Heart Failure with Preserved Ejection Fraction (HFpEF)
Heart Failure with Preserved Ejection Fraction (HFpEF) is a clinical syndrome characterised by symptoms and signs of hea... MRCP exam preparation.
What matters first
Heart Failure with Preserved Ejection Fraction (HFpEF) is a clinical syndrome characterised by symptoms and signs of hea... MRCP exam preparation.
Acute flash pulmonary oedema
9 Jan 2025
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Acute flash pulmonary oedema
- Severe hypertensive emergency (BP less than 180/120 with pulmonary congestion)
- New-onset atrial fibrillation with rapid ventricular response
- Hypotension with signs of low cardiac output
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Heart Failure with Reduced Ejection Fraction (HFrEF)
- Cardiac Amyloidosis
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Heart Failure with Preserved Ejection Fraction (HFpEF)
1. Topic Overview
Summary
Heart Failure with Preserved Ejection Fraction (HFpEF) is a clinical syndrome characterised by symptoms and signs of heart failure with a left ventricular ejection fraction (LVEF) ≥50% and objective evidence of diastolic dysfunction or elevated cardiac filling pressures. HFpEF accounts for approximately 50% of all heart failure cases, with prevalence increasing due to ageing populations and rising rates of obesity, hypertension, and diabetes. [1,2]
Unlike HFrEF, where systolic dysfunction drives the pathophysiology and therapies targeting neurohormonal activation have proven highly effective, HFpEF represents a fundamentally different disease state. The underlying problem is diastolic dysfunction—impaired ventricular relaxation and increased myocardial stiffness—which leads to elevated filling pressures despite preserved contractile function. [3]
For decades, HFpEF lacked evidence-based therapies that reduced mortality or hospitalisations. This changed with the EMPEROR-Preserved (2021) and DELIVER (2022) trials, which demonstrated that SGLT2 inhibitors (empagliflozin and dapagliflozin) significantly reduce heart failure hospitalisations and cardiovascular death, establishing the first Class I recommendation for pharmacological therapy in HFpEF. [4,5]
Management of HFpEF is multifaceted, focusing on three key pillars: (1) treatment of congestion with diuretics, (2) aggressive management of comorbidities (hypertension, diabetes, obesity, atrial fibrillation), and (3) use of SGLT2 inhibitors. Prognosis remains poor, with 5-year mortality rates of 50-60%, similar to HFrEF but with a higher proportion of non-cardiovascular deaths. [6,7]
Key Facts
| Feature | Details |
|---|---|
| Definition | LVEF ≥50% + symptoms/signs of HF + elevated filling pressures or diastolic dysfunction |
| Prevalence | ~50% of all heart failure patients; increasing with population ageing |
| Typical Patient Profile | Elderly (mean age 70-80 years), female predominance (60-65%), multiple comorbidities |
| Mean Comorbidities | Average 5+ concurrent conditions |
| 5-Year Mortality | 50-60% (comparable to HFrEF) |
| Pathophysiology | Diastolic dysfunction, myocardial stiffness, systemic inflammation, microvascular dysfunction |
| Diagnostic Challenge | Requires elevated natriuretic peptides AND/OR echocardiographic/invasive evidence |
| Treatment Breakthrough | SGLT2 inhibitors (empagliflozin, dapagliflozin) - first proven therapy |
| Phenotypic Heterogeneity | Multiple distinct phenotypes (obesity, ischaemic, fibrotic, pulmonary vascular) |
Clinical Pearls
High-Yield Examination Points:
- Diagnosis is challenging: HFpEF is a diagnosis of exclusion requiring symptoms + LVEF ≥50% + elevated natriuretic peptides AND/OR diastolic dysfunction on echo/invasive testing
- Natriuretic peptides lower than HFrEF: Use appropriate thresholds (NT-proBNP > 125 pg/mL in sinus rhythm, > 365 pg/mL in AF); obesity reduces BNP levels
- HFA-PEFF score is key: 6-point diagnostic algorithm (functional + morphological + biomarker domains) with score ≥5 confirming HFpEF
- SGLT2 inhibitors are Class I: Empagliflozin or dapagliflozin reduce HF hospitalisations by ~20% regardless of diabetes status
- Comorbidity treatment is cornerstone: Hypertension, obesity, diabetes, AF, CAD all drive pathophysiology and must be aggressively managed
- What doesn't work: ACE-I/ARB, beta-blockers, and ARNI have NOT shown mortality benefit in HFpEF (unlike HFrEF)
- Phenotypes matter: Obesity phenotype may respond to weight loss/GLP-1 agonists; ischaemic phenotype may need revascularisation
- Exercise intolerance disproportionate: Symptoms often worse than resting echo findings suggest; exercise testing reveals abnormal haemodynamics
- AF is common and problematic: Present in 30-50%; manage with rate control and anticoagulation (rhythm control not superior)
- Prognosis similar to HFrEF: Despite preserved EF, 5-year mortality is 50-60%; most deaths are non-cardiovascular
Why This Matters Clinically
HFpEF represents one of the greatest challenges in modern cardiology and is a major public health burden. With ageing populations, HFpEF prevalence is projected to increase substantially, yet until recently there were no proven therapies to reduce mortality or hospitalisations. [8]
The condition primarily affects elderly patients with multiple comorbidities, making diagnosis complex and management challenging. Unlike HFrEF, where LVEF provides an objective measure of disease severity, HFpEF diagnosis requires integration of clinical, biomarker, and echocardiographic data—often with significant overlap with other conditions such as obesity, deconditioning, and pulmonary disease. [9]
Understanding the pathophysiology of HFpEF as a systemic inflammatory disorder driven by comorbidities (rather than a primary cardiac problem) is essential for effective management. The breakthrough SGLT2 inhibitor trials have finally provided evidence-based therapy, but outcomes remain poor, emphasising the need for early diagnosis, aggressive comorbidity management, and holistic patient care. [10]
2. Epidemiology
Incidence and Prevalence
HFpEF accounts for approximately 50% of all heart failure cases in developed countries, with this proportion increasing over time. [1,11] Population-based studies demonstrate significant variation based on age, sex, and comorbidity burden.
| Metric | Value | Notes |
|---|---|---|
| Proportion of HF Cases | 45-55% | Increasing over time; now equals or exceeds HFrEF |
| Prevalence (General Population) | 1-2% | Age-dependent; increases exponentially with age |
| Prevalence (Age > 65 years) | 4-6% | Particularly high in women |
| Prevalence (Age > 80 years) | 10-12% | Elderly most affected |
| Annual Incidence | 1-3 per 1000 person-years | Age-adjusted; higher in women over 65 |
| Hospital Admissions | ~50% of HF admissions | Equal to HFrEF; readmission rates 20-30% at 30 days |
| Global Burden | > 32 million HF patients worldwide | HFpEF represents ~16 million patients |
The prevalence of HFpEF is increasing at approximately 1% per year, driven by population ageing, improved HFrEF survival (transition from HFrEF to HFpEF/HFmrEF), and rising obesity and diabetes rates. [12,13]
Demographics
Age:
- Mean age at diagnosis: 70-80 years (approximately 5-10 years older than HFrEF patients)
- Age is a strong independent risk factor for HFpEF development
- Elderly patients (> 80 years) have particularly high prevalence and worse outcomes [14]
Sex:
- Female predominance: 60-65% of HFpEF patients are women
- Women develop HFpEF at older ages than men but have similar mortality
- Sex differences in pathophysiology: women have more concentric remodelling, men more ischaemic disease [15]
Ethnicity:
- Higher rates in African Americans compared to Caucasians
- African American patients present at younger ages with worse outcomes
- South Asian populations have high rates of metabolic syndrome-associated HFpEF
- Ethnic differences in comorbidity burden and phenotypic expression [16]
Socioeconomic Factors:
- Lower socioeconomic status associated with higher prevalence
- Healthcare access disparities affect diagnosis and outcomes
- Education level inversely correlated with HFpEF risk
Risk Factors
HFpEF is strongly associated with multiple systemic comorbidities. Most patients have 5 or more concurrent conditions, leading to the conceptualisation of HFpEF as a multisystem disorder rather than an isolated cardiac disease. [17]
Cardiovascular Risk Factors (Prevalence in HFpEF Cohorts):
| Risk Factor | Prevalence | Strength of Association |
|---|---|---|
| Hypertension | 80-95% | Very strong; most important modifiable risk factor |
| Obesity (BMI > 30) | 50-80% | Very strong; mechanistically central to pathophysiology |
| Diabetes Mellitus | 30-50% | Strong; associated with worse prognosis |
| Atrial Fibrillation | 30-50% | Strong; bidirectional relationship (cause and effect) |
| Coronary Artery Disease | 40-60% | Moderate-strong; microvascular dysfunction important |
| Chronic Kidney Disease | 30-50% | Strong; cardiorenal syndrome common |
| Metabolic Syndrome | 60-70% | Very strong; clustering of obesity, hypertension, diabetes |
Other Important Associations:
- Ageing: Independent of comorbidities; age-related myocardial stiffening
- Female sex: Hormonal factors, different response to hypertension
- Sedentary lifestyle: Physical inactivity promotes metabolic dysfunction
- Obstructive sleep apnoea: Present in 50-70% of HFpEF patients; contributes to hypertension and diastolic dysfunction [18]
- Anaemia: Common (20-40%); contributes to symptoms and worse outcomes
- COPD: Present in 20-30%; complicates diagnosis due to overlapping dyspnoea
- Depression: Affects 30-40%; impacts quality of life and adherence
- Frailty: Increasingly recognised; associated with worse outcomes and functional decline
