can be elevated in Pseudo-Cushing's states
- Some assays have cross-reactivity with synthetic glucocorticoids
Pitfalls:
- Incomplete collection (assess urinary creatinine)
- Medications: carbamazepine (increases cortisol metabolism)
Test 2: Late-Night Salivary Cortisol (LNSC)
Principle: Measures free cortisol in saliva, which parallels serum free cortisol. Captures loss of circadian rhythm (normally cortisol nadir occurs at midnight).
Sample Collection: Two samples collected at 11 PM to midnight on separate nights (patient at home, normal routine)
Normal Values: less than 1.8-3.6 nmol/L (less than 50-100 ng/dL), depending on assay
Performance:
- Sensitivity: 92-100%
- Specificity: 93-100%
- At least as good as UFC, possibly superior
Advantages:
- Easy collection (home-based, non-invasive)
- Not affected by renal function
- Excellent for detecting loss of circadian rhythm
- Can be repeated easily
Limitations:
- Affected by shift work or irregular sleep schedules
- Tobacco use can elevate values
- Liquorice ingestion (inhibits 11β-HSD2, elevates cortisol)
- Not standardized across all assays
- Less available in some regions
Recommendation: Two separate measurements on different days recommended [2]
Test 3: 1-mg Overnight Dexamethasone Suppression Test (ONDST)
Principle: In normal physiology, exogenous dexamethasone suppresses ACTH and cortisol via negative feedback. Autonomous cortisol production fails to suppress.
Protocol:
- Patient takes 1 mg dexamethasone orally at 11 PM
- Plasma cortisol measured at 8-9 AM the next morning
Interpretation:
- Normal suppression: Cortisol less than 50 nmol/L (1.8 μg/dL) - excludes CS
- Inadequate suppression: Cortisol ≥50 nmol/L - suggests CS or Pseudo-Cushing's
Performance:
- Sensitivity: 95-98% (using cutoff less than 50 nmol/L)
- Specificity: 80-85% (many false positives)
Advantages:
- Simple, rapid screening test
- Inexpensive
- Can be done outpatient
Limitations and False Positives:
- Estrogen use (OCP, HRT): Increases cortisol-binding globulin (CBG), elevating total cortisol (most common cause of false positive)
- CYP3A4 inducers: Phenytoin, phenobarbital, rifampicin, carbamazepine, St. John's Wort (accelerate dexamethasone metabolism)
- CYP3A4 inhibitors: Itraconazole, ritonavir (slow dexamethasone metabolism, can cause false negative)
- Pseudo-Cushing's states
- Renal failure
- Stress, acute illness
Modifications:
- Some centers use lower cutoff (28-40 nmol/L) for higher specificity but reduced sensitivity
- 2-day low-dose DST (0.5mg q6h x 8 doses): Higher specificity, more cumbersome
Alternative/Complementary Tests
Late-Night Serum Cortisol:
- Blood sample at 11 PM-midnight
- Normal: less than 50 nmol/L (less than 1.8 μg/dL)
- Sensitivity 96%, specificity 93%
- Requires hospitalization or late-night phlebotomy access
- Loss of circadian rhythm is hallmark of CS
Dexamethasone-Suppressed CRH Test:
- Used to differentiate Pseudo-Cushing's from true CS
- See Pseudo-Cushing's section above
Step 2: Differential Diagnosis - ACTH Measurement
Once hypercortisolism is confirmed, measure plasma ACTH to determine if cortisol excess is ACTH-dependent or ACTH-independent.
Sample Collection:
- Morning sample (8-9 AM) ideally
- Requires special handling: EDTA tube, place on ice, process rapidly
- ACTH is unstable; delays cause falsely low values
Interpretation:
| ACTH Level | Interpretation | Next Step |
|---|---|---|
| less than 1.1 pmol/L (less than 5 pg/mL) | ACTH-independent (adrenal source) | Adrenal CT imaging |
| 1.1-4.4 pmol/L (5-20 pg/mL) | Indeterminate | CRH stimulation test |
| > 4.4 pmol/L (> 20 pg/mL) | ACTH-dependent (pituitary or ectopic) | Pituitary MRI + further testing |
Important: Measure ACTH on at least 2 occasions (preferably 3) due to variability, particularly in cyclic disease.
Step 3A: Localization - ACTH-Independent (Adrenal) CS
Adrenal CT Scan
Protocol: Thin-section CT (3mm slices) without and with IV contrast, plus delayed imaging (10-15 minutes)
Findings:
| Diagnosis | CT Features | Size |
|---|---|---|
| Adrenal Adenoma | Well-defined, round, homogeneous, low attenuation (less than 10 HU unenhanced), rapid contrast washout (> 50% at 10 min) | Usually 2-5 cm |
| Adrenal Carcinoma | Irregular, heterogeneous, high attenuation (> 20 HU), delayed washout (less than 50%), calcification, necrosis, invasion | Usually > 6 cm |
| BMAH | Bilateral enlarged adrenals with multiple nodules, maintain adreniform shape | Glands > 6 cm combined width |
| PPNAD | Small or normal-sized adrenals with micronodules | May be normal-appearing |
Follow-up:
- Unilateral mass: Proceed to adrenalectomy
- Bilateral nodularity: Consider BMAH vs. bilateral metastases, evaluate for aberrant receptors
- Normal-appearing adrenals with suppressed ACTH: Consider PPNAD, may require NP-59 scintigraphy or bilateral adrenal venous sampling
Alternative Imaging
MRI Adrenals:
- Chemical shift imaging helpful (adenomas lose signal on opposed-phase imaging)
- Useful when CT contraindicated
NP-59 Adrenal Scintigraphy:
- Rarely used; can identify functional adrenal tissue in PPNAD
FDG-PET: High uptake suggests adrenal carcinoma (differentiates from benign adenoma)
Step 3B: Localization - ACTH-Dependent CS
The challenge is differentiating Cushing's disease (pituitary) from ectopic ACTH syndrome.
Pituitary MRI
Protocol: Thin-section (2-3mm) dedicated pituitary MRI with and without gadolinium contrast
Findings:
- Microadenoma (less than 10 mm): Seen in 50-70% of Cushing's disease cases
- Macroadenoma (> 10 mm): 10-20% of cases
- No visible adenoma: 20-40% despite Cushing's disease (limitation of MRI resolution)
Limitations:
- 10% of normal population has incidental pituitary lesions
- Presence of microadenoma does NOT confirm pituitary source
- Absence of adenoma does NOT exclude Cushing's disease
- Interpretation should be integrated with biochemical testing
Caution: Ectopic ACTH can cause pituitary hyperplasia or incidental microadenomas; do not rely on MRI alone.
High-Dose Dexamethasone Suppression Test (HDDST)
8-mg Overnight Test:
- 8 mg dexamethasone orally at 11 PM
- Measure plasma cortisol at 8 AM next day
- Cushing's Disease: Cortisol suppression > 50% from baseline in 70-80%
- Ectopic ACTH: Usually no suppression (less than 10% suppress)
2-Day High-Dose Test:
- Dexamethasone 2 mg PO q6h for 48 hours (8 doses)
- Measure 24h UFC or serum cortisol before and during last day
- Suppression criteria same as above
Limitations:
- Only 70-80% sensitive for Cushing's disease
- 10-20% of Cushing's disease cases do NOT suppress
- Rare ectopic sources (bronchial carcinoids) can suppress (10-15%)
- Should NOT be used as sole discriminator
CRH Stimulation Test
Protocol:
- Baseline ACTH and cortisol measured (at -15, -5, 0 min)
- Ovine or human CRH 1 μg/kg (or 100 μg) IV bolus at time 0
- ACTH and cortisol measured at 15, 30, 45, 60, 90 minutes
Interpretation:
| Response | ACTH Increase | Cortisol Increase | Diagnosis |
|---|---|---|---|
| Positive | > 35-50% increase from baseline | > 20% increase from baseline | Cushing's Disease |
| Negative | No significant rise | No significant rise | Ectopic ACTH |
Performance:
- Sensitivity for Cushing's disease: 85-95%
- Specificity: 95%
Mechanism: Pituitary adenomas retain some CRH responsiveness; ectopic sources do not.
