Guillain-Barré Syndrome
The pathophysiology involves molecular mimicry, where antibodies generated against infectious agents cross-react with peripheral nerve components, leading to demyelination (in acute inflammatory demyelinating...
What matters first
The pathophysiology involves molecular mimicry, where antibodies generated against infectious agents cross-react with peripheral nerve components, leading to demyelination (in acute inflammatory demyelinating...
Respiratory compromise (FVC declining)
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Respiratory compromise (FVC declining)
- Rapid progression
- Bulbar weakness
- Autonomic instability
Linked comparisons
Differentials and adjacent topics worth opening next.
- Myasthenia Gravis
- Botulism
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Topic family
This concept exists in multiple MedVellum libraries. Use the primary page for the broadest reference view and the others for exam-specific framing.
The pathophysiology involves molecular mimicry, where antibodies generated against infectious agents cross-react with peripheral nerve components, leading to demyelination (in acute inflammatory demyelinating...
GBS is the most common cause of acute flaccid paralysis worldwide, affecting 1-2 people per 100,000 annually. Up to 30% ... ACEM Fellowship Written, ACEM Fellow
\u003e One-liner: Guillain-Barré Syndrome is an acute immune-mediated polyradiculoneuropathy causing rapidly ascending f... CICM Second Part exam preparation.
Clinical explanation and evidence
Guillain-Barré Syndrome
1. Clinical Overview
Summary
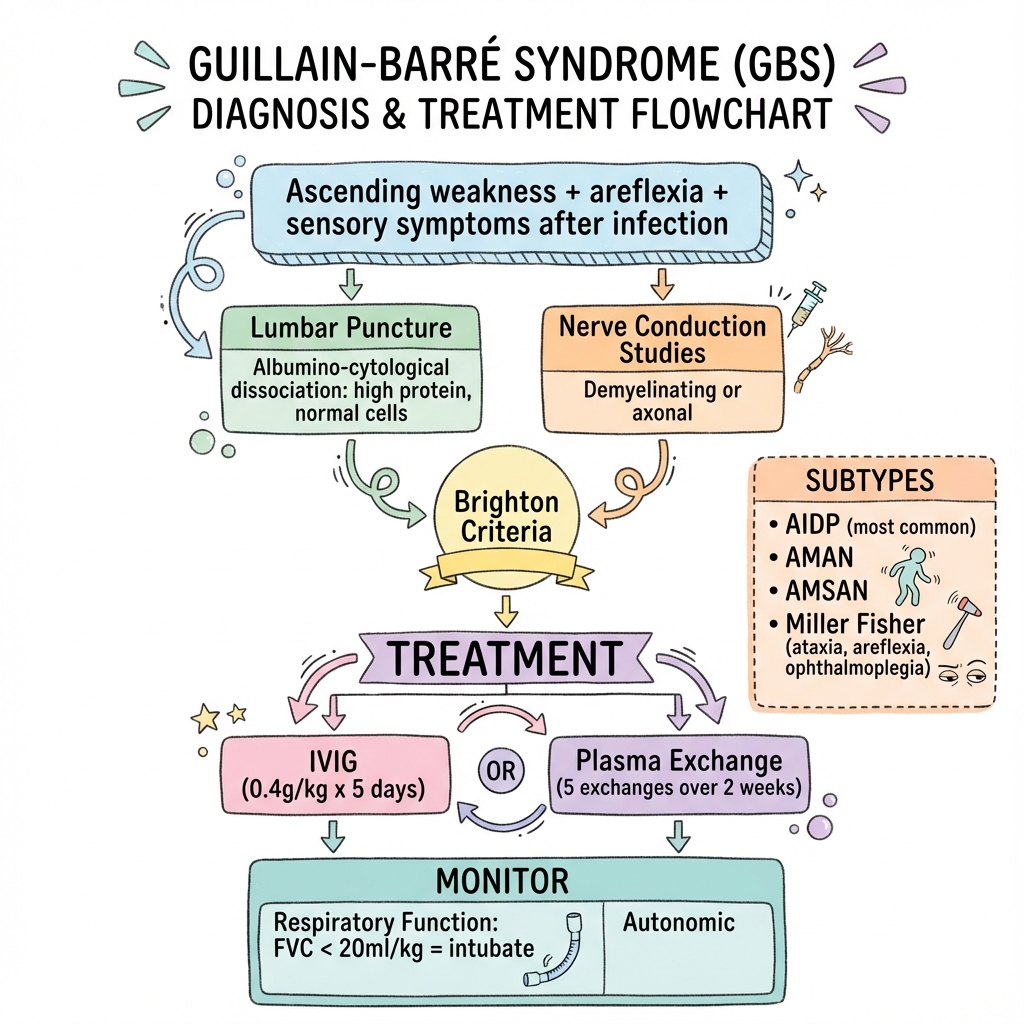
Guillain-Barré syndrome (GBS) is an acute immune-mediated polyradiculoneuropathy characterised by rapidly progressive symmetrical weakness, areflexia, and sensory symptoms. It represents the most common cause of acute flaccid paralysis worldwide, with an incidence of 1-2 per 100,000 per year. [1] GBS typically follows a respiratory or gastrointestinal infection (commonly Campylobacter jejuni) by 1-4 weeks, with approximately 50-70% of cases reporting a preceding infection. [2]
The pathophysiology involves molecular mimicry, where antibodies generated against infectious agents cross-react with peripheral nerve components, leading to demyelination (in acute inflammatory demyelinating polyneuropathy, AIDP) or axonal damage (in acute motor axonal neuropathy, AMAN). [3] The most common form in Western populations is AIDP, accounting for approximately 90% of cases, while axonal variants predominate in Asia and following Campylobacter infection. [4]
GBS can progress to life-threatening respiratory failure in up to 30% of patients, requiring intensive care unit (ICU) admission and mechanical ventilation. [5] Early recognition and treatment with intravenous immunoglobulin (IVIG) or plasma exchange significantly hastens recovery and improves outcomes. [6] Despite treatment, approximately 20% of patients have significant residual disability at 6 months, and mortality remains around 3-5%. [1]
Key Facts
- Definition: Acute immune-mediated polyradiculoneuropathy with ascending weakness
- Incidence: 1-2 per 100,000 per year; bimodal age distribution (young adults and elderly)
- Peak Demographics: All ages; slight male predominance (1.5:1)
- Preceding Infection: 50-70% (Campylobacter 30%, CMV 10%, EBV 10%, Zika virus in outbreaks)
- Pathognomonic Features: Ascending weakness + areflexia + albumino-cytological dissociation
- Gold Standard Investigation: CSF analysis (elevated protein, normal cell count) + nerve conduction studies
- First-line Treatment: IVIG 0.4g/kg/day × 5 days OR plasma exchange (5 exchanges over 10-14 days)
- Respiratory Failure: Occurs in 20-30% of cases; requires close monitoring
- Prognosis: 80% walk independently at 6 months; 60% full recovery at 1 year; 5% mortality
Clinical Pearls
Emergency Pearl: Monitor forced vital capacity (FVC) every 4-6 hours in progressive cases. The "20-30-40 rule": FVC less than 20ml/kg, maximal inspiratory pressure (MIP) less than 30cmH₂O, or maximal expiratory pressure (MEP) less than 40cmH₂O indicates impending respiratory failure. Consider intubation if FVC declines by > 30% or absolute FVC less than 15-20ml/kg. [5]
Diagnostic Pearl: CSF protein may be normal in the first week of illness (40-50% of cases). Lumbar puncture performed on day 7-10 has higher diagnostic yield for demonstrating albumino-cytological dissociation. If clinical suspicion is high and initial LP is normal, repeat after 1 week. [7]
Treatment Pearl: IVIG and plasma exchange are equally effective for moderate-to-severe GBS. Do NOT combine both therapies (no added benefit demonstrated). [6] Corticosteroids alone are ineffective and should NOT be used as monotherapy. However, small molecule complement inhibitors (e.g., eculizumab) are under investigation. [8]
