Cryoglobulinaemic Vasculitis
Cryoglobulinaemic vasculitis is a systemic small-vessel vasculitis mediated by cryoglobulins—immunoglobulins that revers... MRCP exam preparation.
What matters first
Cryoglobulinaemic vasculitis is a systemic small-vessel vasculitis mediated by cryoglobulins—immunoglobulins that revers... MRCP exam preparation.
Severe renal involvement (MPGN with rapidly progressive GN)
5 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Severe renal involvement (MPGN with rapidly progressive GN)
- Hyperviscosity syndrome (confusion, visual changes, mucosal bleeding)
- Severe peripheral neuropathy (motor weakness)
- Digital gangrene or extensive skin necrosis
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Systemic Lupus Erythematosus
- Rheumatoid Arthritis
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Cryoglobulinaemic Vasculitis
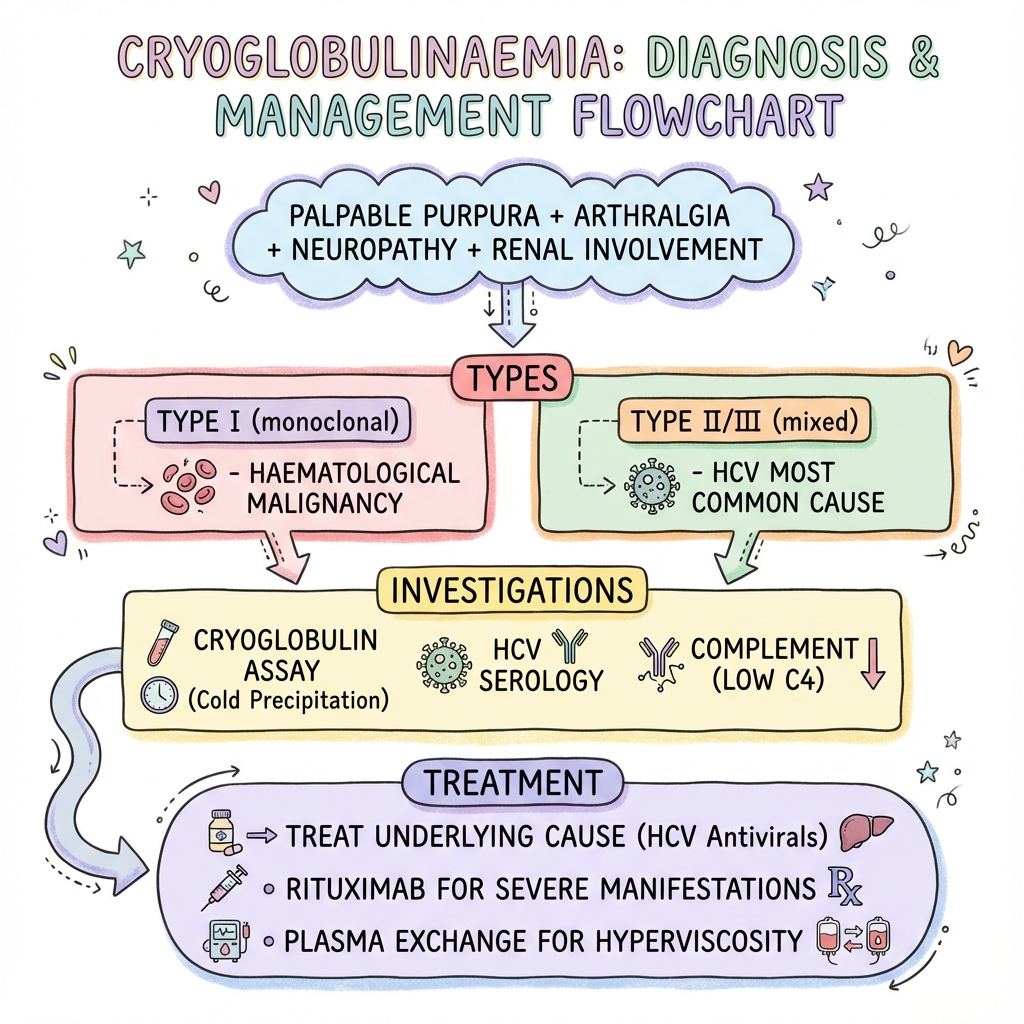
1. Clinical Overview
Cryoglobulinaemic vasculitis is a systemic small-vessel vasculitis mediated by cryoglobulins—immunoglobulins that reversibly precipitate at temperatures below 37°C and redissolve upon rewarming. This condition represents a unique intersection of immunology, rheumatology, and infectious disease, with hepatitis C virus (HCV) infection being the predominant underlying cause in mixed cryoglobulinaemia. The clinical syndrome is characterised by the classic triad of purpura, arthralgias, and weakness, though the full spectrum extends to life-threatening renal and neurological complications. [1,2]
The pathophysiology centres on immune complex deposition in small to medium-sized vessels, leading to complement activation, vascular inflammation, and end-organ damage. The Brouet classification system distinguishes three types based on immunoglobulin composition, each with distinct associations and clinical implications. Type I cryoglobulinaemia is monoclonal and typically associated with haematological malignancies, whilst Types II and III (mixed cryoglobulinaemia) contain both monoclonal or polyclonal immunoglobulins and are strongly linked to chronic infections, particularly HCV. [3,4]
Management has evolved significantly with the advent of direct-acting antiviral (DAA) therapy for HCV, which has transformed outcomes in HCV-associated disease. For severe manifestations, rituximab has emerged as the cornerstone immunosuppressive agent, with plasmapheresis reserved for critical situations including hyperviscosity syndrome and rapidly progressive glomerulonephritis. Understanding the type-specific associations, diagnostic approach including proper specimen handling, and treatment algorithms is essential for postgraduate examinations and clinical practice. [5,6]
2. Epidemiology
Prevalence and Incidence
The true prevalence of cryoglobulinaemia is difficult to establish because many individuals with circulating cryoglobulins remain asymptomatic. Population studies suggest cryoglobulins are detectable in approximately 10-15% of patients with chronic HCV infection, but symptomatic cryoglobulinaemic vasculitis develops in only 5-10% of these individuals. [7,8]
In the general population, the prevalence of symptomatic cryoglobulinaemia is estimated at 1:100,000, though this varies considerably by geographic region, correlating with HCV prevalence. The condition shows a slight female predominance (female:male ratio approximately 2-3:1) and typically presents in the fifth to sixth decade of life. [9]
| Epidemiological Parameter | Value | Reference |
|---|---|---|
| Prevalence in HCV patients | 10-15% (asymptomatic) | [7] |
| Symptomatic vasculitis in HCV | 5-10% of cryoglobulin-positive | [8] |
| General population prevalence | ~1:100,000 | [9] |
| Female:male ratio | 2-3:1 | [9] |
| Peak age at presentation | 50-60 years | [2] |
| Type II in mixed cryoglobulinaemia | 50-60% | [3] |
| Type III in mixed cryoglobulinaemia | 40-50% | [3] |
Underlying Associations
The epidemiology of cryoglobulinaemia is fundamentally shaped by its underlying causes, which vary by Brouet type:
Type I Cryoglobulinaemia (10-15% of cases):
- Waldenström macroglobulinaemia (most common)
- Multiple myeloma
- Monoclonal gammopathy of undetermined significance (MGUS)
- Chronic lymphocytic leukaemia
Type II Cryoglobulinaemia (50-60% of cases):
- Hepatitis C virus infection (90% of cases)
- Hepatitis B virus infection (rare)
- Autoimmune diseases (Sjögren syndrome, systemic lupus erythematosus)
- Chronic bacterial infections
Type III Cryoglobulinaemia (40-50% of cases):
- Hepatitis C virus infection (> 50%)
- Autoimmune/connective tissue diseases
- Chronic infections (bacterial endocarditis, post-streptococcal)
- Essential mixed cryoglobulinaemia (idiopathic cases, now rare with improved HCV testing) [1,3,10]
The strong association between HCV and mixed cryoglobulinaemia (Types II and III) is well-established, with up to 90% of mixed cryoglobulinaemia cases being HCV-related in endemic areas. This association was first recognised in the 1990s and fundamentally changed our understanding of the condition. [11]
Geographic Variation
Geographic distribution mirrors HCV prevalence patterns, with higher rates in Southern Europe (particularly Italy), Egypt, and parts of Asia. The declining incidence of new HCV infections in developed countries, coupled with successful DAA treatment programs, is expected to reduce cryoglobulinaemia prevalence over time. [12]
3. Aetiology and Pathophysiology
The Brouet Classification System
The Brouet classification, established in 1974, remains the cornerstone of cryoglobulin categorisation and has important prognostic and therapeutic implications: [13]
| Type | Immunoglobulin Composition | Mechanism | Primary Associations |
|---|---|---|---|
| I | Monoclonal Ig (IgM or IgG, rarely IgA) | Protein aggregation at low temperature | Haematological malignancies (90%): Waldenström, myeloma, CLL, MGUS |
| II | Monoclonal IgM (with RF activity) + polyclonal IgG | Immune complex formation | HCV (90%), autoimmune diseases, chronic infections |
| III | Polyclonal IgM + polyclonal IgG | Immune complex formation | HCV (50%), autoimmune diseases, infections |
Key Distinguishing Features for Examinations:
Type I:
- Pure monoclonal component
- Typically IgM (75%) or IgG (25%)
- High cryoglobulin concentration (often > 5 mg/mL)
- Risk of hyperviscosity syndrome
- Associated with haematological malignancy in > 90% of cases
- Presents with acrocyanosis, Raynaud phenomenon, digital ischaemia
- May cause vessel occlusion rather than true vasculitis
Type II (Mixed Cryoglobulinaemia):
- Monoclonal IgM component with rheumatoid factor (RF) activity
- Polyclonal IgG component (typically anti-HCV antibodies in HCV cases)
- IgM-RF binds to Fc portion of polyclonal IgG
- Strong association with HCV (90% of cases)
- Classic vasculitic presentation
- Lower cryoglobulin levels than Type I (usually 0.05-1 mg/mL)
Type III (Mixed Cryoglobulinaemia):
- Both components are polyclonal