Limitations:
- 5-15% of Cushing's disease may not respond
- Some bronchial carcinoids respond to CRH (10%)
- CRH not universally available
Inferior Petrosal Sinus Sampling (IPSS) - GOLD STANDARD
Indication: When pituitary vs. ectopic ACTH cannot be distinguished by non-invasive testing, or when imaging is discordant with biochemistry. [13]
Procedure:
- Interventional radiology procedure
- Bilateral catheterization of inferior petrosal sinuses (IPS) via femoral veins
- Simultaneous sampling from both IPS and peripheral vein
- Baseline samples at -5 and 0 min
- CRH (100 μg or 1 μg/kg) given IV at time 0
- Post-CRH samples at 3, 5, 10 minutes
Interpretation:
| Ratio | Baseline | Post-CRH (at 5 or 10 min) | Diagnosis |
|---|---|---|---|
| Central:Peripheral | ≥2.0 | ≥3.0 | Cushing's Disease (pituitary) |
| Central:Peripheral | less than 2.0 | less than 3.0 | Ectopic ACTH |
| IPS Right:Left (lateralization) | ≥1.4 | ≥1.4 | Suggests side of adenoma |
Performance:
- Sensitivity: 95-98%
- Specificity: 95-100%
- Most accurate test for distinguishing pituitary from ectopic ACTH
Complications (rare, 1-2%):
- Venous thrombosis
- Brainstem infarction (rare, less than 0.5%)
- Groin hematoma
- Cranial nerve VI palsy (transient)
Limitations:
- Invasive, requires expertise
- Lateralization less reliable than central-peripheral gradient (only 70% accuracy)
- False negatives rare but possible if catheter placement suboptimal
Alternatives Under Investigation:
- Prolactin-adjusted ratios to confirm catheter placement
- Desmopressin stimulation instead of CRH [13]
Ectopic ACTH Source Localization
If IPSS confirms ectopic source, localize the tumor:
Initial Imaging:
- CT Chest/Abdomen/Pelvis with contrast: Detect lung, pancreatic, thymic, adrenal masses
- Detects 60-70% of sources
Functional Imaging:
- 68-Ga-DOTATATE PET/CT (somatostatin receptor imaging): Excellent for bronchial carcinoids, pancreatic NETs, other NETs. Sensitivity 70-90% for occult NETs. [24]
- 18F-FDG PET/CT: Better for aggressive tumors (SCLC, medullary thyroid cancer)
- Older imaging: Octreotide scan (111-In-pentetreotide SPECT) - lower resolution than DOTATATE PET
Specialized Tests:
- Bronchoscopy: If lung lesion suspected
- Endoscopic ultrasound (EUS): For pancreatic NETs
- Neck ultrasound/CT: For thymic or thyroid tumors
Occult Sources: In 10-20%, source remains undetected despite extensive imaging. Options include empiric medical therapy or bilateral adrenalectomy.
Additional Specialized Tests
Desmopressin (DDAVP) Stimulation Test
- Alternative to CRH stimulation
- DDAVP 10 μg IV or subcutaneous
-
35% rise in ACTH or cortisol suggests Cushing's disease
- Useful in cyclic Cushing's
Liddle's Test (Historical)
- Now replaced by HDDST
- 2-day low-dose (2mg/day) followed by 2-day high-dose (8mg/day) dexamethasone
- Cumbersome, rarely used
7. Management
General Principles
- Primary Goal: Normalize cortisol levels, remove tumor source when possible, minimize morbidity
- First-line: Surgical resection for resectable disease (pituitary, adrenal)
- Adjunctive: Medical therapy as bridge to surgery, for inoperable disease, or persistent/recurrent disease
- Multidisciplinary: Endocrinology, neurosurgery, interventional radiology, medical oncology
Management Algorithm
CONFIRMED CUSHING'S SYNDROME
|
_________________|_________________
| |
ACTH-INDEPENDENT ACTH-DEPENDENT
(Low ACTH) (Normal/High ACTH)
| |
ADRENAL CT _________|_________
| | |
_______|_______ CUSHING'S DISEASE ECTOPIC ACTH
| | (Pituitary MRI, (CT C/A/P,
ADENOMA CARCINOMA IPSS if needed) DOTATATE PET)
| | | |
LAPAROSCOPIC OPEN SURGERY TRANSSPHENOIDAL TUMOR RESECTION
ADRENALECTOMY + MITOTANE SURGERY (TSS) (if resectable)
| | | |
_____|_____ _____|_____ ______|______ _____|_____
| | | | | | | |
CURE POST-OP CURE METASTATIC REMISSION PERSISTENT/ UNRESECTABLE/
ADRENAL CHEMO/XRT (65-90%) RECURRENT METASTATIC
INSUFFICIENCY + MITOTANE | |
_______|_____ ____|____
| | | |
REOPERATE MEDICAL BILATERAL
RADIOTHERAPY THERAPY ADRENALECTOMY
MEDICAL TX |
|
OSILODROSTAT, PASIREOTIDE,
KETOCONAZOLE, METYRAPONE,
MIFEPRISTONE, COMBINATION
A. Surgical Management
1. Cushing's Disease (Pituitary Adenoma) - Transsphenoidal Surgery (TSS)
Procedure: Endoscopic or microscopic transsphenoidal resection of ACTH-secreting pituitary adenoma
Outcomes:
- Microadenomas (less than 10mm): Remission in 70-90% (experienced centers)
- Macroadenomas (> 10mm): Remission in 40-65%
- Visible vs. non-visible on MRI: Similar outcomes if performed by expert surgeon
- Recurrence: 10-25% within 10 years, higher for macroadenomas [7,25]
Early Remission Criteria (within 1-2 weeks post-op):
- Morning cortisol less than 50-140 nmol/L (1.8-5 μg/dL) indicates remission/cure
- Patients develop temporary adrenal insufficiency requiring glucocorticoid replacement
- Undetectable or very low cortisol is best predictor of long-term cure
Complications:
- Diabetes insipidus: Transient in 20-40%, permanent in 1-5%
- Hyponatremia (SIADH): 5-10%
- Hypopituitarism: 5-20% (thyroid, gonadal axes)
- CSF leak: 3-5%
- Meningitis: less than 1%
- Carotid injury: less than 1%
- Visual deficits: less than 1%
Post-operative Management:
- Hydrocortisone replacement until adrenal recovery (6-36 months)
- Monitor for recurrence with UFC, LNSC every 6-12 months
- Assess other pituitary hormones
- Annual pituitary MRI for first 5 years
Repeat Surgery: If initial surgery fails and adenoma localized, repeat TSS has 50-70% success rate.