Variant Recognition Pearl: Miller Fisher syndrome (MFS) presents with the classic triad of ataxia, areflexia, and ophthalmoplegia, and is associated with anti-GQ1b antibodies in > 90% of cases. MFS has an excellent prognosis with recovery typically within 3 months. [9]
Autonomic Pearl: Autonomic dysfunction occurs in 70% of GBS patients and includes cardiac arrhythmias (brady/tachycardia), blood pressure lability, urinary retention, ileus, and pupillary abnormalities. Vagus nerve enlargement on ultrasound may predict autonomic complications. [10]
Why This Matters Clinically
GBS is a neurological emergency requiring rapid diagnosis and management. Respiratory failure can develop within hours to days of presentation, necessitating close monitoring of respiratory parameters. Early initiation of immunotherapy (within 2 weeks of symptom onset) significantly improves outcomes and reduces time to recovery. [6] The condition requires multidisciplinary management involving neurology, critical care, physiotherapy, and rehabilitation medicine. With appropriate treatment and supportive care, the majority of patients make good functional recovery, but 20% are left with significant disability, and 5% die from complications including respiratory failure, autonomic instability, pulmonary embolism, or sepsis. [1,11]
2. Epidemiology
Incidence and Prevalence
Guillain-Barré syndrome occurs worldwide with a relatively consistent incidence of 1-2 cases per 100,000 population per year. [1] The incidence increases with age, with rates doubling from 0.8 per 100,000 in those aged less than 18 years to 1.7 per 100,000 in those aged 50-79 years. There is a slight male predominance with a male:female ratio of approximately 1.5:1. [2]
Exam Detail: Geographic Variation: While the overall incidence is similar worldwide, the subtype distribution varies significantly. In North America and Europe, AIDP accounts for 60-80% of cases, whereas in Asia (particularly China and Japan), axonal forms (AMAN/AMSAN) predominate, accounting for 30-65% of cases. [12] This geographic variation is likely related to differences in triggering infections, particularly the higher prevalence of Campylobacter jejuni enteritis in Asia.
Bimodal Age Distribution
| Age Group | Incidence (per 100,000) | Predominant Subtype |
|---|---|---|
| 0-18 years | 0.6-0.8 | AIDP (Western); AMAN (Asia) |
| 18-50 years | 1.0-1.5 | AIDP |
| 50-79 years | 1.5-2.5 | AIDP |
| ≥80 years | 3.0-4.0 | AIDP, often severe |
Seasonal Variation
Some studies have identified seasonal peaks corresponding to increased respiratory and gastrointestinal infections, with higher incidence in late summer and autumn in temperate climates. [13]
Preceding Infections and Triggers
Approximately 50-70% of GBS patients report a preceding infection 1-4 weeks before symptom onset. [2,14] The most common triggering infections include:
| Organism/Trigger | Frequency | Associated Subtype | Key Features |
|---|---|---|---|
| Campylobacter jejuni | 25-30% | AMAN, AMSAN | Most common bacterial trigger; associated with anti-GM1, anti-GD1a antibodies; worse prognosis with axonal damage |
| Cytomegalovirus (CMV) | 10-15% | AIDP | More common in younger patients; may have bilateral facial palsy |
| Epstein-Barr virus (EBV) | 10% | AIDP | Similar presentation to CMV-associated GBS |
| Mycoplasma pneumoniae | 5% | AIDP | Associated with respiratory illness |
| Influenza A/B | 2-5% | AIDP | Seasonal variation |
| Zika virus | Variable (outbreaks) | AIDP | Epidemic association (French Polynesia 2013-14, Latin America 2015-16) |
| HIV | Rare | AIDP | Usually at seroconversion |
| Hepatitis E | Rare | AIDP, AMAN | More common in endemic areas |
| SARS-CoV-2 (COVID-19) | Emerging | AIDP, MFS variants | Reported association; temporal relationship variable [15] |
| Vaccinations | Very rare (less than 1 in 1 million) | AIDP | Historical association with swine flu vaccine (1976); no confirmed link with modern vaccines |
Exam Detail: Molecular Mimicry in Campylobacter-Associated GBS: Campylobacter jejuni lipooligosaccharides (LOS) contain sialic acid residues that structurally mimic human gangliosides (GM1, GD1a, GD1b, GQ1b) found on peripheral nerves. This molecular mimicry triggers cross-reactive antibody production, leading to antibody-mediated nerve damage. Patients with Campylobacter infection who develop GBS often have more severe disease with axonal involvement (AMAN/AMSAN) and poorer outcomes compared to other triggers. [3,14]
3. Aetiology and Pathophysiology
Molecular Mechanisms
GBS is an autoimmune disorder resulting from aberrant immune responses to infectious agents or other triggers that cross-react with peripheral nerve components. The pathogenesis involves a complex interplay of humoral (antibody-mediated) and cellular (T-cell mediated) immune mechanisms.