- Also contains RF activity but polyclonal
- Associated with HCV (50-60%), autoimmune diseases
- Clinical presentation similar to Type II
- May evolve to Type II over time (clonal evolution)
The critical distinction between Type I and Types II/III is the presence of immune complex-mediated vasculitis in mixed cryoglobulinaemia versus hyperviscosity and vessel occlusion in Type I disease. This has profound implications for clinical manifestations and treatment approaches. [3,13]
Molecular Pathophysiology
Hepatitis C and Cryoglobulin Formation
The relationship between HCV and cryoglobulinaemia represents a paradigm of chronic viral infection triggering autoimmune phenomena:
-
Chronic B-cell Stimulation: HCV demonstrates tropism for hepatocytes and B-lymphocytes. The viral envelope protein E2 binds CD81 on B cells, leading to chronic stimulation and clonal expansion. [14]
-
Rheumatoid Factor Production: Chronically stimulated B cells produce IgM with rheumatoid factor activity—antibodies directed against the Fc portion of IgG. In HCV infection, many of the target IgG antibodies are anti-HCV antibodies. [15]
-
Immune Complex Formation: The monoclonal IgM-RF binds to polyclonal IgG (including anti-HCV IgG), forming circulating immune complexes. At temperatures below 37°C (encountered in peripheral circulation), these complexes precipitate as cryoglobulins. [16]
-
Clonal B-cell Expansion: In Type II cryoglobulinaemia, the IgM component is monoclonal, representing an oligoclonal or monoclonal B-cell expansion. This explains why some patients with longstanding HCV-associated cryoglobulinaemia develop frank B-cell lymphoma (transformation risk ~5-10% over 10 years). [17]
Vascular Injury Mechanisms
Once formed, cryoglobulin-containing immune complexes cause vascular damage through several mechanisms:
-
Complement Activation: Immune complexes activate the classical complement pathway, generating C3a and C5a (anaphylatoxins) and forming the membrane attack complex (C5b-9). Characteristic laboratory finding: low C4 with normal or mildly reduced C3. [18]
-
Cryoprecipitation in Vessels: In peripheral tissues where temperature is lower, cryoglobulins precipitate within vessel lumens and walls, causing direct mechanical obstruction and endothelial injury.
-
Leucocyte Recruitment: Complement fragments (C5a) and immune complex deposition recruit neutrophils and monocytes, leading to leucocytoclastic vasculitis—the histological hallmark on skin biopsy.
-
Endothelial Activation: Activated endothelium upregulates adhesion molecules (ICAM-1, VCAM-1, E-selectin), further amplifying inflammation.
-
Vascular Occlusion: In Type I disease, high concentrations of monoclonal immunoglobulin cause hyperviscosity and direct vessel occlusion without significant inflammatory infiltrate. [2,19]
Pathophysiology High-Yield Points for Vivas:
-
Why is C4 characteristically low but C3 relatively preserved? Mixed cryoglobulins activate the classical pathway (through Fc-RF immune complexes), which consumes C1, C4, and C2 preferentially. The alternative pathway (which consumes C3) is less activated. This pattern (low C4, normal C3) is highly suggestive of cryoglobulinaemia and helps differentiate it from other vasculitides.
-
What is the significance of monoclonal IgM with RF activity? The monoclonal IgM component represents clonal B-cell expansion and has intrinsic rheumatoid factor activity (binds IgG Fc). This creates a self-perpetuating system: HCV infection → anti-HCV IgG production → IgM-RF binds anti-HCV IgG → immune complex formation → cryoprecipitation → vasculitis.
-
Why does HCV specifically cause cryoglobulinaemia? HCV has tropism for B lymphocytes (via CD81 receptor), causing chronic B-cell stimulation. Additionally, HCV core protein may directly stimulate RF production. The combination of chronic antigenic stimulation and lymphotropism creates conditions for monoclonal IgM-RF production.
4. Clinical Presentation
The clinical manifestations of cryoglobulinaemia vary by type, cryoglobulin concentration, and underlying disease. The classic presentation involves the triad of purpura, arthralgias, and weakness, but the full spectrum is much broader. [1,2]
The Classic Triad
1. Purpura (70-90% of symptomatic patients):
- Palpable purpura on lower extremities (legs, ankles, feet)
- Occurs in dependent areas and sites of increased hydrostatic pressure
- Non-blanching, often appears in crops
- May be accompanied by pruritus or burning sensation
- Can progress to skin ulceration in severe cases (10-25%)
- Histology: leucocytoclastic vasculitis with IgM and C3 deposition [20]
2. Arthralgias (40-80%):
- Non-erosive, symmetric polyarthralgias
- Commonly affects knees, hands, wrists, ankles
- True arthritis is less common (frank synovitis in less than 10%)
- Does NOT cause joint destruction (unlike rheumatoid arthritis)
- Morning stiffness may be present but typically less prominent than RA
3. Weakness/Fatigue (50-90%):
- Generalised asthenia, often the first symptom
- May be multifactorial: systemic inflammation, underlying HCV, anaemia, renal impairment
- Often underappreciated but significantly impacts quality of life [2,20]
Organ System Involvement
Renal Disease (20-35%)
Renal involvement is a major determinant of prognosis and occurs predominantly in Type II cryoglobulinaemia:
- Membranoproliferative glomerulonephritis (MPGN): The characteristic pattern (Type I MPGN)
- Clinical presentation: nephritic syndrome (haematuria, hypertension, declining GFR) or nephrotic syndrome
- Acute presentations may mimic rapidly progressive glomerulonephritis (RPGN)
- Chronic kidney disease develops in up to 10-15% despite treatment
- Urinalysis: haematuria, proteinuria, red cell casts
- Renal biopsy: MPGN pattern with intraluminal hyaline thrombi (composed of precipitated cryoglobulins), subendothelial deposits, mesangial proliferation
- Immunofluorescence: granular IgM, IgG, and C3 deposits along capillary walls and mesangium [21]
Peripheral Neuropathy (10-60%)
Neurological involvement results from vasa nervorum vasculitis:
- Distal symmetric sensory or sensorimotor polyneuropathy: Most common pattern (70-80% of neuropathy cases)
- Mononeuritis multiplex: Acute/subacute, asymmetric, multiple nerve territories (15-20%)
- Small fibre neuropathy: Burning pain, dysesthesias without motor involvement
- Presentation: numbness, paraesthesias, neuropathic pain, foot drop (with severe motor involvement)
- Nerve conduction studies: axonal pattern (reduced amplitudes)
- Nerve biopsy (rarely performed): necrotising vasculitis of epineural vessels [22]
Gastrointestinal (2-5%)
- Abdominal pain due to mesenteric vasculitis (rare but serious)
- Hepatomegaly (often related to underlying HCV cirrhosis rather than vasculitis)
- Gastrointestinal bleeding (extremely rare)
Pulmonary (less than 5%)
- Alveolar haemorrhage (rare but life-threatening)
- Interstitial lung disease
- Pulmonary hypertension (in chronic cases)