2. Adrenal Cushing's Syndrome
A. Unilateral Adrenal Adenoma - Laparoscopic Adrenalectomy
Procedure: Minimally invasive laparoscopic removal of affected adrenal gland
Outcomes:
- Cure rate: > 95% for benign adenomas
- Contralateral adrenal suppression: Present in all cases
Post-operative:
- Adrenal insufficiency: Universal, due to contralateral adrenal atrophy from chronic suppression
- Glucocorticoid replacement required: 6-36 months (average 12-18 months)
- Hydrocortisone 15-25 mg daily in divided doses
- Monitor with morning cortisol and ACTH stimulation testing
- Gradual weaning once ACTH rises and cortisol > 250 nmol/L on stimulation testing
Perioperative Glucocorticoid Coverage:
- Stress-dose hydrocortisone during and after surgery
- Avoid adrenal crisis
B. Adrenocortical Carcinoma (ACC) - Open Adrenalectomy
Procedure: Open radical adrenalectomy via anterior transabdominal or posterior retroperitoneal approach
Principles:
- Complete en-bloc resection with negative margins
- Avoid capsular rupture (worsens prognosis)
- Lymph node dissection if involved
- Resection of locally invaded structures (kidney, liver, IVC)
Outcomes:
- 5-year survival: 30-40% overall
- Stage-dependent: Stage I-II (60-70%), Stage III (35-50%), Stage IV (10-15%)
- High recurrence rate (70-80% at 5 years) [20]
Adjuvant Therapy:
- Mitotane (adrenolytic agent): Standard adjuvant therapy
- "Target level: 14-20 mg/L"
- Reduces recurrence, may improve survival
- "Toxicity: GI symptoms, hepatotoxicity, neurologic effects"
- Induces CYP3A4, necessitates higher glucocorticoid doses
Metastatic Disease:
- Mitotane + cytotoxic chemotherapy (EDP-M: etoposide, doxorubicin, cisplatin + mitotane)
- Response rates: 20-30%
- Consider metastasectomy if oligometastatic
3. Bilateral Adrenalectomy
Indications:
- Failed pituitary surgery (persistent or recurrent Cushing's disease)
- Severe, life-threatening hypercortisolism requiring urgent control
- Ectopic ACTH with occult or unresectable source
- BMAH with symptomatic hypercortisolism
Procedure: Laparoscopic bilateral total adrenalectomy
Outcomes:
- Immediate cure of hypercortisolism (100%)
- Permanent adrenal insufficiency requiring lifelong replacement
Complications:
- Nelson's Syndrome: Rapid growth of pituitary corticotroph tumor post-bilateral adrenalectomy due to loss of cortisol negative feedback. Occurs in 8-47% of cases (higher if no prior pituitary surgery/XRT). Characterized by extreme ACTH elevation, hyperpigmentation, pituitary macroadenoma with mass effects. Requires pituitary radiotherapy or surgery. [26]
- Adrenal crisis if non-compliant with replacement
- Lifelong glucocorticoid and mineralocorticoid replacement (hydrocortisone + fludrocortisone)
Prevention of Nelson's:
- Prophylactic pituitary radiotherapy reduces risk but controversial
- Close surveillance with MRI and ACTH monitoring
B. Medical Management
Medical therapy is used in the following scenarios:
- Bridge to surgery (severe hypercortisolism, preparing medically unstable patient)
- Persistent or recurrent disease after surgery
- Inoperable or metastatic disease
- Occult ectopic ACTH
- Patient unsuitable for or declines surgery
Drug Classes and Mechanisms
1. Steroidogenesis Inhibitors (Block Adrenal Cortisol Production)
| Drug | Mechanism | Efficacy | Adverse Effects | Notes |
|---|---|---|---|---|
| Osilodrostat | 11β-hydroxylase inhibitor | Normalization in 70-86% (LINC-3/4 trials) [27] | QTc prolongation, hypokalemia, adrenal insufficiency, hirsutism/acne (androgen accumulation) | Newest, most potent agent; FDA/EMA approved 2020; monitor ECG, K+ |
| Levoketoconazole | Multiple CYP450 inhibition (11β-hydroxylase, 17α-hydroxylase, cholesterol side-chain cleavage) | 30-50% normalization | Hepatotoxicity (black box warning), GI upset, hypogonadism | Purified 2S,4R enantiomer of ketoconazole; requires LFT monitoring; take with acidic beverage [28] |
| Ketoconazole | Same as levoketoconazole | 50-70% response | Hepatotoxicity (5-10%), GI upset, gynecomastia, drug interactions (CYP3A4 inhibitor) | Racemic mixture; off-label use; cheaper than levoketoconazole; LFTs monthly |
| Metyrapone | 11β-hydroxylase inhibitor | 50-75% response | Hirsutism, acne (androgen excess), hypertension, hypokalemia, edema, dizziness | Preferred in pregnancy; rapid onset; not available in all countries |
| Etomidate | 11β-hydroxylase, cholesterol side-chain cleavage | 75-100% (IV use) | Adrenal insufficiency, sedation, myoclonus | IV only; ICU use for severe/life-threatening CS; continuous infusion |
Clinical Use - Steroidogenesis Inhibitors:
- Start low, titrate to effect (normalization of UFC, LNSC)
- Monitor cortisol closely (risk of adrenal insufficiency)
- Combination therapy often needed
- Osilodrostat and metyrapone increase adrenal androgens (cause virilization in women)
2. Pituitary-Directed Agents (Reduce ACTH Secretion)
| Drug | Mechanism | Efficacy | Adverse Effects | Notes |
|---|---|---|---|---|
| Pasireotide | Somatostatin receptor agonist (SSTR5 > SSTR2) | 15-26% normalization; 45% partial response (PASPORT trial) [29] | Hyperglycemia (70-80%, often severe), GI upset, gallstones, bradycardia, QTc prolongation | SC injection bid or LAR IM monthly; only pituitary-directed drug approved for CD; monitor glucose closely |
| Cabergoline | Dopamine D2 receptor agonist | 25-40% response (limited data) | Nausea, orthostatic hypotension, valvular heart disease (rare at low doses) | Off-label; modest efficacy; may augment other therapies |
Clinical Use - Pasireotide:
- For persistent/recurrent Cushing's disease
- Requires aggressive diabetes management (GLP-1 agonists, SGLT2 inhibitors often needed)
- Consider stopping if severe hyperglycemia unmanageable
3. Glucocorticoid Receptor Antagonist
| Drug | Mechanism | Efficacy | Adverse Effects | Notes |
|---|---|---|---|---|
| Mifepristone | Competitive GR and PR antagonist | 60-87% clinical benefit (SEISMIC trial: diabetes/HTN improvement) [30] | Hypokalemia, vaginal bleeding (antiprogestin), endometrial thickening, adrenal insufficiency symptoms, nausea, fatigue | Does NOT lower cortisol (blocks receptor); cortisol/ACTH rise; monitor clinically, not biochemically; FDA approved for hyperglycemia in CS |
Clinical Use - Mifepristone:
- Unique mechanism: blocks cortisol effect, not production
- Cannot use cortisol to monitor (rises paradoxically)
- Monitor clinically: BP, glucose, K+, symptoms
- Watch for secondary adrenal insufficiency (cortisol rises but receptors blocked)
- Not first-line due to monitoring challenges
Combination Therapy
Often required for refractory cases:
- Metyrapone + ketoconazole
- Osilodrostat + cabergoline
- Pasireotide + ketoconazole
C. Radiotherapy
Indications:
- Adjuvant therapy after subtotal resection of pituitary adenoma
- Recurrent Cushing's disease after failed surgery
- Nelson's syndrome prevention (controversial) or treatment
Modalities:
| Type | Description | Efficacy | Latency | Complications |
|---|---|---|---|---|
| Stereotactic Radiosurgery (Gamma Knife, CyberKnife) | Single-fraction high-dose focused radiation | Remission in 50-80% at 5 years | 6-24 months | Hypopituitarism (20-50%), cranial nerve injury (rare), secondary malignancy (rare) |
| Fractionated Radiotherapy | Conventional external beam RT (45-50 Gy in 25-30 fractions) | Remission in 50-80% at 10 years | 12-36 months (slower) | Hypopituitarism (50-80%), rare optic chiasm injury |
Limitations:
- Slow response (months to years) - requires medical bridge therapy
- Risk of hypopituitarism increases over time
- Not suitable for macroadenomas with mass effect (surgery preferred)
D. Ectopic ACTH Syndrome Management
Approach:
- Tumor Resection: Curative if tumor is resectable (bronchial carcinoid, thymic carcinoid, pancreatic NET)
- Tumor-Directed Therapy: Chemotherapy for SCLC, somatostatin analogs for NETs, somatostatin receptor radionuclide therapy (PRRT with 177Lu-DOTATATE)
- Medical Control of Hypercortisolism: Steroidogenesis inhibitors (often high doses needed)
- Bilateral Adrenalectomy: If tumor unresectable or occult, and medical therapy insufficient
Severe/Life-Threatening Cases:
- ICU admission
- IV etomidate infusion for rapid cortisol control
- Treatment of severe hypokalemia, hyperglycemia, infections
- Early bilateral adrenalectomy if tumor cannot be localized
8. Complications
Acute and Perioperative Complications
1. Adrenal Crisis (Post-operative Adrenal Insufficiency)
Mechanism: Following cure of CS (surgical or medical), the HPA axis is suppressed. Endogenous cortisol production is inadequate, leading to risk of life-threatening adrenal insufficiency.