Step 1: Infection and Immune Activation
Following infection (typically respiratory or gastrointestinal), the host immune system generates antibodies and activated T-cells against pathogen-specific antigens. In susceptible individuals, structural similarities between pathogen epitopes and peripheral nerve gangliosides or myelin proteins lead to cross-reactive immune responses (molecular mimicry). [3]
Step 2: Cross-Reactive Antibody Production
Anti-ganglioside antibodies are detected in 40-60% of GBS patients and are highly specific for certain subtypes:
| Antibody | Associated Subtype | Target | Clinical Features |
|---|---|---|---|
| Anti-GM1 | AMAN | Motor axons | Pure motor involvement; associated with Campylobacter |
| Anti-GD1a | AMAN, AMSAN | Motor and sensory axons | Severe axonal damage |
| Anti-GQ1b | Miller Fisher syndrome | Oculomotor nerves | Ophthalmoplegia, ataxia, areflexia (> 90% sensitivity) [9] |
| Anti-GT1a | MFS, pharyngeal-cervical-brachial variant | Cranial nerves | Bulbar weakness |
| Anti-GD1b | Sensory ataxic GBS | Sensory neurons | Prominent sensory ataxia |
Step 3: Nerve Damage Mechanisms
Demyelinating Form (AIDP):
- Autoantibodies and complement attack Schwann cells and myelin
- Macrophages infiltrate nerve roots and peripheral nerves
- Myelin stripping occurs with relative axonal preservation
- Conduction block develops from demyelination
- Remyelination typically occurs during recovery
Axonal Form (AMAN/AMSAN):
- Anti-ganglioside antibodies bind to nodal and paranodal regions of axons
- Complement activation leads to formation of membrane attack complex (MAC)
- Direct axonal injury occurs without significant demyelination
- Macrophage invasion at nodes of Ranvier
- Recovery depends on axonal regeneration (slower than remyelination)
Exam Detail: Complement Cascade in GBS: Following anti-ganglioside antibody binding to nerve membranes, the classical complement pathway is activated. C1q binds to antibody-antigen complexes, triggering sequential activation of C4, C2, C3, and C5-C9, culminating in formation of the membrane attack complex (MAC, C5b-9). MAC insertion into nerve cell membranes creates pores leading to cell lysis and nerve damage. This mechanism is supported by the presence of complement components in nerve biopsies from GBS patients and forms the rationale for complement-targeted therapies currently in development (e.g., eculizumab, an anti-C5 monoclonal antibody). [8]
Step 4: Clinical Progression
- Onset (Days 0-7): Progressive ascending weakness, sensory symptoms, pain
- Progression Phase (Days 7-14): Continued worsening; nadir typically reached by 2 weeks (90% of cases) or 4 weeks (98% of cases)
- Plateau Phase (Weeks 2-4): Stabilisation of symptoms
- Recovery Phase (Weeks to Months): Remyelination (AIDP) or axonal regeneration (AMAN/AMSAN); recovery may take 6-12 months or longer
Genetic Susceptibility
While GBS is not inherited, certain HLA alleles may confer susceptibility or resistance to specific GBS subtypes, though genetic factors play a minor role compared to environmental triggers. [16]
4. Clinical Presentation
Symptoms
Motor Symptoms (Universal)
- Ascending symmetrical weakness: Typically begins in legs (distal or proximal) and ascends to arms over hours to days
- Weakness peaks by 2 weeks in 50% of patients, by 4 weeks in 90% [1]
- Proximal and distal muscles equally affected
- Facial weakness (bilateral) in 50% of cases
- Bulbar weakness (dysphagia, dysarthria) in 30-40%
- Respiratory muscle weakness in 20-30%, may require ventilation [5]
Sensory Symptoms (80%)
- Paraesthesias (tingling, numbness) in hands and feet
- Sensory ataxia (impaired proprioception)
- Neuropathic pain (back pain, radicular pain, muscle pain) in 60-80%
- Pain is often severe and undertreated; neuropathic pain medications (gabapentin, pregabalin) are indicated
Autonomic Symptoms (70%)
- Cardiac: Tachycardia, bradycardia, arrhythmias
- Blood pressure lability (hyper/hypotension)
- Urinary retention
- Ileus, constipation
- Pupillary abnormalities
- Excessive sweating or anhidrosis [10]
Signs
Motor Signs
- Symmetrical flaccid weakness (proximal and distal)
- Areflexia or hyporeflexia (hallmark feature; > 90% of cases)
- Facial diplegia (bilateral facial weakness)
- Bulbar palsy (reduced gag reflex, palatal weakness, dysarthria, dysphagia)
- Respiratory insufficiency (tachypnoea, use of accessory muscles, paradoxical breathing)
- Ophthalmoplegia (in Miller Fisher syndrome and related variants)
Sensory Signs
- Distal sensory loss (glove-and-stocking distribution)
- Reduced vibration and proprioception
- Sensory ataxia (impaired joint position sense)
- Romberg's sign positive
Autonomic Signs
- Resting tachycardia or bradycardia
- Postural hypotension
- Abdominal distension (ileus)
- Urinary retention
Diagnostic Criteria (Brighton Criteria)
The Brighton Collaboration case definition for GBS provides standardised diagnostic criteria:
Level 1 (Diagnostic Certainty):
- Bilateral flaccid weakness of limbs
- Decreased or absent deep tendon reflexes in weak limbs
- Monophasic illness pattern
- Interval between onset and nadir: 12 hours to 28 days
- CSF: Cell count less than 50 cells/μL, protein elevated
- NCS: Consistent with GBS (demyelinating or axonal pattern)
- Absence of alternative diagnosis
Level 2 (High Diagnostic Certainty):
- As above but with CSF or NCS not performed or with equivocal findings
Subtypes and Variants
| Subtype/Variant | Frequency | Clinical Features | Electrophysiology | Antibodies | Prognosis |
|---|---|---|---|---|---|
| AIDP | 60-80% (West), 20-30% (Asia) | Classic ascending weakness, areflexia, sensory symptoms | Demyelinating: prolonged distal latencies, slowed conduction velocity, conduction block, temporal dispersion | Variable | Good; 80% walk at 6 months |
| AMAN | 5-10% (West), 30-50% (Asia) | Pure motor, severe weakness, rapid progression | Axonal: reduced CMAP amplitude, normal sensory responses | Anti-GM1, anti-GD1a | Moderate; slower recovery due to axonal regeneration |
| AMSAN | 5-10% | Severe motor and sensory involvement, respiratory failure common | Axonal: reduced CMAP and SNAP amplitudes | Anti-GM1, anti-GD1a, anti-GD1b | Poor; prolonged recovery, significant residual disability |
| Miller Fisher Syndrome (MFS) | 5-10% | Ataxia, areflexia, ophthalmoplegia (AAO triad); descending pattern | May be normal or mild demyelinating | Anti-GQ1b (> 90%) [9] | Excellent; recovery in 2-3 months without treatment |
| Pharyngeal-Cervical-Brachial | Rare | Bulbar weakness, neck weakness, arm weakness; leg-sparing | Variable | Anti-GT1a | Good with treatment |
| Acute Sensory Neuropathy | Rare | Pure sensory; severe sensory ataxia, areflexia | Reduced or absent SNAPs | Anti-GD1b | Variable |
| Bickerstaff Brainstem Encephalitis | Rare | Ophthalmoplegia, ataxia, altered consciousness, hyperreflexia | Variable | Anti-GQ1b | Good with treatment |
Clinical Pearl: Recognising Atypical Presentations: Not all GBS patients present with classic ascending weakness. Variant forms include:
- Descending GBS: Weakness starts in cranial nerves (facial weakness, ophthalmoplegia, bulbar weakness) before progressing to limbs
- Paraparetic GBS: Weakness confined to legs; can mimic spinal cord pathology
- Pure motor or pure sensory forms: Isolated motor or sensory involvement
- GBS with preserved reflexes: Rare; hyperreflexia or normal reflexes may occur, especially early in the course
Maintain a high index of suspicion even when the presentation is atypical, especially if there is a history of antecedent infection.
Red Flags
[!CAUTION] Immediate Interventions Required:
- Rapidly progressive weakness: Ascending from legs to arms within 24 hours — consider ICU admission
- FVC less than 20ml/kg or declining > 30%: Impending respiratory failure — prepare for intubation [5]
- Bulbar weakness: High risk of aspiration pneumonia — NBM, consider NG tube or intubation
- Autonomic instability: Severe tachycardia (> 120bpm), bradycardia (less than 40bpm), BP lability — continuous cardiac monitoring required [10]
- Inability to walk unaided: Higher risk of prolonged disability and ICU admission
- Absent gag reflex or inability to handle secretions: Airway protection compromised
5. Investigations
Cerebrospinal Fluid (CSF) Analysis
Albumino-Cytological Dissociation is the hallmark CSF finding in GBS:
| Parameter | Finding | Interpretation |
|---|---|---|
| Protein | Elevated (typically 0.55-2.5g/L) | Absent in first week in 30-50% of cases; peaks at 4-6 weeks [7] |
| White cell count (WCC) | Normal (less than 5 cells/μL) | Typically 0-10 cells/μL; if > 50 cells/μL, consider alternative diagnosis (HIV, Lyme, sarcoidosis) |
| Pattern | High protein, normal cells | Albumino-cytological dissociation |
| Glucose | Normal | |
| Opening pressure | Normal or mildly elevated |
Clinical Pearl: Timing of LP: CSF protein is normal in the first week in 30-50% of patients. If GBS is suspected but initial CSF is normal, repeat LP after 1 week for improved diagnostic sensitivity. However, clinical diagnosis should not be delayed pending LP results if the clinical picture is consistent with GBS.
