Tuberculosis (Adult)
Tuberculosis (TB) is a chronic granulomatous infectious disease caused by organisms of the Mycobacterium tuberculosis co... MRCP exam preparation.
What matters first
Tuberculosis (TB) is a chronic granulomatous infectious disease caused by organisms of the Mycobacterium tuberculosis co... MRCP exam preparation.
MDR-TB or XDR-TB
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- MDR-TB or XDR-TB
- HIV co-infection
- Miliary TB
- TB meningitis
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Lung Cancer
- Non-tuberculous Mycobacteria
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Tuberculosis (Adult)
1. Clinical Overview
Summary
Tuberculosis (TB) is a chronic granulomatous infectious disease caused by organisms of the Mycobacterium tuberculosis complex, primarily affecting the lungs but capable of disseminating to virtually any organ system. [1] Despite being preventable and curable, TB remains a leading cause of infectious disease mortality worldwide, with approximately 10.6 million new cases and 1.3 million deaths annually. [2] The pathogen is transmitted via respiratory droplets, and clinical disease develops in approximately 5-10% of immunocompetent individuals who become infected, with the remainder developing latent TB infection (LTBI). [3]
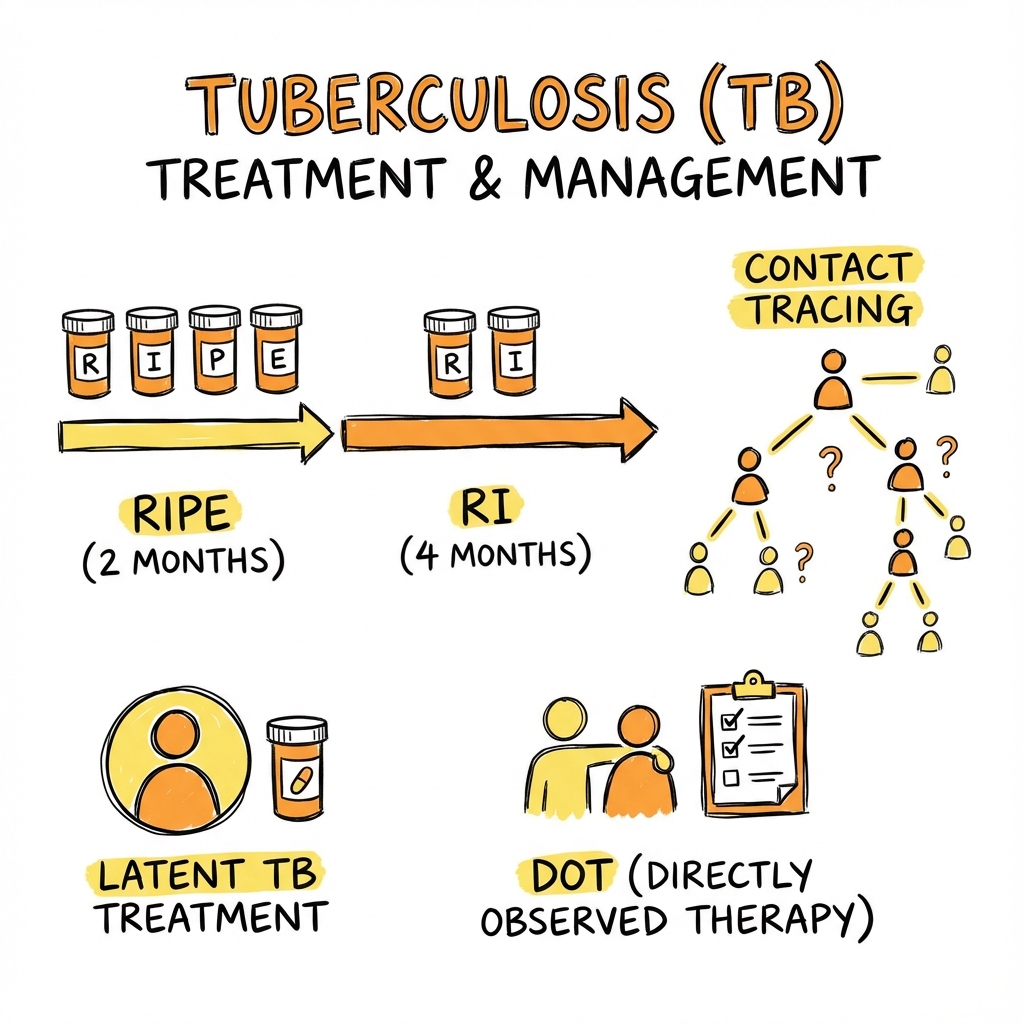
The classic clinical presentation includes chronic productive cough (greater than 2-3 weeks duration), constitutional symptoms (fever, night sweats, weight loss), and haemoptysis. [1] Diagnosis requires integration of clinical suspicion, chest radiography (typically showing upper lobe infiltrates with or without cavitation), microbiological confirmation through sputum smear microscopy, molecular testing (Xpert MTB/RIF), and mycobacterial culture. [4] Standard treatment comprises a 6-month regimen of four first-line drugs: Rifampicin, Isoniazid, Pyrazinamide, and Ethambutol for the initial 2 months (intensive phase), followed by Rifampicin and Isoniazid for 4 months (continuation phase). [5]
Drug-resistant TB, particularly multidrug-resistant TB (MDR-TB) and extensively drug-resistant TB (XDR-TB), represents a significant global health threat requiring prolonged treatment with second-line agents. [6] All TB cases require statutory notification and public health follow-up including contact tracing. The intersection of TB with HIV remains critical, as TB is the leading cause of death among people living with HIV. [2]
Key Facts
| Parameter | Value |
|---|---|
| Definition | Infection with Mycobacterium tuberculosis complex |
| Global Incidence | 10.6 million new cases/year (2022) [2] |
| Global Mortality | 1.3 million deaths/year (2022) [2] |
| UK Incidence | 4,425 cases (2022); 6.6 per 100,000 population [7] |
| MDR-TB Globally | 450,000 cases/year [2] |
| Lifetime Risk LTBI→Active | 5-10% (immunocompetent); 10% per year if HIV+ [3] |
| Transmission | Airborne droplet nuclei (1-5 μm) |
| Pathognomonic Triad | Cough > 2 weeks + upper lobe cavitation + positive AFB |
| Gold Standard Diagnosis | Mycobacterial culture + drug susceptibility testing |
| First-line Treatment | 2HRZE/4HR (6 months total) |
| Treatment Success Rate | > 95% drug-sensitive TB with adherence [5] |
Clinical Pearls
Xpert MTB/RIF Pearl: GeneXpert MTB/RIF Ultra provides sensitivity of 88% (smear-negative, culture-positive) and detects rifampicin resistance mutations within 2 hours. It is the WHO-recommended initial diagnostic test for all patients with suspected TB. [4,8]
HIV Testing Pearl: HIV testing is mandatory in ALL patients diagnosed with TB. Globally, 12% of people with TB are co-infected with HIV, rising to over 50% in parts of sub-Saharan Africa. TB is the leading cause of death in people living with HIV. [2]
Pyridoxine Pearl: Co-administer pyridoxine (vitamin B6) 10-25 mg daily with isoniazid to prevent peripheral neuropathy. Essential in high-risk groups: HIV infection, diabetes, malnutrition, alcohol dependency, chronic renal failure, and pregnancy. [5]
Rifampicin Interactions Pearl: Rifampicin is a potent CYP450 inducer. Critical interactions include reduced efficacy of oral contraceptives, warfarin, antiretrovirals (particularly protease inhibitors and NNRTIs), and calcineurin inhibitors. Dose adjustments or alternative agents required. [9]
Contact Tracing Pearl: All household and close contacts require screening within 2 weeks of index case notification. Screen with symptom inquiry, chest radiograph, and IGRA (or TST). Treat LTBI if positive to break transmission chain. [10]
Smear-Negative TB Pearl: Up to 50% of culture-confirmed pulmonary TB cases are smear-negative. Clinical-radiological suspicion should prompt treatment initiation while awaiting culture, particularly in HIV-positive patients. [11]
Why This Matters Clinically
TB remains a re-emerging disease in high-income countries, driven by immigration from endemic regions, HIV co-infection, and increasing rates of drug resistance. [7] Delayed diagnosis leads to ongoing transmission, with each untreated case potentially infecting 10-15 contacts annually. [1] A high index of suspicion, particularly in at-risk populations, combined with rapid molecular diagnostics, is essential to reduce mortality and prevent transmission. Early appropriate treatment is cost-effective and prevents the development of resistance.