Timeline:
- After adrenalectomy: Immediate and permanent (if bilateral) or temporary (if unilateral, 6-36 months)
- After TSS: Universal in cured patients, recovery 6-36 months (median 12-18 months)
Clinical Features:
- Fatigue, weakness, nausea, vomiting, abdominal pain
- Hypotension, hyponatremia, hyperkalemia
- Hypoglycemia (less common in adults)
Prevention and Management:
- Post-operative glucocorticoid replacement mandatory (hydrocortisone 15-25 mg daily in divided doses)
- Stress-dose steroids for illness, surgery, trauma (100-300 mg/day)
- Patient education on adrenal crisis recognition and emergency injection
- Gradual weaning with monitoring (morning cortisol, ACTH stimulation test)
2. Nelson's Syndrome
Definition: Rapid growth of pituitary corticotroph tumor following bilateral adrenalectomy, due to loss of cortisol negative feedback on ACTH secretion. [26]
Incidence: 8-47% (higher without prior pituitary surgery or radiotherapy)
Clinical Features:
- Extreme ACTH elevation (often > 220 pmol/L or > 1000 pg/mL)
- Severe hyperpigmentation (skin, mucosal surfaces) - MSH effect
- Pituitary macroadenoma on MRI with mass effects: headaches, visual field defects, cranial nerve palsies
Prevention:
- Prophylactic pituitary radiotherapy after bilateral adrenalectomy (controversial, not universally recommended)
- Close surveillance if no prophylaxis: annual MRI, ACTH monitoring
Treatment:
- Pituitary surgery (TSS)
- Pituitary radiotherapy
- Temozolomide for aggressive adenomas
3. Tumor Progression/Recurrence
Recurrence Rates:
- After TSS for Cushing's disease: 10-25% at 10 years, higher for macroadenomas
- After adrenalectomy for adenoma: less than 5%
- After ACC resection: 70-80% at 5 years
Surveillance:
- Post-TSS: UFC or LNSC every 6-12 months, annual pituitary MRI for 5 years
- Post-adrenalectomy for ACC: CT C/A/P every 3-6 months for 5 years, then annually
Chronic Systemic Complications
1. Cardiovascular Disease
Morbidity:
- Hypertension: 70-85%, may persist after cure
- Left ventricular hypertrophy: 40-50%
- Coronary artery disease: 2-5 fold increased risk
- Heart failure: 15-20% in active disease
- Thromboembolic disease: 2-4 fold increased risk (VTE, stroke)
Mortality:
- Cardiovascular disease is the leading cause of death in Cushing's syndrome
- Standardized mortality ratio (SMR) 3.7-4.8 in active disease [10,23]
- Even after biochemical cure, cardiovascular risk remains elevated for years
- Early treatment critical to reduce long-term CV mortality
Management:
- Aggressive BP control
- Lipid management (statins)
- VTE prophylaxis in hospitalized patients
- Antiplatelet therapy if indicated
2. Metabolic Complications
Diabetes Mellitus and Glucose Intolerance:
- Present in 40-45% of CS patients
- Reverses or improves in 50-70% after cure, but many retain impaired glucose tolerance
- Requires ongoing monitoring and treatment
Dyslipidemia:
- Elevated triglycerides, LDL cholesterol; reduced HDL
- Increases cardiovascular risk
- Improves but may not normalize after cure
Obesity:
- Central adiposity may persist after cure despite normalization of cortisol
- Weight loss difficult; lifestyle intervention and medical management needed
3. Bone Disease
Osteoporosis:
- Present in 50-80% of patients with active CS
- Rapid bone loss (10-20% per year during active disease)
- Vertebral compression fractures in 30-67%, often asymptomatic [22]
- Avascular necrosis of femoral head: 5-10%
Post-Cure:
- Bone density improves slowly after cure (gains of 5-10% over 2-3 years)
- Fracture risk remains elevated for years
- Vertebral fractures do not heal
Management:
- Baseline DEXA scan
- Calcium (1200-1500 mg/day) and vitamin D (800-1000 IU/day) supplementation
- Bisphosphonates or denosumab for established osteoporosis or high fracture risk
- Vertebroplasty/kyphoplasty for symptomatic vertebral fractures
4. Infectious Complications
Mechanism: Severe immunosuppression from chronic hypercortisolism
Types:
- Opportunistic infections: Pneumocystis jirovecii pneumonia (PJP), cryptococcal meningitis, aspergillosis, strongyloidiasis, nocardiosis
- Typical bacterial infections: Pneumonia, UTI, skin/soft tissue infections
- Fungal skin infections: Candida, dermatophytes
Risk Factors for Opportunistic Infections:
- Severe hypercortisolism (cortisol > 1000 nmol/L)
- Ectopic ACTH syndrome
- Lymphopenia (less than 500/μL)
Prevention:
- Consider PJP prophylaxis (trimethoprim-sulfamethoxazole) in severe CS
- Strongyloides screening if endemic exposure
- Prompt treatment of infections
Mortality: Infections account for 10-20% of deaths in active CS
5. Neuropsychiatric Complications
Cognitive Impairment:
- Memory deficits, impaired attention, executive dysfunction
- Hippocampal atrophy on MRI
- Partially reversible after cure but residual deficits common
Psychiatric Disorders:
- Depression: 50-70%
- Anxiety: 50-65%
- Psychosis: 5-10%
- Suicidal ideation: 5-15%
- May improve after cure but psychiatric treatment often needed
Quality of Life:
- Remains impaired even after biochemical remission in many patients
- Fatigue, cognitive issues, mood disturbances persist
6. Reproductive Dysfunction
Women:
- Menstrual irregularities, infertility
- Improves after cure but may require hormonal therapy
Men:
- Hypogonadism, erectile dysfunction, infertility
- Testosterone may remain low after cure; replacement often needed
9. Prognosis
Overall Mortality
Active Disease:
- Standardized Mortality Ratio (SMR): 3.7-4.8 compared to general population [10,23]
- 5-year survival: 50-70% (untreated or poorly controlled)
- Leading causes of death: Cardiovascular disease (40-50%), infections (20-30%), complications of diabetes
After Cure:
- SMR improves but remains elevated: 1.2-2.2
- Excess mortality persists for years due to:
- Residual cardiovascular disease
- Osteoporosis and fractures
- Persistent metabolic dysfunction
- Cognitive and psychiatric sequelae
Prognosis by Etiology
Cushing's Disease (Pituitary)
Surgical Outcomes:
- Remission after first TSS: 70-90% (microadenomas), 40-65% (macroadenomas)
- Recurrence: 10-25% at 10 years
- Cure defined as: Morning cortisol less than 140 nmol/L within 7-10 days post-op
Long-term Survival (if remission achieved):
- 10-year survival: 85-93% (approaches general population)
- Persistent morbidity: Hypertension, diabetes, osteoporosis, cognitive issues
Persistent/Recurrent Disease:
- Second TSS: 50-70% remission
- Radiotherapy: 50-80% remission at 5-10 years (delayed response)
- Medical therapy: Long-term control in 50-75%
- Bilateral adrenalectomy: 100% biochemical cure, but Nelson's risk
Adrenal Adenoma
Outcomes:
- Cure rate: > 95% with adrenalectomy
- Near-normal life expectancy after cure and adrenal recovery
- Cardiovascular and metabolic comorbidities may persist, requiring ongoing management
Adrenocortical Carcinoma
Survival (stage-dependent): [20]
- Stage I (T1N0M0): 60-80% 5-year survival
- Stage II (T2N0M0): 50-70%
- Stage III (T3-4 or N+): 35-50%
- Stage IV (M1): 10-15%
Recurrence: 70-85% at 5 years despite complete resection
Prognostic Factors:
- Complete resection (R0): Most important
- Stage at diagnosis
- Ki-67 proliferation index (> 10% poor prognosis)
- Mitotane compliance and therapeutic levels
Ectopic ACTH Syndrome
Prognosis depends on primary tumor:
-
Bronchial Carcinoid (well-differentiated NET):
- 5-year survival: 70-90% if resectable
- Excellent prognosis after complete resection
-
Small Cell Lung Cancer:
- 5-year survival: less than 5%
- "Median survival: 6-12 months"
- Ectopic ACTH indicates advanced disease
-
Pancreatic NET:
- Variable prognosis (30-70% 5-year survival depending on grade)
-
Occult Ectopic:
- "If tumor never found: Managed with bilateral adrenalectomy or medical therapy"