Mechanistic Risk Factors:
- Systemic inflammation: Obesity, diabetes, autoimmune conditions drive inflammatory state
- Endothelial dysfunction: Precedes clinical HFpEF; affects microvasculature
- Insulin resistance: Central to metabolic phenotype
- Volume overload states: Renal disease, dietary sodium excess
Temporal Trends
The epidemiology of HFpEF has changed significantly over the past two decades:
- Increasing proportion of HF: HFpEF has increased from ~40% to > 50% of all HF cases
- Shifting phenotypes: Ischaemic HF declining; metabolic/obesity phenotypes increasing
- Survival improvements: HFrEF mortality declining faster than HFpEF (due to effective therapies)
- Transition patients: Improved HFrEF survival leads to EF recovery and transition to HFpEF/HFmrEF ("phenotype drift")
- Healthcare burden: HFpEF now represents majority of HF hospitalisations in elderly populations [19]
3. Pathophysiology
Paradigm Shift: From "Diastolic Dysfunction" to "Systemic Inflammatory Disorder"
Historically, HFpEF was conceptualised as isolated "diastolic heart failure" with impaired ventricular relaxation. This view has evolved dramatically over the past decade. HFpEF is now understood as a systemic inflammatory disorder driven by multiple comorbidities that cause endothelial dysfunction, microvascular inflammation, and ultimately myocardial structural and functional changes. [3,20]
The Paulus-Tschöpe paradigm (2013) revolutionised understanding of HFpEF pathophysiology, proposing a mechanistic sequence from comorbidity burden to myocardial disease. [20]
The Pathophysiological Cascade
Stage 1: Comorbidity-Induced Systemic Inflammation
Multiple comorbidities (obesity, diabetes, hypertension, metabolic syndrome) create a pro-inflammatory state characterised by:
- Elevated circulating cytokines (IL-6, TNF-α, IL-1β)
- Oxidative stress and reactive oxygen species production
- Activation of NLRP3 inflammasome
- Endothelial activation with upregulation of adhesion molecules (VCAM-1, ICAM-1) [20,21]
Stage 2: Coronary Microvascular Endothelial Dysfunction
Systemic inflammation affects the coronary microcirculation:
- Reduced nitric oxide (NO) bioavailability due to endothelial inflammation
- Impaired endothelium-dependent vasodilation
- Increased endothelial permeability and leucocyte infiltration
- Microvascular rarefaction (reduced capillary density)
- Impaired coronary flow reserve despite normal epicardial coronary arteries [22]
Stage 3: Cardiomyocyte Dysfunction
Microvascular dysfunction leads to cardiomyocyte-level changes:
a) Titin Dysfunction:
- Titin is a giant sarcomeric protein providing passive myocardial elasticity
- Reduced protein kinase G (PKG) activity (due to low NO) leads to titin hypophosphorylation
- Hypophosphorylated titin is stiffer, increasing passive cardiomyocyte stiffness
- This is a key reversible mechanism of diastolic dysfunction [23]
b) Hypertrophy:
- Concentric left ventricular hypertrophy (LVH) develops in response to hypertension
- Increased wall stress triggers myocyte hypertrophy
- Cardiomyocyte cross-sectional area increases
- Contributes to reduced chamber compliance
c) Metabolic Derangements:
- Impaired myocardial glucose and fatty acid metabolism
- Mitochondrial dysfunction and reduced ATP production
- Accumulation of advanced glycation end-products (AGEs) in diabetic patients
- Altered calcium handling affecting relaxation [24]
Stage 4: Extracellular Matrix Remodelling
Chronic inflammation drives fibrosis:
- Cardiac fibroblast activation and transformation to myofibroblasts
- Increased collagen deposition (types I and III)
- Matrix metalloproteinase (MMP) imbalance favouring fibrosis
- Perivascular and interstitial fibrosis increase chamber stiffness
- Replacement fibrosis in areas of microvascular ischaemia [25]
Stage 5: Left Atrial Remodelling
The left atrium faces chronically elevated pressures:
- LA dilation (left atrial volume index > 34 mL/m²)
- LA fibrosis and electrical remodelling
- Impaired LA reservoir, conduit, and booster pump function
- Increased susceptibility to atrial fibrillation
- AF further impairs diastolic filling and worsens HFpEF [26]
Stage 6: Pulmonary Vascular Remodelling
Chronically elevated LA pressures cause pulmonary consequences:
- Pulmonary venous hypertension (post-capillary)
- Over time, pulmonary arterial remodelling develops (combined pre- and post-capillary PH)
- Right ventricular pressure overload
- RV dysfunction and remodelling
- Tricuspid regurgitation worsens RV function
- Severe RV dysfunction confers very poor prognosis [27]
Diastolic Dysfunction: The Final Common Pathway
All the above mechanisms converge to produce diastolic dysfunction, characterised by:
1. Impaired Active Relaxation:
- Energy-dependent process requiring ATP-dependent calcium reuptake into sarcoplasmic reticulum
- Slowed relaxation prolongs isovolumetric relaxation time (IVRT)
- Reduced early diastolic filling (reduced E wave velocity)
- Echocardiographic pattern: impaired relaxation (Grade I diastolic dysfunction)
2. Increased Passive Stiffness:
- Structural changes (fibrosis, hypertrophy, titin hypophosphorylation) increase chamber stiffness
- Reduced chamber compliance means higher pressures needed for equivalent filling volumes
- Steep end-diastolic pressure-volume relationship
- Even small volume changes cause large pressure increases
3. Elevated Filling Pressures:
- Combination of impaired relaxation and increased stiffness
- Elevated left ventricular end-diastolic pressure (LVEDP > 16 mmHg)
- Elevated LA pressure (mean LAP > 12 mmHg at rest, > 25 mmHg with exercise)
- Pulmonary venous pressure rises, causing dyspnoea and pulmonary congestion
- Reduced filling during diastole impairs cardiac output, especially with exertion [28]
Systolic-Diastolic Coupling
While LVEF is preserved (≥50%), subtle systolic dysfunction exists in HFpEF:
- Reduced longitudinal strain (global longitudinal strain less negative than normal)
- Impaired myocardial contractile reserve (limited ability to increase EF with exercise)
- Ventricular-arterial uncoupling (stiff ventricle interacting with stiff vasculature)
- These abnormalities worsen exercise capacity despite "normal" resting EF [29]
Exercise Intolerance: Integrative Pathophysiology
HFpEF patients have marked exercise intolerance disproportionate to resting haemodynamics:
Cardiac Factors:
- Inability to augment cardiac output normally with exercise (chronotropic incompetence, impaired stroke volume reserve)
- Exaggerated rise in filling pressures with minimal exertion
- Atrial fibrillation limits rate response and eliminates atrial kick
- Pulmonary hypertension and RV dysfunction limit cardiac output
Peripheral Factors:
- Skeletal muscle abnormalities (fibre-type shift, mitochondrial dysfunction, capillary rarefaction)
- Reduced peripheral oxygen extraction
- Impaired vasodilatory capacity
- Deconditioning exacerbates myopathy
- Anaemia reduces oxygen delivery [30]
Ventilatory Factors:
- High ventilatory equivalent for CO₂ (VE/VCO₂)
- Abnormal ventilatory response to exercise
- Pulmonary congestion and reduced diffusion capacity
- COPD co-existence
Phenotypic Heterogeneity
HFpEF is not a single disease but rather a clinical syndrome with multiple distinct phenotypes, each with different dominant mechanisms: [17,31]
1. Metabolic/Obesity Phenotype:
- Central obesity, insulin resistance, metabolic syndrome
- High plasma volume and systemic venous congestion
- Epicardial and pericardial adiposity
- Systemic inflammation driven by adipokines
- May respond to weight loss and GLP-1 receptor agonists
2. Ischaemic/CAD Phenotype:
- Prior myocardial infarction, obstructive CAD
- Microvascular dysfunction and impaired coronary flow reserve
- Regional wall motion abnormalities and replacement fibrosis
- May benefit from revascularisation if ischaemia demonstrated
3. Elderly/Fibrotic Phenotype:
- Advanced age, frailty, multimorbidity
- Diffuse myocardial fibrosis and myocardial stiffness
- Chronotropic incompetence
- LA fibrosis and AF
- Poorest functional capacity and prognosis
4. Pulmonary Vascular Phenotype:
- Elevated pulmonary vascular resistance
- RV dysfunction and remodelling
- Severe exercise intolerance
- High mortality; may need pulmonary vasodilators
5. Hypertensive Phenotype:
- Long-standing hypertension
- Concentric LVH
- Good blood pressure control may improve symptoms
- Responsive to afterload reduction
6. Atrial Fibrillation-Dominant Phenotype:
- AF as primary driver of symptoms
- Loss of atrial contraction worsens filling
- Rate control and rhythm management critical
Phenotyping is clinically important because targeted therapy based on dominant phenotype may improve outcomes. [32]
4. Clinical Presentation
Typical Presentation
The classic HFpEF patient is an elderly, obese, hypertensive woman with diabetes presenting with progressive exertional dyspnoea and exercise intolerance. [14]
Cardinal Symptoms:
| Symptom | Frequency | Characteristics |
|---|---|---|
| Exertional Dyspnoea | > 90% | Progressive, disproportionate to cardiac findings, worse than HFrEF for same NYHA class |
| Exercise Intolerance | > 85% | Severe fatigue, leg heaviness, reduced functional capacity |
| Orthopnoea | 40-60% | Less prominent than HFrEF; develops later in disease course |
| Paroxysmal Nocturnal Dyspnoea | 30-40% | Less common than HFrEF |
| Peripheral Oedema | 50-70% | Ankle and lower leg swelling, worse at end of day |
| Abdominal Bloating/Discomfort | 40-50% | Hepatic congestion, ascites in advanced cases |
| Nocturia | 50-60% | Redistribution of fluid while supine |
| Palpitations | 30-50% | Often due to paroxysmal or permanent AF |
Physical Examination Findings:
General Inspection:
- Obesity (BMI > 30 kg/m² in 50-80%)