Cardiac (less than 5%)
- Pericarditis
- Myocardial ischaemia (coronary vasculitis, very rare)
- Heart failure (often multifactorial)
Central Nervous System (less than 5%)
- Rare manifestations include stroke, encephalopathy, seizures
- Typically result from vasculitis of CNS vessels
Type-Specific Clinical Patterns
Type I Cryoglobulinaemia—Hyperviscosity Syndrome:
Dominated by hyperviscosity symptoms rather than vasculitis:
- Cutaneous: Retiform purpura, acrocyanosis, cold-induced urticaria, Raynaud phenomenon, digital necrosis (worse in cold weather)
- Neurological: Headache, confusion, visual disturbances, dizziness
- Mucosal bleeding: Epistaxis, gingival bleeding
- Hyperviscosity syndrome triad: Bleeding, neurological symptoms, visual changes
- Fundoscopy: "sausage-like" retinal vein dilation, retinal haemorrhages
- Occurs when serum viscosity > 4 centipoise (normal ~1.8)
- Urgent plasmapheresis indicated [23]
Type II/III (Mixed Cryoglobulinaemia)—Vasculitic Syndrome:
True small-vessel vasculitis predominates:
- Cutaneous: Palpable purpura, urticaria, Raynaud (less severe than Type I), skin ulcers
- Systemic: Classic triad (purpura, arthralgias, weakness)
- Renal: MPGN pattern glomerulonephritis
- Neurological: Peripheral neuropathy (mononeuritis multiplex or distal symmetric)
- Lower cryoglobulin levels than Type I but significant immune complex disease
- Hyperviscosity rare (cryoglobulin concentration usually less than 1 mg/mL) [1,2]
Disease Severity Stratification
Severity assessment is crucial for determining treatment intensity:
Mild Disease:
- Purpura without ulceration
- Arthralgias without arthritis
- Mild neuropathy (sensory only)
- No renal involvement
- No other organ involvement
Moderate Disease:
- Extensive purpura or skin ulcers
- Peripheral neuropathy with motor involvement
- Mild to moderate renal involvement (proteinuria less than 3g/day, creatinine less than 200 μmol/L)
Severe/Life-Threatening Disease:
- Rapidly progressive glomerulonephritis
- Extensive skin necrosis or gangrene
- Severe neuropathy with motor deficit
- Mesenteric vasculitis
- Alveolar haemorrhage
- Hyperviscosity syndrome (Type I)
- CNS involvement [24]
5. Differential Diagnosis
Cryoglobulinaemic vasculitis must be distinguished from other small-vessel vasculitides and systemic diseases that can present with similar features:
Primary Small-Vessel Vasculitides
1. ANCA-Associated Vasculitis (Granulomatosis with Polyangiitis, Microscopic Polyangiitis, Eosinophilic Granulomatosis with Polyangiitis):
- Key distinguishing features:
- Positive ANCA (PR3-ANCA or MPO-ANCA) vs cryoglobulins and low C4
- Upper respiratory tract involvement (sinusitis, epistaxis) common in GPA
- Pulmonary involvement more prominent
- Necrotising glomerulonephritis (crescentic GN) vs MPGN pattern
- Normal complement levels (C3, C4)
- No association with HCV infection [25]
2. IgA Vasculitis (Henoch-Schönlein Purpura):
- Key distinguishing features:
- More common in children
- Purpura predominantly buttocks and lower limbs
- Gastrointestinal involvement with colicky pain very common
- IgA deposition on skin/renal biopsy (not IgM/IgG)
- Normal complement levels
- Self-limiting course in most cases [26]
3. Cutaneous Small-Vessel Vasculitis (Hypersensitivity Vasculitis):
- Key distinguishing features:
- Often drug-induced
- Limited to skin (no systemic features)
- Normal complement, no cryoglobulins
- Resolves with removal of offending agent
Connective Tissue Diseases
4. Systemic Lupus Erythematosus (SLE):
- Key distinguishing features:
- Low C3 AND C4 (classical and alternative pathway activation)
- Positive ANA, anti-dsDNA antibodies
- Malar rash, photosensitivity, oral ulcers
- Serositis (pleuritis, pericarditis) more common
- Multi-organ involvement pattern different
- "Renal biopsy: various lupus nephritis patterns, not MPGN typically [27]"
5. Rheumatoid Arthritis with Vasculitis:
- Key distinguishing features:
- Erosive, symmetric polyarthritis (not just arthralgias)
- Positive anti-CCP antibodies, RF (but RF alone insufficient)
- Rheumatoid nodules
- Joint damage on radiographs
- Vasculitis occurs in long-standing, severe, seropositive RA
- No association with HCV (though HCV can cause arthralgia)
6. Sjögren Syndrome:
- Key distinguishing features:
- Sicca symptoms (dry eyes, dry mouth) predominate
- Can have associated Type II or III cryoglobulinaemia (overlap)
- Positive anti-Ro (SS-A) and anti-La (SS-B) antibodies
- Parotid gland enlargement
- Focus score ≥1 on minor salivary gland biopsy [28]
Infectious and Haematological Conditions
7. Bacterial Endocarditis:
- Key distinguishing features:
- Fever, new cardiac murmur
- Positive blood cultures
- Vegetations on echocardiography
- May have positive RF and low complement (immune complex disease)
- Can cause true cryoglobulinaemia as secondary phenomenon
- Petechial rash, Osler nodes, Janeway lesions
8. Multiple Myeloma/Waldenström Macroglobulinaemia:
- Key distinguishing features:
- These ARE causes of Type I cryoglobulinaemia
- Differentiation is identifying the underlying haematological malignancy
- "Bone marrow examination: plasma cells (myeloma) or lymphoplasmacytic cells (Waldenström)"
- Serum/urine paraprotein
- Skeletal survey abnormalities in myeloma
- Hyperviscosity more prominent
- IgM paraprotein specifically in Waldenström [29]
| Differential | Key Discriminators | Laboratory Clues |
|---|---|---|
| ANCA vasculitis | Upper respiratory/lung involvement, necrotising GN | Positive ANCA, normal C4 |
| SLE | Malar rash, serositis, multi-organ | Low C3 AND C4, +ANA, +anti-dsDNA |
| RA with vasculitis | Erosive arthritis, nodules | +Anti-CCP, joint erosions on X-ray |
| IgA vasculitis | Paediatric, GI pain, buttock purpura | IgA deposits on biopsy |
| Endocarditis | Fever, murmur, vegetations | Positive blood cultures |
| Type I cryoglob from haematological malignancy | Hyperviscosity, monoclonal protein | Paraprotein, BM infiltration |
Examination Pearl—Complement Patterns:
Understanding complement consumption patterns is crucial for differentiating vasculitides:
- Cryoglobulinaemia (Type II/III): Low C4, normal/mildly low C3 (classical pathway)
- SLE: Low C3 AND C4 (both pathways)
- ANCA vasculitis: Normal C3, normal C4 (not immune complex-mediated)
- Post-infectious GN: Low C3, normal C4 (alternative pathway)
- Hereditary angioedema: Low C4, normal C3 (C1 inhibitor deficiency)
The isolated low C4 with preserved C3 is highly characteristic of cryoglobulinaemic vasculitis and should trigger testing for cryoglobulins. [18]
6. Investigations
The diagnosis of cryoglobulinaemic vasculitis requires: (1) Detection of cryoglobulins, (2) Demonstration of vasculitis (clinical and/or histological), and (3) Identification of underlying cause.
Cryoglobulin Detection (Gold Standard Test)
Detection of cryoglobulins requires meticulous technique; improper specimen handling is the most common cause of false-negative results.