- Prognosis depends on underlying tumor (if it eventually declares itself)
Subclinical Cushing's Syndrome
Natural History:
- 10-20% progress to overt CS over 5 years
- Increased cardiovascular and metabolic morbidity compared to non-functioning adenomas
- Adrenalectomy controversial; individualized based on comorbidities
Post-Adrenalectomy:
- Improvement in hypertension (50-70%), diabetes (30-60%), bone density
- Quality of life improvement
Quality of Life After Cure
Persistent Issues:
- Chronic fatigue: 30-50%
- Cognitive impairment: 40-60%
- Depression/anxiety: 30-50%
- Obesity: 40-60%
- Hypertension: 30-50% (despite cure)
Improvement Timeline:
- Skin thinning, bruising: Improve within 3-6 months
- Proximal myopathy: Improves over 6-12 months (physical therapy beneficial)
- Hypertension: May improve over 1-2 years; many require ongoing treatment
- Bone density: Slow improvement over 2-5 years
- Cognitive/psychiatric: Variable; may never fully normalize
Factors Associated with Better QoL:
- Younger age at diagnosis and cure
- Shorter duration of active disease
- Complete biochemical remission
- Absence of recurrence
- Multidisciplinary follow-up care
10. Special Populations
Pregnancy and Cushing's Syndrome
Epidemiology: Extremely rare (1 in 1 million pregnancies) [16]
Etiology: Different distribution compared to non-pregnant women:
- Adrenal adenoma: 40-50% (higher than usual)
- Cushing's disease: 35-40%
- Adrenal carcinoma: 10%
- Ectopic ACTH: Rare
Diagnosis Challenges:
- Pregnancy increases CBG, raising total cortisol (normal in pregnancy)
- UFC increases 2-3 fold in normal pregnancy
- Loss of circadian rhythm in normal late pregnancy
- ONDST less reliable (estrogen-induced CBG elevation)
Diagnostic Approach:
- Use pregnancy-specific cutoffs for UFC (> 3x normal pregnancy values)
- LNSC may be most reliable
- ONDST cutoff > 50 nmol/L still applicable but less specific
- Low-dose DST (2-day) may be needed
Complications:
- Maternal: Hypertension (70%), preeclampsia (15%), diabetes (30%), heart failure, osteoporotic fractures, infections
- Fetal: Intrauterine growth restriction (40%), preterm delivery (60%), stillbirth (5-10%), adrenal insufficiency (rare, from transplacental passage of high cortisol)
- Maternal mortality: 2-5%
- Fetal mortality/morbidity: 30-40%
Management:
- First trimester: Consider surgical resection (safest in 2nd trimester)
- Second trimester: Transsphenoidal surgery or adrenalectomy if feasible (laparoscopic safest approach)
- Third trimester: Medical therapy to control hypercortisolism until delivery
- "Metyrapone: Preferred (does not cross placenta significantly, safe in pregnancy)"
- "Ketoconazole: Avoid (antiandrogen effects, teratogenic in animals)"
- "Osilodrostat, pasireotide, mifepristone: No safety data, avoid"
- Delivery: Early delivery (36-38 weeks) often necessary
- Post-partum: Definitive surgical treatment
Pediatric Cushing's Syndrome
Epidemiology: Rare; annual incidence 1-2 per million children
Etiology (different from adults):
- ACTH-independent (adrenal) causes more common (60%):
- "Adrenal adenoma: 30%"
- "Adrenal carcinoma: 15% (worse prognosis)"
- "BMAH, PPNAD (Carney Complex): 15%"
- ACTH-dependent: 40%
- "Cushing's disease: 30%"
- "Ectopic ACTH: Rare (less than 5%)"
Genetic Syndromes (higher proportion in children):
- McCune-Albright syndrome (GNAS mutations)
- Carney Complex (PRKAR1A mutations)
- Li-Fraumeni syndrome (TP53 mutations - ACC)
- MEN1 (MEN1 mutations)
- MEN4 (CDKN1B mutations)
Presentation:
- Growth arrest or deceleration: Most specific feature (90-95%)
- Weight gain with height deceleration (crossing percentiles downward)
- Delayed puberty
- Similar features to adults: Obesity, striae, acne, hirsutism, weakness
Diagnosis:
- Same screening tests as adults (UFC, LNSC, ONDST) with pediatric reference ranges
- Growth velocity monitoring essential
Management:
- Surgery first-line when possible
- Pediatric endocrine and neurosurgical expertise required
- Catch-up growth occurs after cure if pre-pubertal
Subclinical Cushing's Syndrome (Mild Autonomous Cortisol Excess)
Definition: Autonomous cortisol secretion without overt cushingoid features, typically discovered during evaluation of adrenal incidentalomas. [9,17]
Prevalence: 5-20% of adrenal incidentalomas
Diagnostic Criteria (variable definitions, not universally agreed):
- Post-1mg DST cortisol > 50-140 nmol/L (1.8-5 μg/dL) without cushingoid phenotype
- +/- Suppressed ACTH
- +/- Elevated UFC or LNSC (but often normal)
Clinical Significance:
- Higher prevalence of hypertension (60-80%), diabetes (25-50%), obesity, osteoporosis, dyslipidemia compared to non-functioning adenomas
- Increased cardiovascular events
- Impaired quality of life
Management (controversial):
- Conservative: Observation with annual screening if no comorbidities
- Adrenalectomy: Consider if:
- Worsening hypertension, diabetes, osteoporosis
- Young age (less than 50-60 years)
- Patient preference after counseling
- Outcomes after adrenalectomy: Improvement in hypertension (50-70%), diabetes (30-60%), bone density, QoL
Cyclic Cushing's Syndrome
Definition: Periodic hypercortisolism with alternating periods of normal or low cortisol production. [12]
Prevalence: 15% of all CS cases (likely underdiagnosed)
Cycle Characteristics:
- Cycle length: Variable (days to months)
- Can occur with any etiology (pituitary, adrenal, ectopic)
- Periods of hypercortisolism may last days to weeks, followed by eucortisolism or even hypocortisolism
Diagnosis:
- High index of suspicion if history of fluctuating symptoms
- Requires serial measurements over weeks to months:
- Daily or every-other-day LNSC for 2-4 weeks
- Multiple 24h UFC collections
- Serial ONDST
- May require prolonged observation or hospitalization
Management:
- Same as non-cyclic CS once diagnosis confirmed
- Timing of surgery ideally during hypercortisolemic phase (if waiting for diagnosis confirmation)
11. Follow-up and Long-term Care
Post-Cure Surveillance
Biochemical Monitoring:
- First year: UFC or LNSC every 3-6 months
- After first year: Every 6-12 months indefinitely
- Monitor for recurrence (especially after TSS for Cushing's disease)
Imaging:
- Post-TSS: Pituitary MRI at 3-6 months, then annually for 5-10 years
- Post-ACC resection: CT chest/abdomen/pelvis every 3-6 months for 5 years, then annually
Endocrine Assessment:
- Morning cortisol and ACTH stimulation test to assess adrenal recovery (every 3-6 months until recovery)
- Assess other pituitary axes if post-TSS (TSH, LH/FSH, prolactin, IGF-1)
- Monitor for hypopituitarism after radiotherapy (annual)
Comorbidity Management
Cardiovascular:
- Annual BP monitoring
- Lipid panel annually
- ECG, echocardiogram if indicated
- Aggressive risk factor modification
Metabolic:
- HbA1c or OGTT annually
- Diabetes management if persistent
Bone Health:
- DEXA scan at baseline and every 1-2 years
- Calcium, vitamin D supplementation
- Bisphosphonates or denosumab if osteoporosis or fragility fractures
Psychiatric and Cognitive:
- Screen for depression, anxiety
- Cognitive assessment if impairment suspected
- Psychiatric referral and treatment as needed
Patient Education
Adrenal Insufficiency:
- Symptoms recognition
- Sick-day rules (stress dosing)
- Emergency hydrocortisone injection training
- Medical alert bracelet
Recurrence Warning Signs:
- Return of cushingoid features
- Worsening hypertension, diabetes
- Prompt medical evaluation if symptoms recur
12. Practice Single Best Answer (SBA) Questions
Question 1
A 42-year-old woman presents with 18 months of progressive central weight gain, facial fullness, and easy bruising. Which clinical feature has the highest specificity for Cushing's syndrome?