2. Epidemiology
Global Burden
Tuberculosis is the second leading infectious cause of death globally after COVID-19 and disproportionately affects low- and middle-income countries. [2]
| Metric | Value (2022) | Source |
|---|---|---|
| Estimated TB cases globally | 10.6 million | WHO 2023 [2] |
| TB deaths (HIV-negative) | 1.13 million | WHO 2023 [2] |
| TB deaths (HIV-positive) | 167,000 | WHO 2023 [2] |
| Rifampicin-resistant TB | 410,000 | WHO 2023 [2] |
| MDR-TB (RIF+INH resistant) | ~175,000 | WHO 2023 [2] |
| TB-HIV co-infection | 12% of all cases | WHO 2023 [2] |
| Countries with highest burden | India, Indonesia, China, Philippines, Pakistan | WHO 2023 [2] |
Regional Distribution:
- 44% of cases occur in South-East Asia Region
- 23% occur in African Region
- 8 countries account for two-thirds of global burden: India (28%), Indonesia (9.2%), China (7.4%), Philippines (7.0%), Pakistan (5.7%), Nigeria (4.5%), Bangladesh (3.6%), Democratic Republic of Congo (2.9%) [2]
UK Epidemiology
The UK is classified as a low-incidence country (less than 10 cases per 100,000 population) with marked geographic and demographic heterogeneity. [7]
| Metric | Value (2022) | Notes |
|---|---|---|
| Total cases notified | 4,425 | Provisional data |
| Incidence rate | 6.6 per 100,000 | Declining trend |
| Cases in London | ~35% | Higher urban burden |
| Non-UK born proportion | 71% | Predominantly migrants from high-burden countries |
| Pulmonary TB | 50% | Remainder extrapulmonary |
| Drug-resistant cases | 1.5% isoniazid; 0.6% MDR | Lower than global average |
High-Risk Groups in UK:
- Born in high-incidence country (India, Pakistan, Bangladesh, sub-Saharan Africa)
- Recent arrival from endemic area (within 5 years)
- Homeless individuals
- Prison population
- People who inject drugs
- HIV-positive individuals
- Close contacts of known TB cases
- Healthcare workers in high-risk settings
- Immunocompromised (biologics, transplant, chemotherapy)
Risk Factors for TB Disease
| Category | Risk Factors | Relative Risk |
|---|---|---|
| Exposure | Close contact with smear-positive case | 10-15× [1] |
| Healthcare worker in high-incidence setting | 2-3× | |
| Born/residence in endemic country | Variable by country | |
| Homeless shelter, prison, refugee camp | 10-50× | |
| Immunosuppression | HIV/AIDS (most potent risk) | 20-37× [12] |
| TNF-α inhibitors | 4-8× [13] | |
| Systemic corticosteroids (> 15mg/day prednisolone) | 2-4× | |
| Solid organ transplant | 20-74× | |
| Haematological malignancy | 4-8× | |
| Medical Conditions | Diabetes mellitus | 2-3× [14] |
| Chronic kidney disease/dialysis | 10-25× | |
| Silicosis | 30× | |
| Gastrectomy/jejunoileal bypass | 2-5× | |
| Head and neck cancer | 10× | |
| Lifestyle | Tobacco smoking | 2-3× [15] |
| Alcohol excess (> 40g/day) | 2-4× | |
| Malnutrition (BMI less than 18.5) | 2-3× | |
| Age | Elderly (> 65 years) | Reactivation risk |
| Infants (less than 5 years) | Rapid progression risk |
Latent TB Infection vs Active TB Disease
Understanding this distinction is fundamental to TB management:
| Feature | Latent TB Infection (LTBI) | Active TB Disease |
|---|---|---|
| Definition | Persistent immune response to M. tuberculosis without clinical/radiological/microbiological evidence of active disease | Clinical disease with or without microbiological confirmation |
| Symptoms | None | Present (cough, fever, weight loss, sweats) |
| Infectious | No | Yes (if pulmonary/laryngeal) |
| Chest X-ray | Normal or healed TB (calcified nodule, fibrosis) | Abnormal (infiltrates, cavitation, effusion) |
| TST/IGRA | Positive | Usually positive (may be negative if anergic) |
| Sputum smear/culture | Negative | May be positive |
| Treatment | Preventive therapy (3-9 months) | Full treatment regimen (6+ months) |
| Public health | No notification | Statutory notification and contact tracing |
| Lifetime progression risk | 5-10% (immunocompetent) | N/A |
3. Aetiology & Pathophysiology
Causative Organisms
The Mycobacterium tuberculosis complex comprises several closely related species:
| Species | Characteristics | Geographic Distribution |
|---|---|---|
| M. tuberculosis | Primary human pathogen; 99% of human TB | Worldwide |
| M. bovis | Bovine TB; transmitted via unpasteurised dairy | Declining in developed countries |
| M. africanum | Causes up to 40% of TB in West Africa | West Africa |
| M. microti | Rare human pathogen | Europe |
| M. canetti | Extremely rare; smooth colony variant | Horn of Africa |
| M. bovis BCG | Vaccine strain; can cause disease in immunocompromised | Iatrogenic |
Microbiological Characteristics:
- Aerobic, non-spore-forming bacillus
- Slow-growing (doubling time 12-24 hours; visible colonies 3-8 weeks)
- Acid-fast on Ziehl-Neelsen staining (mycolic acid cell wall)
- Obligate intracellular pathogen (survives within macrophages)
- Optimal growth at 37°C with 5-10% CO₂
- Resistant to desiccation, weak acids, and many disinfectants
Transmission
TB is transmitted via the airborne route through droplet nuclei:
Step 1: Generation of Infectious Aerosols
- Produced by coughing, sneezing, speaking, singing
- Each cough generates 3,000 droplet nuclei
- Droplet nuclei are 1-5 μm diameter (optimal for alveolar deposition)
- Remain suspended in air for hours in enclosed spaces
Step 2: Inhalation and Deposition
- Particles > 5 μm trapped in upper airway mucosa
- Particles 1-5 μm reach terminal alveoli
- Bacilli engulfed by alveolar macrophages
Factors Affecting Transmission:
| Factor | Effect |
|---|---|
| Smear positivity | Smear-positive 3× more infectious than smear-negative |
| Cavitary disease | 100× more bacilli than non-cavitary |
| Cough frequency | Direct correlation with infectivity |
| Ventilation | Poor ventilation increases transmission |
| Duration of exposure | > 8 hours cumulative exposure high risk |
| Proximity | less than 1 metre highest risk |
Pathogenesis of Primary TB Infection
Stage 1: Initial Infection (Day 0-21)
- Inhaled bacilli reach terminal alveoli (usually lower/middle lobes)
- Alveolar macrophages engulf bacilli via complement and surfactant receptors
- Bacilli survive and replicate within phagosomes by preventing phagosome-lysosome fusion
- Infected macrophages release chemokines, recruiting additional innate immune cells
- Bacilli spread via lymphatics to hilar lymph nodes
Stage 2: Adaptive Immune Response (Week 2-8)
- Dendritic cells present mycobacterial antigens to T cells in lymph nodes
- CD4+ Th1 lymphocytes activated; produce IFN-γ and TNF-α
- IFN-γ activates macrophages to kill intracellular bacilli
- TNF-α essential for granuloma formation and maintenance
- TST/IGRA becomes positive (indicates immune sensitisation)
Stage 3: Granuloma Formation
The hallmark of TB is the granuloma - an organised structure designed to contain infection:
| Layer | Components | Function |
|---|---|---|
| Centre | Caseous necrosis (cheese-like) | Contains bacilli; limits spread |
| Inner zone | Epithelioid macrophages, Langhans giant cells | Active immune response |
| Outer zone | CD4+ and CD8+ T lymphocytes | Cytokine production, immune regulation |
| Periphery | Fibroblasts, fibrous capsule | Structural containment |
Primary Complex (Ghon Complex):
- Ghon focus: Primary pulmonary lesion (usually subpleural, lower/middle lobes)
- Ghon complex: Ghon focus + draining hilar lymphadenopathy
- Ranke complex: Healed, calcified Ghon complex
Stage 4: Outcomes of Primary Infection
| Outcome | Frequency | Mechanism |