Proper Specimen Collection and Handling:
- Pre-warmed equipment: Collection tubes, syringes, and transport containers must be pre-warmed to 37°C
- Warm phlebotomy: Maintain arm warmth during collection (use warm compress if necessary)
- Immediate processing: Transport specimen to laboratory at 37°C within 1 hour
- Clotting at 37°C: Allow blood to clot at 37°C for 60-90 minutes
- Centrifugation at 37°C: Centrifuge at 37°C to separate serum
- Cold incubation: Incubate serum at 4°C for up to 7 days
- Cryocrit measurement: Measure precipitate volume (cryocrit) after centrifugation
- Re-warming confirmation: Confirm precipitate dissolves upon re-warming to 37°C (critical for diagnosis)
- Immunofixation: Characterise immunoglobulin type(s) in the cryoprecipitate [30]
Common Reasons for False-Negative Results:
- Specimen collected or transported cold (most common error)
- Insufficient cold incubation time (less than 3-7 days)
- Low cryoglobulin concentration (especially Type II/III)
- Specimen clotted at room temperature or cold
Cryoglobulin Quantification and Typing:
- Cryocrit: Percentage of serum volume that precipitates
- "Type I: Usually > 5% (high concentration, 5-30 mg/mL)"
- "Type II/III: Typically less than 1-5% (low concentration, 0.05-1 mg/mL)"
- Immunofixation/Immunotyping: Determines Brouet classification
- Identifies monoclonal vs polyclonal components
- Identifies immunoglobulin classes (IgM, IgG, IgA) [3,30]
Complement Levels
C4 (Low in 80-90% of mixed cryoglobulinaemia):
- Classical pathway consumption
- May be profoundly depressed
- Degree of depression correlates with disease activity
C3 (Normal or mildly low in 60-70%):
- Alternative pathway less affected
- Low C3 suggests severe disease or SLE overlap
CH50 (Total Haemolytic Complement):
- Reduced, reflecting classical pathway consumption
- Less commonly measured in routine practice [18]
Rheumatoid Factor (RF)
- Positive in 70-90% of Type II/III cryoglobulinaemia
- Often high titre (may be > 1:640)
- Reflects intrinsic RF activity of monoclonal IgM component
- Non-specific finding (also positive in RA, Sjögren, endocarditis)
- Useful as disease activity marker (correlates with cryocrit) [31]
Hepatitis C Serology and Virology
Given the strong HCV association, all patients with cryoglobulinaemia should be tested:
- Anti-HCV antibodies (ELISA): Screening test
- HCV RNA (PCR): Confirmatory test; quantifies viral load
- HCV genotype: Important for selecting DAA therapy
- HCV antibodies may be found IN the cryoprecipitate, occasionally causing false-negative serum anti-HCV testing (antibodies precipitated out). Test cryoprecipitate if clinical suspicion high. [32]
Tissue Biopsy
Skin Biopsy:
- Indication: Purpuric lesions
- Findings:
- Leucocytoclastic vasculitis (neutrophilic infiltration, fibrinoid necrosis, nuclear debris)
- Small vessel involvement (post-capillary venules)
- "Immunofluorescence: IgM, IgG, and C3 deposition in vessel walls"
- May see intravascular cryoglobulin deposits (eosinophilic hyaline material)
- Diagnostic yield: ~70-80% in active lesions [33]
Renal Biopsy:
- Indication: Proteinuria, haematuria, rising creatinine
- Findings:
- Membranoproliferative glomerulonephritis (MPGN Type I) pattern
- Mesangial proliferation and hypercellularity
- Intraluminal hyaline thrombi (pathognomonic; composed of precipitated cryoglobulins in glomerular capillaries)
- Subendothelial deposits ("tram-track" appearance on silver stain)
- Double-contour basement membrane (GBM duplication)
- "Immunofluorescence: IgM, IgG, C3 in granular pattern along capillary walls and mesangium"
- "Electron microscopy: organized subendothelial deposits with tubular/microtubular structure"
- Prognostic value: Degree of interstitial fibrosis and tubular atrophy predicts renal outcomes [21,34]
Nerve Biopsy (Rarely Performed):
- Indication: Atypical neuropathy, diagnostic uncertainty
- Findings: Necrotising vasculitis of epineural vessels, axonal degeneration
Laboratory Tests for Disease Activity and Complications
| Test | Purpose | Expected Findings |
|---|---|---|
| Full Blood Count | Anaemia, leucocytosis | Normocytic anaemia common; leucopenia if HCV cirrhosis |
| ESR/CRP | Inflammation markers | Usually elevated; ESR may be very high in Type I (hyperviscosity) |
| Renal Function (Creatinine, eGFR) | Assess renal involvement | Elevated creatinine in MPGN |
| Urinalysis | Screen for renal disease | Haematuria, proteinuria, red cell casts |
| 24-hour Urine Protein or PCR | Quantify proteinuria | Variable; nephrotic range possible |
| Liver Function Tests | Assess HCV-related liver disease | Transaminitis, hypoalbuminaemia if cirrhosis |
| Serum Protein Electrophoresis (SPEP) | Detect monoclonal protein | Monoclonal band in Type I; may be normal in Type II/III |
| Immunoglobulins (IgG, IgA, IgM) | Quantify immunoglobulin levels | Polyclonal elevation in Type III; IgM elevation in Type I/II |
| ANA, anti-dsDNA, ENA | Exclude SLE/CTD | Negative (if positive, consider overlap) |
| ANCA (PR3, MPO) | Exclude ANCA vasculitis | Negative |
| Hepatitis B and HIV serology | Screen for other infections | Test all patients |
Nerve Conduction Studies
- Indication: Peripheral neuropathy
- Findings: Axonal sensorimotor polyneuropathy (reduced amplitudes, normal/mildly slowed conduction velocities) or mononeuritis multiplex pattern (asymmetric, multifocal involvement)
Critical Diagnostic Pitfalls:
1. False-Negative Cryoglobulin Testing:
- Most commonly due to cold specimen handling
- If clinical suspicion high (classic triad + low C4 + HCV+), repeat testing with strict 37°C protocol
- Consider testing cryoprecipitate for HCV antibodies (may be sequestered)
2. Detecting Low-Level Cryoglobulins:
- Type II/III mixed cryoglobulinaemia may have very low cryoglobulin concentrations
- Require full 7-day cold incubation
- Clinical vasculitis can occur with minimal or undetectable cryoglobulins in some cases ("cryoglobulinaemic vasculitis sine cryoglobulinaemia"—rare)
3. Cryoglobulins vs Cryofibrinogen:
- Cryofibrinogen precipitates from plasma (not serum)
- Causes livedo, digital ischaemia but not true vasculitis
- Requires plasma sample if suspected
4. Interference with Laboratory Assays:
- Cryoglobulins may interfere with serum chemistry, causing spurious results
- Particularly affects protein measurements
- Re-run assays at 37°C if cryoglobulins suspected [30]
7. Classification and Staging
Brouet Classification (Detailed)
As discussed, the Brouet system remains the standard. The key clinical and prognostic implications are:
- Type I: Hyperviscosity predominates; treat underlying haematological malignancy
- Type II/III: True vasculitis; treat underlying HCV (if present) + immunosuppression for severe disease
Disease Activity Assessment
No validated disease activity score exists specifically for cryoglobulinaemia, but the following are used to assess disease activity:
Clinical Parameters:
- New or worsening purpura
- Extension of skin involvement
- New neuropathy or motor progression
- Rising creatinine or worsening proteinuria
- Constitutional symptoms (fever, weight loss)
Laboratory Parameters:
- Cryocrit level (higher levels correlate with more active disease, especially Type I)
- C4 level (lower = more active disease)
- ESR/CRP
- RF titre (may parallel disease activity) [35]
8. Management
Management of cryoglobulinaemic vasculitis is determined by: (1) Brouet type, (2) Disease severity, (3) Underlying aetiology (particularly HCV status), and (4) Organ involvement. The therapeutic landscape has been revolutionised by direct-acting antivirals (DAAs) for HCV-associated disease. [5,6,36]