Atypical CSF Findings Suggesting Alternative Diagnoses:
- Pleocytosis (WCC > 50 cells/μL): HIV-associated GBS, Lyme disease, sarcoidosis, lymphomatous meningitis
- Low glucose: Infectious meningitis, carcinomatous meningitis
- Markedly elevated protein (> 5g/L): Spinal block, chronic inflammatory demyelinating polyneuropathy (CIDP)
Nerve Conduction Studies (NCS)
NCS help confirm the diagnosis, classify the subtype (demyelinating vs. axonal), and predict prognosis.
Demyelinating Pattern (AIDP)
| Parameter | Finding | Significance |
|---|---|---|
| Motor conduction velocity | Reduced (less than 70-80% of lower limit of normal) | Demyelination |
| Distal motor latency | Prolonged (> 125% of upper limit of normal) | Distal demyelination |
| F-wave latency | Prolonged or absent | Early finding; proximal demyelination |
| Conduction block | Partial (> 20% reduction in CMAP amplitude between distal and proximal stimulation) | Focal demyelination causing conduction failure |
| Temporal dispersion | Prolonged CMAP duration | Non-uniform demyelination |
| Sensory nerve action potential (SNAP) | Reduced amplitude or absent | Sensory nerve involvement |
Axonal Pattern (AMAN/AMSAN)
| Parameter | Finding | Significance |
|---|---|---|
| Compound muscle action potential (CMAP) | Reduced amplitude | Axonal loss; poor prognostic sign |
| Motor conduction velocity | Normal or mildly reduced | Axons intact but fewer |
| F-waves | Absent or prolonged | Proximal involvement |
| SNAP (AMSAN) | Reduced or absent | Sensory axonal loss |
| SNAP (AMAN) | Normal | Pure motor form |
Exam Detail: Early NCS Findings: Nerve conduction studies may be normal in the first week of illness in 10-20% of cases. The earliest electrophysiological abnormality is often prolonged or absent F-waves, which reflect proximal nerve root dysfunction. Serial NCS may be required to confirm the diagnosis and classify the subtype accurately.
Antibody Testing
Anti-ganglioside antibodies support the diagnosis and predict subtype:
| Antibody | Specificity for GBS | Associated Subtype | Clinical Utility |
|---|---|---|---|
| Anti-GQ1b | > 90% for MFS [9] | Miller Fisher syndrome | Highly specific; confirms MFS diagnosis |
| Anti-GM1 | 20-50% | AMAN (especially post-Campylobacter) | Supports axonal motor variant |
| Anti-GD1a | 20-40% | AMAN, AMSAN | Severe axonal forms |
| Anti-GT1a | Variable | Pharyngeal-cervical-brachial variant | Bulbar predominant GBS |
| Anti-GD1b | Rare | Sensory ataxic variant | Pure sensory forms |
Note: Absence of anti-ganglioside antibodies does NOT exclude GBS (40-60% of cases are antibody-negative).
Respiratory Function Monitoring
Critical for all GBS patients, especially those with bulbar weakness, rapidly progressive disease, or autonomic instability:
| Test | Normal Value | Threshold for Concern | Action |
|---|---|---|---|
| Forced vital capacity (FVC) | > 60ml/kg | less than 30ml/kg | Close monitoring; consider ICU |
| less than 20ml/kg | High risk of respiratory failure; prepare for intubation [5] | ||
| Negative inspiratory force (NIF/MIP) | < -60cmH₂O | >-30cmH₂O | Weak inspiratory muscles; impending failure |
| Peak expiratory flow (PEF/MEP) | > 300L/min | less than 40cmH₂O | Weak expiratory muscles; ineffective cough |
| Oxygen saturation (SpO₂) | > 95% | less than 92% | Late sign of hypoventilation; urgent action required |
| Arterial blood gas (ABG) | pH 7.35-7.45, PaCO₂ 35-45mmHg | PaCO₂ > 50mmHg | Hypercapnic respiratory failure |
Clinical Pearl: The 20-30-40 Rule for Respiratory Failure:
- FVC less than 20ml/kg
- MIP less than 30cmH₂O (less negative than -30)
- MEP less than 40cmH₂O
Any one of these indicates high risk of respiratory failure and need for intubation. Do not wait for hypoxia or hypercapnia — these are late signs. [5]
Additional Investigations
| Investigation | Purpose | Findings |
|---|---|---|
| MRI spine | Exclude spinal cord pathology (myelitis, cord compression) | GBS: Nerve root enhancement with gadolinium (cauda equina, cervical roots) |
| Blood tests | Identify trigger, exclude mimics | FBC, U&E, LFT, TFT, vitamin B12, anti-GQ1b antibodies, serology for CMV/EBV/Mycoplasma/HIV |
| Stool/throat culture | Identify Campylobacter jejuni | Positive in 20-30% of cases |
| ECG, cardiac telemetry | Monitor autonomic dysfunction | Arrhythmias (brady/tachycardia, heart block) [10] |
| Nerve/muscle ultrasound | Emerging tool; distinguishes demyelinating vs. axonal | Nerve enlargement (cross-sectional area) in AIDP; vagus nerve enlargement predicts autonomic dysfunction [10] |
6. Differential Diagnosis
Acute flaccid paralysis has a broad differential diagnosis. Key conditions to exclude:
| Condition | Key Distinguishing Features | Diagnostic Tests |
|---|---|---|
| Myasthenia gravis | Fatigable weakness, fluctuating symptoms, ptosis, diplopia, normal reflexes, respiratory muscles involved late | Acetylcholine receptor antibodies, anti-MuSK antibodies, repetitive nerve stimulation, edrophonium test |
| Botulism | Descending paralysis, dilated unreactive pupils, no sensory symptoms, normal CSF | Stool/serum botulinum toxin assay, EMG (small amplitude CMAPs, incremental response to high-frequency stimulation) |
| Acute transverse myelitis | Sensory level, bladder/bowel dysfunction early, hyperreflexia, Babinski sign positive | MRI spine (cord signal abnormality), CSF pleocytosis |
| Spinal cord compression | Back pain, sensory level, sphincter dysfunction, hyperreflexia below lesion | MRI spine (urgent) |
| Acute intermittent porphyria | Abdominal pain, psychiatric symptoms, seizures, hyponatraemia | Urine porphobilinogen, elevated urinary ALA and PBG |