|---|---|---|
| Contained LTBI | 90-95% (immunocompetent) | Effective granuloma containment; bacilli dormant but viable |
| Primary progressive disease | 5-10% | Inadequate immune response; immediate progression to active TB |
| Disseminated disease | less than 1% (unless immunocompromised) | Haematogenous/lymphatic spread before containment |
Pathogenesis of Reactivation (Post-Primary) TB
Reactivation occurs when immune control fails, typically years to decades after primary infection:
Risk Factors for Reactivation:
- HIV infection (10% per year vs 10% lifetime)
- TNF-α inhibitor therapy
- Immunosuppressive medications
- Malnutrition
- Diabetes mellitus
- Advanced age
- Malignancy
Characteristics of Reactivation TB:
- Location: Upper lobes preferred (higher oxygen tension, reduced lymphatic drainage)
- Cavitation: Common (tissue destruction by immune response)
- Infectivity: High (large numbers of bacilli in cavities)
- Spread: Via bronchi (bronchogenic spread) or blood (miliary TB)
Virulence Factors
| Factor | Function |
|---|---|
| Cord factor (Trehalose 6,6'-dimycolate) | Prevents phagosome-lysosome fusion; toxic to mitochondria |
| Lipoarabinomannan (LAM) | Inhibits macrophage activation; scavenges reactive oxygen species |
| Mycolic acids | Impermeability; resistance to killing; acid-fastness |
| ESX-1 secretion system (ESAT-6, CFP-10) | Phagosomal escape; granuloma formation; cell-to-cell spread |
| Catalase-peroxidase (KatG) | Neutralises reactive oxygen species; activates isoniazid (prodrug) |
| Sulfolipids | Inhibit macrophage activation |
Extrapulmonary TB - Pathogenesis
Extrapulmonary TB results from haematogenous or lymphatic dissemination:
| Site | Mechanism | Frequency |
|---|---|---|
| Lymph node | Direct lymphatic spread | Most common EPTB (35%) |
| Pleural | Rupture of subpleural focus; hypersensitivity reaction | 15-20% |
| CNS | Haematogenous spread; rupture of Rich focus | 5-10% |
| Genitourinary | Haematogenous to kidneys | 5-10% |
| Bone/joint | Haematogenous; Batson's venous plexus (spine) | 10% |
| Miliary | Massive haematogenous dissemination | 1-2% |
| Pericardial | Direct spread from mediastinal nodes | 1-2% |
| Abdominal | Swallowed sputum; haematogenous | 3-5% |
4. Clinical Presentation
Pulmonary Tuberculosis
Constitutional Symptoms:
| Symptom | Frequency | Characteristics |
|---|---|---|
| Fever | 60-85% | Low-grade, often evening rise (Hippocratic fever pattern) |
| Night sweats | 50-70% | Drenching; require change of night clothes |
| Weight loss | 60-80% | Often > 5% body weight; anorexia common |
| Fatigue/malaise | 70-80% | Non-specific; may be profound |
Respiratory Symptoms:
| Symptom | Frequency | Clinical Features |
|---|---|---|
| Chronic cough | 90-95% | Productive; duration > 2-3 weeks; progressive |
| Sputum production | 70-80% | Mucopurulent; may be blood-streaked |
| Haemoptysis | 20-30% | From cavitary disease; risk of massive haemoptysis |
| Chest pain | 20-30% | Pleuritic if pleural involvement |
| Dyspnoea | 20-30% | Indicates extensive disease or complication |
Physical Signs:
| Sign | Significance |
|---|---|
| Cachexia | Chronic disease; poor prognosis indicator |
| Fever | May be absent; low-grade if present |
| Tachycardia | Sepsis; advanced disease |
| Pallor | Anaemia of chronic disease |
| Finger clubbing | Chronic suppurative disease; bronchiectasis |
| Reduced chest expansion | Extensive fibrosis/effusion |
| Dull percussion | Consolidation; effusion; collapse |
| Bronchial breathing | Consolidation; cavitation |
| Crackles | Over affected areas |
| Amphoric breathing | Large cavity (rare classical sign) |
Atypical Presentations
| Population | Atypical Features |
|---|---|
| HIV-positive | Lower lobe disease; minimal cavitation; smear-negative; atypical CXR; disseminated disease common; mediastinal lymphadenopathy |
| Elderly | Non-specific symptoms; confusion; minimal cough; normal temperature; may mimic malignancy |
| Children | Primary complex; hilar lymphadenopathy; miliary pattern; CNS involvement; paucibacillary (smear often negative) |
| Diabetics | Lower lobe involvement; more extensive disease; increased mortality |
| Immunosuppressed | Rapid progression; atypical sites; disseminated disease |
Extrapulmonary Tuberculosis
Extrapulmonary TB accounts for 15-20% of all TB cases and up to 50% in HIV-positive individuals. [16]
Lymph Node TB (Scrofula)
Most common form of EPTB:
| Feature | Details |
|---|---|
| Sites | Cervical (most common), axillary, mediastinal, mesenteric |
| Presentation | Painless, progressive lymphadenopathy; matted nodes; cold abscess |
| Examination | Firm, non-tender, may be matted; sinus formation if advanced |
| Diagnosis | Fine needle aspiration/biopsy; granulomas; AFB; culture |
| Treatment | Standard 6-month regimen; paradoxical reaction common |
Pleural TB
| Feature | Details |
|---|---|
| Mechanism | Rupture of subpleural focus; hypersensitivity reaction |
| Presentation | Pleuritic chest pain; dyspnoea; dry cough; fever |
| Pleural fluid | Exudate; lymphocyte-predominant; low glucose; high ADA (> 40 U/L) |
| Diagnosis | Pleural biopsy (histology + culture) most sensitive; fluid culture low yield |
| Treatment | Standard regimen; drainage if symptomatic; steroids controversial |
Central Nervous System TB
CNS TB carries high mortality (25-30%) and morbidity if untreated. [17]
TB Meningitis:
| Feature | Details |
|---|---|
| Presentation | Subacute meningitis (weeks); headache; fever; confusion; cranial nerve palsies (VI most common) |
| CSF findings | Lymphocytic pleocytosis; elevated protein (1-5 g/L); low glucose (less than 50% plasma); positive culture (50-70%) |
| Imaging | Basal meningeal enhancement; hydrocephalus; tuberculomas; infarcts |
| Treatment | 12-month regimen; dexamethasone reduces mortality (NNT=8) [18] |
| Complications | Hydrocephalus; stroke; cranial nerve palsies; seizures; SIADH |
Tuberculoma:
- Space-occupying lesion(s); seizures; focal deficits
- May occur during treatment (paradoxical reaction)
- Ring-enhancing lesion(s) on MRI
Bone and Joint TB (Pott's Disease)
| Site | Features |
|---|---|
| Spine (most common) | Lower thoracic/lumbar most affected; back pain; kyphosis (gibbus); cold abscess; neurological deficit (paraplegia if cord compression) |
| Hip/knee | Monoarticular; chronic arthritis; pain; swelling; destruction |
| Diagnosis | MRI (disc/vertebral body involvement); CT-guided biopsy; culture |
| Treatment | 6-12 months (may extend for slow response); surgical decompression if neurological deficit |
Miliary TB
Disseminated haematogenous TB representing failure of containment:
| Feature | Details |
|---|---|
| Presentation | Fever; weight loss; hepatosplenomegaly; pancytopenia; multiorgan dysfunction |
| CXR | Diffuse 1-3 mm nodules ("millet seeds") in all lung fields |
| HRCT | Random micronodules; ground-glass opacity |
| Diagnosis | Bronchoscopy with BAL; liver/bone marrow biopsy; cultures from multiple sites |
| Prognosis | High mortality without treatment; associated with HIV |
Genitourinary TB
| Feature | Details |
|---|---|
| Presentation | Sterile pyuria; haematuria; frequency; renal colic; epididymitis; infertility |
| Imaging | Calcified kidneys; hydronephrosis; ureteric strictures; "thimble bladder" |
| Diagnosis | Early morning urine × 3 (AFB and culture); characteristic imaging |
| Treatment | Standard regimen; surgical intervention for obstruction |
Pericardial TB
| Feature | Details |