- Respiratory distress with minimal exertion
- Peripheral oedema (pitting ankle and lower leg oedema)
- Elevated body mass index and central adiposity
Cardiovascular Examination:
| Sign | Frequency | Significance |
|---|---|---|
| Elevated Blood Pressure | 60-80% | Often hypertensive at presentation; target less than 130/80 mmHg |
| Elevated JVP | 30-50% at rest | May only become elevated with exercise or hepatojugular reflux |
| S4 Gallop | 40-60% | Suggests impaired LV compliance; stiff ventricle requires atrial "kick" |
| S3 Gallop | less than 20% | Less common than HFrEF; suggests severe diastolic dysfunction |
| Displaced Apex Beat | Uncommon | LV usually normal size or small; concentric hypertrophy |
| Murmurs | 30-40% | Functional mitral regurgitation; aortic stenosis (mimic/cause) |
| Atrial Fibrillation | 30-50% | Irregularly irregular pulse; loss of 'a' wave in JVP |
Respiratory Examination:
- Bibasal crackles: May be present but often absent at rest (appear with exertion or fluid overload)
- Pleural effusions: In decompensated HFpEF
- Wheezing: Cardiac asthma in pulmonary oedema
Abdominal Examination:
- Hepatomegaly: Tender, pulsatile liver in severe RV failure
- Ascites: Advanced right-sided failure
- Hepatojugular reflux: Positive test suggests elevated right atrial pressure
Peripheral Examination:
- Dependent oedema: Pitting oedema of ankles, legs, sacrum (if bedbound)
- Pulses: Usually normal; assess for peripheral arterial disease
- Skin changes: Venous stasis dermatitis in chronic oedema
Atypical Presentations
1. Flash Pulmonary Oedema:
- Sudden onset severe dyspnoea with frothy pink sputum
- Often triggered by acute hypertensive crisis (BP > 180/110 mmHg)
- Relatively preserved cardiac output but severe pulmonary congestion
- May require non-invasive ventilation (CPAP/BiPAP)
- Distinguished from HFrEF by preserved EF and hypertension [33]
2. Exercise-Induced Symptoms with Normal Resting Findings:
- Completely normal physical examination at rest
- Normal or near-normal resting echocardiography
- Symptoms only manifest with exertion
- Requires exercise echocardiography or invasive exercise haemodynamic testing
- Elevated PCWP with exercise (> 25 mmHg) confirms HFpEF [34]
3. Obesity-Attributed Dyspnoea:
- Symptoms incorrectly attributed to deconditioning or obesity alone
- Elevated natriuretic peptides provide diagnostic clue
- High BMI paradoxically suppresses BNP levels (lower threshold needed)
- Exercise testing reveals cardiac limitation (not just deconditioning)
4. AF-Dominant Presentation:
- New-onset or poorly controlled atrial fibrillation
- Rapid ventricular rate (> 110 bpm) causes symptoms
- AF may be both cause and consequence of HFpEF
- Rate control and anticoagulation critical
5. Right-Sided Heart Failure:
- Predominant peripheral oedema, hepatomegaly, ascites
- Pulmonary hypertension and RV dysfunction
- Minimal lung findings
- Requires differentiation from isolated right heart disease
6. Elderly Frailty Syndrome:
- Gradual functional decline and fatigue
- Minimal dyspnoea at rest due to sedentary lifestyle
- Multimorbidity obscures diagnosis
- Symptoms attributed to "old age" rather than HFpEF
Symptom Severity Classification
NYHA Functional Classification:
| Class | Description | Typical HFpEF Patient |
|---|---|---|
| I | No limitation; ordinary activity does not cause symptoms | Rare; early HFpEF or compensated |
| II | Slight limitation; comfortable at rest; ordinary activity causes dyspnoea/fatigue | Most common presentation; symptomatic on moderate exertion |
| III | Marked limitation; comfortable at rest; less than ordinary activity causes symptoms | Advanced HFpEF; dyspnoea on minimal exertion (walking short distances) |
| IV | Unable to carry out any activity without symptoms; symptoms at rest | Severe HFpEF; often hospitalised; refractory symptoms |
HFpEF patients often report more severe symptoms for a given NYHA class compared to HFrEF patients, possibly due to chronotropic incompetence, pulmonary vascular disease, and peripheral factors. [35]
Red Flags Requiring Urgent Assessment
IMMEDIATE EVALUATION NEEDED:
- Acute pulmonary oedema: Severe dyspnoea, hypoxia, pink frothy sputum, widespread crackles
- Hypertensive emergency with pulmonary congestion: BP > 180/120 mmHg + respiratory distress
- Rapid atrial fibrillation with haemodynamic compromise: Heart rate > 130 bpm, hypotension, chest pain
- Syncope: May indicate ventricular arrhythmia, complete heart block, or severe aortic stenosis
- Signs of cardiogenic shock: Hypotension, cool peripheries, oliguria, altered mental status
- Acute kidney injury with fluid overload: Rising creatinine, oliguria, worsening oedema despite diuretics
- Chest pain suggestive of ACS: Rule out acute coronary syndrome as precipitant
5. Differential Diagnosis
HFpEF is a diagnosis of exclusion. Many conditions mimic or coexist with HFpEF. Careful evaluation is required to distinguish true HFpEF from alternative diagnoses. [9]
Cardiac Causes of Dyspnoea with Preserved EF
| Condition | Key Distinguishing Features | Investigations |
|---|---|---|
| Cardiac Amyloidosis | Thick LV walls + low voltage ECG; restrictive physiology; multisystem involvement (neuropathy, renal) | Echocardiographic "sparkling" appearance, strain pattern (apical sparing), technetium-99m PYP scan, serum-free light chains, cardiac MRI with LGE |
| Constrictive Pericarditis | Prior pericarditis, cardiac surgery, or radiation; rapid x and y descents on JVP; pericardial calcification on CT | CT/MRI showing thickened pericardium, septal bounce on echo, respiratory variation in mitral inflow, invasive haemodynamics (equalisation of diastolic pressures) |
| Hypertrophic Cardiomyopathy (HCM) | Family history, asymmetric septal hypertrophy (≥15 mm), SAM of mitral valve, dynamic LVOT obstruction | ECG (LVH, deep Q waves), echo (ASH, SAM, gradient), cardiac MRI, genetic testing |
| Aortic Stenosis (Severe) | Systolic ejection murmur, low pulse pressure, concentric LVH, critical valve area less than 1.0 cm² | Echocardiography with Doppler (valve area, mean gradient > 40 mmHg), CT calcium score |
| Restrictive Cardiomyopathy | Infiltrative diseases (amyloid, sarcoid, haemochromatosis); biatrial enlargement; normal LV size | Endomyocardial biopsy (gold standard), cardiac MRI with tissue characterisation, specific biomarkers |
| High-Output Heart Failure | Hyperdynamic circulation (tachycardia, bounding pulses, wide pulse pressure); specific cause (anaemia, thyrotoxicosis, AV fistula, Paget's disease) | High cardiac output on echo/invasive testing, identify underlying cause |
Non-Cardiac Causes of Dyspnoea
| Condition | Key Features | Investigations |
|---|---|---|
| COPD/Emphysema | Smoking history, obstructive spirometry, hyperinflation, low DLCO | Spirometry (FEV1/FVC less than 0.7), chest X-ray/CT (hyperinflation, bullae), low BNP |
| Pulmonary Fibrosis (ILD) | Restrictive physiology, bibasal crackles, clubbing, dry cough | HRCT (reticular opacities, honeycombing), restrictive spirometry, low DLCO |
| Pulmonary Hypertension (PAH) | Younger, female, loud P2, elevated JVP, RV heave, group 1 PH risk factors | Echocardiography (high RVSP, RV dilation), right heart catheterisation (mPAP ≥25 mmHg, PCWP less than 15 mmHg), V/Q scan |
| Obesity Hypoventilation Syndrome | Severe obesity (BMI > 35), hypercapnia, daytime somnolence, OSA | ABG (↑PaCO₂, ↓PaO₂), overnight oximetry/sleep study, low BNP |
| Deconditioning | Sedentary lifestyle, normal cardiac and pulmonary testing, low peak VO₂ without cardiac limitation | Cardiopulmonary exercise testing (RER > 1.1, no O₂ pulse plateau, normal VE/VCO₂ slope) |
| Anaemia | Fatigue, pallor, low haemoglobin (less than 10 g/dL) | Full blood count, iron studies, B12/folate |
| Thyroid Disease | Hyperthyroidism (weight loss, tremor, AF) or hypothyroidism (weight gain, bradycardia) | Thyroid function tests (TSH, free T4) |
Conditions Commonly Coexisting with HFpEF
Many patients have multiple concurrent diagnoses, making management complex:
- HFpEF + COPD (20-30%)
- HFpEF + Obesity hypoventilation syndrome (15-25%)
- HFpEF + Pulmonary hypertension (30-40%)
- HFpEF + Atrial fibrillation (30-50%)
- HFpEF + Chronic kidney disease (30-50%)
Coexistence does not exclude HFpEF; both conditions may require treatment. [36]
6. Investigations
Diagnostic Approach
The 2019 HFA-PEFF diagnostic algorithm provides a stepwise approach to HFpEF diagnosis, integrating clinical, echocardiographic, and biomarker data. [9]
Step 1: Pre-Test Assessment (P-Step)
Establish clinical suspicion for HFpEF. Requires:
- Symptoms and/or signs of heart failure
- Preserved LVEF: ≥50%
- Absence of alternative diagnosis explaining symptoms
Calculate HFA-PEFF Pre-test Score:
- Major criteria (2 points each): AF, BMI > 30 kg/m²
- Minor criteria (1 point each): Age > 60, hypertension requiring ≥2 drugs, diabetes, OSA
Score:
- 0-1 points: HFpEF unlikely; consider alternative diagnoses
- ≥2 points: Proceed to Step 2 (functional/morphological/biomarker assessment)
Step 2: Echocardiography and Natriuretic Peptides (E and F Steps)
HFA-PEFF Diagnostic Score integrates three domains:
Domain 1: Functional (Diastolic Function)
| Parameter | Major Criterion (2 points) | Minor Criterion (1 point) |
|---|---|---|
| Septal e' velocity | less than 7 cm/s | 7-9 cm/s |
| Lateral e' velocity | less than 10 cm/s | 10-12 cm/s |
| Average E/e' ratio | ≥15 | 9-14 |
| TR velocity | > 2.8 m/s | 2.5-2.8 m/s |
| Global Longitudinal Strain | less than 16% (absolute value) | - |
e' velocity: Tissue Doppler imaging of mitral annulus; reflects active LV relaxation (reduced in diastolic dysfunction) E/e' ratio: Estimates LV filling pressures; E/e' ≥15 strongly suggests elevated LAP TR velocity: Reflects pulmonary artery systolic pressure; elevated in PH secondary to HFpEF