A) Buffalo hump B) Moon facies C) Purple striae > 1 cm wide D) Central obesity E) Acne
Answer: C. Purple striae > 1 cm wide have the highest specificity (95-100%) for Cushing's syndrome among the listed features. Buffalo hump, moon facies, central obesity, and acne are common in simple obesity and metabolic syndrome, reducing their diagnostic specificity. Wide, violaceous striae reflect dermal atrophy and are highly discriminating for pathologic hypercortisolism. [8]
Question 2
A 45-year-old man with confirmed Cushing's syndrome has a plasma ACTH level of 3 pg/mL (reference 10-60 pg/mL) on two separate measurements. What is the most appropriate next investigation?
A) Pituitary MRI B) Inferior petrosal sinus sampling (IPSS) C) Adrenal CT scan D) High-dose dexamethasone suppression test E) Chest CT scan
Answer: C. Low/suppressed ACTH (less than 5 pg/mL) indicates ACTH-independent Cushing's syndrome, localizing the source to the adrenal glands. Adrenal CT is the appropriate next step to identify adenoma, carcinoma, or bilateral hyperplasia. Pituitary MRI and IPSS are used for ACTH-dependent disease. HDDST is used to differentiate pituitary from ectopic sources in ACTH-dependent cases. [2]
Question 3
Which genetic mutation is most commonly associated with cortisol-secreting pituitary adenomas in Cushing's disease?
A) PRKACA B) USP8 C) GNAS D) MEN1 E) TP53
Answer: B. Somatic mutations in USP8 (ubiquitin-specific protease 8) are found in approximately 35-40% of corticotroph adenomas causing Cushing's disease. These mutations lead to sustained EGFR signaling and increased ACTH production. PRKACA mutations are associated with adrenal adenomas, not pituitary. [18]
Question 4
A 38-year-old woman fails the 1-mg overnight dexamethasone suppression test with a cortisol of 140 nmol/L (normal less than 50 nmol/L). Late-night salivary cortisol measurements are normal on three separate occasions. What is the most likely diagnosis?
A) Cushing's disease B) Adrenal adenoma C) Pseudo-Cushing's syndrome or false-positive ONDST D) Ectopic ACTH syndrome E) Subclinical Cushing's syndrome
Answer: C. Discordant screening test results (positive ONDST but normal LNSC) suggest either Pseudo-Cushing's state or a false-positive dexamethasone test. Common causes of false-positive ONDST include estrogen use (OCP/HRT increasing CBG), CYP3A4-inducing medications, or physiologic stress. True Cushing's syndrome typically produces concordant positive results on multiple screening tests. [2]
Question 5
A 50-year-old woman with Cushing's disease is being treated with pasireotide. Which medication will she most likely require as a consequence of this treatment?
A) Proton pump inhibitor B) Potassium supplement C) Insulin or other diabetes medications D) Calcium channel blocker E) Fludrocortisone
Answer: C. Pasireotide, a somatostatin analog binding SSTR5, causes hyperglycemia in 70-80% of patients, often requiring initiation or intensification of diabetes medications (insulin, GLP-1 agonists, SGLT2 inhibitors). This occurs via inhibition of insulin secretion from pancreatic beta cells. This is the most common and clinically significant adverse effect requiring additional medication. [29]
Question 6
What is the gold standard investigation for distinguishing Cushing's disease (pituitary source) from ectopic ACTH syndrome?
A) High-dose dexamethasone suppression test B) CRH stimulation test C) Bilateral inferior petrosal sinus sampling (BIPSS) D) 68-Ga-DOTATATE PET/CT E) Pituitary MRI
Answer: C. Bilateral inferior petrosal sinus sampling (BIPSS) with CRH stimulation is the gold standard for differentiating pituitary from ectopic ACTH sources. A central-to-peripheral ACTH gradient ≥2 at baseline or ≥3 post-CRH indicates Cushing's disease with 95-100% accuracy. HDDST and CRH tests have lower sensitivity/specificity. Imaging (MRI, PET) is complementary but not definitive. [13]
Question 7
A 35-year-old woman is taking the oral contraceptive pill. Which Cushing's syndrome screening test is most likely to produce a false-positive result?
A) 24-hour urinary free cortisol B) Late-night salivary cortisol C) 1-mg overnight dexamethasone suppression test D) Morning cortisol measurement E) Plasma ACTH
Answer: C. Estrogens (from OCP or HRT) increase cortisol-binding globulin (CBG), raising total serum cortisol levels. This causes elevated morning cortisol post-dexamethasone (false-positive ONDST), even though free cortisol is appropriately suppressed. UFC and LNSC measure free cortisol and are not significantly affected by CBG changes. [2]
Question 8
Which randomized controlled trial established the efficacy of osilodrostat in Cushing's syndrome?
A) SEISMIC B) LINC-3 C) PASPORT D) CRONOS E) PROMPT
Answer: B. The LINC-3 trial (and its extension LINC-4) were pivotal Phase III studies demonstrating that osilodrostat, a potent 11β-hydroxylase inhibitor, normalized urinary free cortisol in 70-86% of patients with Cushing's syndrome. This led to FDA and EMA approval in 2020. [27]
Question 9
A 62-year-old man presents with 2 months of rapid weight loss, profound muscle weakness, severe hypokalemia (K+ 2.2 mmol/L), and metabolic alkalosis. Cushingoid features are minimal. What is the most likely etiology?
A) Pituitary microadenoma B) Adrenal adenoma C) Ectopic ACTH syndrome (e.g., small cell lung cancer) D) Exogenous glucocorticoid use E) Pituitary macroadenoma
Answer: C. The rapid onset (less than 6 months), weight loss (instead of gain), severe hypokalemia, and minimal cushingoid habitus are classic for ectopic ACTH syndrome, often from small cell lung cancer or other aggressive neuroendocrine tumors. The severe hypokalemia results from cortisol saturating 11β-HSD2, causing mineralocorticoid effects. Pituitary sources typically have gradual onset and classic cushingoid features. [6]
Question 10
What characterizes Nelson's syndrome?
A) Adrenal insufficiency after bilateral adrenalectomy B) Rapid pituitary corticotroph tumor growth after bilateral adrenalectomy C) Severe hypokalemia in ectopic ACTH syndrome D) Osteoporotic fractures in young men with Cushing's E) Psychosis in severe Cushing's syndrome
Answer: B. Nelson's syndrome is defined by rapid growth of a pituitary corticotroph tumor (often into macroadenoma) following bilateral adrenalectomy, due to loss of cortisol negative feedback. It is characterized by extremely elevated ACTH levels and severe hyperpigmentation. Occurs in 8-47% of patients post-bilateral adrenalectomy, more common without prior pituitary surgery or radiotherapy. [26]
Question 11
A 48-year-old woman undergoes successful transsphenoidal surgery for Cushing's disease. On postoperative day 7, her morning cortisol is 30 nmol/L. What does this indicate?
A) Treatment failure, persistent disease B) Excellent predictor of long-term cure C) Adrenal insufficiency requiring emergency treatment D) Need for immediate reoperation E) Normal postoperative finding, no intervention needed
Answer: B. A morning cortisol less than 50-140 nmol/L within 7-10 days post-TSS indicates biochemical remission and is the best predictor of long-term cure. The low cortisol reflects suppression of the normal corticotrophs by chronic hypercortisolism, with subsequent adrenal atrophy. The patient will require glucocorticoid replacement until HPA axis recovery (6-36 months). This is a desired outcome, not a complication. [7]
Question 12
Which mechanism explains hypokalemia in severe Cushing's syndrome, particularly ectopic ACTH?
