Systemic Lupus Erythematosus
Systemic lupus erythematosus (SLE) is a chronic, relapsing-remitting multisystem autoimmune disease characterised by los... MRCP exam preparation.
What matters first
Systemic lupus erythematosus (SLE) is a chronic, relapsing-remitting multisystem autoimmune disease characterised by los... MRCP exam preparation.
Lupus nephritis (Class III-IV)
10 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Lupus nephritis (Class III-IV)
- Neuropsychiatric lupus
- Catastrophic antiphospholipid syndrome
- Severe haematological crisis
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Drug-Induced Lupus
- Mixed Connective Tissue Disease
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Systemic Lupus Erythematosus
1. Clinical Overview
Summary
Systemic lupus erythematosus (SLE) is a chronic, relapsing-remitting multisystem autoimmune disease characterised by loss of tolerance to nuclear antigens, production of pathogenic autoantibodies (particularly anti-dsDNA and anti-Sm), immune complex deposition, and complement-mediated inflammation affecting virtually any organ system. [1,2] It predominantly affects women of childbearing age with a striking female-to-male ratio of 9:1, and shows marked ethnic variation with higher incidence and severity in African, Asian, and Hispanic populations. [3] The disease exhibits remarkable heterogeneity in presentation, ranging from mild mucocutaneous and musculoskeletal involvement to life-threatening renal, neurological, and haematological manifestations. [4]
Classification is based on the 2019 ACR/EULAR criteria, which require antinuclear antibody (ANA) positivity at titre ≥1:80 as an entry criterion, followed by weighted scoring across clinical and immunological domains with a threshold of 10 or more points for classification as SLE. [5] This represents a significant improvement over previous criteria with 96% sensitivity and 93% specificity. The SLICC (Systemic Lupus International Collaborating Clinics) 2012 criteria remain an alternative validated classification system. [6]
Hydroxychloroquine is the cornerstone of therapy for all patients with SLE unless contraindicated, with robust evidence demonstrating reduced disease flares by 50%, prevention of irreversible organ damage, reduced mortality, reduced thrombosis risk, favourable lipid and glycaemic effects, and safety in pregnancy. [7,8] Additional immunosuppression is tailored to specific organ involvement and disease severity, with mycophenolate mofetil and cyclophosphamide serving as primary agents for lupus nephritis, while newer biologics including belimumab (anti-BAFF), anifrolumab (anti-type I interferon receptor), and voclosporin (calcineurin inhibitor) provide additional therapeutic options. [9,10,11]
Modern management has transformed SLE prognosis, with 10-year survival now exceeding 90% compared to less than 50% in the 1950s. [12] However, lupus nephritis remains a major determinant of long-term outcomes, with 10-20% of patients with Class III-IV nephritis progressing to end-stage renal disease despite treatment. [13] Premature cardiovascular disease is now the leading cause of late mortality, occurring at 2-10 times the rate of age-matched controls. [14]
Key Facts
- Definition: Chronic multisystem autoimmune disease with loss of tolerance to nuclear antigens, pathogenic autoantibody production, and immune complex-mediated inflammation
- Incidence: 1-10 per 100,000 per year (marked ethnic variation); prevalence 20-150 per 100,000 worldwide
- Demographics: F:M ratio 9:1 (reproductive years), 2:1 in children and elderly; peak onset 15-45 years; 2-4x higher incidence in African, Asian, Hispanic populations
- Pathognomonic Features: ANA positivity (95-99%) + anti-dsDNA antibodies (60-70%) + multi-organ involvement + low complement (C3/C4)
- Gold Standard Diagnosis: 2019 ACR/EULAR criteria (ANA ≥1:80 entry + weighted domains ≥10 points)
- Gold Standard Investigation: ANA (screening), anti-dsDNA and anti-Sm (specificity), C3/C4 (activity), urinalysis (nephritis)
- First-line Treatment: Hydroxychloroquine 5mg/kg/day (maximum 400mg) for ALL patients
- Severe Organ Involvement: Mycophenolate mofetil or cyclophosphamide for nephritis/CNS disease
- Prognosis: 10-year survival >90%; 20-year survival 75-85%; nephritis and neuropsychiatric disease major determinants
- Leading Causes of Death: Infection (early), cardiovascular disease (late), malignancy
Clinical Pearls
HCQ Universal Pearl: Hydroxychloroquine reduces flares by 50%, prevents irreversible organ damage accumulation (SLICC Damage Index), reduces mortality by up to 70%, and is safe in pregnancy. [7,8] ALL SLE patients should be on HCQ at 5mg/kg actual body weight (≤400mg/day) unless contraindicated. Retinopathy risk is less than 1% with proper dosing but annual ophthalmology screening is required after 5 years of use.