Domain 2: Morphological (Structural Changes)
| Parameter | Major Criterion (2 points) | Minor Criterion (1 point) |
|---|---|---|
| LA volume index (LAVi) | > 34 mL/m² | 29-34 mL/m² |
| LV mass index (LVMi) | > 149 g/m² (men), > 122 g/m² (women) | 115-149 (men), 95-122 (women) |
| RWT (relative wall thickness) | > 0.42 | - |
| LV wall thickness | ≥12 mm | - |
LAVi: LA dilation reflects chronic elevation of LA pressure; most sensitive marker of diastolic dysfunction chronicity LVMi: Concentric LVH common in hypertensive HFpEF RWT: (2 × posterior wall thickness)/LV internal diameter; > 0.42 indicates concentric remodelling
Domain 3: Biomarkers
| Parameter | Sinus Rhythm | Atrial Fibrillation |
|---|---|---|
| NT-proBNP (Major) | > 220 pg/mL | > 660 pg/mL |
| NT-proBNP (Minor) | 125-220 pg/mL | 365-660 pg/mL |
| BNP (Major) | > 80 pg/mL | > 240 pg/mL |
| BNP (Minor) | 35-80 pg/mL | 105-240 pg/mL |
Important Considerations:
- Obesity reduces natriuretic peptide levels; use lower thresholds if BMI > 35 kg/m²
- Atrial fibrillation increases natriuretic peptides; higher thresholds applied
- Renal dysfunction elevates NT-proBNP (less affected for BNP)
- Age increases baseline levels (higher in elderly)
HFA-PEFF Score Interpretation:
| Total Score | Interpretation | Next Steps |
|---|---|---|
| 0-1 points | HFpEF unlikely | Consider alternative diagnoses; stop workup |
| 2-4 points | Intermediate probability | Proceed to Step 3: Functional testing (exercise echo, stress testing, or invasive haemodynamics) |
| 5-6 points | HFpEF confirmed | Diagnose HFpEF; initiate treatment |
Step 3: Functional/Stress Testing (If Score 2-4)
When HFA-PEFF score is 2-4, additional testing to provoke elevation in filling pressures:
a) Exercise Echocardiography:
- Diastolic parameters measured at rest and peak exercise (bicycle or treadmill)
- Abnormal response: E/e' > 15, septal e' less than 7 cm/s, TR velocity > 2.8 m/s at peak exercise
- Demonstrates exercise-induced elevation in filling pressures [34]
b) Invasive Exercise Haemodynamics (Gold Standard):
- Right heart catheterisation with exercise (supine bicycle)
- Diagnostic criteria: PCWP > 25 mmHg at peak exercise OR PCWP > 15 mmHg at rest
- Confirms HFpEF definitively
- Particularly useful when diagnosis uncertain [37]
c) Diastolic Stress Test:
- Pharmacological stress (dobutamine) with echo
- Less validated than exercise but useful if patient cannot exercise
Core Investigations
| Investigation | Findings in HFpEF | Purpose |
|---|---|---|
| ECG | LVH (Sokolow-Lyon, Cornell criteria), LAE (P mitrale), AF, non-specific ST-T changes | Identify arrhythmias, ischaemia, LVH |
| Chest X-ray | Normal heart size or mild cardiomegaly, pulmonary venous congestion, Kerley B lines, pleural effusions | Assess pulmonary congestion, exclude lung pathology |
| Transthoracic Echo | LVEF ≥50%, diastolic dysfunction, LAE, LVH, assess valves, estimate RVSP | Cornerstone of diagnosis; comprehensive structural/functional assessment |
| NT-proBNP/BNP | Elevated but often lower than HFrEF (NT-proBNP > 125 pg/mL in SR, > 365 in AF) | Diagnosis, rule-out (high NPV), prognosis |
| Full Blood Count | Anaemia (Hb less than 12 g/dL women, less than 13 men) in 20-40% | Identify anaemia as contributor to symptoms |
| Renal Function (eGFR, creatinine) | CKD common (eGFR less than 60 in 30-50%) | Cardiorenal syndrome; affects prognosis and diuretic dosing |
| Electrolytes | Hypokalaemia (diuretics), hyperkalaemia (ACE-I/ARB, CKD) | Monitor with diuretics and RAAS inhibitors |
| HbA1c/Glucose | Diabetes in 30-50% | Screen for diabetes; guide SGLT2i use |
| Lipid Profile | Assess cardiovascular risk | Optimise statin therapy |
| Thyroid Function (TSH) | Exclude hyper/hypothyroidism | AF risk, rule out thyroid-related symptoms |
| Liver Function Tests | Hepatic congestion (elevated bilirubin, ALT/AST) | Assess RV failure severity |
Advanced/Specialist Investigations
| Investigation | Indication | Findings |
|---|---|---|
| Cardiac MRI | Unclear diagnosis, suspected infiltrative disease, assess fibrosis | LGE pattern (amyloid, sarcoid), quantify fibrosis (ECV), rule out HCM |
| Cardiopulmonary Exercise Testing (CPET) | Quantify exercise limitation, differentiate cardiac vs. pulmonary vs. deconditioning | Reduced peak VO₂, elevated VE/VCO₂ slope, reduced O₂ pulse, low anaerobic threshold |
| Right Heart Catheterisation | Diagnostic uncertainty, assessment of PH, pre-transplant evaluation | Elevated PCWP (> 15 mmHg rest, > 25 mmHg exercise), assess PVR, CO |
| Coronary Angiography | Exclude obstructive CAD, chest pain, prior MI | Assess for revascularisation targets |
| Genetic Testing | Family history of cardiomyopathy, young onset, unexplained LVH | HCM, amyloidosis, Fabry disease genes |
| Endomyocardial Biopsy | Suspected infiltrative disease (amyloid, sarcoid), unclear restrictive cardiomyopathy | Histological diagnosis (rarely needed) |
| Technetium-99m PYP Scan | Suspected cardiac amyloidosis (thick walls, low voltage ECG, neuropathy) | Positive uptake (Perugini grade 2-3) in ATTR amyloidosis |
| Sleep Study (Polysomnography) | Suspected OSA (snoring, daytime somnolence, obesity) | AHI ≥5 confirms OSA; common in HFpEF (50-70%) |
Diagnostic Pitfalls
Common Errors:
- Diagnosing HFpEF based on symptoms + LVEF ≥50% alone (must have elevated natriuretic peptides or diastolic dysfunction)
- Ignoring alternative diagnoses (amyloidosis, constrictive pericarditis, aortic stenosis)
- Over-relying on resting echo (exercise testing often needed)
- Using wrong BNP thresholds (obesity lowers BNP; AF raises it)
- Missing concomitant pathology (HFpEF + COPD, HFpEF + pulmonary hypertension)
7. Management
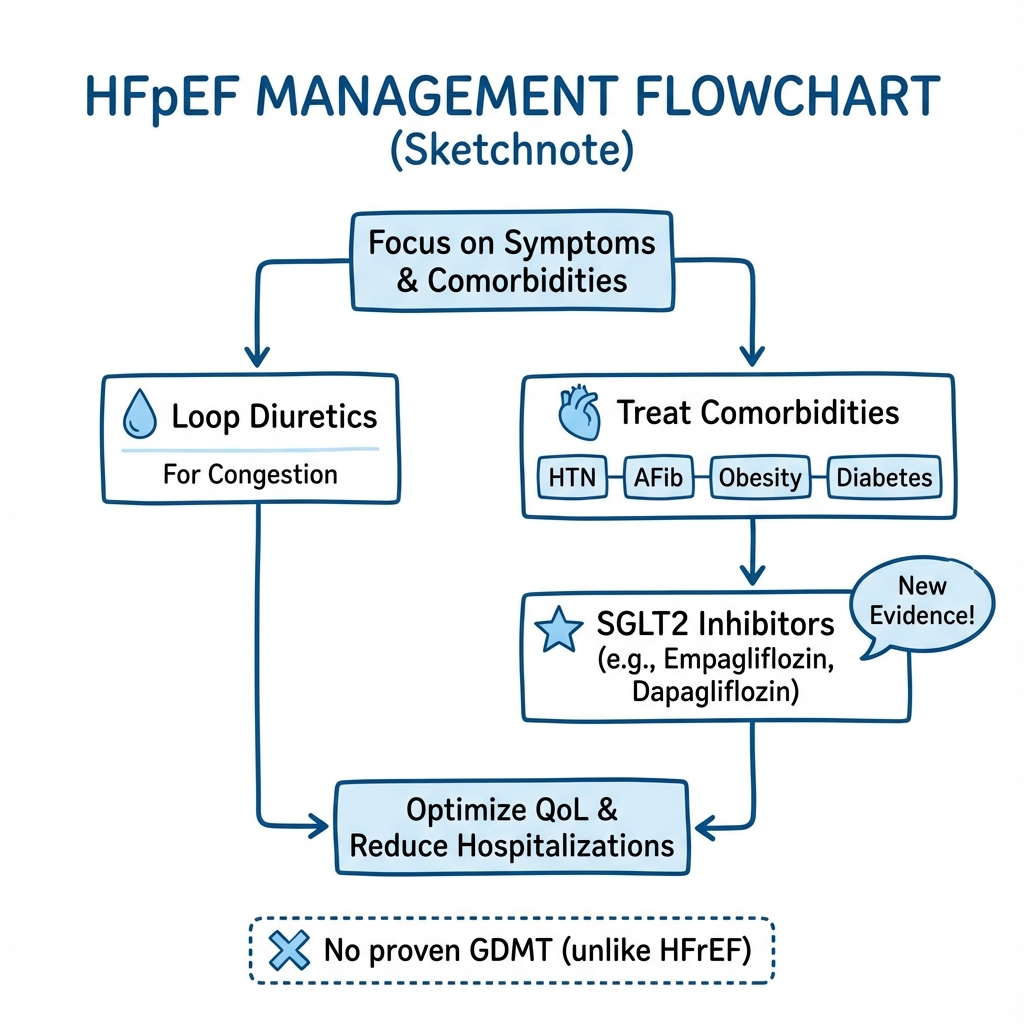
Management of HFpEF is fundamentally different from HFrEF. Unlike HFrEF, where neurohormonal blockade with ACE-I/ARB, beta-blockers, MRAs, and ARNI form the foundation of therapy, HFpEF management focuses on three pillars: (1) symptomatic relief with diuretics, (2) aggressive comorbidity treatment, and (3) SGLT2 inhibitors. [38,39]
Treatment Algorithm
Step 1: Confirm Diagnosis and Exclude Mimics
- Apply HFA-PEFF diagnostic algorithm (score ≥5 or score 2-4 with positive functional testing)
- Rule out cardiac amyloidosis, constrictive pericarditis, severe aortic stenosis, HCM
- Assess for coexisting conditions (COPD, pulmonary fibrosis, renal disease, obesity)
Step 2: Treat Congestion (Diuretics)
Loop Diuretics (Symptomatic Relief):
- Indication: Volume overload, peripheral oedema, dyspnoea due to pulmonary congestion
- Goal: Achieve and maintain euvolaemia (dry weight)
- Choice: Furosemide, bumetanide, torasemide
| Drug | Starting Dose | Usual Maintenance | Notes |
|---|---|---|---|
| Furosemide | 20-40 mg OD | 40-120 mg OD-BD | Most commonly used; bioavailability 50% (variable) |
| Bumetanide | 0.5-1 mg OD | 1-3 mg OD | 40× more potent than furosemide; better bioavailability |
| Torasemide | 5-10 mg OD | 10-20 mg OD | Longer half-life; more consistent absorption |
Principles:
- Use lowest effective dose to avoid over-diuresis (causes hypotension, AKI, electrolyte disturbance)
- Monitor weight daily (target stable dry weight)
- Monitor renal function and electrolytes (weekly initially, then monthly)
- Educate patients on flexible diuretic dosing based on weight and symptoms
- Combination therapy: Add thiazide (metolazone 2.5-5 mg) for diuretic resistance (sequential nephron blockade) [40]
Caution:
- Avoid over-diuresis: HFpEF patients are preload-sensitive (stiff LV requires adequate filling)
- Excessive diuresis → hypotension, AKI, fatigue, poor quality of life
- Monitor for hypokalaemia and hypomagnesaemia (replace aggressively)
Step 3: Initiate SGLT2 Inhibitor (Class I Recommendation)
Breakthrough Therapy for HFpEF:
The EMPEROR-Preserved and DELIVER trials demonstrated SGLT2 inhibitors significantly reduce heart failure hospitalisations and cardiovascular death in HFpEF, establishing Class I, Level A evidence. [4,5,38]
Recommended Agents:
| Drug | Dose | Key Trial | Primary Outcome |
|---|---|---|---|
| Empagliflozin | 10 mg OD | EMPEROR-Preserved (2021) | 21% reduction in CV death or HF hospitalisation (HR 0.79, 95% CI 0.69-0.90) |
| Dapagliflozin | 10 mg OD | DELIVER (2022) | 18% reduction in CV death or worsening HF (HR 0.82, 95% CI 0.73-0.92) |
Key Points:
- Benefit independent of diabetes status (effective in non-diabetics)
- Benefit across entire EF spectrum (HFpEF, HFmrEF, HFrEF)
- NNT ~20-25 to prevent one HF hospitalisation over 2 years
- Generally well-tolerated; low risk of hypoglycaemia (if not on insulin/sulfonylureas)
- Initiate in all HFpEF patients unless contraindicated
Mechanism (not fully understood):
- Natriuresis and osmotic diuresis (modest BP and volume reduction)
- Improved myocardial energetics (shift to ketone metabolism)
- Reduced oxidative stress and inflammation
- Improved endothelial function
- Reduced epicardial adipose tissue
- Effects on kidney (reduced intraglomerular pressure, kidney protection) [41]
Contraindications/Cautions:
- eGFR less than 20 mL/min/1.73m² (limited efficacy, though not harmful)
- Type 1 diabetes (risk of DKA; use with caution)
- Recurrent UTIs or genital infections (counsel on hygiene)
- Euglycaemic DKA risk (rare; educate patients on sick-day rules)
Monitoring:
- Baseline: eGFR, electrolytes, BP
- 2-4 weeks: Repeat renal function (small transient eGFR dip acceptable)
- Ongoing: Monitor for genital infections, dehydration
Step 4: Aggressive Comorbidity Management
Comorbidities drive HFpEF pathophysiology. Optimal management of each is essential and may improve symptoms and prognosis. [17,42]
a) Hypertension (Present in 80-95%):