|---|---|
| Presentation | Chest pain; dyspnoea; pericardial friction rub; signs of tamponade or constriction |
| Diagnosis | Pericardiocentesis (lymphocytic exudate, high ADA); pericardial biopsy |
| Complications | Tamponade (emergency drainage); constrictive pericarditis |
| Treatment | 6-month regimen; adjunctive steroids reduce mortality (controversial); pericardiectomy for constriction |
Abdominal TB
| Feature | Details |
|---|---|
| Forms | Intestinal (ileocaecal most common); peritoneal (ascites); lymphadenopathy |
| Presentation | Abdominal pain; distension; weight loss; fever; bowel obstruction |
| Ascitic fluid | High protein; lymphocytic; high ADA |
| Diagnosis | CT findings; laparoscopy; tissue biopsy |
| Differential | Crohn's disease (major differential for ileocaecal TB) |
Red Flags and Emergency Presentations
[!CAUTION] Immediate Action Required:
- Massive haemoptysis (> 200 mL): Risk of asphyxiation; lateral decubitus (affected side down); urgent bronchial artery embolisation
- Respiratory failure: ICU; mechanical ventilation; high mortality
- TB meningitis: Immediate treatment; do not await confirmation; adjunctive steroids
- Spinal TB with neurological deficit: Urgent MRI; neurosurgical review; decompression
- Pericardial tamponade: Emergency pericardiocentesis
- Miliary TB with multiorgan failure: ICU; consider MDR-TB
5. Investigations
Diagnostic Algorithm
SUSPECTED TUBERCULOSIS
↓
┌────────────────────────────────────────────────┐
│ INITIAL ASSESSMENT │
│ • Symptoms: cough > 2 weeks, fever, │
│ night sweats, weight loss, haemoptysis │
│ • Risk factors: endemic country, HIV, │
│ contact, immunosuppression │
│ • Examination: signs of disease/complications │
└────────────────────────────────────────────────┘
↓
┌────────────────────────────────────────────────┐
│ RESPIRATORY ISOLATION │
│ • Single room, negative pressure if available │
│ • N95/FFP2 for staff; surgical mask for patient│
└────────────────────────────────────────────────┘
↓
┌────────────────────────────────────────────────┐
│ IMMEDIATE INVESTIGATIONS │
│ • Sputum × 3 (spot, early morning, spot) │
│ - Smear microscopy (ZN/auramine) │
│ - Xpert MTB/RIF (first-line molecular) │
│ - Mycobacterial culture + DST │
│ • Chest X-ray (PA) │
│ • HIV test (MANDATORY in all patients) │
│ • Baseline bloods: FBC, LFTs, U&E, HbA1c │
│ • Hepatitis B/C serology │
└────────────────────────────────────────────────┘
↓
Xpert MTB/RIF or Smear positive?
↓ Yes ↓ No
┌───────────────┐ ┌─────────────────────────┐
│ START │ │ High clinical suspicion?│
│ TREATMENT │ │ Consider: │
│ IMMEDIATELY │ │ • Induced sputum │
│ │ │ • Bronchoscopy + BAL │
│ • Notify │ │ • Empirical treatment │
│ public │ │ while awaiting culture│
│ health │ │ │
└───────────────┘ └─────────────────────────┘
Microbiological Investigations
Sputum Testing
| Test | Principle | Sensitivity | Specificity | Time to Result | Notes |
|---|---|---|---|---|---|
| AFB Smear (ZN/auramine) | Direct visualisation | 50-80% | 95-99% | Hours | Requires 5,000-10,000 bacilli/mL; auramine (fluorescent) superior |
| Xpert MTB/RIF | PCR for TB DNA + rpoB mutations | 89% (smear+); 67% (smear−) | 98% | 2 hours | Detects rifampicin resistance; WHO-recommended first-line molecular test [4] |
| Xpert MTB/RIF Ultra | Enhanced PCR | 95% (smear+); 88% (smear−) | 96% | 2 hours | Improved sensitivity for smear-negative and HIV+ [8] |
| Culture (liquid - MGIT) | Mycobacterial growth detection | 80-85% | 100% | 1-3 weeks | Gold standard; allows DST |
| Culture (solid - LJ) | Growth on egg-based medium | 80-85% | 100% | 4-8 weeks | Less sensitive; longer time |
| Line Probe Assay (LPA) | Molecular detection of resistance mutations | High | High | 24-48 hours | First/second-line resistance detection; GenoType MTBDRplus/sl |
Sample Collection:
- Collect 3 sputum specimens (spot → early morning → spot) on consecutive days
- Minimum 5 mL volume per specimen
- If unable to expectorate: induced sputum (3% hypertonic saline nebulisation)
- If still unsuccessful: bronchoscopy with BAL
Specimen Handling:
- Transport to laboratory within 24 hours
- Refrigerate at 4°C if delayed
- Decontamination (NaOH-NALC) before culture
Drug Susceptibility Testing (DST)
Essential for all culture-positive cases:
| Method | Drugs Tested | Time | Notes |
|---|---|---|---|
| Phenotypic (culture-based) | All first/second-line | 2-4 weeks after positive culture | Gold standard; critical concentration method |
| Xpert MTB/RIF | Rifampicin | 2 hours | 95% accuracy for RIF resistance |
| Line Probe Assays | INH, RIF, FQ, aminoglycosides | 24-48 hours | Direct from smear+ specimens |
| Whole genome sequencing | All | Days | Comprehensive resistance profiling; emerging standard |
Chest Radiography
Classic CXR Findings in Pulmonary TB:
| Finding | Description | Significance |
|---|---|---|
| Upper lobe infiltrates | Apical/posterior segments; patchy consolidation | Most characteristic; high oxygen tension favours bacilli |
| Cavitation | Air-fluid level within consolidation; thick wall | Indicates high bacillary load; highly infectious; treatment response monitor |
| Fibrosis | Volume loss; traction bronchiectasis; architectural distortion | Healing/healed disease |
| Hilar/mediastinal lymphadenopathy | Lymph node enlargement | More common in children, HIV+, primary TB |
| Pleural effusion | Usually unilateral | Pleural TB; typically lymphocytic exudate |
| Miliary pattern | Diffuse 1-3 mm nodules | Haematogenous dissemination; high mortality |
| Lower lobe disease | Consolidation in lower lobes | Atypical; consider HIV, diabetes, endobronchial spread |
Atypical CXR Findings (HIV-positive):
- Normal CXR (up to 10-15%)
- Mediastinal lymphadenopathy without parenchymal disease
- Lower/middle lobe disease
- Minimal cavitation
- Miliary pattern more common
Additional Investigations
Baseline Blood Tests
| Test | Purpose | Expected Findings |
|---|---|---|
| Full blood count | Anaemia; leukocytosis | Normocytic anaemia (chronic disease); lymphopenia (if HIV); monocytosis |
| Liver function tests | Baseline; hepatotoxicity monitoring | Often normal; mild ALP elevation; deranged in hepatic TB |
| Urea and electrolytes | Baseline; hyponatraemia | SIADH (CNS TB); renal TB |
| HbA1c | Screen for diabetes | Diabetes increases TB risk 2-3× |
| C-reactive protein | Inflammatory marker; treatment response | Elevated; useful for monitoring |
| Vitamin D | Often deficient in TB | Low levels associated with TB risk |
| Hepatitis B/C serology | Baseline before hepatotoxic drugs | Screen for chronic hepatitis |
HIV Testing
MANDATORY in all TB patients:
- Offer rapid HIV test at diagnosis
- 12% global co-infection rate; higher in endemic areas
- Co-infection affects treatment timing, regimen, prognosis
- If HIV-positive: CD4 count, viral load, start ART
Tuberculin Skin Test (TST) and Interferon-Gamma Release Assays (IGRA)
| Feature | TST (Mantoux) | IGRA (QuantiFERON-TB Gold, T-SPOT.TB) |
|---|---|---|
| Principle | Delayed-type hypersensitivity to PPD | In vitro IFN-γ release to TB-specific antigens (ESAT-6, CFP-10) |
| Administration | Intradermal injection; read at 48-72 hours | Single blood sample |
| Interpretation | Induration ≥5 mm (HIV+), ≥10 mm (high-risk), ≥15 mm (low-risk) | Positive/negative/indeterminate |
| False positives | BCG vaccination; NTM infection | Minimal (antigens specific to M. tuberculosis) |
| False negatives | Anergy (HIV, malnutrition, miliary TB) | Anergy; technical failure |