| Tick paralysis | Ascending paralysis, history of tick exposure, rapid recovery after tick removal | Clinical; identification and removal of tick |
| Chronic inflammatory demyelinating polyneuropathy (CIDP) | Progressive or relapsing-remitting course > 8 weeks, often > 2 months to nadir | NCS (demyelinating), CSF protein elevated, treatment-responsive to steroids |
| Critical illness polyneuropathy/myopathy | Occurs in ICU setting after sepsis, multi-organ failure; flaccid weakness, difficulty weaning from ventilator | EMG/NCS (axonal neuropathy or myopathy), CK normal or elevated |
| Acute stroke (brainstem or bilateral hemispheric) | Sudden onset, altered consciousness, hyperreflexia, Babinski positive, cranial nerve signs | CT/MRI brain |
| Hypokalaemic periodic paralysis | Sudden onset, potassium less than 2.5mmol/L, normal reflexes, no sensory symptoms | Serum potassium, ECG (U waves, T wave flattening) |
| Poliomyelitis (or enterovirus D68) | Asymmetric weakness, fever, meningismus, CSF pleocytosis, hyperreflexia in some cases | CSF PCR (enterovirus), stool viral culture |
Exam Detail: Viva Approach to Acute Flaccid Paralysis:
- Pattern of weakness: Ascending (GBS), descending (botulism, MFS), proximal (myopathy), distal (neuropathy)
- Reflexes: Areflexia (GBS, botulism), hyperreflexia (myelopathy, stroke), normal (myasthenia)
- Sensory involvement: Present (GBS, AMAN spares sensory), absent (botulism, motor neuron disease)
- Cranial nerves: Facial diplegia (GBS), ophthalmoplegia (MFS, myasthenia, botulism)
- Sphincter involvement: Early (myelopathy), late or absent (GBS)
- Time course: Hours to days (GBS, myasthenia crisis), sudden (stroke), weeks (CIDP)
7. Management
Initial Assessment and Stabilisation
A. Emergency Assessment
-
Airway, Breathing, Circulation (ABC):
- Assess for bulbar weakness (drooling, pooling of secretions, weak gag, dysarthria)
- Measure respiratory function: FVC, MIP, MEP every 4-6 hours
- Continuous cardiac monitoring for autonomic dysfunction
-
Neurological Assessment:
- Document muscle strength (MRC grading 0-5) for all muscle groups
- Assess sensory function, reflexes, cranial nerves
- Establish baseline for monitoring progression
-
Level of Care:
- ICU admission if:
- FVC less than 30ml/kg or declining rapidly
- Bulbar weakness
- Autonomic instability
- Inability to walk unaided
- Rapidly progressive disease (ascending to arms within 24-48 hours)
- ICU admission if:
Respiratory Management
Indications for Intubation and Mechanical Ventilation [5]
Absolute Indications:
- FVC less than 15ml/kg
- MIP >-30cmH₂O (i.e., less negative than -30)
- Hypoxia (SpO₂ less than 90% on room air) despite supplemental O₂
- Hypercapnia (PaCO₂ > 50mmHg)
- Respiratory rate > 30/min with accessory muscle use
- Inability to clear secretions
- Severe bulbar weakness with aspiration risk
Relative Indications:
- FVC 15-20ml/kg with declining trend
- Autonomic instability
- Rapid progression
Clinical Pearl: Elective vs. Emergency Intubation: In GBS, aim for elective intubation when respiratory parameters are declining but before respiratory failure occurs. Emergency intubation in hypoxic, hypercapnic patients carries higher risk of complications (aspiration, cardiac arrest from vagal stimulation). Use of rapid sequence intubation with appropriate premedication (avoid succinylcholine due to risk of hyperkalaemia in denervated muscle).
Mechanical Ventilation:
- Lung-protective strategies (tidal volume 6-8ml/kg ideal body weight)
- Avoid hyperventilation (may worsen autonomic instability)
- Early tracheostomy (within 2 weeks) if prolonged ventilation anticipated (reduces laryngeal injury, facilitates weaning) [5]
Weaning from Ventilation:
- When FVC > 15-20ml/kg and improving
- MIP < -30cmH₂O
- Stable autonomic function
- Able to protect airway (intact gag, able to clear secretions)
Immunotherapy
Early immunotherapy (within 2 weeks of symptom onset) hastens recovery, reduces time to independent walking, and improves outcomes. [6]
First-Line Treatments (Equally Effective)
| Treatment | Regimen | Mechanism | Evidence | Adverse Effects |
|---|---|---|---|---|
| Intravenous Immunoglobulin (IVIG) | 0.4g/kg/day × 5 days (total 2g/kg) | Neutralises pathogenic antibodies, modulates complement, anti-inflammatory effects | Cochrane review: Equivalent to plasma exchange for moderate-severe GBS [6] | Headache, aseptic meningitis, hyperviscosity, thrombosis, renal impairment, anaphylaxis (IgA deficiency) |
| Plasma Exchange (PLEX) | 5 exchanges over 10-14 days (200-250ml/kg total volume) | Removes pathogenic antibodies, complement, inflammatory mediators | Cochrane review: Effective for moderate-severe GBS; reduces time to recover walking [6] | Hypotension, line sepsis, coagulopathy, hypocalcaemia, thrombosis |
Choice of Treatment:
- IVIG is more widely available, easier to administer, and preferred in most centres
- PLEX may be preferred if:
- IVIG contraindicated (IgA deficiency with anti-IgA antibodies, renal impairment)
- Rapid access to apheresis facilities
- Cost considerations (in some countries, PLEX is cheaper)
Clinical Pearl: Do NOT Combine IVIG and PLEX: Sequential or combined therapy with IVIG and plasma exchange does NOT provide additional benefit over either treatment alone and may increase adverse effects. [6] Choose one modality and complete the full course.
Corticosteroids: NOT Recommended
- Oral corticosteroids alone are INEFFECTIVE in GBS and should NOT be used as monotherapy. [6]
- Combination therapy (IVIG or PLEX + corticosteroids) does NOT improve outcomes and is NOT recommended.
- Exception: Some clinicians use IV methylprednisolone in severe AIDP or as a trial in treatment-resistant cases, though evidence is weak.