- Target BP: less than 130/80 mmHg (lower may be better if tolerated)
- First-line: RAAS inhibitors (ACE-I or ARB) despite lack of mortality benefit; control BP and regression of LVH
- Add: Thiazide diuretics, calcium channel blockers (amlodipine, diltiazem)
- Avoid excessive reduction: Hypotension worsens symptoms in HFpEF (preload-dependent)
ACE-I/ARB in HFpEF:
- Do NOT reduce mortality (PEP-CHF, I-PRESERVE, CHARM-Preserved trials neutral) [43]
- May improve symptoms and quality of life
- Useful for BP control and LVH regression
- Indication: Hypertension, post-MI, CKD, diabetes (not for HFpEF-specific mortality benefit)
b) Diabetes Mellitus (30-50%):
- Target HbA1c: less than 7% (individualise based on age, frailty)
- Preferred agent: SGLT2 inhibitor (dual benefit: diabetes + HFpEF)
- Add metformin if eGFR > 30 mL/min (first-line for glycaemia)
- Consider GLP-1 receptor agonist if obese (see below)
- Avoid: Thiazolidinediones (pioglitazone) – cause fluid retention and worsen HF
c) Obesity (50-80%):
- Target: 5-10% weight loss improves symptoms, exercise capacity, and quality of life
- Lifestyle: Diet (caloric restriction, low sodium less than 2 g/day), exercise (aerobic and resistance training)
- GLP-1 receptor agonists: Semaglutide, liraglutide, tirzepatide
- Promote weight loss (10-15% body weight)
- "Emerging evidence suggests benefit in HFpEF with obesity (STEP-HFpEF trial: semaglutide improved symptoms and exercise capacity) [44]"
- May reduce inflammation, epicardial fat, improve metabolic profile
- Bariatric surgery: Consider in severe obesity (BMI > 40 or > 35 with comorbidities) refractory to medical therapy
d) Atrial Fibrillation (30-50%):
AF is both a cause and consequence of HFpEF. Loss of atrial contraction worsens diastolic filling. [26]
Rate Control (Preferred Strategy):
- Target: Resting HR less than 110 bpm (lenient) or less than 80 bpm (strict, if symptomatic)
- Agents:
- Beta-blockers (bisoprolol 2.5-10 mg OD, carvedilol 3.125-25 mg BD) – first-line
- Non-dihydropyridine CCB (diltiazem 60-120 mg BD, verapamil) – if beta-blockers contraindicated
- Digoxin 62.5-250 mcg OD – adjunct for rate control (especially if LV systolic dysfunction develops)
- Evidence: RACE II trial showed lenient rate control non-inferior to strict [45]
Rhythm Control:
- Not superior to rate control in HFpEF (AFFIRM, RACE trials)
- Consider in young, symptomatic, paroxysmal AF, first episode
- Agents: Amiodarone (most effective but toxicity), sotalol, dronedarone
- Catheter ablation: Consider in highly symptomatic patients refractory to medical therapy
Anticoagulation (Essential):
- CHA₂DS₂-VASc score ≥2 (men) or ≥3 (women): Anticoagulate
- Preferred: Direct oral anticoagulants (DOACs) – apixaban, rivaroxaban, edoxaban, dabigatran
- Warfarin: If mechanical valve, severe mitral stenosis, or eGFR less than 15 mL/min
- DOACs preferred over warfarin (easier, safer, no INR monitoring) [46]
e) Coronary Artery Disease (40-60%):
- Optimise medical therapy: Aspirin, statin (atorvastatin 80 mg), ACE-I/ARB
- Revascularisation: If obstructive CAD with evidence of ischaemia (stress imaging showing inducible ischaemia)
- Microvascular dysfunction: Common even without obstructive CAD; no specific therapy proven
f) Chronic Kidney Disease (30-50%):
- Monitor renal function closely (diuretics, ACE-I/ARB, SGLT2i all affect GFR)
- Target BP less than 130/80 mmHg; use ACE-I/ARB for proteinuria
- SGLT2 inhibitors provide kidney protection (slow eGFR decline)
- Adjust drug doses for GFR (digoxin, DOAC doses)
- Avoid over-diuresis (cardiorenal syndrome)
- Nephrology referral if eGFR less than 30 mL/min or rapidly declining
g) Obstructive Sleep Apnoea (50-70%):
- Screen all HFpEF patients (Epworth Sleepiness Scale, STOP-BANG questionnaire)
- Confirm with overnight oximetry or polysomnography
- Treat with CPAP (improves BP, reduces AF burden, may improve symptoms)
- Weight loss if obese
h) Anaemia (20-40%):
- Investigate: Iron deficiency (serum ferritin, transferrin saturation), B12/folate, haemolysis, chronic disease
- Treat iron deficiency: IV iron if ferritin less than 100 ng/mL or ferritin 100-300 with TSAT less than 20% (AFFIRM-AHF trial showed benefit in HFrEF; extrapolate to HFpEF) [47]
- Erythropoietin-stimulating agents: Not recommended (no benefit, potential harm in TREAT, RED-HF trials)
Step 5: Consider Additional Therapies
a) Mineralocorticoid Receptor Antagonists (MRA):
- Spironolactone 12.5-50 mg OD or eplerenone 25-50 mg OD
- Evidence: TOPCAT trial (2014) showed modest reduction in HF hospitalisations (no mortality benefit) [48]
- "Americas cohort: Positive effect"
- "Russia/Georgia cohort: No effect (suspected adherence issues)"
- Meta-analysis suggests genuine but modest benefit
- Indications: Consider if symptoms persist despite SGLT2i and diuretics; particularly if hypokalaemia from loop diuretics
- Contraindications: eGFR less than 30 mL/min, K⁺ > 5.0 mmol/L
- Monitoring: Check K⁺ and creatinine at 1 week, 4 weeks, then 3-monthly (risk of hyperkalaemia)
b) Sacubitril/Valsartan (ARNI):
- PARAGON-HF trial (2019): Borderline benefit in HFpEF (HR 0.87, p=0.06) [49]
- Subgroup with LVEF 45-57% (HFmrEF/lower HFpEF range) showed benefit
- Women and younger patients appeared to benefit more
- Current role: Not routinely recommended for HFpEF (LVEF ≥50%); consider for HFmrEF (LVEF 41-49%)
- Dose: Sacubitril/valsartan 24/26 mg BD, titrate to 97/103 mg BD
c) Exercise Rehabilitation:
- Strong evidence for improving exercise capacity, quality of life, and functional status [50]
- Recommend: Supervised cardiac rehabilitation (aerobic + resistance training)
- Dose: 30-60 minutes, 3-5 times per week, moderate intensity
- Benefits: Improved peak VO₂, muscle function, QoL; reduces hospitalisations
- Underutilised: Only 10-20% of HFpEF patients participate
d) Sodium and Fluid Restriction:
- Sodium: less than 2 g/day (moderate restriction; severe restriction may worsen QoL without clear benefit)
- Fluid restriction: Generally less than 2 L/day if hyponatraemic or severe volume overload (otherwise not mandatory)
- Patient education: Low-salt diet, avoid processed foods, read labels
e) Devices:
- No role for ICD or CRT in HFpEF (unlike HFrEF)
- Pacemaker: Only if conventional indication (symptomatic bradycardia, high-degree AV block)
- Investigational devices: InterAtrial shunt devices (reduce LA pressure; Phase III trials ongoing)
Step 6: Advanced/Refractory HFpEF
Escalation for Persistent NYHA III-IV Symptoms Despite Optimal Therapy:
- Pulmonary vasodilators: If significant pulmonary hypertension and RV dysfunction (sildenafil, riociguat – limited evidence)
- Cardiac transplantation: Rarely indicated (age, comorbidities usually preclude); reserved for young patients
- Palliative care: For elderly, frail patients with refractory symptoms; focus on symptom control and quality of life
What Does NOT Work in HFpEF
Unlike HFrEF, the following therapies have failed to show mortality benefit and are not indicated specifically for HFpEF:
| Therapy | Trial | Outcome | Role in HFpEF |
|---|---|---|---|
| ACE-I (Perindopril) | PEP-CHF | Neutral | Use only for hypertension, post-MI, CKD |
| ARB (Irbesartan) | I-PRESERVE | Neutral | Use only for hypertension or ACE-I intolerance |
| ARB (Candesartan) | CHARM-Preserved | Neutral (modest HF hosp reduction) | Weak indication; use for BP control |
| Beta-blockers | Meta-analyses | No mortality benefit | Use only for AF rate control, post-MI, or angina |
| Digoxin | DIG trial | No benefit in HFpEF subgroup | Use only for AF rate control |
| Ivabradine | No HFpEF trial | Not studied | No role |
| Nitrates/Hydralazine | No HFpEF trial | Not studied | No role |
| Sildenafil | RELAX trial | Neutral | Not recommended routinely |
Summary: Recommended Medications in HFpEF
For ALL HFpEF Patients:
- Loop diuretic (titrate to euvolaemia)
- SGLT2 inhibitor (empagliflozin 10 mg OD or dapagliflozin 10 mg OD) – Class I
For Comorbidities: 3. ACE-I or ARB (if hypertension, diabetes, CKD, post-MI) 4. Statin (if CAD, diabetes, or cardiovascular risk) 5. Anticoagulation (if AF with CHA₂DS₂-VASc ≥2) 6. Beta-blocker or CCB (if AF for rate control, or hypertension)
Consider: 7. MRA (spironolactone/eplerenone) (if persistent symptoms, recurrent hospitalisations) 8. GLP-1 agonist (if obesity and diabetes)
8. Prognosis and Outcomes
Mortality
HFpEF carries a prognosis as poor as HFrEF, challenging the misconception that "preserved EF" implies better outcomes. [6,7]
| Timeframe | Mortality | Notes |
|---|---|---|
| 30-day (post-hospitalisation) | 5-10% | Similar to HFrEF |
| 1-year | 15-25% | High early mortality after first hospitalisation |
| 5-year | 50-65% | Comparable to many cancers |
| 10-year | 75-85% | Very poor long-term survival |
Comparative Mortality:
- HFpEF vs. HFrEF: 5-year mortality ~50-60% for both (historically HFrEF worse, but gap narrowing as HFrEF therapies improve)
- HFpEF vs. General Population: Age-matched mortality 3-4 times higher than those without HF
- HFpEF vs. Cancer: Similar 5-year mortality to metastatic colon cancer [51]
Cause of Death
Unlike HFrEF (where cardiovascular deaths dominate), HFpEF patients have a higher proportion of non-cardiovascular deaths: [52]
| Cause of Death | HFpEF | HFrEF |
|---|---|---|
| Cardiovascular | 40-50% | 60-70% |
| Sudden cardiac death | 20-30% | 40-50% |
| Progressive heart failure | 15-20% | 20-30% |
| Myocardial infarction/stroke | 10-15% | 10-15% |
| Non-cardiovascular | 50-60% | 30-40% |
| Cancer | 15-20% | 10-15% |
| Infection/sepsis | 10-15% | 5-10% |
| Renal failure | 5-10% | 5-10% |
| Respiratory disease | 5-10% | 5-10% |
This has important implications: therapies targeting cardiovascular risk alone may have limited impact on overall mortality.