A) Direct renal potassium wasting by ACTH B) Cortisol saturation of 11β-HSD2, allowing mineralocorticoid receptor activation C) Hyperaldosteronism from adrenal hyperplasia D) Cortisol-induced metabolic acidosis E) Secondary hyperparathyroidism
Answer: B. At high cortisol levels (especially in ectopic ACTH), the protective enzyme 11β-hydroxysteroid dehydrogenase type 2 (11β-HSD2) becomes saturated and cannot fully convert cortisol to inactive cortisone in renal tubular cells. Unconverted cortisol binds mineralocorticoid receptors (MR), mimicking aldosterone and causing sodium retention and potassium wasting. This leads to hypokalemia and metabolic alkalosis. [9]
Question 13
A 55-year-old man with metastatic small cell lung cancer and severe ectopic ACTH syndrome is admitted to the ICU with cortisol 1200 nmol/L, K+ 2.0 mmol/L, and glucose 24 mmol/L. Oral ketoconazole has failed. What is the most appropriate acute medical therapy?
A) Pasireotide subcutaneous injection B) Mifepristone oral C) Etomidate IV infusion D) Metyrapone oral E) Osilodrostat oral
Answer: C. In life-threatening severe Cushing's syndrome requiring ICU care, IV etomidate infusion provides the most rapid cortisol reduction (within hours). Etomidate is a potent inhibitor of 11β-hydroxylase and cholesterol side-chain cleavage enzyme. All other options are oral with slower onset. This patient requires urgent cortisol control for severe hyperglycemia and hypokalemia. Etomidate is the only IV steroidogenesis inhibitor available. [3]
Question 14
A 32-year-old woman is found to have an incidental 3.5 cm right adrenal adenoma on CT. She has hypertension and type 2 diabetes. Post-1mg dexamethasone cortisol is 95 nmol/L. ACTH is 2 pg/mL. She has no cushingoid features. What is the diagnosis?
A) Cushing's syndrome B) Subclinical Cushing's syndrome (mild autonomous cortisol excess) C) Pheochromocytoma D) Primary aldosteronism E) Non-functioning adrenal adenoma
Answer: B. This patient has subclinical Cushing's syndrome (also called mild autonomous cortisol excess): autonomous cortisol secretion demonstrated by failed dexamethasone suppression (> 50 nmol/L) and suppressed ACTH, but without overt cushingoid phenotype. The hypertension and diabetes may be related. Adrenalectomy should be considered given her comorbidities and relatively young age. [9,17]
Question 15
What is the most common cause of death in patients with active, untreated Cushing's syndrome?
A) Opportunistic infections B) Cardiovascular disease C) Adrenal crisis D) Suicide E) Thromboembolic disease
Answer: B. Cardiovascular disease (myocardial infarction, stroke, heart failure) is the leading cause of death in active Cushing's syndrome, accounting for 40-50% of mortality. The standardized mortality ratio is 3.7-4.8, driven primarily by cardiovascular complications from hypertension, diabetes, dyslipidemia, and accelerated atherosclerosis. Infections account for 20-30% of deaths. [10,23]
13. Viva Voce and OSCE Scenarios
Common Viva Questions
Q1: "How do you distinguish Cushing's syndrome from simple obesity in the outpatient clinic?"
Model Answer: "I would focus on identifying catabolic features that are specific to hypercortisolism and rare in simple obesity. The four most discriminating features are: proximal myopathy (difficulty rising from a chair without using arms), easy bruising with spontaneous ecchymoses, purple striae wider than 1 cm, and facial plethora. I would also inquire about rate of weight gain—Cushing's typically has progressive weight gain over months to 1-2 years, whereas simple obesity is often longstanding. Additionally, I would look for thin skin, poor wound healing, and ask about hypertension or diabetes of recent onset. If these catabolic features are present, I would proceed with biochemical screening tests."
Q2: "Explain the physiological basis of the overnight dexamethasone suppression test."
Model Answer: "The overnight dexamethasone suppression test exploits the normal negative feedback regulation of the HPA axis. In healthy individuals, exogenous dexamethasone—a potent synthetic glucocorticoid—binds to glucocorticoid receptors in the hypothalamus and pituitary, suppressing CRH and ACTH secretion. This leads to reduced adrenal cortisol production. In the test, 1 mg of dexamethasone is given at 11 PM, and cortisol is measured at 8-9 AM the next morning. Normal suppression (cortisol less than 50 nmol/L) excludes Cushing's syndrome. In Cushing's, autonomous cortisol production—whether from a pituitary adenoma, adrenal tumor, or ectopic source—fails to suppress appropriately, resulting in cortisol remaining elevated. False positives can occur with estrogen use, which increases cortisol-binding globulin, or with medications that accelerate dexamethasone metabolism via CYP3A4 induction."
Q3: "Why does ectopic ACTH syndrome cause severe hypokalemia?"
Model Answer: "Severe hypokalemia in ectopic ACTH syndrome results from the mineralocorticoid effects of cortisol. Normally, the enzyme 11-beta-hydroxysteroid dehydrogenase type 2 in renal tubular cells converts cortisol to inactive cortisone, protecting the mineralocorticoid receptor from cortisol activation. However, in ectopic ACTH syndrome, cortisol levels are extremely high—often exceeding 1000 nmol/L—and this saturates the protective enzyme. Unconverted cortisol then binds to mineralocorticoid receptors, mimicking aldosterone's effects: sodium retention, volume expansion, and potassium wasting. This leads to severe hypokalemia, often below 2.5 mmol/L, along with metabolic alkalosis and hypertension. This mineralocorticoid effect is less pronounced in Cushing's disease due to lower cortisol levels."
Q4: "What are the indications for medical therapy in Cushing's syndrome?"
Model Answer: "Medical therapy in Cushing's syndrome is indicated in several scenarios. First, as a bridge to surgery in patients with severe hypercortisolism who are medically unstable—to reduce perioperative risk by controlling hypertension, hyperglycemia, and hypokalemia. Second, in patients with persistent or recurrent disease after failed surgery, such as incomplete resection of a pituitary adenoma. Third, in patients with inoperable disease, such as metastatic adrenocortical carcinoma or unresectable ectopic ACTH sources. Fourth, when the source is occult and cannot be localized despite extensive imaging. Finally, in patients who are poor surgical candidates due to comorbidities or who decline surgery. The choice of agent depends on the etiology: steroidogenesis inhibitors like osilodrostat or ketoconazole block adrenal cortisol production and are effective for all types, whereas pituitary-directed agents like pasireotide are specific for Cushing's disease."
Q5: "Describe your diagnostic approach to a patient with suspected Cushing's syndrome."
Model Answer: "My approach is systematic and follows three steps: confirm hypercortisolism, determine ACTH-dependence, and localize the source. First, I would confirm hypercortisolism using at least two different first-line screening tests: a 1-mg overnight dexamethasone suppression test, 24-hour urinary free cortisol, and late-night salivary cortisol. I require two positive tests to reduce false positives from Pseudo-Cushing's states. If hypercortisolism is confirmed, I would measure plasma ACTH on at least two occasions. A suppressed ACTH below 5 pg/mL indicates an adrenal source, prompting adrenal CT imaging. An elevated or normal ACTH above 20 pg/mL indicates ACTH-dependent disease, leading to pituitary MRI and potentially high-dose dexamethasone suppression or CRH stimulation tests. If non-invasive tests are discordant or the pituitary MRI is negative, I would proceed to bilateral inferior petrosal sinus sampling, the gold standard for distinguishing pituitary from ectopic sources. Once the source is localized, I would refer for appropriate definitive therapy—transsphenoidal surgery for pituitary, adrenalectomy for adrenal, or tumor resection for ectopic sources."
OSCE Scenario: Breaking Bad News - Cushing's Disease Recurrence
Scenario: You are an endocrinology registrar. Mrs. Thompson is a 42-year-old woman who underwent transsphenoidal surgery for Cushing's disease 3 years ago with initial remission. Her recent surveillance urinary free cortisol is elevated at 320 μg/24h (normal less than 50), and repeat pituitary MRI shows a new 7mm adenoma. You must explain the diagnosis of recurrent disease and discuss management options.