Emerging Therapies
| Therapy | Mechanism | Status | Evidence |
|---|---|---|---|
| Eculizumab | Anti-C5 monoclonal antibody; blocks complement activation | Phase 2 trials completed; not yet approved [8] | ICA-GBS trial: Showed trend towards benefit but did not meet primary endpoint |
| Complement C1 inhibitors | Inhibits classical complement pathway | Investigational | Preclinical and early clinical studies ongoing [8] |
| Anti-neonatal Fc receptor (FcRn) antagonists | Reduces pathogenic IgG levels | Investigational | Trials in other antibody-mediated diseases; potential for GBS |
Supportive Care
Autonomic Dysfunction Management [10]
| Manifestation | Management |
|---|---|
| Tachycardia | Usually benign and self-limiting; avoid beta-blockers unless symptomatic or HR > 140bpm; treat underlying causes (pain, anxiety, hypoxia) |
| Bradycardia/heart block | Temporary pacing if symptomatic (syncope, hypotension, pauses > 3 seconds); atropine for acute episodes (use cautiously — may precipitate tachycardia) |
| Hypertension | Short-acting agents (labetalol, hydralazine); avoid long-acting agents (risk of rebound hypotension) |
| Hypotension | IV fluids; vasopressors if refractory (noradrenaline); avoid excessive fluid boluses (risk of pulmonary oedema) |
| Urinary retention | Intermittent catheterisation preferred over indwelling catheter (reduces UTI risk) |
| Ileus | NBM, NG decompression, prokinetics (metoclopramide, erythromycin), early enteral nutrition once ileus resolves |
Venous Thromboembolism (VTE) Prophylaxis
- High risk: Immobile patients require pharmacological prophylaxis (LMWH) plus mechanical prophylaxis (compression stockings, intermittent pneumatic compression devices)
- DVT/PE incidence: 3-5% despite prophylaxis
Pain Management
- Neuropathic pain (60-80% of patients): Gabapentin (300-1200mg TDS), pregabalin (75-150mg BD), duloxetine, amitriptyline
- Musculoskeletal pain: Paracetamol, NSAIDs (if no contraindication)
- Severe pain: Opioids (morphine, oxycodone) may be required; use cautiously (risk of ileus, respiratory depression)
Physiotherapy and Rehabilitation
- Early mobilisation: As soon as medically stable and improving
- Passive range-of-motion exercises: Prevent contractures
- Respiratory physiotherapy: Chest physiotherapy, assisted cough, suctioning
- Multidisciplinary rehabilitation: Physiotherapy, occupational therapy, speech and language therapy (for dysphagia), psychology
- Orthoses and aids: Ankle-foot orthoses (AFOs) for foot drop, walking aids
Nutritional Support
- Enteral nutrition: NG feeding if dysphagia or reduced consciousness; aim for early enteral nutrition (within 48 hours)
- Parenteral nutrition: If enteral route not feasible (ileus, aspiration risk)
Psychological Support
- Anxiety and depression are common (up to 30% of patients)
- Provide psychological support, reassurance about prognosis
- Consider psychiatric referral if severe or persistent mood disturbance
Monitoring During Hospital Stay
| Parameter | Frequency | Action Threshold |
|---|---|---|
| FVC, MIP, MEP | Every 4-6 hours (if declining) | FVC less than 20ml/kg → prepare for intubation |
| Muscle strength (MRC sum score) | Daily | Document progression/stabilisation |
| Cardiac monitoring | Continuous (if autonomic dysfunction) | Treat arrhythmias as per protocol |
| Blood pressure | Every 1-4 hours | Lability → avoid long-acting agents |
| Oxygen saturation | Continuous | less than 92% → ABG, consider ventilation |
| Bladder function | Daily | Retention → catheterise |
| Bowel function | Daily | Ileus → NBM, NG decompression |
8. Prognosis
Outcomes at 6-12 Months
| Outcome | Percentage |
|---|---|
| Walk independently at 6 months | 80% [1] |
| Full functional recovery at 1 year | 60% [1] |
| Significant residual disability (unable to walk independently) | 20% [11] |
| Mortality | 3-5% [1,11] |
Recovery Timeline
- Nadir (peak weakness): 50% by 2 weeks, 90% by 4 weeks
- Plateau phase: 2-4 weeks
- Recovery phase: Begins 2-4 weeks after nadir; continues for 6-18 months
- Demyelinating (AIDP): Faster recovery (remyelination occurs over weeks to months)
- Axonal (AMAN/AMSAN): Slower recovery (axonal regeneration at 1mm/day; may take 12-24 months)
Predictors of Poor Outcome
| Factor | Impact on Prognosis |
|---|---|
| Age > 60 years | Slower recovery, higher disability [11] |
| Preceding Campylobacter infection | Associated with axonal forms (AMAN/AMSAN); poorer outcome [14] |
| Axonal subtype (AMAN/AMSAN) | Reduced CMAP amplitude predicts prolonged recovery and residual disability [12] |
| Rapid progression to nadir | Severe disease; higher risk of ventilation and poor outcome |
| Requirement for mechanical ventilation | 50% still disabled at 1 year [5] |
| Inability to walk unaided at 2 weeks | Predicts prolonged disability [11] |
| Low CMAP amplitude (less than 20% of normal) | Severe axonal loss; poor prognosis |
| Elevated CSF protein (> 2.5g/L) | May correlate with disease severity |
| Delayed treatment (> 2 weeks from onset) | Reduced efficacy of immunotherapy [6] |
Causes of Mortality (3-5%)
- Respiratory failure (despite ventilation)
- Autonomic instability (cardiac arrhythmias, cardiac arrest)
- Pulmonary embolism
- Sepsis (nosocomial infections, aspiration pneumonia)
- Multi-organ failure
Long-Term Sequelae
- Residual weakness and fatigue: 10-20% have persistent weakness, limiting activities
- Neuropathic pain: May persist for months to years
- Sensory ataxia: Impaired proprioception affecting balance and gait
- Foot drop: Requires long-term use of AFOs
- Psychological: Anxiety, depression, PTSD (especially in those who required ICU care)
Recurrence
- Recurrence rate: 2-5% of patients experience a second episode of GBS, often after another infection
- Recurrent episodes should prompt consideration of chronic inflammatory demyelinating polyneuropathy (CIDP), which is a relapsing-remitting or progressive condition requiring different treatment (corticosteroids, IVIG maintenance).
9. Complications
| Complication | Incidence | Prevention/Management |
|---|---|---|
| Respiratory failure | 20-30% [5] | Monitor FVC; early intubation if FVC less than 20ml/kg |
| Pneumonia (aspiration, ventilator-associated) | 20-30% | Airway protection; NG feeding; chest physiotherapy; VAP bundle |
| Venous thromboembolism (DVT/PE) | 3-5% | LMWH prophylaxis + mechanical compression |
| Cardiac arrhythmias | 20-40% [10] | Continuous cardiac monitoring; treat arrhythmias; avoid beta-blockers/calcium channel blockers unless essential |
| Urinary tract infection | 10-20% | Intermittent catheterisation preferred; remove catheter as soon as possible |
| Pressure ulcers | 5-10% | Regular repositioning; pressure-relieving mattresses |
| Contractures | Variable | Passive range-of-motion exercises; physiotherapy |
| ICU-acquired weakness | Variable (in ventilated patients) | Early mobilisation; minimise sedation; avoid neuromuscular blockers |
| Psychological (anxiety, depression, PTSD) | 20-30% | Psychological support; psychiatric referral if needed |
| Neuropathic pain | 60-80% | Gabapentin, pregabalin, duloxetine, amitriptyline |
10. Guidelines and Evidence
Key Guidelines
- European Federation of Neurological Societies/Peripheral Nerve Society (EFNS/PNS) Guidelines on Management of GBS (van Doorn et al., 2008; updated regularly)
- American Academy of Neurology (AAN) Practice Parameter: GBS in adults (Hughes et al.)
- National Institute for Health and Care Excellence (NICE) — no specific guideline for GBS; managed as per neurology best practice
Landmark Trials and Systematic Reviews
| Study/Review | Key Finding | Reference |
|---|---|---|
| Hughes et al., Cochrane Review (2014) | IVIG and plasma exchange are equally effective for moderate-to-severe GBS. Corticosteroids alone are ineffective. [6] | PMID: 25238327 |
| Willison et al., Lancet (2016) | Comprehensive review of GBS pathophysiology, clinical features, diagnosis, and treatment. [1] | PMID: 26948435 |
| Leonhard et al., Nature Reviews Disease Primers (2024) | Updated primer on GBS: epidemiology, pathogenesis, diagnosis, treatment, and future directions. [2] | PMID: 39702645 |
| Shang et al., Expert Rev Clin Immunol (2020) | Review of mechanical ventilation in GBS; 20-30% require ventilation; early tracheostomy beneficial. [5] | PMID: 33112177 |
| Yao et al., Hum Vaccin Immunother (2023) | Review of emerging immunotherapies for GBS, including complement inhibitors. [8] | PMID: 37278272 |
11. Examination Focus
Viva Preparation
"Tell me about Guillain-Barré syndrome."
Model Answer:
"Guillain-Barré syndrome is an acute immune-mediated polyradiculoneuropathy characterised by rapidly progressive symmetrical weakness, areflexia, and sensory symptoms. It is the most common cause of acute flaccid paralysis, with an incidence of 1-2 per 100,000 per year.