Hospitalisation Burden
- First HF hospitalisation: 30-day readmission rate 20-30%; 1-year readmission rate 50-60%
- Recurrent hospitalisations: Each hospitalisation accelerates functional decline and worsens prognosis
- Length of stay: Average 5-7 days per admission
- Cost: HF hospitalisations account for 60-70% of total HF healthcare costs
- SGLT2 inhibitors reduce HF hospitalisations by ~20-25%, a major benefit [4,5]
Prognostic Factors
Poor Prognosis Associated With:
| Factor | Hazard Ratio (Approximate) | Comment |
|---|---|---|
| Older age | 1.05 per year | Exponential increase with age |
| Male sex | 1.2-1.5 | Women have better survival despite higher prevalence |
| NYHA Class III-IV | 2.0-3.0 vs. Class I-II | Functional status predicts mortality |
| Elevated NT-proBNP | 1.3-2.0 per doubling | Strong biomarker of prognosis |
| Renal dysfunction (eGFR less than 60) | 1.5-2.5 | Cardiorenal syndrome; worse with lower eGFR |
| Atrial fibrillation | 1.3-1.6 | Associated with worse outcomes |
| Pulmonary hypertension | 2.0-3.0 | RV dysfunction especially poor prognosis |
| Hyponatraemia (Na less than 135) | 1.5-2.0 | Marker of severity and neurohormonal activation |
| Anaemia (Hb less than 12 g/dL) | 1.3-1.8 | Common and under-treated |
| Low LVEF (even within "preserved" range) | 1.2 per 5% reduction | LVEF 50-55% worse than > 60% |
| Diabetes mellitus | 1.3-1.5 | Metabolic burden worsens outcomes |
| Prior HF hospitalisation | 2.0-2.5 | Each hospitalisation worsens prognosis |
Better Prognosis Associated With:
- Younger age
- Female sex
- Higher BMI (obesity paradox: overweight/obese patients have better survival than underweight/normal weight – possibly due to nutritional reserve)
- Higher blood pressure (systolic BP > 120 mmHg)
- Absence of comorbidities
- Higher eGFR
- Sinus rhythm
Functional Decline and Quality of Life
Functional Capacity:
- Peak VO₂: Severely reduced (typically less than 15 mL/kg/min; less than 12 mL/kg/min indicates very poor prognosis)
- 6-minute walk distance: less than 300 metres indicates poor functional status
- Progressive decline: Accelerates with each hospitalisation and advancing age
- Frailty: Highly prevalent (40-60%); associated with poor outcomes and reduced independence [53]
Quality of Life:
- Kansas City Cardiomyopathy Questionnaire (KCCQ) scores: Markedly impaired; comparable to HFrEF
- Domains affected: Physical limitation, social limitation, symptom burden, self-efficacy
- Depression and anxiety: Present in 30-50%; undertreated and associated with worse QoL and outcomes
- Caregiver burden: High; patients often require assistance with ADLs
Prognostic Models and Risk Scores
Several validated risk scores exist for HFpEF:
MAGGIC Risk Score:
- Variables: Age, sex, EF, NYHA class, creatinine, diabetes, COPD, HF duration, BMI, beta-blocker use
- Predicts 1- and 3-year mortality
- Widely validated in HF populations including HFpEF [54]
Meta-Analysis Global Group in Chronic Heart Failure (MAGGIC):
- Online calculator available
- Useful for prognostication and shared decision-making
Biomarkers:
- NT-proBNP and high-sensitivity troponin: Serial measurements improve risk stratification
- Rising levels indicate worsening prognosis
9. Complications
Acute Complications
| Complication | Incidence | Pathophysiology | Management |
|---|---|---|---|
| Acute Pulmonary Oedema | 10-20% of presentations | Sudden rise in BP → acute rise in LVEDP → pulmonary venous congestion → alveolar oedema | IV loop diuretics (furosemide 40-80 mg bolus), nitrates (GTN infusion), non-invasive ventilation (CPAP/BiPAP), BP control |
| Flash Pulmonary Oedema | 5-10% | Hypertensive emergency; very rapid onset; often recurrent | As above + aggressive BP control (IV nitrates, labetalol); consider renal artery stenosis if recurrent |
| Rapid Atrial Fibrillation | 15-25% | New-onset AF or loss of rate control; loss of atrial kick worsens diastolic filling | Rate control (IV beta-blocker or diltiazem), anticoagulation, consider DCCV if haemodynamically unstable |
| Acute Kidney Injury | 20-30% during hospitalisation | Over-diuresis, hypoperfusion, contrast nephropathy, cardiorenal syndrome | Stop/reduce diuretics temporarily, IV fluids if hypovolaemic, avoid nephrotoxins, monitor closely |
| Hypotension | 10-15% | Over-diuresis, excessive RAAS inhibition, sepsis | Reduce/stop diuretics and vasodilators, IV fluids cautiously, identify cause |
| Cardiogenic Shock | less than 5% | Rare in pure HFpEF; consider acute MI, severe valvular disease, or arrhythmia | ICU care, inotropes (dobutamine, milrinone), treat underlying cause, mechanical support rarely needed |
Chronic Complications
1. Atrial Fibrillation:
- Develops in 30-50% of HFpEF patients over time
- Loss of atrial contraction reduces cardiac output by 15-20% (critical in HFpEF where atrial kick is important)
- Increases thromboembolic risk (stroke, systemic embolism)
- Management: Rate control, anticoagulation, consider rhythm control if highly symptomatic
2. Pulmonary Hypertension and Right Ventricular Dysfunction:
- Chronic LA hypertension → pulmonary venous hypertension → pulmonary arterial remodelling → RV pressure overload
- RV dysfunction develops in 30-40% of advanced HFpEF
- Associated with severe symptoms, poor exercise capacity, and very poor prognosis
- Management: Optimise LV filling pressures, diuretics, consider pulmonary vasodilators (limited evidence)
3. Recurrent Hospitalisations:
- 50-60% readmitted within 1 year of index HF hospitalisation
- Each hospitalisation accelerates functional decline, increases frailty, and worsens prognosis
- Prevention: Optimal medical therapy (SGLT2i, diuretics), comorbidity control, HF nurse-led follow-up, patient education
4. Cardiorenal Syndrome:
- Bidirectional interaction between heart and kidneys
- HFpEF → renal hypoperfusion, venous congestion → worsening renal function → fluid retention → worsening HF
- Diuretic resistance develops
- Management: Careful diuretic dosing, SGLT2i (kidney-protective), avoid over-diuresis, nephrology input
5. Thromboembolic Events:
- Stroke risk increased (especially with AF)
- Pulmonary embolism (due to immobility, venous stasis)
- Systemic embolism
- Prevention: Anticoagulation if AF, mobilisation, compression stockings in hospitalised patients
6. Depression and Cognitive Decline:
- Depression in 30-50% (undertreated)
- Cognitive impairment and dementia risk increased (due to cerebral hypoperfusion, microemboli)
- Impact on adherence, self-care, and quality of life
- Management: Screen for depression (PHQ-9), consider SSRI, cognitive behavioural therapy, optimise cardiovascular risk factors
7. Functional Decline and Frailty:
- Progressive loss of functional capacity
- Sarcopenia and muscle wasting
- Increased falls risk
- Loss of independence and need for institutional care
- Management: Exercise rehabilitation, nutritional support, multidisciplinary care
8. Sudden Cardiac Death:
- Accounts for 20-30% of deaths in HFpEF
- Mechanisms: Ventricular arrhythmias (VT/VF), bradyarrhythmias (high-degree AV block, sinus arrest)
- No proven benefit of ICD in HFpEF (unlike HFrEF)
10. Prevention
Primary Prevention (Preventing HFpEF Development)
Modifiable Risk Factor Control:
Given that HFpEF is driven by comorbidities, aggressive risk factor management may prevent or delay disease onset:
1. Hypertension Control:
- Maintain BP less than 130/80 mmHg throughout life
- Prevents LV hypertrophy and arterial stiffening
- SPRINT trial: Intensive BP control (less than 120 mmHg) reduced HF incidence by 38% [55]
2. Obesity Prevention and Management:
- Maintain healthy BMI (18.5-24.9 kg/m²)
- Weight loss if overweight/obese
- Reduces systemic inflammation, insulin resistance, and metabolic burden
3. Diabetes Prevention:
- Lifestyle modification (diet, exercise)
- Metformin for high-risk individuals (impaired glucose tolerance)
- Tight glycaemic control if diabetes develops
4. Physical Activity:
- Regular aerobic and resistance exercise (150 minutes/week moderate intensity)
- Improves cardiovascular fitness, reduces obesity, lowers BP
- Strong inverse association between cardiorespiratory fitness and HFpEF risk
5. Dietary Interventions:
- Low sodium intake (less than 2 g/day)
- Mediterranean diet or DASH diet
- Limit alcohol (less than 14 units/week)
6. Smoking Cessation:
- Smoking increases cardiovascular risk and oxidative stress
- Cessation reduces HF risk by 20-30%
7. Management of Sleep Apnoea:
- Screen high-risk individuals (obese, snoring, daytime somnolence)
- Treat with CPAP if OSA confirmed
Secondary Prevention (Preventing Progression and Hospitalisations)
Once HFpEF is diagnosed, focus shifts to preventing disease progression and reducing hospitalisations:
1. Optimal Medical Therapy:
- SGLT2 inhibitors (reduce HF hospitalisations by 20-25%)
- Diuretics (maintain euvolaemia)
- Comorbidity control (hypertension, diabetes, AF, CAD)
2. Self-Management and Patient Education:
- Daily weight monitoring (alert clinician if > 2 kg gain over 3 days)
- Recognise early signs of decompensation (increasing dyspnoea, oedema, fatigue)
- Medication adherence
- Dietary sodium restriction
- Fluid restriction if appropriate
3. Heart Failure Nurse-Led Follow-Up:
- Regular phone or clinic follow-up
- Titration of diuretics based on symptoms and weight
- Early identification of decompensation
- Reduces readmissions by 20-30%
4. Cardiac Rehabilitation:
- Supervised exercise programme
- Improves functional capacity and quality of life
- Reduces hospitalisations
5. Multidisciplinary Care:
- Cardiology, heart failure nursing, pharmacy, physiotherapy, dietetics, palliative care
- Comprehensive management of complex multimorbidity
6. Treat Acute Infections Promptly:
- Respiratory infections precipitate decompensation
- Influenza and pneumococcal vaccination
- Early antibiotics for infections
7. Medication Review:
- Avoid NSAIDs (fluid retention, renal impairment)
- Avoid thiazolidinediones (glitazones – fluid retention)
- Minimise polypharmacy
11. Special Populations
Elderly and Frail Patients
HFpEF predominantly affects the elderly (mean age 75-80 years). Management must be individualised:
Challenges:
- Multimorbidity (average 5-8 chronic conditions)
- Polypharmacy (10-15 medications)
- Frailty (40-60%)
- Cognitive impairment (20-30%)
- Falls risk
- Limited life expectancy
Management Principles:
- Symptom relief prioritised over mortality reduction (pragmatic goals)
- Avoid over-treatment: Risk of hypotension, falls, AKI with aggressive therapy
- Simplify regimens: Minimise pill burden, once-daily dosing preferred
- Palliative care approach: Focus on quality of life, symptom control, advanced care planning
- Multidisciplinary input: Geriatricians, HF nurses, physiotherapy, occupational therapy
- Functional assessment: Comprehensive geriatric assessment; address frailty, sarcopenia, nutrition
Pregnant Women with HFpEF
Pregnancy in HFpEF is rare (disease predominantly affects post-menopausal women) but high-risk:
Physiological Challenges:
- Pregnancy increases plasma volume by 40-50%
- Cardiac output increases by 30-50%
- Systemic vascular resistance falls
- HFpEF patients poorly tolerate volume expansion
Management:
- Pre-pregnancy counselling: High-risk; consider avoiding pregnancy if severe HFpEF
- Multidisciplinary care: Cardiology, obstetrics, anaesthetics
- Medication adjustments: Stop ACE-I/ARB (teratogenic); stop MRA; continue diuretics cautiously
- Monitoring: Frequent echocardiography, close symptom surveillance
- Delivery planning: Elective induction or caesarean section; avoid fluid overload peripartum
- Postpartum: High-risk period for decompensation (volume shifts); close monitoring
HFpEF in Younger Patients
HFpEF in patients less than 60 years is uncommon but increasingly recognised:
Causes:
- Severe obesity (metabolic syndrome phenotype)
- Hypertrophic cardiomyopathy
- Infiltrative diseases (amyloidosis, sarcoidosis, Fabry disease)
- Genetic cardiomyopathies
- Chronic hypertension
Approach:
- Investigate thoroughly: Exclude secondary causes (amyloidosis, HCM, constrictive pericarditis)
- Aggressive risk factor modification: Weight loss, exercise, BP control (decades of life at stake)
- Genetic counselling: If familial cardiomyopathy suspected
- Consider advanced therapies: May be transplant candidates if severe and refractory
12. Patient and Layperson Explanation
What is Heart Failure with Preserved Ejection Fraction (HFpEF)?