| Use | LTBI screening; contact tracing | LTBI screening (preferred in BCG-vaccinated) |
| Limitation | Cannot distinguish LTBI from active TB | Cannot distinguish LTBI from active TB |
Investigations for Extrapulmonary TB
| Site | Key Investigations |
|---|---|
| Lymph node | FNA/excision biopsy for AFB, culture, histology (granulomas) |
| Pleural | Thoracentesis (lymphocytic exudate, ADA > 40 U/L); pleural biopsy |
| CNS | Lumbar puncture (lymphocytic, high protein, low glucose); MRI brain/spine |
| Bone/joint | MRI; CT-guided biopsy; synovial fluid culture |
| Genitourinary | Early morning urine × 3; CT urogram; cystoscopy |
| Pericardial | Echocardiography; pericardiocentesis (ADA > 40); pericardial biopsy |
| Abdominal | CT abdomen/pelvis; ascitic tap (ADA > 40); colonoscopy/laparoscopy |
| Miliary | HRCT chest; bronchoscopy + BAL; liver/bone marrow biopsy |
6. Management
Management Algorithm
CONFIRMED/PROBABLE PULMONARY TB
↓
┌──────────────────────────────────────────────────┐
│ INITIAL STEPS (Day 1) │
│ • Respiratory isolation (airborne precautions) │
│ • HIV test (MANDATORY) │
│ • Baseline bloods (FBC, LFT, U&E, HbA1c) │
│ • Notify public health (statutory) │
│ • Assess for drug resistance risk │
│ • Assess drug interactions │
│ • Initiate contact tracing │
└──────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────┐
│ STANDARD TREATMENT (2HRZE/4HR) │
│ │
│ INTENSIVE PHASE (2 months): │
│ • Rifampicin 10 mg/kg (max 600 mg) daily │
│ • Isoniazid 5 mg/kg (max 300 mg) daily │
│ • Pyrazinamide 25 mg/kg (max 2 g) daily │
│ • Ethambutol 15 mg/kg daily │
│ + Pyridoxine 10-25 mg daily │
│ │
│ CONTINUATION PHASE (4 months): │
│ • Rifampicin 10 mg/kg (max 600 mg) daily │
│ • Isoniazid 5 mg/kg (max 300 mg) daily │
│ + Pyridoxine 10-25 mg daily │
│ │
│ TOTAL DURATION: 6 months │
└──────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────┐
│ MONITORING │
│ • Clinical: symptoms, weight, adherence │
│ • Sputum at 2 months (should be culture-negative)│
│ • LFTs: baseline, 2 weeks, then monthly │
│ • Visual acuity: baseline, monthly (ethambutol) │
│ • Drug levels: if poor response/toxicity │
│ • CXR: at completion │
└──────────────────────────────────────────────────┘
↓
End of treatment
↓
┌──────────────────────────────────────────────────┐
│ TREATMENT OUTCOMES │
│ • Cured: culture-negative at end of treatment │
│ • Completed: finished treatment, no culture │
│ • Failed: culture-positive at 5 months │
│ • Died: during treatment │
│ • Lost to follow-up: interrupted > 2 months │
└──────────────────────────────────────────────────┘
First-Line Anti-Tuberculosis Drugs
| Drug | Mechanism | Dose | Max Dose | Key Side Effects | Monitoring |
|---|---|---|---|---|---|
| Rifampicin (R) | Inhibits RNA polymerase | 10 mg/kg | 600 mg | Hepatotoxicity; orange secretions; drug interactions (CYP450 inducer); flu-like syndrome | LFTs monthly |
| Isoniazid (H) | Inhibits mycolic acid synthesis | 5 mg/kg | 300 mg | Hepatotoxicity; peripheral neuropathy; rash; lupus-like syndrome | LFTs monthly; give pyridoxine |
| Pyrazinamide (Z) | Unknown; active at low pH | 25 mg/kg | 2 g | Hepatotoxicity (most hepatotoxic); hyperuricaemia; arthralgia; rash | LFTs monthly; uric acid |
| Ethambutol (E) | Inhibits arabinosyltransferase | 15 mg/kg | - | Optic neuritis (dose-related); colour blindness (red-green) | Visual acuity/colour at baseline then monthly |
Pyridoxine Supplementation:
- Prevents isoniazid-induced peripheral neuropathy
- Dose: 10-25 mg daily
- Essential for high-risk groups: HIV, diabetes, alcohol excess, malnutrition, pregnancy, chronic renal failure
Fixed-Dose Combinations (FDCs)
WHO recommends FDCs to improve adherence:
- Intensive phase: RHZE tablet (e.g., Rimstar)
- Continuation phase: RH tablet
- Dose by weight bands
Treatment of Specific Situations
Extrapulmonary TB
| Site | Duration | Special Considerations |
|---|---|---|
| Lymph node | 6 months | Paradoxical reactions common (new/enlarging nodes) |
| Pleural | 6 months | Drainage if symptomatic; steroids controversial |
| CNS (meningitis/tuberculoma) | 12 months | Dexamethasone 0.4 mg/kg/day (2 weeks) tapering over 6-8 weeks [18]; reduces mortality |
| Bone/joint | 6-12 months | May extend if slow response; surgical intervention if instability |
| Pericardial | 6 months | Steroids may reduce constriction risk; pericardiectomy if constrictive |
| Miliary | 6-12 months | Prolonged treatment if CNS involvement |
| Genitourinary | 6 months | Urology input for obstruction |
TB in HIV Co-infection
| Aspect | Recommendation |
|---|---|
| TB treatment | Standard regimens; 6 months (12 if CNS) |
| ART initiation | Within 2 weeks if CD4 less than 50; within 8 weeks if CD4 > 50 [19] |
| Drug interactions | Rifabutin substituted for rifampicin with PIs; NNRTI dose adjustment |
| IRIS | Immune reconstitution inflammatory syndrome; steroids if severe |
| Cotrimoxazole | Continue prophylaxis until immune reconstitution |
Drug-Resistant TB
| Type | Definition | Treatment Approach |
|---|---|---|
| Isoniazid monoresistance | Resistant to INH only | 6REZ or 9RE; levofloxacin may be added [5] |
| Rifampicin-resistant (RR-TB) | Resistant to RIF ± other drugs | Treat as MDR-TB until full DST available |
| MDR-TB | Resistant to at least RIF and INH | WHO Group A, B, C drugs; 9-18 months; specialist management [6] |
| Pre-XDR-TB | MDR + resistance to fluoroquinolone | Bedaquiline-based regimens |
| XDR-TB | MDR + resistance to FQ + bedaquiline/linezolid | Highly individualised; limited options |
WHO Drug Groups for MDR-TB:
- Group A (preferred): Levofloxacin/moxifloxacin, bedaquiline, linezolid
- Group B (add to A): Clofazimine, cycloserine/terizidone
- Group C (if A/B cannot be used): Ethambutol, delamanid, pyrazinamide, imipenem, amikacin, ethionamide
Key Points:
- Bedaquiline-based shorter regimens (BPaLM/BPaL): 6 months total; high efficacy [20]
- All MDR-TB patients should be managed by specialist MDR-TB services
- DOT mandatory
- Monthly monitoring for adverse effects
Directly Observed Therapy (DOT)
| Aspect | Recommendation |
|---|---|
| Definition | Trained observer watches patient swallow each dose |
| Indications | All MDR-TB; homeless; previous default; prison; substance use; concern about adherence |
| Setting | Clinic, home, workplace, pharmacy |
| Benefit | Improves treatment completion; reduces resistance |
Drug Interactions
| Drug | Interaction with Rifampicin | Management |
|---|---|---|
| Warfarin | Reduced effect (CYP2C9/3A4 induction) | Increase warfarin dose; monitor INR closely |
| Oral contraceptives | Reduced efficacy | Use alternative contraception |
| Methadone | Reduced levels; withdrawal | Increase methadone dose |
| Corticosteroids | Reduced effect | Double steroid dose |
| Statins | Reduced effect | Monitor; may need higher doses |
| Antiretrovirals (PIs) | Drastically reduced PI levels | Substitute rifabutin for rifampicin |
| Antiretrovirals (NNRTIs) | Reduced NNRTI levels | Efavirenz dose adjust; avoid nevirapine |
| Calcineurin inhibitors | Subtherapeutic levels | Monitor levels; increase dose significantly |
| Azole antifungals | Reduced effect | Increase dose or use alternative |
| DOACs | Reduced effect | Avoid; use warfarin with monitoring |
Hepatotoxicity Management
Definition: ALT > 3× ULN with symptoms OR ALT > 5× ULN without symptoms
Management Algorithm:
- Stop all hepatotoxic drugs (RIF, INH, PZA)
- Exclude other causes (viral hepatitis, alcohol)
- Wait for LFTs to normalise