Approximately 50-70% of cases follow an antecedent infection, most commonly Campylobacter jejuni, CMV, or EBV, occurring 1-4 weeks prior. The pathophysiology involves molecular mimicry, where antibodies generated against infectious agents cross-react with peripheral nerve gangliosides or myelin components, leading to either demyelination (in AIDP) or axonal damage (in AMAN/AMSAN).
Clinically, patients present with ascending symmetrical weakness that progresses over hours to days, reaching a nadir by 2 weeks in 50% and by 4 weeks in 90%. Associated features include areflexia, distal paraesthesias, neuropathic pain, facial diplegia, bulbar weakness, and autonomic dysfunction. The hallmark CSF finding is albumino-cytological dissociation: elevated protein with normal cell count, though this may be absent in the first week.
Life-threatening complications include respiratory failure (20-30% of cases) and autonomic instability. Close monitoring of respiratory function with serial FVC measurements is critical. The 20-30-40 rule guides intubation: FVC less than 20ml/kg, MIP less than 30cmH₂O, or MEP less than 40cmH₂O indicate impending respiratory failure.
Treatment involves early immunotherapy with IVIG (0.4g/kg/day for 5 days) or plasma exchange (5 exchanges over 10-14 days), which are equally effective. Corticosteroids alone are ineffective. Supportive care includes respiratory support, management of autonomic dysfunction, VTE prophylaxis, pain control, and multidisciplinary rehabilitation.
Prognosis is generally favourable: 80% walk independently at 6 months, though 20% have significant residual disability, and mortality is 3-5%. Poor prognostic factors include older age, axonal subtype, preceding Campylobacter infection, requirement for ventilation, and low CMAP amplitude on nerve conduction studies."
Key Viva Points
- Definition: Acute immune-mediated polyradiculoneuropathy; ascending weakness + areflexia
- Incidence: 1-2 per 100,000 per year
- Triggers: 50-70% post-infectious (Campylobacter most common); molecular mimicry
- Pathophysiology: Cross-reactive antibodies attack peripheral nerves (demyelination or axonal damage)
- Subtypes: AIDP (most common in West), AMAN/AMSAN (axonal), Miller Fisher (anti-GQ1b)
- Clinical features: Ascending weakness, areflexia, sensory symptoms, autonomic dysfunction
- Respiratory monitoring: FVC every 4-6 hours; intubate if less than 20ml/kg or declining > 30% (20-30-40 rule)
- CSF: Albumino-cytological dissociation (high protein, normal cells); may be normal in first week
- NCS: Demyelinating (AIDP) vs. axonal (AMAN/AMSAN) pattern
- Treatment: IVIG or plasma exchange (equally effective); NOT steroids alone; do NOT combine IVIG + PLEX
- Prognosis: 80% walk at 6 months; 20% disabled; 5% mortality
- Poor prognosis: Older age, axonal subtype, Campylobacter infection, ventilation, low CMAP
Common Viva Questions
Q1. What is the most common preceding infection in GBS? A: Campylobacter jejuni (25-30%), followed by CMV (10-15%) and EBV (10%). [14]
Q2. What is albumino-cytological dissociation? A: Elevated CSF protein with normal white cell count (less than 10 cells/μL), characteristic of GBS. If WCC > 50, consider alternative diagnoses (HIV, Lyme, sarcoid). [7]
Q3. When would you intubate a patient with GBS? A: Absolute indications: FVC less than 15ml/kg, MIP >-30cmH₂O, hypoxia/hypercapnia, inability to clear secretions, severe bulbar weakness. Relative: FVC 15-20ml/kg with declining trend. Aim for elective intubation before respiratory failure. [5]
Q4. What is the difference between AIDP and AMAN? A: AIDP (acute inflammatory demyelinating polyneuropathy) is the demyelinating form (90% in West) with better prognosis due to remyelination. AMAN (acute motor axonal neuropathy) is the axonal motor form (more common in Asia and post-Campylobacter) with slower recovery due to axonal regeneration. [12]
Q5. What antibody is associated with Miller Fisher syndrome? A: Anti-GQ1b antibodies are present in > 90% of Miller Fisher syndrome cases. MFS presents with ataxia, areflexia, and ophthalmoplegia (AAO triad). [9]
Q6. Why should you not give corticosteroids in GBS? A: Cochrane review demonstrated that corticosteroids alone are ineffective in GBS and do not improve outcomes. Combination therapy (IVIG/PLEX + steroids) also shows no added benefit. [6]
Q7. What are poor prognostic factors in GBS? A: Age > 60 years, preceding Campylobacter infection, axonal subtype (AMAN/AMSAN), requirement for mechanical ventilation, inability to walk at 2 weeks, low CMAP amplitude (less than 20% of normal), delayed treatment (> 2 weeks). [11]
Q8. What is the role of complement inhibitors in GBS? A: Eculizumab (anti-C5 antibody) blocks complement activation, which is central to nerve damage in GBS. Phase 2 trials (ICA-GBS) showed a trend towards benefit but did not meet the primary endpoint. Further studies are ongoing. [8]
Clinical Case Scenario (MRCP PACES Style)
Scenario: A 45-year-old man presents with 3 days of progressive leg weakness and difficulty walking. He reports a diarrhoeal illness 2 weeks ago. On examination, he has symmetrical proximal and distal weakness (MRC grade 3/5 in legs, 4/5 in arms), areflexia, and reduced sensation to light touch in a glove-and-stocking distribution.
Task: Discuss your immediate management.
Model Answer:
-
Assess severity and admit: This patient has likely GBS. Assess airway, breathing, circulation. Measure FVC, MIP, MEP. Admit to a monitored bed (HDU/ICU if FVC less than 30ml/kg or rapidly progressive).
-
Confirm diagnosis:
- Lumbar puncture: Look for albumino-cytological dissociation (may be normal in first week; repeat if negative).
- Nerve conduction studies: Confirm GBS and classify subtype (demyelinating vs. axonal).
- Blood tests: FBC, U&E, anti-GQ1b antibodies (to exclude Miller Fisher), serology for CMV/EBV/Mycoplasma, stool culture for Campylobacter.
-
Monitor for complications:
- Respiratory: FVC, MIP, MEP every 4-6 hours. Intubate if FVC less than 20ml/kg or declining > 30%.
- Autonomic: Continuous cardiac monitoring for arrhythmias; monitor BP.
- Mobility: Assess ability to walk (inability to walk unaided = poor prognostic sign).
-
Initiate immunotherapy:
- IVIG 0.4g/kg/day for 5 days OR plasma exchange (5 exchanges over 10-14 days).
- Start as soon as diagnosis is made (ideally within 2 weeks of symptom onset).
-
Supportive care:
- VTE prophylaxis: LMWH + mechanical compression.
- Pain management: Neuropathic pain agents (gabapentin, pregabalin).
- Physiotherapy: Passive range-of-motion exercises to prevent contractures.
- Nutrition: Enteral feeding if dysphagia develops.
- Psychological support: Reassurance about prognosis.
-
Multidisciplinary approach: Involve neurology, critical care, physiotherapy, dietetics, speech and language therapy.
12. Patient/Layperson Explanation
"What is Guillain-Barré syndrome?"
Guillain-Barré syndrome (GBS) is a rare condition where the body's immune system mistakenly attacks the nerves outside the brain and spinal cord (called peripheral nerves). This causes sudden weakness that usually starts in the legs and can spread upwards to the arms and face.