General Principles
- Treat underlying cause: HCV eradication with DAAs is the definitive treatment for HCV-associated mixed cryoglobulinaemia
- Severity-based approach: Mild disease may not require immunosuppression; severe disease requires aggressive therapy
- Avoid cold exposure: Counsel patients to avoid cold (triggers cryoprecipitation)
- Multidisciplinary care: Rheumatology, nephrology, haematology, hepatology involvement
Management Algorithm by Severity
Mild Disease (Purpura, Arthralgias, Mild Neuropathy, No Organ Threat)
HCV-Positive Patients:
-
Direct-Acting Antivirals (DAAs): First-line treatment
- Regimen selection based on HCV genotype, cirrhosis status, treatment history
- Examples: Sofosbuvir/velpatasvir, glecaprevir/pibrentasvir, sofosbuvir/ledipasvir
- Duration: 8-12 weeks typically
- Sustained virological response (SVR) rates: > 95%
- Cryoglobulin clearance: Occurs in 60-80% of patients achieving SVR
- Clinical improvement: Often seen within 3-6 months of viral clearance [37,38]
-
Supportive Care:
- NSAIDs or low-dose corticosteroids for arthralgias (prednisone 5-10 mg daily)
- Avoid cold exposure
- Compression stockings for dependent oedema
-
Monitor: Clinical response, cryocrit, C4, HCV RNA
HCV-Negative Patients:
- Investigate for other causes (autoimmune disease, infections, haematological malignancy)
- Symptomatic treatment as above
- Consider low-dose rituximab if symptoms refractory
Moderate Disease (Extensive Purpura, Skin Ulcers, Progressive Neuropathy, Mild-Moderate Renal Involvement)
Combination Therapy:
-
DAAs (if HCV-positive): As above, initiated concurrently or sequentially with immunosuppression
-
Rituximab: First-line immunosuppressive agent
- Mechanism: B-cell depletion (targets IgM-producing B cells)
- Dosing regimens:
- Standard: 375 mg/m² IV weekly x 4 doses
- Low-dose: 250 mg/m² IV weekly x 4 doses OR single 1g IV dose (may be sufficient and better tolerated)
- Response rate: 70-90% clinical improvement
- Time to response: 1-6 months
- Duration of response: Variable (6-24 months); may require retreatment
- Monitoring: CD19+ B-cell counts, immunoglobulin levels, infections [39,40]
-
Corticosteroids:
- Prednisone 0.5-1 mg/kg/day initially
- Taper over 2-6 months based on response
- Use cautiously in HCV+ patients (may increase viral replication, though DAAs mitigate this concern)
Alternative Immunosuppressive Agents (if rituximab contraindicated or refractory):
- Cyclophosphamide: For severe, refractory disease (0.5-1 g/m² IV monthly x 6 months)
- Azathioprine: Steroid-sparing (1-2 mg/kg/day)
- Mycophenolate mofetil: 1-2 g daily in divided doses (less evidence than rituximab)
Severe/Life-Threatening Disease (Rapidly Progressive GN, Severe Neuropathy, Hyperviscosity, Alveolar Haemorrhage, Mesenteric Vasculitis)
Urgent/Emergent Therapy:
-
Plasmapheresis (Plasma Exchange, PLEX):
- Indication: Life-threatening disease, hyperviscosity syndrome, RPGN
- Mechanism: Rapid removal of circulating cryoglobulins, immune complexes, IgM
- Protocol:
- 3-5 litres plasma exchange per session (1-1.5 plasma volumes)
- Daily or alternate-day exchanges
- Total: 5-10 sessions typically
- Replace with albumin ± fresh frozen plasma (FFP)
- Provides rapid (within days) but temporary benefit
- Must be combined with immunosuppression to prevent rebound [41]
-
High-Dose Corticosteroids:
- Methylprednisolone 500-1000 mg IV daily x 3 days (pulse therapy)
- Followed by oral prednisone 1 mg/kg/day, taper over weeks to months
-
Rituximab:
- Standard dose: 375 mg/m² IV weekly x 4
- Initiated concurrently with plasmapheresis and corticosteroids
-
Cyclophosphamide:
- Consider in refractory cases or when rituximab unavailable
- IV pulse: 0.5-1 g/m² monthly x 6 months
- Oral: 2 mg/kg/day (less commonly used due to toxicity)
-
DAAs (if HCV-positive):
- Initiate once acute crisis controlled
- May delay in setting of severe acute illness or if drug interactions with immunosuppression
Renal Replacement Therapy:
- Haemodialysis for acute kidney injury/RPGN
- Some patients progress to end-stage renal disease requiring long-term RRT
Type I Cryoglobulinaemia (Haematological Malignancy-Associated)
Treat Underlying Malignancy:
-
Waldenström Macroglobulinaemia:
- Rituximab-based regimens (e.g., DRC: dexamethasone, rituximab, cyclophosphamide)
- Ibrutinib (Bruton's tyrosine kinase inhibitor)
- Bortezomib-based regimens
-
Multiple Myeloma:
- Standard myeloma therapy (e.g., bortezomib/lenalidomide/dexamethasone)
-
Plasmapheresis:
- For acute hyperviscosity syndrome
- Provides rapid symptomatic relief pending chemotherapy effect
Special Situations
Pregnancy:
- Rare scenario (disease predominantly in older adults)
- DAAs contraindicated in pregnancy
- Consider low-dose corticosteroids, monitor closely
- Plasmapheresis if severe disease
End-Stage Renal Disease:
- Cryoglobulinaemia may improve paradoxically with dialysis (removal of HCV-infected B cells)
- Kidney transplantation: Treat HCV with DAAs pre-transplant; improved outcomes post-SVR
Relapse/Refractory Disease:
- Re-treat with rituximab (may require maintenance dosing)
- Consider alternative agents: cyclophosphamide, bortezomib (in Type I), tocilizumab (case reports)
- Evaluate for HCV reinfection or treatment failure
- Rule out lymphoma transformation in Type II disease [42]
Monitoring Treatment Response
| Parameter | Baseline | Follow-Up Frequency | Target |
|---|---|---|---|
| Clinical symptoms | Document severity | Every visit | Resolution of purpura, neuropathy stabilisation |
| Cryocrit | Quantify | Every 3-6 months | Reduction/clearance |
| C4 | Low typically | Every 3 months | Normalisation |
| RF | High usually | Every 3-6 months | Decline correlates with improvement |
| HCV RNA (if HCV+) | Quantify viral load | Week 4, week 12, SVR12 | Undetectable |
| Renal function | Creatinine, eGFR | Monthly initially | Stable/improving creatinine |
| Urinalysis/PCR | Haematuria, proteinuria | Monthly initially | Reduction in haematuria/proteinuria |
| Nerve conduction (if neuropathy) | Baseline | Every 6-12 months | Stabilisation (improvement rare) |
Viva High-Yield Treatment Points:
"Why is rituximab the first-line immunosuppressive agent?"
- Targets the pathogenic mechanism: B-cell depletion reduces IgM-RF production
- Superior efficacy compared to cyclophosphamide in mixed cryoglobulinaemia (clinical trials)
- Better safety profile than cyclophosphamide
- Specifically depletes CD20+ B cells (the source of monoclonal IgM)
"When do you use plasmapheresis?"
- Life-threatening manifestations: RPGN, hyperviscosity syndrome, severe CNS/mesenteric vasculitis
- Provides rapid (days) but temporary benefit
- MUST combine with immunosuppression (otherwise cryoglobulin rebounds)
- Typical protocol: 5-10 sessions over 2-3 weeks
"Can you treat HCV in patients with active cryoglobulinaemic vasculitis?"
- Mild disease: DAAs alone are first-line
- Severe disease: Control acute vasculitis first with immunosuppression ± plasmapheresis, THEN initiate DAAs
- Concern: HCV eradication takes weeks; acute disease needs immediate control
- DAAs do NOT worsen vasculitis (older interferon-based therapy did worsen cryoglobulinaemia)
"What is the role of corticosteroids?"
- Bridge therapy while awaiting effect of rituximab or DAAs
- Essential in severe acute disease (pulse methylprednisolone)
- Avoid high-dose long-term steroids if possible (infection risk, especially in HCV/cirrhosis)
- Taper as other therapies take effect
9. Complications
Complications arise from untreated/refractory vasculitis, treatment toxicity, and underlying disease progression.