Heart failure means your heart isn't working as well as it should. In HFpEF, the heart muscle has become stiff and doesn't relax properly between beats. This means the heart can't fill with enough blood, especially when you're active.
The heart still squeezes normally (this is what "preserved ejection fraction" means), but because it's stiff, it can't fill properly. This causes fluid to back up into your lungs and body, making you breathless and tired.
What Causes HFpEF?
HFpEF is usually caused by several conditions working together over many years:
- High blood pressure: The most common cause; forces the heart to work harder, making it thicker and stiffer
- Being overweight or obese: Extra weight puts strain on the heart and causes inflammation
- Diabetes: High blood sugar damages blood vessels and the heart muscle
- Getting older: The heart naturally becomes stiffer with age
- Irregular heartbeat (atrial fibrillation): Affects how well the heart fills with blood
What Are the Symptoms?
Common symptoms include:
- Breathlessness – especially when walking, climbing stairs, or lying flat
- Tiredness and low energy – difficulty doing activities you used to manage easily
- Swollen ankles and legs – fluid builds up, especially at the end of the day
- Needing to urinate at night – fluid redistributes when you lie down
- Difficulty exercising – you may feel out of proportion to what you'd expect
How is HFpEF Diagnosed?
Your doctor will:
- Ask about symptoms and examine you (check blood pressure, listen to heart and lungs, check for ankle swelling)
- Blood tests – including a test called "BNP" or "NT-proBNP" that shows if the heart is under strain
- Echocardiogram (heart ultrasound) – shows that the heart is squeezing normally but is stiff and not relaxing well
- ECG (heart tracing) – checks heart rhythm and looks for signs of thickening
- Sometimes exercise tests or other scans if the diagnosis is unclear
How is HFpEF Treated?
There is no cure for HFpEF, but treatment can improve symptoms and help you live longer:
1. Water Tablets (Diuretics):
- Help your body get rid of extra fluid
- Reduce breathlessness and ankle swelling
- You'll need to take these daily and adjust the dose based on your weight and symptoms
2. SGLT2 Inhibitors (Empagliflozin or Dapagliflozin):
- New breakthrough medication proven to reduce hospitalisations
- Originally diabetes medication but works even if you don't have diabetes
- Protects your heart and kidneys
- Take once daily
3. Treating Other Conditions:
- Blood pressure tablets – keep blood pressure under control (less than 130/80 mmHg)
- Diabetes medication – control blood sugar
- Blood thinners – if you have atrial fibrillation (irregular heartbeat)
- Cholesterol tablets (statins) – if you have heart disease or diabetes
4. Lifestyle Changes:
- Lose weight if overweight (even 5-10% weight loss helps)
- Low-salt diet – avoid processed foods, don't add salt to meals
- Regular exercise – walking, swimming, cycling (start slowly and build up)
- Stop smoking – critical for heart health
- Limit alcohol – no more than 14 units per week
5. Monitoring:
- Weigh yourself daily – alert your doctor if you gain > 2 kg in 3 days (may mean fluid building up)
- Watch for worsening symptoms – increased breathlessness, more ankle swelling, extreme tiredness
What Should I Avoid?
- Anti-inflammatory painkillers (NSAIDs) like ibuprofen – cause fluid retention and worsen heart failure
- Too much salt – read food labels; aim for less than 2 g salt per day
- Excessive fluids – limit to 1.5-2 litres per day if advised by doctor
- Skipping medications – take all medications as prescribed
When Should I Seek Help Urgently?
Go to A&E (Emergency Department) or call 999 if you experience:
- Sudden severe breathlessness – difficulty breathing even at rest
- Chest pain – could be a heart attack
- Fainting or dizziness – may indicate abnormal heart rhythm
- Coughing up pink frothy liquid – sign of fluid in the lungs
- Rapid weight gain (> 2-3 kg in 2-3 days) with worsening breathlessness
What is the Outlook?
HFpEF is a serious condition, and unfortunately, long-term outlook is similar to many cancers. However:
- Treatments have improved – SGLT2 inhibitors reduce hospital admissions and help you live longer
- Symptom control – with the right medications and lifestyle changes, many people feel much better
- Quality of life – focus on what's important to you; stay as active as possible
- Support: Heart failure nurses, cardiac rehabilitation, and support groups can help
Key message: HFpEF is a long-term condition requiring lifelong treatment, but with the right approach, you can live well and manage your symptoms.
13. Summary for Examinations (MRCP/Postgraduate Level)
High-Yield Exam Facts
Definition:
- LVEF ≥50% + symptoms/signs of HF + elevated natriuretic peptides (NT-proBNP > 125 pg/mL SR, > 365 AF) AND/OR diastolic dysfunction on echo/invasive testing
Epidemiology:
- 50% of all HF cases; increasing prevalence
- Mean age 70-80; 60-65% female
- 5-year mortality 50-60% (similar to HFrEF)
Pathophysiology:
- Comorbidity-driven systemic inflammation → coronary microvascular dysfunction → titin hypophosphorylation → cardiomyocyte stiffness + fibrosis → diastolic dysfunction → elevated filling pressures
Diagnosis (HFA-PEFF Algorithm):
- P-Step: Pre-test score (AF, obesity, age, hypertension, diabetes, OSA)
- E-Step: Echo (E/e', e' velocity, LAVi, LVMi, TR velocity)
- F-Step: Biomarkers (NT-proBNP, BNP)
- Score ≥5: HFpEF confirmed
- Score 2-4: Functional testing (exercise echo or invasive haemodynamics)
Key Investigations:
- Echo: LVEF ≥50%, E/e' ≥15 (or 9-14), e' less than 7 cm/s, LAVi > 34 mL/m², LVH
- NT-proBNP: > 125 pg/mL (SR), > 365 pg/mL (AF); lower in obesity
- Exercise testing: PCWP > 25 mmHg with exercise (gold standard)
Management (Class I):
- Loop diuretics (symptom control)
- SGLT2 inhibitors (empagliflozin 10 mg or dapagliflozin 10 mg) – reduce HF hospitalisations by 20%
- Aggressive comorbidity control: Hypertension (less than 130/80), diabetes, obesity, AF
What Doesn't Work:
- ACE-I/ARB (no mortality benefit; use only for BP control)
- Beta-blockers (no mortality benefit; use only for AF rate control or post-MI)
- ARNI (borderline; only consider in HFmrEF LVEF 41-49%)
Key Trials:
- EMPEROR-Preserved: Empagliflozin reduced CV death/HF hosp by 21%
- DELIVER: Dapagliflozin reduced CV death/worsening HF by 18%
- TOPCAT: Spironolactone modest reduction in HF hosp (no mortality benefit)
- PARAGON-HF: Sacubitril/valsartan borderline (benefit in LVEF 45-57%)
Complications:
- AF (30-50%), pulmonary hypertension, RV dysfunction, cardiorenal syndrome, recurrent hospitalisations
Prognosis:
- 5-year mortality 50-60%
- Non-CV deaths (50-60%) > CV deaths (40-50%)
- Poor prognostic factors: Age, male sex, NYHA III-IV, high NT-proBNP, renal dysfunction, AF, pulmonary hypertension
14. References
Primary Literature
-
Borlaug BA. Evaluation and management of heart failure with preserved ejection fraction. Nat Rev Cardiol. 2020;17(9):559-573. PMID: 32433711
-
Dunlay SM, Roger VL, Redfield MM. Epidemiology of heart failure with preserved ejection fraction. Nat Rev Cardiol. 2017;14(10):591-602. PMID: 28492288
-
Shah SJ, Kitzman DW, Borlaug BA, et al. Phenotype-Specific Treatment of Heart Failure With Preserved Ejection Fraction: A Multiorgan Roadmap. Circulation. 2016;134(1):73-90. PMID: 27358439
-
Anker SD, Butler J, Filippatos G, et al. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N Engl J Med. 2021;385(16):1451-1461. PMID: 34449189
-
Solomon SD, McMurray JJV, Claggett B, et al. Dapagliflozin in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N Engl J Med. 2022;387(12):1089-1098. PMID: 36027570
-
Owan TE, Hodge DO, Herges RM, et al. Trends in prevalence and outcome of heart failure with preserved ejection fraction. N Engl J Med. 2006;355(3):251-259. PMID: 16855265
-
Meta-analysis Global Group in Chronic Heart Failure (MAGGIC). The survival of patients with heart failure with preserved or reduced left ventricular ejection fraction: an individual patient data meta-analysis. Eur Heart J. 2012;33(14):1750-1757. PMID: 21821849
-
Tsao CW, Lyass A, Enserro D, et al. Temporal Trends in the Incidence of and Mortality Associated With Heart Failure With Preserved and Reduced Ejection Fraction. JACC Heart Fail. 2018;6(8):678-685. PMID: 30007554
-
Pieske B, Tschöpe C, de Boer RA, et al. How to diagnose heart failure with preserved ejection fraction: the HFA-PEFF diagnostic algorithm: a consensus recommendation from the Heart Failure Association (HFA) of the European Society of Cardiology (ESC). Eur Heart J. 2019;40(40):3297-3317. PMID: 31504452
-
Redfield MM, Borlaug BA. Heart Failure With Preserved Ejection Fraction: A Review. JAMA. 2023;329(10):827-838. PMID: 36892832
-
Dunlay SM, Roger VL, Weston SA, et al. Longitudinal changes in ejection fraction in heart failure patients with preserved and reduced ejection fraction. Circ Heart Fail. 2012;5(6):720-726. PMID: 22936826