What causes it? GBS often happens after an infection, such as a stomach bug or flu. The infection triggers the immune system, but in some people, the immune system accidentally attacks the nerves instead of just the infection. The most common infection linked to GBS is a bacterial infection called Campylobacter, which causes food poisoning.
What are the symptoms?
- Weakness in the legs that spreads to the arms (over hours to days)
- Numbness or tingling in the hands and feet
- Difficulty walking or standing
- In severe cases, difficulty breathing, swallowing, or facial weakness
Is it serious? Yes, GBS can be serious. About 1 in 4 people need help with breathing and may need a machine (ventilator) to help them breathe. However, with treatment and support, most people recover. About 8 out of 10 people can walk on their own again within 6 months.
How is it treated?
- Immunotherapy: Special treatments to calm down the immune system and stop it attacking the nerves. The two main treatments are:
- "Intravenous immunoglobulin (IVIG): Antibodies given through a drip."
- "Plasma exchange: A procedure to clean the blood."
- Supportive care: Help with breathing if needed, pain relief, and physiotherapy.
What is the outlook? Most people recover, though it can take months. About 60% make a full recovery within a year. Some people are left with weakness or tiredness that lasts longer. Sadly, about 1 in 20 people do not survive, usually due to complications like breathing problems or infections.
What should I do if I think I have GBS? If you develop sudden weakness in your legs or arms, especially after a recent infection, seek urgent medical attention. GBS is a medical emergency and early treatment improves recovery.
13. References
-
Willison HJ, Jacobs BC, van Doorn PA. Guillain-Barré syndrome. Lancet. 2016;388(10045):717-727. doi:10.1016/S0140-6736(16)00339-1. PMID: 26948435
-
Leonhard SE, Papri N, Querol L, Rinaldi S, Shahrizaila N, Jacobs BC. Guillain-Barré syndrome. Nat Rev Dis Primers. 2024;10(1):97. doi:10.1038/s41572-024-00580-4. PMID: 39702645
-
Yuki N, Hartung HP. Guillain-Barré syndrome. N Engl J Med. 2012;366(24):2294-2304. doi:10.1056/NEJMra1114525. PMID: 22738097
-
Davalos L, Kissel JT, Barohn RJ, Dimachkie MM. Guillain-Barré syndrome and variants. Neurol Clin. 2025;43(4):743-760. doi:10.1016/j.ncl.2025.05.011. PMID: 41161994
-
Shang P, Zhu M, Baker M, Feng J, Zhou C, Zhang HL. Mechanical ventilation in Guillain-Barré syndrome. Expert Rev Clin Immunol. 2020;16(11):1053-1064. doi:10.1080/1744666X.2021.1840355. PMID: 33112177
-
Hughes RAC, Swan AV, van Doorn PA. Intravenous immunoglobulin for Guillain-Barré syndrome. Cochrane Database Syst Rev. 2014;(9):CD002063. doi:10.1002/14651858.CD002063.pub6. PMID: 25238327
-
Fokke C, van den Berg B, Drenthen J, Walgaard C, van Doorn PA, Jacobs BC. Diagnosis of Guillain-Barré syndrome and validation of Brighton criteria. Brain. 2014;137(Pt 1):33-43. doi:10.1093/brain/awt285. PMID: 24163275
-
Yao J, Zhou R, Liu Y, Lu Z. Progress in Guillain-Barré syndrome immunotherapy—A narrative review of new strategies in recent years. Hum Vaccin Immunother. 2023;19(2):2215153. doi:10.1080/21645515.2023.2215153. PMID: 37278272
-
Pan J, Zheng N, Yu D, Jiang H, Zhou Y. Unilateral ophthalmoplegia in anti-GQ1b antibody syndrome: case report and systematic literature review. Front Immunol. 2025;16:1669821. doi:10.3389/fimmu.2025.1669821. PMID: 41142776
-
Liu L, Ye Y, Wang L, Song X, Cao J, Qi Y, Xing Y. Nerve ultrasound evaluation of Guillain-Barré syndrome subtypes in northern China. Muscle Nerve. 2021;64(5):560-566. doi:10.1002/mus.27386. PMID: 34355400
-
Van den Berg B, Walgaard C, Drenthen J, Fokke C, Jacobs BC, van Doorn PA. Guillain-Barré syndrome: pathogenesis, diagnosis, treatment and prognosis. Nat Rev Neurol. 2014;10(8):469-482. doi:10.1038/nrneurol.2014.121. PMID: 25023340
-
Uncini A, Kuwabara S. The electrodiagnosis of Guillain-Barré syndrome subtypes: where do we stand? Clin Neurophysiol. 2018;129(12):2586-2593. doi:10.1016/j.clinph.2018.09.025. PMID: 30388583
-
Esposito S, Longo MR. Guillain-Barré syndrome. Autoimmun Rev. 2017;16(1):96-101. doi:10.1016/j.autrev.2016.09.022. PMID: 27666816
-
Paintsil EK, Masanta WO, Dreyer A, et al. Campylobacter in Africa—A specific viewpoint. Eur J Microbiol Immunol (Bp). 2023;13(4):107-124. doi:10.1556/1886.2023.00043. PMID: 38051352
-
Filosto M, Cotti Piccinelli S, Gazzina S, et al. Guillain-Barré syndrome and COVID-19: an observational multicentre study from two Italian hotspot regions. J Neurol Neurosurg Psychiatry. 2021;92(7):751-756. doi:10.1136/jnnp-2020-324837. PMID: 33419890
-
Hadden RDM, Karch H, Hartung HP, et al. Preceding infections, immune factors, and outcome in Guillain-Barré syndrome. Neurology. 2001;56(6):758-765. doi:10.1212/wnl.56.6.758. PMID: 11274311
-
Donofrio PD. Guillain-Barré syndrome. Continuum (Minneap Minn). 2017;23(5):1295-1309. doi:10.1212/CON.0000000000000513. PMID: 28968363
-
Ha LD, Abbas F, Rao M. Guillain-Barré syndrome presenting with sinus node dysfunction and refractory shock. Am J Case Rep. 2017;18:251-254. doi:10.12659/ajcr.902668. PMID: 28283677
-
Athreya Krishnamurthy R, Digpal SS, Rafai I, Kanhangad D. Antibody positive Miller-Fisher syndrome and acute motor sensory axonal neuropathy with respiratory failure: a rare overlap. Cureus. 2025;17(11):e96422. doi:10.7759/cureus.96422. PMID: 41376764
-
Hafsteinsdottir B, Gonzalez-Ortiz F, Gleisner N, et al. Brain-derived tau as an outcome marker in Guillain-Barré syndrome: a retrospective cohort study. Eur J Neurol. 2025;32(4):e70155. doi:10.1111/ene.70155. PMID: 40237241
Last Updated: 2026-01-06
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for guillain-barré syndrome?
Seek immediate emergency care if you experience any of the following warning signs: Respiratory compromise (FVC declining), Rapid progression, Bulbar weakness, Autonomic instability, Unable to walk unaided.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Peripheral Nervous System Anatomy
- Autoimmune Mechanisms
Differentials
Competing diagnoses and look-alikes to compare.
- Myasthenia Gravis
- Botulism
- Acute Transverse Myelitis
- Chronic Inflammatory Demyelinating Polyneuropathy
Consequences
Complications and downstream problems to keep in mind.
- Respiratory Failure
- Autonomic Dysfunction