- Reintroduce one drug at a time (RIF first, then INH, then PZA)
- If PZA implicated, may omit and extend treatment to 9 months (2HRE/7HR)
- If severe/recurrent hepatotoxicity, use non-hepatotoxic regimen (fluoroquinolone, aminoglycoside, ethambutol, cycloserine)
Infection Control
| Measure | Implementation |
|---|---|
| Airborne precautions | Negative pressure room; 6-12 air changes/hour |
| PPE for staff | N95/FFP2 respirator; fit-tested |
| Patient mask | Surgical mask when leaving room |
| Duration of isolation | Until smear-negative on treatment (usually 2 weeks if drug-sensitive) |
| Visitor restrictions | No immunocompromised visitors |
| Transport | Surgical mask; minimise time in shared areas |
Public Health Measures
| Action | Details |
|---|---|
| Statutory notification | Notify local public health within 3 days |
| Contact tracing | Identify household and close contacts; screen within 2 weeks |
| Contact screening | Symptom inquiry, CXR, IGRA (or TST) |
| LTBI treatment | Treat contacts with LTBI to prevent progression |
| Outbreak investigation | If linked cases identified |
7. Latent TB Infection (LTBI)
Definition and Significance
LTBI represents a state of persistent immune response to TB antigens without clinical, radiological, or microbiological evidence of active disease. [21]
| Aspect | Details |
|---|---|
| Global prevalence | ~2 billion (25% world population) |
| Lifetime reactivation risk | 5-10% (immunocompetent); higher with immunosuppression |
| Importance | Reservoir for future active TB; treating LTBI prevents disease |
Who to Screen for LTBI
| Group | Rationale |
|---|---|
| Close contacts of smear-positive TB | High transmission risk |
| HIV-positive individuals | High reactivation risk |
| Before starting TNF-α inhibitors | Reactivation risk |
| Before transplantation | Immunosuppression risk |
| New entrants from high-incidence countries | LTBI prevalence |
| Healthcare workers in high-risk settings | Occupational exposure |
| Prisoners | Institutional transmission |
| Homeless individuals | High-risk population |
Diagnosis of LTBI
| Test | Sensitivity | Specificity | Advantages | Disadvantages |
|---|---|---|---|---|
| IGRA | 80-90% | 95-99% | Single visit; unaffected by BCG; objective | Cost; laboratory required |
| TST | 70-80% | 56-95% (BCG-dependent) | Low cost; no laboratory | False positives with BCG; requires return visit |
NICE Recommendation (UK):
- Use IGRA as first-line for LTBI screening
- TST acceptable if IGRA unavailable
- Single positive test sufficient to diagnose LTBI (if active TB excluded)
LTBI Treatment Regimens
| Regimen | Duration | Efficacy | Notes |
|---|---|---|---|
| 3HP (Rifapentine + INH weekly) | 3 months | 85-90% | Preferred; DOT; fewer hepatotoxicity |
| 4R (Rifampicin daily) | 4 months | 85% | Good adherence; fewer adverse effects |
| 3HR (Rifampicin + INH daily) | 3 months | 80% | Alternative option |
| 6H/9H (INH daily) | 6-9 months | 60-90% | Traditional; hepatotoxicity risk; poor adherence |
Key Considerations:
- Exclude active TB before starting LTBI treatment
- Check for drug interactions
- Monitor LFTs (baseline, monthly)
- Counsel on hepatotoxicity symptoms
8. BCG Vaccination
BCG Vaccine Overview
| Aspect | Details |
|---|---|
| Composition | Live attenuated M. bovis (Bacille Calmette-Guérin) |
| Efficacy | 70-80% protection against severe TB (miliary, meningitis) in children; variable protection against pulmonary TB (0-80%) [22] |
| Duration of protection | Wanes over 10-15 years |
| Route | Intradermal injection (deltoid) |
| Dose | 0.05 mL (less than 12 months); 0.1 mL (≥12 months) |
UK BCG Policy (Since 2005)
Universal neonatal BCG was discontinued; targeted vaccination:
| Group | Recommendation |
|---|---|
| Neonates in high-incidence areas (≥40/100,000) | BCG at birth |
| Infants with parent/grandparent from high-incidence country (≥40/100,000) | BCG at birth |
| Previously unvaccinated children less than 16 from high-incidence countries | BCG after negative TST/IGRA |
| Healthcare workers at risk | BCG if TST/IGRA negative |
| Laboratory workers handling TB specimens | BCG if TST/IGRA negative |
BCG Complications
| Complication | Frequency | Management |
|---|---|---|
| Local abscess | 1-5% | Usually self-limiting; drain if fluctuant |
| Regional lymphadenitis | 0.1-1% | Observation; aspirate if suppurative |
| BCG-itis (disseminated) | Rare (immunocompromised) | Anti-TB treatment (INH + RIF; not PZA) |
| Keloid scarring | Common | Cosmetic concern |
9. Complications of Tuberculosis
Pulmonary Complications
| Complication | Mechanism | Incidence | Management |
|---|---|---|---|
| Massive haemoptysis | Rasmussen aneurysm erosion; bronchiectasis | 5-10% | Airway protection; bronchial artery embolisation; surgery if recurrent |
| Respiratory failure | Extensive disease; ARDS | Variable | ICU; mechanical ventilation; high mortality |
| Pneumothorax | Cavity rupture | Rare | Chest drain; may require pleurodesis |
| Bronchiectasis | Post-TB fibrosis | 30-50% survivors | Airway clearance; treat exacerbations |
| Aspergilloma | Fungal ball in residual cavity | 10-15% of cavities | Observation; surgery/embolisation if symptomatic |
| Pleural empyema | Secondary infection of effusion | Rare | Chest drain; prolonged antibiotics |
| Cor pulmonale | Pulmonary destruction | Late complication | Oxygen; treat underlying disease |
Drug-Related Complications
| Drug | Complication | Frequency | Prevention/Management |
|---|---|---|---|
| INH/RIF/PZA | Hepatotoxicity | 2-8% | Monitor LFTs; stop drugs if ALT > 5× ULN |
| Isoniazid | Peripheral neuropathy | 2% | Pyridoxine supplementation |
| Isoniazid | Drug-induced lupus | Rare | Stop isoniazid; usually reversible |
| Ethambutol | Optic neuritis | 1-5% (dose-dependent) | Baseline visual acuity; monthly monitoring; stop drug immediately |
| Pyrazinamide | Hyperuricaemia/gout | Common | Allopurinol if symptomatic |
| Rifampicin | Drug interactions | Universal | Review all medications |
| Rifampicin | Orange secretions | Universal | Patient counselling |
| Aminoglycosides | Ototoxicity, nephrotoxicity | Dose-dependent | Audiometry; renal function monitoring |
Paradoxical Reactions
Worsening or new TB symptoms/lesions despite appropriate treatment:
| Feature | Details |
|---|---|
| Mechanism | Immune reconstitution (especially with ART initiation in HIV) |
| Timing | 2 weeks to 3 months after starting treatment |
| Manifestations | Fever; enlarging lymph nodes; new effusions; worsening CXR |
| Management | Continue TB treatment; NSAIDs; corticosteroids if severe |
| Differentiate from | Treatment failure (positive cultures); drug resistance |
10. Prognosis and Outcomes
Treatment Outcomes (Drug-Sensitive TB)
| Outcome | Definition | Target Rate |
|---|---|---|
| Treatment success | Cured + completed | ≥90% |
| Cured | Culture-negative at end of treatment | - |
| Completed | Finished treatment without culture confirmation | - |
| Failed | Culture-positive at 5 months or later | less than 5% |
| Died | Death from any cause during treatment | less than 5% |
| Lost to follow-up | Treatment interrupted ≥2 consecutive months | less than 5% |
Prognostic Factors
Poor Prognostic Factors:
| Factor | Impact |
|---|---|
| HIV co-infection (untreated) | Mortality 2-4× higher |
| MDR/XDR-TB | Lower cure rates; higher mortality |
| Delayed diagnosis | More extensive disease; higher mortality |
| Advanced disease (extensive CXR changes) | Longer time to culture conversion |