-
Gerber Y, Weston SA, Redfield MM, et al. A contemporary appraisal of the heart failure epidemic in Olmsted County, Minnesota, 2000 to 2010. JAMA Intern Med. 2015;175(6):996-1004. PMID: 25895156
-
Savarese G, Lund LH. Global Public Health Burden of Heart Failure. Card Fail Rev. 2017;3(1):7-11. PMID: 28785469
-
Borlaug BA, Redfield MM. Diastolic and systolic heart failure are distinct phenotypes within the heart failure spectrum. Circulation. 2011;123(18):2006-2013. PMID: 21555723
-
Lam CSP, Arnott C, Beale AL, et al. Sex differences in heart failure. Eur Heart J. 2019;40(47):3859-3868c. PMID: 31385935
-
Bahrami H, Kronmal R, Bluemke DA, et al. Differences in the incidence of congestive heart failure by ethnicity: the multi-ethnic study of atherosclerosis. Arch Intern Med. 2008;168(19):2138-2145. PMID: 18955644
-
Uijl A, Savarese G, Vaartjes I, et al. Identification of distinct phenotypic clusters in heart failure with preserved ejection fraction. Eur J Heart Fail. 2021;23(6):973-982. PMID: 33779119
-
Oldenburg O, Wellmann B, Buchholz A, et al. Sleep-disordered breathing in patients with symptomatic heart failure: a contemporary study of prevalence in and characteristics of 700 patients. Eur J Heart Fail. 2007;9(3):251-257. PMID: 17027333
-
Shah KS, Xu H, Matsouaka RA, et al. Heart Failure With Preserved, Borderline, and Reduced Ejection Fraction: 5-Year Outcomes. J Am Coll Cardiol. 2017;70(20):2476-2486. PMID: 29141781
-
Paulus WJ, Tschöpe C. A novel paradigm for heart failure with preserved ejection fraction: comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. J Am Coll Cardiol. 2013;62(4):263-271. PMID: 23684677
-
Sanders-van Wijk S, van Empel V, Davarzani N, et al. Circulating biomarkers of distinct pathophysiological pathways in heart failure with preserved vs. reduced left ventricular ejection fraction. Eur J Heart Fail. 2015;17(10):1006-1014. PMID: 26472682
-
Mohammed SF, Hussain S, Mirzoyev SA, et al. Coronary microvascular rarefaction and myocardial fibrosis in heart failure with preserved ejection fraction. Circulation. 2015;131(6):550-559. PMID: 25552356
-
Hamdani N, Franssen C, Lourenço A, et al. Myocardial titin hypophosphorylation importantly contributes to heart failure with preserved ejection fraction in a rat metabolic risk model. Circ Heart Fail. 2013;6(6):1239-1249. PMID: 24014826
-
Schiattarella GG, Altamirano F, Tong D, et al. Nitrosative stress drives heart failure with preserved ejection fraction. Nature. 2019;568(7752):351-356. PMID: 30971821
-
González A, Schelbert EB, Díez J, Butler J. Myocardial Interstitial Fibrosis in Heart Failure: Biological and Translational Perspectives. J Am Coll Cardiol. 2018;71(15):1696-1706. PMID: 29650126
-
Zakeri R, Chamberlain AM, Roger VL, Redfield MM. Temporal relationship and prognostic significance of atrial fibrillation in heart failure patients with preserved ejection fraction: a community-based study. Circulation. 2013;128(10):1085-1093. PMID: 23908348
-
Gorter TM, van Veldhuisen DJ, Bauersachs J, et al. Right heart dysfunction and failure in heart failure with preserved ejection fraction: mechanisms and management. Position statement on behalf of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2018;20(1):16-37. PMID: 29023887
-
Borlaug BA, Nishimura RA, Sorajja P, Lam CS, Redfield MM. Exercise hemodynamics enhance diagnosis of early heart failure with preserved ejection fraction. Circ Heart Fail. 2010;3(5):588-595. PMID: 20543134
-
Kraigher-Krainer E, Shah AM, Gupta DK, et al. Impaired systolic function by strain imaging in heart failure with preserved ejection fraction. J Am Coll Cardiol. 2014;63(5):447-456. PMID: 24184245
-
Kitzman DW, Nicklas B, Kraus WE, et al. Skeletal muscle abnormalities and exercise intolerance in older patients with heart failure and preserved ejection fraction. Am J Physiol Heart Circ Physiol. 2014;306(9):H1364-H1370. PMID: 24658015
-
Cohen JB, Schrauben SJ, Zhao L, et al. Clinical Phenogroups in Heart Failure With Preserved Ejection Fraction: Detailed Phenotypes, Prognosis, and Response to Spironolactone. JACC Heart Fail. 2020;8(3):172-184. PMID: 31838043
-
Segar MW, Patel KV, Ayers C, et al. Phenomapping of patients with heart failure with preserved ejection fraction using machine learning-based unsupervised cluster analysis. Eur J Heart Fail. 2020;22(1):148-158. PMID: 31520473
-
Gandhi SK, Powers JC, Nomeir AM, et al. The pathogenesis of acute pulmonary edema associated with hypertension. N Engl J Med. 2001;344(1):17-22. PMID: 11136955
-
Obokata M, Kane GC, Reddy YNV, Olson TP, Melenovsky V, Borlaug BA. Role of Diastolic Stress Testing in the Evaluation for Heart Failure With Preserved Ejection Fraction: A Simultaneous Invasive-Echocardiographic Study. Circulation. 2017;135(9):825-838. PMID: 28039229
-
Hogg K, Swedberg K, McMurray J. Heart failure with preserved left ventricular systolic function; epidemiology, clinical characteristics, and prognosis. J Am Coll Cardiol. 2004;43(3):317-327. PMID: 15013109
-
Mentz RJ, Kelly JP, von Lueder TG, et al. Noncardiac comorbidities in heart failure with reduced versus preserved ejection fraction. J Am Coll Cardiol. 2014;64(21):2281-2293. PMID: 25456761
-
Reddy YNV, Carter RE, Obokata M, Redfield MM, Borlaug BA. A Simple, Evidence-Based Approach to Help Guide Diagnosis of Heart Failure With Preserved Ejection Fraction. Circulation. 2018;138(9):861-870. PMID: 29792299
-
McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42(36):3599-3726. PMID: 34447992
-
Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145(18):e895-e1032. PMID: 35363499
-
Felker GM, Lee KL, Bull DA, et al. Diuretic strategies in patients with acute decompensated heart failure. N Engl J Med. 2011;364(9):797-805. PMID: 21366472
-
Lopaschuk GD, Verma S. Mechanisms of Cardiovascular Benefits of Sodium Glucose Co-Transporter 2 (SGLT2) Inhibitors: A State-of-the-Art Review. JACC Basic Transl Sci. 2020;5(6):632-644. PMID: 32613148
-
Somaratne JB, Berry C, McMurray JJ, Poppe KK, Doughty RN, Whalley GA. The prognostic significance of heart failure with preserved left ventricular ejection fraction: a literature-based meta-analysis. Eur J Heart Fail. 2009;11(9):855-862. PMID: 19654140
-
Cleland JG, Tendera M, Adamus J, et al. The perindopril in elderly people with chronic heart failure (PEP-CHF) study. Eur Heart J. 2006;27(19):2338-2345. PMID: 16963472
-
Kosiborod MN, Abildstrøm SZ, Borlaug BA, et al. Semaglutide in Patients with Heart Failure with Preserved Ejection Fraction and Obesity. N Engl J Med. 2023;389(12):1069-1084. PMID: 37622681
-
Van Gelder IC, Groenveld HF, Crijns HJ, et al. Lenient versus strict rate control in patients with atrial fibrillation. N Engl J Med. 2010;362(15):1363-1373. PMID: 20231232
-
Ruff CT, Giugliano RP, Braunwald E, et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet. 2014;383(9921):955-962. PMID: 24315724
-
Ponikowski P, Kirwan BA, Anker SD, et al. Ferric carboxymaltose for iron deficiency at discharge after acute heart failure: a multicentre, double-blind, randomised, controlled trial. Lancet. 2020;396(10266):1895-1904. PMID: 33197395
-
Pitt B, Pfeffer MA, Assmann SF, et al. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2014;370(15):1383-1392. PMID: 24716680
-
Solomon SD, McMurray JJV, Anand IS, et al. Angiotensin-Neprilysin Inhibition in Heart Failure with Preserved Ejection Fraction. N Engl J Med. 2019;381(17):1609-1620. PMID: 31475794
-
Pandey A, Parashar A, Kumbhani D, et al. Exercise training in patients with heart failure and preserved ejection fraction: meta-analysis of randomized control trials. Circ Heart Fail. 2015;8(1):33-40. PMID: 25399909
-
Stewart S, MacIntyre K, Hole DJ, Capewell S, McMurray JJ. More 'malignant' than cancer? Five-year survival following a first admission for heart failure. Eur J Heart Fail. 2001;3(3):315-322. PMID: 11378002
-
Henkel DM, Redfield MM, Weston SA, Gerber Y, Roger VL. Death in heart failure: a community perspective. Circ Heart Fail. 2008;1(2):91-97. PMID: 19300532
-
Pandey A, Kitzman D, Reeves G. Frailty Is Intertwined With Heart Failure: Mechanisms, Prevalence, Prognosis, Assessment, and Management. JACC Heart Fail. 2019;7(12):1001-1011. PMID: 31779921
-
Pocock SJ, Ariti CA, McMurray JJ, et al. Predicting survival in heart failure: a risk score based on 39 372 patients from 30 studies. Eur Heart J. 2013;34(19):1404-1413. PMID: 23095984
-
SPRINT Research Group. A Randomized Trial of Intensive versus Standard Blood-Pressure Control. N Engl J Med. 2015;373(22):2103-2116. PMID: 26551272
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. It does not replace professional medical judgement. Always verify critical information and consider individual patient factors.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Cardiac Physiology - Diastolic Function
- Echocardiography - Diastolic Assessment
Differentials
Competing diagnoses and look-alikes to compare.