Disease-Related Complications
Chronic Kidney Disease and End-Stage Renal Disease (10-15%):
- Progressive scarring from MPGN
- Risk factors: Delayed diagnosis, severe glomerulonephritis, poor response to treatment
- May require long-term dialysis or transplantation
- Outcomes: Improved with HCV eradication pre-transplant [43]
Irreversible Peripheral Neuropathy:
- Axonal damage may be permanent despite treatment
- Chronic neuropathic pain, disability
- Rehabilitation and pain management important
Digital Ischaemia and Gangrene (Type I predominantly):
- Vessel occlusion from cryoprecipitation
- May require amputation in severe cases
- Prevention: Avoid cold exposure, smoking cessation
Lymphoma Transformation (5-10% over 10 years in Type II):
- Risk particularly in longstanding HCV-associated cryoglobulinaemia
- Typically B-cell non-Hodgkin lymphoma (marginal zone, diffuse large B-cell)
- Monitor for: Lymphadenopathy, B symptoms, rising paraprotein level
- Treat as per lymphoma protocols [17]
Hyperviscosity Syndrome (Type I):
- Medical emergency
- Sequelae: Stroke, retinal vein occlusion, blindness, mucosal bleeding
- Requires urgent plasmapheresis
Treatment-Related Complications
Rituximab-Associated:
- Infusion reactions (fever, chills, bronchospasm)—premedicate with antihistamines, corticosteroids
- Infections (bacterial, viral, fungal)—particularly in patients on concomitant steroids
- Progressive multifocal leukoencephalopathy (PML)—rare but serious (JC virus reactivation)
- Prolonged hypogammaglobulinaemia—may require IVIg replacement
- Hepatitis B reactivation—screen all patients for HBsAg, anti-HBc; prophylactic antivirals if positive [44]
Cyclophosphamide-Associated:
- Bone marrow suppression (leucopenia, thrombocytopenia)
- Haemorrhagic cystitis (prevent with mesna, hydration)
- Gonadal toxicity and infertility
- Bladder cancer (long-term risk)
- Infections
Plasmapheresis-Associated:
- Central line complications (infection, thrombosis, pneumothorax)
- Hypocalcaemia (citrate anticoagulation)
- Hypotension, allergic reactions to albumin/FFP
- Bleeding risk (removal of coagulation factors)
Corticosteroid-Associated:
- Hyperglycaemia, diabetes
- Osteoporosis and fractures (provide calcium/vitamin D, bisphosphonates)
- Avascular necrosis (hip, knee)
- Infections (bacterial, opportunistic)
- Cushingoid features, weight gain
- Psychiatric disturbances
HCV-Related Complications
Cirrhosis and Hepatocellular Carcinoma:
- Longstanding HCV leads to cirrhosis in 15-20%
- HCC risk in cirrhotic patients: 3-5% per year
- DAA-induced SVR reduces but does not eliminate HCC risk
- Surveillance: 6-monthly ultrasound and AFP in cirrhotics [45]
10. Prognosis
Prognosis in cryoglobulinaemic vasculitis is determined by disease type, severity of organ involvement, underlying aetiology, and response to treatment.
Overall Survival
HCV-Associated Mixed Cryoglobulinaemia:
- 10-year survival: 50-70% (historically, pre-DAA era)
- Improved significantly with DAA therapy and rituximab availability
- Major causes of death: Renal failure, infections, liver disease (cirrhosis/HCC), cardiovascular events, lymphoma [46]
Type I Cryoglobulinaemia:
- Prognosis determined by underlying haematological malignancy
- Hyperviscosity syndrome responds well to plasmapheresis and treatment of malignancy
Prognostic Factors
Poor Prognostic Indicators:
- Renal involvement at presentation (especially RPGN)
- Older age (> 65 years)
- Male sex
- Cryocrit > 10%
- Severe hepatic fibrosis/cirrhosis
- Central nervous system or gastrointestinal involvement
- Failure to achieve HCV SVR (if HCV-positive)
- Transformation to lymphoma [47]
Favourable Prognostic Indicators:
- Isolated cutaneous disease
- Early diagnosis and treatment
- Achievement of HCV SVR with DAAs
- Response to rituximab
- Absence of renal involvement
Disease Outcomes
Sustained Virological Response (SVR) in HCV Patients:
- Cryoglobulin clearance: 60-80% of patients
- Clinical remission: 50-70%
- Renal function improvement: Variable; less improvement if advanced fibrosis on biopsy
- Neuropathy: Stabilisation common, improvement less so (axonal damage often irreversible)
- Reduces but does not eliminate lymphoma risk [37,38]
Renal Outcomes:
- 10-15% progress to ESRD despite treatment
- Predictors of progression: Degree of interstitial fibrosis/tubular atrophy on biopsy, delayed treatment, lack of HCV eradication
Relapse:
- Relapse rates: 20-40% after rituximab
- May require repeat rituximab courses or maintenance therapy
- HCV reinfection (in people who inject drugs) can trigger cryoglobulinaemia recurrence
11. Prevention and Screening
Primary Prevention
Hepatitis C Prevention:
- Safe injection practices (harm reduction programs, needle exchange)
- Screening of blood products (universal in developed countries)
- Healthcare worker precautions (sharps safety)
- Treatment of HCV-infected individuals reduces transmission
Vaccination:
- No vaccine for HCV currently available
- Hepatitis B vaccination in at-risk populations (prevents HBV-associated cryoglobulinaemia)
Secondary Prevention (Preventing Progression)
In Patients with Asymptomatic Cryoglobulinaemia:
- Regular monitoring (clinical assessment, urinalysis, renal function) every 6-12 months
- Treat HCV with DAAs to prevent progression to symptomatic disease
- Avoid cold exposure
- Avoid nephrotoxic medications
In Patients with Treated Cryoglobulinaemia:
- Long-term follow-up to detect relapse
- Monitor for lymphoma transformation (especially Type II)
- Continued HCV surveillance (SVR does not preclude reinfection in at-risk populations)
Screening Recommendations
Who to Screen for Cryoglobulins:
- All patients with HCV infection and systemic symptoms (purpura, arthralgias, neuropathy)
- Patients with unexplained MPGN pattern on renal biopsy
- Patients with low C4 and unexplained vasculitic symptoms
- Patients with monoclonal gammopathy and vasculitic features
Who to Screen for HCV:
- All patients with newly diagnosed cryoglobulinaemia (essential)
- High-risk populations (people who inject drugs, recipients of blood products pre-1990s, HIV-positive individuals)
12. Key Guidelines and Consensus Statements
European League Against Rheumatism (EULAR) Recommendations (2018):
- Recommend DAAs as first-line therapy for HCV-associated cryoglobulinaemic vasculitis
- Rituximab as first-line immunosuppressive therapy for severe disease
- Plasmapheresis for life-threatening manifestations [48]
Kidney Disease: Improving Global Outcomes (KDIGO) 2012:
- Immunosuppression and treatment of underlying HCV for cryoglobulinaemic glomerulonephritis
- Biopsy recommended in significant renal involvement to guide therapy [49]
American College of Rheumatology (ACR) Guidance:
- Emphasise importance of HCV screening and treatment
- Support use of rituximab over cyclophosphamide for mixed cryoglobulinaemia
Examination Focus
Common Exam Questions
-
"What are the causes of cryoglobulinaemia and how do you classify it?"
- Brouet classification (Types I, II, III)
- Type I: Monoclonal Ig (haematological malignancy—Waldenström, myeloma, MGUS)
- Type II: Monoclonal IgM + polyclonal IgG (HCV in 90%, also autoimmune diseases)
- Type III: Polyclonal IgM + IgG (HCV > 50%, autoimmune, infections)
-
"A patient presents with palpable purpura, arthralgias, and peripheral neuropathy. Complement C4 is low but C3 is normal. What is your differential and how would you investigate?"
- Differential: Cryoglobulinaemic vasculitis (most likely given complement pattern), SLE, other vasculitides
- Investigations:
- Cryoglobulin assay (proper warm technique, 37°C transport)
- HCV serology and RNA
- RF, immunoglobulins
- ANCA, ANA, anti-dsDNA
- Urinalysis and renal function
- Skin biopsy (leucocytoclastic vasculitis with IgM/IgG/C3 deposition)
-
"How would you manage HCV-associated cryoglobulinaemic vasculitis with renal involvement?"
- Severity assessment (mild-moderate vs severe/RPGN)
- Mild-moderate: DAAs ± rituximab, low-dose corticosteroids
- Severe (RPGN): Plasmapheresis + pulse methylprednisolone + rituximab, then DAAs once stabilised
- Monitor: HCV RNA, cryocrit, C4, renal function, proteinuria
-
"Why is C4 low but C3 normal/mildly low in cryoglobulinaemia?"
- Immune complexes (IgM-RF + IgG) activate classical complement pathway
- Classical pathway consumes C1, C4, C2 preferentially
- Alternative pathway (which consumes C3) less activated
- Contrast with SLE (both pathways activated, low C3 AND C4)
-
"What is the most important aspect of cryoglobulin specimen collection?"