| Poor nutritional status | Impaired immune response |
| Diabetes mellitus | Higher failure and relapse rates |
| Older age | Increased mortality |
| Poor adherence | Treatment failure; resistance |
| Drug toxicity | Treatment interruption |
Long-Term Sequelae
| Sequela | Description |
|---|---|
| Post-TB lung disease | Bronchiectasis; fibrosis; restrictive defect; chronic respiratory symptoms |
| Chronic respiratory failure | May require long-term oxygen |
| Recurrent infections | Bronchiectasis-related |
| Reduced quality of life | Persistent symptoms; psychological impact |
| Increased mortality risk | Persists beyond treatment completion |
Follow-Up
| Timepoint | Assessment |
|---|---|
| End of treatment | Clinical assessment; CXR; document outcome |
| 12 months post-treatment | Clinical review; consider CXR if high-risk for relapse |
| Long-term | Respiratory follow-up if significant post-TB lung disease |
11. Prevention and Control
Hierarchy of TB Prevention
- Primary prevention: BCG vaccination (protects against severe childhood TB)
- Secondary prevention: Early case detection; contact tracing; LTBI treatment
- Tertiary prevention: Effective treatment of active disease; infection control
Contact Tracing Protocol
| Step | Action |
|---|---|
| 1 | Identify index case (especially smear-positive pulmonary) |
| 2 | Define infectious period (3 months before diagnosis; until 2 weeks on treatment) |
| 3 | Identify contacts (household, close, workplace, social) |
| 4 | Prioritise: household contacts; immunocompromised; children less than 5 years |
| 5 | Screen contacts: symptom inquiry, CXR, IGRA/TST |
| 6 | If active TB: notify; treat |
| 7 | If LTBI: offer preventive treatment |
| 8 | If negative: repeat screening at 8-12 weeks (window period) |
New Entrant Screening
UK policy for migrants from high-incidence countries (≥40/100,000):
| Age | Screening |
|---|---|
| All ages | CXR at port of entry (if > 11 years old) |
| Children less than 16 years | BCG if unvaccinated and IGRA negative |
| Adults | IGRA; treat LTBI if positive |
12. Key Guidelines
Major Guidelines Referenced
| Guideline | Organisation | Year | Key Recommendations |
|---|---|---|---|
| Tuberculosis (NG33) | NICE (UK) | 2016 (updated 2023) | Comprehensive UK guidance; diagnosis, treatment, contact tracing, LTBI [10] |
| WHO Consolidated Guidelines on TB | WHO | 2022 | Global standards; treatment regimens; drug-resistant TB [5,6] |
| WHO Rapid Communication on TB | WHO | 2023 | Updated recommendations including shorter MDR-TB regimens [20] |
| Official ATS/CDC/IDSA Clinical Practice Guidelines | ATS/CDC/IDSA | 2016 | North American guidance on drug-susceptible TB [23] |
| Guidelines for Treatment of Drug-Susceptible TB | WHO | 2022 | Updated first-line treatment recommendations |
| BHIVA Guidelines for TB/HIV Co-infection | BHIVA | 2019 | ART timing; drug interactions; IRIS management [19] |
13. Examination Focus
Common Exam Questions
- "What are the risk factors for tuberculosis?"
- "How would you investigate a patient with suspected pulmonary TB?"
- "Describe the first-line treatment regimen for TB and the mechanism of each drug."
- "What are the side effects of anti-TB medications and how would you monitor for them?"
- "How do you differentiate latent TB infection from active TB disease?"
- "What are the features of drug-resistant TB and how would you manage it?"
- "Describe the pathogenesis of TB from primary infection to reactivation."
- "What are the indications for and options for LTBI treatment?"
- "How would you manage a patient with TB meningitis?"
- "What public health measures are required for a patient diagnosed with TB?"
Viva Points
Opening Statement: "Tuberculosis is a chronic granulomatous infection caused by Mycobacterium tuberculosis, primarily affecting the lungs but capable of extrapulmonary dissemination. It affects 10.6 million people annually and causes 1.3 million deaths globally. Diagnosis relies on clinical suspicion, chest radiography, and microbiological confirmation including molecular testing (Xpert MTB/RIF) and culture. Standard treatment is a 6-month regimen: 2 months of RIPE (Rifampicin, Isoniazid, Pyrazinamide, Ethambutol) followed by 4 months of Rifampicin and Isoniazid."
Key Statistics to Quote:
- Global burden: 10.6 million cases, 1.3 million deaths/year
- UK incidence: 6.6 per 100,000 (4,425 cases in 2022)
- HIV co-infection: 12% globally
- LTBI → active TB: 5-10% lifetime risk
- Xpert MTB/RIF sensitivity: 89% (smear+), 67% (smear−)
- Treatment success rate: > 95% for drug-sensitive TB
Classification to Know: Drug resistance categories (monoresistant, RR-TB, MDR-TB, pre-XDR, XDR-TB)
Evidence to Cite:
- Xpert MTB/RIF landmark study (NEJM 2010) [4]
- Theron et al. Xpert Ultra (Lancet ID 2018) [8]
- Prasad et al. steroids in TB meningitis Cochrane review [18]
Common Mistakes
❌ Mistakes that fail candidates:
- Not testing for HIV in every TB patient
- Forgetting pyridoxine supplementation with isoniazid
- Not monitoring LFTs during treatment
- Failing to notify public health
- Not recognising that TST/IGRA cannot distinguish LTBI from active TB
- Missing the need for 12-month treatment in TB meningitis
- Not adjusting for drug interactions with rifampicin
- Treating XDR-TB without specialist input
- Forgetting to ask about prior TB treatment (resistance risk)
- Not considering extrapulmonary TB in HIV-positive patients with fever
Model Answers
Q: A 35-year-old man from India presents with 4 weeks of productive cough, fever, night sweats, and 5 kg weight loss. How would you investigate and manage him?
A: "This presentation raises high clinical suspicion for pulmonary tuberculosis given the chronic cough, constitutional symptoms, and epidemiological risk factor of birth in a high-incidence country.
My immediate management would be:
- Respiratory isolation with airborne precautions
- Sputum samples × 3 for AFB smear, Xpert MTB/RIF, and mycobacterial culture
- Chest X-ray looking for upper lobe infiltrates, cavitation, or effusion
- HIV test - this is mandatory in all TB patients
- Baseline blood tests: FBC, LFTs, U&E, HbA1c, hepatitis serology
If Xpert is positive or there is high clinical suspicion, I would start standard treatment: 2 months of Rifampicin, Isoniazid, Pyrazinamide, and Ethambutol, followed by 4 months of Rifampicin and Isoniazid. I would add Pyridoxine to prevent isoniazid-induced neuropathy.
I would notify public health within 3 days and initiate contact tracing for household contacts.
Monitoring would include clinical review, sputum culture at 2 months (expecting culture conversion), monthly LFTs, and visual acuity testing for ethambutol toxicity."
Differentials to Consider
| Differential | Key Distinguishing Features |
|---|---|
| Lung cancer | Older age; smoking history; mass lesion; no response to TB treatment |
| Community-acquired pneumonia | Acute onset; lobar consolidation; responds to antibiotics |
| Non-tuberculous mycobacteria | Similar presentation; different species on culture; often structural lung disease |
| Sarcoidosis | Bilateral hilar lymphadenopathy; elevated ACE; non-caseating granulomas |
| Histoplasmosis/other fungi | Travel to endemic areas; immunosuppression; specific fungal testing |
| Nocardiosis | Immunosuppression; branching gram-positive rods |
| Lymphoma | Mediastinal lymphadenopathy; B symptoms; biopsy diagnostic |
14. Patient Communication
What is Tuberculosis?