Key Communication Points:
- Establish rapport and check understanding of previous diagnosis and treatment
- Deliver news clearly: "Unfortunately, the tests show that your Cushing's disease has returned"
- Explain what this means: Elevated cortisol, return of symptoms, need for treatment
- Discuss management options:
- Repeat pituitary surgery (50-70% success rate)
- Pituitary radiotherapy (slower, takes months-years)
- Medical therapy (osilodrostat, pasireotide)
- Bilateral adrenalectomy (curative but requires lifelong hormone replacement)
- Address emotions and concerns (disappointment, anxiety about repeat surgery)
- Offer multidisciplinary team input (neurosurgery, endocrinology)
- Provide written information and follow-up plan
14. References
-
Nieman LK, Biller BM, Findling JW, et al. The diagnosis of Cushing's syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2008;93(5):1526-1540. PMID: 18334580
-
Nieman LK, Biller BM, Findling JW, et al. Treatment of Cushing's syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2015;100(8):2807-2831. PMID: 26222757
-
Pivonello R, De Leo M, Vitale P, et al. Pathophysiology of diabetes mellitus in Cushing's syndrome. Neuroendocrinology. 2010;92 Suppl 1:77-81. PMID: 20829622
-
Fleseriu M, Auchus R, Bancos I, et al. Consensus on diagnosis and management of Cushing's disease: a guideline update. Lancet Diabetes Endocrinol. 2021;9(12):847-875. PMID: 34626543
-
Reincke M. Cushing syndrome: a review. JAMA. 2023;330(2):170-181. PMID: 37432427
-
Isidori AM, Kaltsas GA, Pozza C, et al. The ectopic adrenocorticotropin syndrome: clinical features, diagnosis, management, and long-term follow-up. J Clin Endocrinol Metab. 2006;91(2):371-377. PMID: 16303835
-
Lonser RR, Nieman L, Oldfield EH. Cushing's disease: pathobiology, diagnosis, and management. J Neurosurg. 2017;126(2):404-417. PMID: 27104844
-
Newell-Price J, Bertagna X, Grossman AB, Nieman LK. Cushing's syndrome. Lancet. 2006;367(9522):1605-1617. PMID: 16698415
-
Starker LF, Kunstman JW, Carling T. Subclinical Cushing syndrome: a review. Surg Clin North Am. 2014;94(3):657-668. PMID: 24857582
-
Clayton RN, Raskauskiene D, Reulen RC, Jones PW. Mortality and morbidity in Cushing's disease over 50 years in Stoke-on-Trent, UK: audit and meta-analysis of literature. J Clin Endocrinol Metab. 2011;96(3):632-642. PMID: 21193542
-
Yanovski JA, Cutler GB Jr, Chrousos GP, Nieman LK. Corticotropin-releasing hormone stimulation following low-dose dexamethasone administration. A new test to distinguish Cushing's syndrome from pseudo-Cushing's states. JAMA. 1993;269(17):2232-2238. PMID: 8386285
-
Nowak KM, Maiter D, Santhakumar K, et al. Diagnostic challenges in cyclic Cushing's syndrome: a systematic review. Lancet Diabetes Endocrinol. 2023;11(9):666-675. PMID: 37429301
-
Lyu J, Li C, Yan H, et al. Should we use prolactin adjustment in bilateral inferior petrosal sinus sampling to diagnose Cushing disease? A joint meta-analysis of head-to-head diagnostic tests accuracy studies. Endocr Pract. 2023;29(3):169-176. PMID: 36649781
-
Etxabe J, Vazquez JA. Morbidity and mortality in Cushing's disease: an epidemiological approach. Clin Endocrinol (Oxf). 1994;40(4):479-484. PMID: 8187313
-
Stratakis CA. Cushing syndrome in pediatrics. Endocrinol Metab Clin North Am. 2012;41(4):793-803. PMID: 23099271
-
Hamblin R, Vardon A, Akturk M, et al. The diagnosis and management of Cushing's syndrome in pregnancy. J Neuroendocrinol. 2022;34(7):e13118. PMID: 35491087
-
Raffaelli M, Brunaud L, De Crea C, et al. Outcome of adrenalectomy for subclinical hypercortisolism and Cushing syndrome. Surgery. 2017;161(1):264-271. PMID: 27865591
-
Reincke M, Sbiera S, Hayakawa A, et al. Mutations in the deubiquitinase gene USP8 cause Cushing's disease. Nat Genet. 2015;47(1):31-38. PMID: 25485749
-
Cao Y, He M, Gao Z, et al. Activating hotspot L205R mutation in PRKACA and adrenal Cushing's syndrome. Science. 2014;344(6186):913-917. PMID: 24855271
-
Fassnacht M, Dekkers OM, Else T, et al. European Society of Endocrinology Clinical Practice Guidelines on the management of adrenocortical carcinoma in adults, in collaboration with the European Network for the Study of Adrenal Tumors. Eur J Endocrinol. 2018;179(4):G1-G46. PMID: 30299884
-
Assié G, Libé R, Espiard S, et al. ARMC5 mutations in macronodular adrenal hyperplasia with Cushing's syndrome. N Engl J Med. 2013;369(22):2105-2114. PMID: 24283226
-
Mazziotti G, Angeli A, Bilezikian JP, et al. Glucocorticoid-induced osteoporosis: an update. Trends Endocrinol Metab. 2006;17(4):144-149. PMID: 16678739
-
Dekkers OM, Horváth-Puhó E, Jørgensen JO, et al. Multisystem morbidity and mortality in Cushing's syndrome: a cohort study. J Clin Endocrinol Metab. 2013;98(6):2277-2284. PMID: 23533241
-
Hofman MS, Lau WF, Hicks RJ. Somatostatin receptor imaging with 68Ga DOTATATE PET/CT: clinical utility, normal patterns, pearls, and pitfalls in interpretation. Radiographics. 2015;35(2):500-516. PMID: 25763733
-
Tritos NA, Biller BM. Diagnosis and management of pituitary adenomas: a review. JAMA. 2023;329(16):1386-1398. PMID: 37097352
-
Assié G, Bahurel H, Coste J, et al. Corticotroph tumor progression after adrenalectomy in Cushing's Disease: a reappraisal of Nelson's Syndrome. J Clin Endocrinol Metab. 2007;92(1):172-179. PMID: 17062760
-
Pivonello R, Fleseriu M, Newell-Price J, et al. Efficacy and safety of osilodrostat in patients with Cushing's disease (LINC 3): a multicentre phase III study with a double-blind, randomised withdrawal phase. Lancet Diabetes Endocrinol. 2020;8(9):748-761. PMID: 32702306
-
Creemers SG, Feelders RA, de Jong FH, et al. Levoketoconazole, the 2S,4R enantiomer of ketoconazole: a new steroidogenesis inhibitor for Cushing's syndrome treatment. J Clin Endocrinol Metab. 2021;106(4):e1618-e1629. PMID: 33399817
-
Colao A, Petersenn S, Newell-Price J, et al. A 12-month phase 3 study of pasireotide in Cushing's disease. N Engl J Med. 2012;366(10):914-924. PMID: 22397653
-
Fleseriu M, Biller BM, Findling JW, et al. Mifepristone, a glucocorticoid receptor antagonist, produces clinical and metabolic benefits in patients with Cushing's syndrome. J Clin Endocrinol Metab. 2012;97(6):2039-2049. PMID: 22466348
Last Updated: 2026-01-06 | MedVellum Editorial Team Evidence Level: High (30 PubMed citations) Word Count: ~13,500 words Line Count: 1050 lines
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Hypothalamic-Pituitary-Adrenal (HPA) Axis Physiology
- Steroidogenesis Pathways
- Adrenal Gland Anatomy
Differentials
Competing diagnoses and look-alikes to compare.
- Pseudo-Cushing's Syndrome (Alcoholism/Depression)
- Polycystic Ovary Syndrome (PCOS)
- Metabolic Syndrome
- Primary Aldosteronism
- Exogenous Glucocorticoid Use
Consequences
Complications and downstream problems to keep in mind.
- Secondary Osteoporosis
- Steroid-Induced Diabetes Mellitus
- Cardiovascular Risk in Endocrine Disorders
- Adrenal Insufficiency
- Nelson Syndrome