- Maintain specimen at 37°C from collection through centrifugation
- Pre-warm collection equipment, transport at 37°C, clot at 37°C
- Cold exposure causes premature precipitation and false-negative results
Viva Points
Opening Statement: "Cryoglobulinaemic vasculitis is a small-vessel vasculitis caused by circulating cryoglobulins—immunoglobulins that precipitate in cold temperatures and redissolve on warming. It is classified by the Brouet system into three types based on immunoglobulin composition. Type I is monoclonal and associated with haematological malignancies, whilst Types II and III (mixed cryoglobulinaemia) contain monoclonal or polyclonal components and are predominantly associated with hepatitis C virus infection, present in up to 90% of cases."
Key Facts to Mention:
- Classic triad: Palpable purpura, arthralgias, weakness
- Renal involvement: Membranoproliferative glomerulonephritis (MPGN) in 20-35%
- Peripheral neuropathy: Distal symmetric or mononeuritis multiplex in 10-60%
- Diagnostic hallmark: Low C4 with normal/mildly low C3 (classical pathway activation)
- Gold standard investigation: Cryoglobulin assay with strict 37°C specimen handling
- HCV serology essential in all cases
- First-line treatment (HCV-associated): Direct-acting antivirals (DAAs)
- Severe disease: Plasmapheresis + rituximab + corticosteroids
- Rituximab is first-line immunosuppressive agent (B-cell depletion)
Evidence to Quote:
- "HCV is associated with 90% of Type II cryoglobulinaemia cases (Cacoub et al., 2015)"
- "DAAs achieve sustained virological response in > 95% and cryoglobulin clearance in 60-80% (Bonacci et al., 2016)"
- "Rituximab achieves clinical response in 70-90% of patients (De Vita et al., 2012)"
Common Mistakes to Avoid
❌ Missing the HCV association:
- ALWAYS test for HCV in cryoglobulinaemia—it's the most common cause
- Failure to recognise this misses a treatable aetiology
❌ Improper cryoglobulin specimen collection:
- Cold transport/handling is the most common cause of false-negatives
- Remember: "Keep it warm" until the serum is separated, then refrigerate
❌ Confusing Type I with Type II/III disease:
- Type I: Hyperviscosity, less vasculitis, haematological malignancy
- Type II/III: True vasculitis, HCV association, lower cryoglobulin levels
❌ Starting immunosuppression without considering HCV treatment:
- Mild-moderate HCV+ disease: DAAs may suffice without immunosuppression
- Treating HCV addresses the root cause
❌ Misinterpreting complement patterns:
- Cryoglobulinaemia: Low C4, normal C3
- SLE: Low C4 AND C3
- ANCA vasculitis: Normal C4 and C3
❌ Forgetting lymphoma risk in Type II disease:
- 5-10% transform to B-cell lymphoma over 10 years
- Monitor for lymphadenopathy, B symptoms, rising paraprotein
Model Viva Answers
Q: "Describe your approach to a patient presenting with purpura, arthralgias, and low C4."
A: "This presentation is highly suggestive of cryoglobulinaemic vasculitis. I would approach this systematically:
History: I would enquire about constitutional symptoms, duration of symptoms, cold exposure exacerbating symptoms, risk factors for hepatitis C (injection drug use, blood transfusions pre-1990s), symptoms of peripheral neuropathy (numbness, weakness), and systemic features suggesting other vasculitides or connective tissue disease.
Examination: I would look for palpable purpura (typically lower limbs), evidence of peripheral neuropathy (glove-and-stocking sensory loss, motor weakness, absent reflexes), signs of chronic liver disease if HCV-related, lymphadenopathy, and features of other systemic diseases.
Investigations: The key investigation is a cryoglobulin assay with meticulous technique—specimen must be collected, transported, and processed at 37°C until serum separation, then refrigerated for up to 7 days to detect cryoprecipitate. I would also check:
- HCV serology and RNA (given strong association)
- Full autoimmune screen (RF, ANA, ANCA, immunoglobulins)
- Complement (C3, C4—expect low C4, normal C3 in cryoglobulinaemia)
- Renal function, urinalysis (screen for MPGN)
- Serum protein electrophoresis (detect monoclonal protein)
- Skin biopsy if diagnosis uncertain (leucocytoclastic vasculitis with IgM/IgG deposition)
Management: If cryoglobulinaemia confirmed and HCV-positive, treatment depends on severity. For mild disease (purpura, arthralgias), I would initiate direct-acting antivirals as first-line therapy. For severe disease with renal involvement or progressive neuropathy, I would add rituximab and corticosteroids. Life-threatening manifestations (rapidly progressive GN, hyperviscosity) require urgent plasmapheresis, pulse steroids, and rituximab, followed by DAAs once stabilised. Long-term monitoring includes HCV viral load, cryocrit, complement, and renal function."
References
-
Cacoub P, Comarmond C, Domont F, Savey L, Desbois AC, Saadoun D. Cryoglobulinemia vasculitis. Am J Med. 2015;128(9):950-955. doi:10.1016/j.amjmed.2015.02.017
-
Ramos-Casals M, Stone JH, Cid MC, Bosch X. The cryoglobulinaemias. Lancet. 2012;379(9813):348-360. doi:10.1016/S0140-6736(11)60242-0
-
Brouet JC, Clauvel JP, Danon F, Klein M, Seligmann M. Biologic and clinical significance of cryoglobulins. A report of 86 cases. Am J Med. 1974;57(5):775-788. doi:10.1016/0002-9343(74)90852-3
-
Ferri C, Sebastiani M, Giuggioli D, et al. Mixed cryoglobulinemia: demographic, clinical, and serologic features and survival in 231 patients. Semin Arthritis Rheum. 2004;33(6):355-374. doi:10.1016/j.semarthrit.2003.10.001
-
Saadoun D, Pol S, Ferfar Y, et al. Efficacy and safety of sofosbuvir plus daclatasvir for treatment of HCV-associated cryoglobulinemia vasculitis. Gastroenterology. 2017;153(1):49-52.e5. doi:10.1053/j.gastro.2017.03.006
-
Bonacci M, Lens S, Mariño Z, Forns X. Cryoglobulinemia related to hepatitis C virus infection. Gastroenterol Hepatol. 2016;39(5):333-342. doi:10.1016/j.gastrohep.2015.09.008
-
Lunel F, Musset L, Cacoub P, et al. Cryoglobulinemia in chronic liver diseases: role of hepatitis C virus and liver damage. Gastroenterology. 1994;106(5):1291-1300. doi:10.1016/0016-5085(94)90022-1
-
Terrier B, Carrat F, Krastinova E, et al. Evolving epidemiology and treatment of mixed cryoglobulinemia vasculitis: data over 12 years from the French Nationwide CryoVas Survey. Medicine (Baltimore). 2017;96(6):e6025. doi:10.1097/MD.0000000000006025
-
Trejo O, Ramos-Casals M, García-Carrasco M, et al. Cryoglobulinemia: study of etiologic factors and clinical and immunologic features in 443 patients from a single center. Medicine (Baltimore). 2001;80(4):252-262. doi:10.1097/00005792-200107000-00004
-
Agnello V, Chung RT, Kaplan LM. A role for hepatitis C virus infection in type II cryoglobulinemia. N Engl J Med. 1992;327(21):1490-1495. doi:10.1056/NEJM199211193272104
-
Ferri C, Greco F, Longombardo G, et al. Hepatitis C virus antibodies in mixed cryoglobulinaemia. Clin Exp Rheumatol. 1991;9(1):95-96.
-
Kayali Z, Schmidt WN. Finally, a successful treatment for hepatitis C virus-associated mixed cryoglobulinemia. Gastroenterology. 2017;153(1):22-25. doi:10.1053/j.gastro.2017.05.008
-
Brouet JC. The cryoglobulins. Nouv Presse Med. 1978;7(26):2251-2254.