"TB is a bacterial infection that mainly affects your lungs. It's caused by a germ called Mycobacterium tuberculosis that spreads through the air when someone with TB coughs or sneezes. TB has been around for thousands of years, and while it's a serious infection, it's completely curable with the right treatment."
Is It Curable?
"Yes, TB is curable with a course of antibiotics taken for 6 months. With proper treatment, over 95% of people with TB are cured. The key is taking all your medications exactly as prescribed, even when you start feeling better."
The Treatment
"Your treatment involves taking several tablets every day for 6 months:
- For the first 2 months, you'll take 4 different medications
- For the remaining 4 months, you'll take 2 medications
- You'll also take vitamin B6 to protect your nerves
It's very important to:
- Take your medications at the same time every day
- Never miss doses - this can cause the bacteria to become resistant
- Complete the full course even when you feel well"
Side Effects to Report
"Please tell us immediately if you experience:
- Feeling sick, vomiting, or losing your appetite
- Yellow skin or eyes (jaundice)
- Dark urine or pale stools
- Numbness or tingling in your hands or feet
- Visual problems - any change in your vision or colour perception
- Unusual bruising or bleeding
- Skin rash or itching
Your urine, sweat, and tears may turn orange-red from the medication - this is normal and harmless."
Am I Infectious?
"If you have TB in your lungs, you can spread it to others through coughing. However, after 2 weeks of treatment, you're usually no longer infectious to others. We'll test your sputum to confirm. Until then:
- Cover your mouth when coughing
- Open windows for ventilation
- Wear a mask around others
- Avoid close contact with babies or people with weak immune systems"
What About My Family and Contacts?
"We need to check people you've been in close contact with, especially those living with you. They'll be offered screening which may include:
- Questions about symptoms
- A chest X-ray
- A blood test
This is to protect them and prevent spread. If they've been infected but aren't sick, we can offer treatment to prevent them developing TB."
15. References
-
Pai M, Behr MA, Dowdy D, et al. Tuberculosis. Nat Rev Dis Primers. 2016;2:16076. doi:10.1038/nrdp.2016.76
-
World Health Organization. Global Tuberculosis Report 2023. Geneva: WHO; 2023. Available at: https://www.who.int/publications/i/item/9789240083851
-
Houben RM, Dodd PJ. The Global Burden of Latent Tuberculosis Infection: A Re-estimation Using Mathematical Modelling. PLoS Med. 2016;13(10):e1002152. doi:10.1371/journal.pmed.1002152
-
Boehme CC, Nabeta P, Hillemann D, et al. Rapid Molecular Detection of Tuberculosis and Rifampin Resistance. N Engl J Med. 2010;363(11):1005-1015. doi:10.1056/NEJMoa0907847
-
World Health Organization. WHO consolidated guidelines on tuberculosis. Module 4: treatment - drug-susceptible tuberculosis treatment. Geneva: WHO; 2022. Available at: https://www.who.int/publications/i/item/9789240048126
-
World Health Organization. WHO consolidated guidelines on tuberculosis. Module 4: treatment - drug-resistant tuberculosis treatment, 2022 update. Geneva: WHO; 2022. doi:10.2139/ssrn.4074549
-
UK Health Security Agency. Tuberculosis in England: 2023 report. London: UKHSA; 2023. Available at: https://www.gov.uk/government/publications/tuberculosis-in-england-2023-report
-
Dorman SE, Schumacher SG, Alland D, et al. Xpert MTB/RIF Ultra for detection of Mycobacterium tuberculosis and rifampicin resistance: a prospective multicentre diagnostic accuracy study. Lancet Infect Dis. 2018;18(1):76-84. doi:10.1016/S1473-3099(17)30691-6
-
Niemi M, Backman JT, Fromm MF, Neuvonen PJ, Kivistö KT. Pharmacokinetic interactions with rifampicin: clinical relevance. Clin Pharmacokinet. 2003;42(9):819-850. doi:10.2165/00003088-200342090-00003
-
National Institute for Health and Care Excellence. Tuberculosis (NG33). London: NICE; 2016 (updated 2023). Available at: https://www.nice.org.uk/guidance/ng33
-
Getahun H, Harrington M, O'Brien R, Nunn P. Diagnosis of smear-negative pulmonary tuberculosis in people with HIV infection or AIDS in resource-constrained settings: informing urgent policy changes. Lancet. 2007;369(9578):2042-2049. doi:10.1016/S0140-6736(07)60284-0
-
Lawn SD, Churchyard G. Epidemiology of HIV-associated tuberculosis. Curr Opin HIV AIDS. 2009;4(4):325-333. doi:10.1097/COH.0b013e32832c7d61
-
Baddley JW, Cantini F, Goletti D, et al. ESCMID Study Group for Infections in Compromised Hosts (ESGICH) Consensus Document on the safety of targeted and biological therapies: an infectious diseases perspective (Soluble immune effector molecules [I]: anti-tumor necrosis factor-α agents). Clin Microbiol Infect. 2018;24 Suppl 2:S10-S20. doi:10.1016/j.cmi.2017.12.025
-
Jeon CY, Murray MB. Diabetes mellitus increases the risk of active tuberculosis: a systematic review of 13 observational studies. PLoS Med. 2008;5(7):e152. doi:10.1371/journal.pmed.0050152
-
Lin HH, Ezzati M, Murray M. Tobacco smoke, indoor air pollution and tuberculosis: a systematic review and meta-analysis. PLoS Med. 2007;4(1):e20. doi:10.1371/journal.pmed.0040020
-
Peto HM, Pratt RH, Harrington TA, LoBue PA, Armstrong LR. Epidemiology of extrapulmonary tuberculosis in the United States, 1993-2006. Clin Infect Dis. 2009;49(9):1350-1357. doi:10.1086/605559
-
Thwaites GE, van Toorn R, Schoeman J. Tuberculous meningitis: more questions, still too few answers. Lancet Neurol. 2013;12(10):999-1010. doi:10.1016/S1474-4422(13)70168-6
-
Prasad K, Singh MB, Ryan H. Corticosteroids for managing tuberculous meningitis. Cochrane Database Syst Rev. 2016;4(4):CD002244. doi:10.1002/14651858.CD002244.pub4
-
Gopalan N, Andrade BB, Swaminathan S. Tuberculosis-immune reconstitution inflammatory syndrome in HIV: from pathogenesis to prediction. Expert Rev Clin Immunol. 2014;10(5):631-645. doi:10.1586/1744666X.2014.892828
-
Conradie F, Bagdasaryan TR, Borisov S, et al. Bedaquiline-Pretomanid-Linezolid Regimens for Drug-Resistant Tuberculosis. N Engl J Med. 2022;387(9):810-823. doi:10.1056/NEJMoa2119430
-
Getahun H, Matteelli A, Chaisson RE, Raviglione M. Latent Mycobacterium tuberculosis infection. N Engl J Med. 2015;372(22):2127-2135. doi:10.1056/NEJMra1405427
-
Roy A, Eisenhut M, Harris RJ, et al. Effect of BCG vaccination against Mycobacterium tuberculosis infection in children: systematic review and meta-analysis. BMJ. 2014;349:g4643. doi:10.1136/bmj.g4643
-
Nahid P, Dorman SE, Alipanah N, et al. Official American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America Clinical Practice Guidelines: Treatment of Drug-Susceptible Tuberculosis. Clin Infect Dis. 2016;63(7):e147-e195. doi:10.1093/cid/ciw376
-
Lawn SD, Zumla AI. Tuberculosis. Lancet. 2011;378(9785):57-72. doi:10.1016/S0140-6736(10)62173-3
-
Theron G, Venter R, Calligaro G, et al. Xpert MTB/RIF Results in Patients With Previous Tuberculosis: Can We Distinguish True From False Positive Results? Clin Infect Dis. 2016;62(8):995-1001. doi:10.1093/cid/civ1223
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Immunology of Granulomatous Inflammation
- Respiratory Physiology
Differentials
Competing diagnoses and look-alikes to compare.
- Lung Cancer
- Non-tuberculous Mycobacteria
- Sarcoidosis
Consequences
Complications and downstream problems to keep in mind.
- Bronchiectasis
- Chronic Respiratory Failure