Anti-dsDNA Activity Pearl: Anti-dsDNA antibodies correlate with disease activity and are particularly associated with lupus nephritis. [15] Rising anti-dsDNA titres often precede clinical flares by 4-8 weeks, allowing proactive intervention. However, approximately 30-40% of SLE patients remain anti-dsDNA negative throughout their disease course.
Complement Consumption Pearl: Low C3 and C4 levels suggest active disease with immune complex consumption via classical complement pathway activation. [16] Persistently low complement levels with normal or negative anti-dsDNA may indicate hereditary complement deficiency (C1q, C2, C4), which itself is a strong SLE risk factor (25-fold increased risk).
Nephritis Surveillance Pearl: All SLE patients require regular urinalysis (every 3-6 months minimum). Any new proteinuria (spot urine protein:creatinine ratio >50mg/mmol), active urinary sediment (dysmorphic RBCs, RBC casts), or rising creatinine warrants urgent nephrology assessment and strong consideration of renal biopsy. [17] The ISN/RPS classification guides treatment intensity.
Pregnancy Planning Pearl: Plan all pregnancies during disease quiescence (ideally 6-12 months of remission). Anti-Ro/SSA antibodies carry 1-2% risk of congenital heart block and 15-20% risk of neonatal lupus. [18] Fetal echocardiography monitoring is essential. Continue hydroxychloroquine throughout pregnancy. Avoid mycophenolate and cyclophosphamide (teratogenic).
Steroid-Sparing Pearl: Prolonged glucocorticoid use (>7.5mg prednisolone equivalent daily for >6 months) causes irreversible organ damage. [19] Always use steroid-sparing immunosuppressants (azathioprine, methotrexate, mycophenolate) to enable rapid steroid taper to less than 7.5mg/day maintenance or discontinuation.
Type I Interferon Pearl: Type I interferon signature is present in approximately 60-80% of SLE patients and correlates with disease activity and severity. [20] Anifrolumab (anti-type I IFN receptor antibody) shows particular efficacy in high interferon signature patients.
Why This Matters Clinically
SLE is the prototypical systemic autoimmune disease and the "great imitator" in medicine, capable of affecting any organ system with presentations ranging from subtle constitutional symptoms to life-threatening multi-organ failure. Rheumatologists, nephrologists, dermatologists, neurologists, haematologists, and general physicians must all recognise and manage SLE and its complications.
Delayed diagnosis leads to irreversible organ damage, quantified by the SLICC/ACR Damage Index, which predicts mortality and morbidity. [21] Early diagnosis and treat-to-target strategies (aiming for remission or low disease activity using validated instruments like SLEDAI-2K or BILAG) have transformed outcomes. Understanding the immunology, autoantibody profiles, complement system, and emerging biologics is essential for modern SLE management and exam success.
2. Epidemiology
Incidence and Prevalence
SLE shows marked geographic and ethnic variation in incidence and prevalence. [3,22]
| Population | Incidence (per 100,000/year) | Prevalence (per 100,000) | Notes |
|---|---|---|---|
| Overall (global) | 1-10 | 20-150 | Wide variation by region |
| African descent | 8-11 | 200-300 | Higher severity, earlier onset |
| Asian descent | 4-8 | 50-100 | Particularly East/Southeast Asian |
| European descent | 1-5 | 20-50 | Lower incidence baseline |
| Hispanic/Latino | 4-7 | 100-200 | Intermediate-high risk |
| UK (general) | 3-4 | 50-70 | Higher in urban areas |
| USA (general) | 5-6 | 60-100 | Regional variation |
The incidence of SLE has increased over the past 50 years, likely due to improved case ascertainment, better diagnostic criteria, increased disease awareness, and true increases in disease incidence due to environmental factors. [22]
Demographics
- Sex: Profound female predominance 9:1 during reproductive years (ages 15-45); ratio narrows to 2:1 in prepubertal children and post-menopausal women, implicating hormonal factors (oestrogen) in pathogenesis
- Age: Peak incidence 15-45 years; 15-20% present in childhood (less than 16 years) with more severe disease; late-onset SLE (>50 years) shows less female predominance and milder disease
- Race/Ethnicity:
- "African ancestry: 2-4x higher incidence, younger age at onset, more severe disease, higher nephritis rates (50-70% vs 30-40%), worse outcomes"
- "Asian ancestry (particularly East/Southeast Asian): 2-3x higher incidence, increased neuropsychiatric manifestations"
- "Hispanic/Latino: 2-3x higher incidence, increased nephritis"
- "Native American: High incidence in some populations"
- "European ancestry: Lowest incidence, later age at onset"
- Geography: Higher prevalence in urban vs rural areas; higher prevalence at latitudes closer to equator (UV exposure hypothesis); clustering in certain geographic regions suggests environmental triggers
- Socioeconomic factors: Lower socioeconomic status associated with higher incidence, more severe disease, worse outcomes (likely multifactorial: access to care, medication adherence, comorbidities)
Risk Factors
| Category | Factors | Relative Risk | Notes |
|---|---|---|---|
| Genetic | Family history of SLE | 10-20x | Concordance 25-50% in monozygotic twins |
| HLA-DR2 (DRB1*1501) | 2-3x | Associated with anti-Ro/La antibodies | |
| HLA-DR3 (DRB1*0301) | 2-3x | Associated with anti-dsDNA, nephritis | |
| Complement deficiencies (C1q, C2, C4) | 10-90x | C1q deficiency: >90% develop SLE | |
| TREX1 mutations | Variable | Defective DNA degradation → type I IFN | |
| IRF5, STAT4, BLK, PTPN22 | 1.5-2x | Type I interferon and B cell signalling | |
| Hormonal | Female sex | 9x | Oestrogen enhances B cell activation |
| Early menarche | 1.5x | Prolonged oestrogen exposure | |
| Exogenous oestrogen (HRT, OCP) | 1.2-2x | Controversial, may trigger flares | |
| Pregnancy/postpartum period | Variable | 30% flare during pregnancy | |
| Environmental | Ultraviolet (UV) light exposure | 2-3x | Triggers apoptosis, exposes nuclear Ags |
| Epstein-Barr virus (EBV) infection | 2-10x | Molecular mimicry with nuclear antigens | |
| Smoking (current) | 1.5-2x | Dose-dependent; worsens outcomes | |
| Silica exposure (occupational) | 2-3x | Mining, construction, sandblasting | |
| Crystalline silica dust | 2-5x | Enhances autoimmunity | |
| Medications | Hydralazine | Variable | Drug-induced lupus (anti-histone+) |
| Procainamide | Variable | Drug-induced lupus (anti-histone+) | |
| Isoniazid, minocycline | Variable | Usually resolves with drug cessation | |
| TNF inhibitors | Rare | Paradoxical drug-induced lupus | |
| Interferon-alpha therapy | Variable | Unmasks SLE in predisposed |
Temporal Trends
- Improved survival: 5-year survival increased from less than 50% (1950s) to >95% (2020s); 10-year survival now >90% [12]
- Changing mortality patterns: Early deaths from active disease/infection declining; late deaths from cardiovascular disease and malignancy increasing
- Organ involvement trends: Nephritis rates may be decreasing in some populations (earlier diagnosis and treatment); neuropsychiatric manifestations increasingly recognised
- Treatment evolution: Introduction of biologics (belimumab 2011, anifrolumab 2021, voclosporin 2021) expanding therapeutic options [9,10,11]
3. Aetiology and Pathophysiology
Genetic Predisposition
SLE is a polygenic disease with >100 genetic loci identified through genome-wide association studies (GWAS). [23] Key genetic factors include:
HLA Associations:
- HLA-DR2 (DRB1*1501): Associated with anti-Ro/SSA and anti-La/SSB antibodies, photosensitivity, subacute cutaneous lupus
- HLA-DR3 (DRB1*0301): Associated with anti-dsDNA antibodies, lupus nephritis, younger age at onset, more severe disease
- HLA-DR4: Some protective effects against SLE
Complement Deficiencies:
- C1q deficiency: >90% develop SLE; defective clearance of apoptotic debris and immune complexes
- C2 deficiency: 10-30% develop SLE; most common complement deficiency
- C4 deficiency: 75% develop SLE or lupus-like disease
Type I Interferon Pathway:
- IRF5 (interferon regulatory factor 5): Increased type I interferon production
- STAT4: Enhanced interferon signalling and Th1 differentiation
- TYK2: Type I interferon receptor signalling
B Cell Signalling:
- BLK (B lymphocyte kinase): Altered B cell receptor signalling
- BANK1: B cell activation and proliferation
- LYN: Inhibitory signalling in B cells
Clearance of Apoptotic Cells:
- TREX1: Three prime repair exonuclease; defects lead to accumulation of intracellular DNA → type I interferon activation