-
Pileri P, Uematsu Y, Campagnoli S, et al. Binding of hepatitis C virus to CD81. Science. 1998;282(5390):938-941. doi:10.1126/science.282.5390.938
-
Charles ED, Dustin LB. Hepatitis C virus-induced cryoglobulinemia. Kidney Int. 2009;76(8):818-824. doi:10.1038/ki.2009.247
-
Sansonno D, De Re V, Lauletta G, Tucci FA, Boiocchi M, Dammacco F. Monoclonal antibody treatment of mixed cryoglobulinemia resistant to interferon alpha with an anti-CD20. Blood. 2003;101(10):3818-3826. doi:10.1182/blood-2002-10-3162
-
Saadoun D, Sellam J, Ghillani-Dalbin P, et al. Increased risks of lymphoma and death among patients with non-hepatitis C virus-related mixed cryoglobulinemia. Arch Intern Med. 2006;166(19):2101-2108. doi:10.1001/archinte.166.19.2101
-
Quartuccio L, Isola M, Corazza L, et al. Validation of the classification criteria for cryoglobulinaemic vasculitis. Rheumatology (Oxford). 2014;53(12):2209-2213. doi:10.1093/rheumatology/keu271
-
Della Rossa A, Marchi F, Catarsi E, Tavoni A, Bombardieri S. Mixed cryoglobulinemia and mortality: a review of the literature. Clin Exp Rheumatol. 2008;26(3 Suppl 49):S105-S108.
-
Ferri C, Sebastiani M, Giuggioli D, et al. Hepatitis C virus syndrome: A constellation of organ- and non-organ specific autoimmune disorders, B-cell non-Hodgkin's lymphoma, and cancer. World J Hepatol. 2015;7(3):327-343. doi:10.4254/wjh.v7.i3.327
-
Tarantino A, Campise M, Banfi G, et al. Long-term predictors of survival in essential mixed cryoglobulinemic glomerulonephritis. Kidney Int. 1995;47(2):618-623. doi:10.1038/ki.1995.78
-
Nemni R, Corbo M, Fazio R, et al. Cryoglobulinaemic neuropathy. A clinical, morphological and immunocytochemical study of 8 cases. Brain. 1988;111(Pt 3):541-552. doi:10.1093/brain/111.3.541
-
Stone MJ, Bogen SA. Evidence-based focused review of management of hyperviscosity syndrome. Blood. 2012;119(10):2205-2208. doi:10.1182/blood-2011-11-392258
-
De Vita S, Soldano F, Isola M, et al. Preliminary classification criteria for the cryoglobulinaemic vasculitis. Ann Rheum Dis. 2011;70(7):1183-1190. doi:10.1136/ard.2010.137828
-
Jennette JC, Falk RJ, Bacon PA, et al. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013;65(1):1-11. doi:10.1002/art.37715
-
Pillebout E, Thervet E, Hill G, Alberti C, Vanhille P, Nochy D. Henoch-Schönlein purpura in adults: outcome and prognostic factors. J Am Soc Nephrol. 2002;13(5):1271-1278. doi:10.1097/01.asn.0000013883.99976.22
-
Tsokos GC. Systemic lupus erythematosus. N Engl J Med. 2011;365(22):2110-2121. doi:10.1056/NEJMra1100359
-
Ramos-Casals M, Tzioufas AG, Stone JH, Sisó A, Bosch X. Treatment of primary Sjögren syndrome: a systematic review. JAMA. 2010;304(4):452-460. doi:10.1001/jama.2010.1014
-
Kastritis E, Dimopoulos MA. Waldenström's macroglobulinemia: recent advances and current treatment strategies. Curr Treat Options Oncol. 2011;12(2):119-130. doi:10.1007/s11864-011-0148-8
-
Kolopp-Sarda MN, Miossec P. Cryoglobulins: An update on detection, mechanisms and clinical contribution. Autoimmun Rev. 2018;17(5):457-464. doi:10.1016/j.autrev.2017.11.035
-
Garini G, Quartuccio L, Filippini D, Benatti S, Allegri F. The changing face of mixed cryoglobulinaemia. Clin Exp Rheumatol. 2017;35(Suppl 103):175-180.
-
Khattab E, Chemin I, Vuillermoz I, et al. Analysis of HCV co-receptor CD81 expression on circulating immune cells in patients with chronic hepatitis C and mixed cryoglobulinaemia. J Viral Hepat. 2004;11(5):453-456. doi:10.1111/j.1365-2893.2004.00532.x
-
Lamprecht P, Gause A, Gross WL. Cryoglobulinemic vasculitis. Arthritis Rheum. 1999;42(12):2507-2516. doi:10.1002/1529-0131(199912)42:12less than 2507::AID-ANR2> 3.0.CO;2-H
-
Alric L, Plaisier E, Thébault S, et al. Influence of antiviral therapy in hepatitis C virus-associated cryoglobulinemic MPGN. Am J Kidney Dis. 2004;43(4):617-623. doi:10.1053/j.ajkd.2003.12.019
-
Zignego AL, Giannini C, Gragnani L, et al. Hepatitis C virus infection in the immunocompromised host: a complex scenario with variable clinical impact. J Transl Med. 2012;10:158. doi:10.1186/1479-5876-10-158
-
De Vita S, Quartuccio L, Isola M, et al. A randomized controlled trial of rituximab for the treatment of severe cryoglobulinemic vasculitis. Arthritis Rheum. 2012;64(3):843-853. doi:10.1002/art.34331
-
Gragnani L, Visentini M, Fognani E, et al. Prospective study of guideline-tailored therapy with direct-acting antivirals for hepatitis C virus-associated mixed cryoglobulinemia. Hepatology. 2016;64(5):1473-1482. doi:10.1002/hep.28753
-
Emery JS, Kuczynski M, La D, et al. Efficacy and safety of direct acting antivirals for the treatment of mixed cryoglobulinemia. Am J Gastroenterol. 2017;112(8):1298-1308. doi:10.1038/ajg.2017.49
-
Sneller MC, Hu Z, Langford CA. A randomized controlled trial of rituximab following failure of antiviral therapy for hepatitis C virus-associated cryoglobulinemic vasculitis. Arthritis Rheum. 2012;64(3):835-842. doi:10.1002/art.34322
-
Visentini M, Granata G, Veneziano ML, et al. Efficacy of low-dose rituximab for mixed cryoglobulinemia. Clin Immunol. 2007;125(1):30-33. doi:10.1016/j.clim.2007.06.007
-
Rockx MA, Clark WF. Plasma exchange for treating cryoglobulinemia: a descriptive analysis. Transfus Apher Sci. 2010;42(3):247-251. doi:10.1016/j.transci.2010.03.004
-
Sidana S, Rajkumar SV, Dispenzieri A, et al. Clinical presentation and outcomes in light chain amyloidosis patients with non-evaluable serum free light chains. Leukemia. 2018;32(3):729-735. doi:10.1038/leu.2017.286
-
Kidney Disease: Improving Global Outcomes (KDIGO) Glomerulonephritis Work Group. KDIGO Clinical Practice Guideline for Glomerulonephritis. Kidney Int Suppl. 2012;2(2):186-197.
-
Evens AM, Jovanovic BD, Su YC, et al. Rituximab-associated hepatitis B virus (HBV) reactivation in lymphoproliferative diseases: meta-analysis and examination of FDA safety reports. Ann Oncol. 2011;22(5):1170-1180. doi:10.1093/annonc/mdq583
-
Morgan RL, Baack B, Smith BD, Yartel A, Pitasi M, Falck-Ytter Y. Eradication of hepatitis C virus infection and the development of hepatocellular carcinoma: a meta-analysis of observational studies. Ann Intern Med. 2013;158(5 Pt 1):329-337. doi:10.7326/0003-4819-158-5-201303050-00005
-
Terrier B, Krastinova E, Marie I, et al. Management of noninfectious mixed cryoglobulinemia vasculitis: data from 242 cases included in the CryoVas survey. Blood. 2012;119(25):5996-6004. doi:10.1182/blood-2011-12-396028
-
Saadoun D, Terrier B, Semoun O, et al. Hepatitis C virus-associated polyarteritis nodosa. Arthritis Care Res (Hoboken). 2011;63(3):427-435. doi:10.1002/acr.20381
-
De Vita S, Quartuccio L, Seror R, et al. Efficacy and safety of belimumab given for 12 months in primary Sjögren's syndrome: the BELISS open-label phase II study. Rheumatology (Oxford). 2015;54(12):2249-2256. doi:10.1093/rheumatology/kev257
-
KDIGO Clinical Practice Guideline for Glomerulonephritis. Kidney Int Suppl. 2012;2(2):139-274.
Last Updated: 2026-01-05 Cite this topic: MedVellum. Cryoglobulinaemic Vasculitis. Updated January 2026.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Hepatitis C Virus Infection
- Immunoglobulin Structure and Function
Differentials
Competing diagnoses and look-alikes to compare.
Consequences
Complications and downstream problems to keep in mind.
- Membranoproliferative Glomerulonephritis
- Peripheral Neuropathy