- DNASE1: Degrades extracellular DNA; deficiency impairs clearance
Other Pathways:
- PTPN22: T cell receptor signalling; also associated with RA, T1DM
- FCGR2A, FCGR3A: Fc receptor variants affecting immune complex clearance
- TNF-AIP3: NF-κB signalling regulation
Pathophysiology Overview
SLE pathogenesis involves a complex interplay of genetic susceptibility, environmental triggers, immune dysregulation, autoantibody production, immune complex deposition, and tissue inflammation. [1,2,24]
Step 1: Environmental Triggers in Genetically Susceptible Individuals
- UV light: Induces keratinocyte apoptosis, exposes nuclear antigens (especially Ro/SSA on apoptotic blebs), upregulates type I interferons
- Infections: EBV molecular mimicry with nuclear antigens (EBNA-1 shares homology with Sm and Ro antigens); other viruses may trigger via TLR activation
- Smoking: Oxidative stress, DNA damage, altered immune responses
- Hormonal factors: Oestrogen enhances B cell survival and antibody production, reduces regulatory T cell function
- Medications: Direct oxidative damage (procainamide, hydralazine), altered self-antigen presentation
Step 2: Loss of Immune Tolerance
- Defective clearance of apoptotic cells: Complement deficiencies and defects in phagocyte clearance lead to accumulation of apoptotic material and secondary necrosis, releasing nuclear antigens
- Dendritic cell activation: Plasmacytoid dendritic cells (pDCs) sense nucleic acids via TLR7/TLR9, produce massive amounts of type I interferons (IFN-α, IFN-β)
- Type I interferon signature: Present in 60-80% of SLE patients; drives disease through multiple mechanisms: [20]
- Enhanced dendritic cell maturation and antigen presentation
- B cell activation and differentiation into plasma cells
- T cell activation and Th1/Th17 skewing
- Decreased regulatory T cell function
- Direct tissue effects (vascular damage)
Step 3: B Cell Hyperactivity and Autoantibody Production
- B cell abnormalities: Increased B cell survival (elevated BAFF/BLyS levels), reduced B cell activation thresholds, autoreactive B cell escape from negative selection
- T cell help: CD4+ T helper cells provide co-stimulation (CD40-CD40L) and cytokines (IL-21, IL-6) driving B cell differentiation
- Autoantibody production: [25]
- "Anti-nuclear antibodies (ANA): 95-99% sensitive, screen via indirect immunofluorescence on HEp-2 cells; patterns (homogeneous, speckled, nucleolar) have limited specificity"
- "Anti-dsDNA: 60-70% of SLE; highly specific (>95%); correlates with disease activity and nephritis; pathogenic via direct binding to glomerular basement membrane and immune complex formation"
- "Anti-Sm: 20-30% of SLE; highly specific (pathognomonic, >98%); targets spliceosomal proteins; does not correlate with activity"
- "Anti-Ro/SSA (52kD, 60kD): 30-40%; photosensitivity, subacute cutaneous lupus, neonatal lupus, congenital heart block; overlaps with Sjögren's"
- "Anti-La/SSB: 10-15%; usually with anti-Ro; Sjögren's overlap"
- "Anti-RNP: 25-30%; overlaps with mixed connective tissue disease (MCTD); may be protective against nephritis"
- "Anti-ribosomal P: 10-20%; neuropsychiatric lupus (especially depression, psychosis), hepatic involvement"
- "Antiphospholipid antibodies: 30-40%; lupus anticoagulant, anticardiolipin, anti-β2 glycoprotein I; thrombosis and pregnancy morbidity"
Step 4: Immune Complex Formation and Deposition
- Circulating immune complexes: Autoantibodies bind nuclear antigens released from apoptotic cells forming immune complexes
- Tissue deposition: Immune complexes deposit in tissues with high blood flow and/or basement membranes:
- Kidney glomeruli (lupus nephritis)
- Skin-dermal junction (cutaneous lupus)
- Choroid plexus and blood-brain barrier (neuropsychiatric lupus)
- Serosal surfaces (pleuritis, pericarditis)
- Joints (arthritis)
Step 5: Complement Activation and Inflammation
- Classical pathway activation: Immune complexes activate C1q → classical complement cascade
- C3 and C4 consumption: Active disease shows low C3/C4 due to ongoing consumption
- Complement fragments: C3a and C5a are anaphylatoxins promoting inflammation; C3b opsonises immune complexes
- Membrane attack complex (C5b-9): Direct cell lysis and tissue damage
Step 6: Neutrophil and Macrophage Recruitment
- Neutrophil extracellular traps (NETs): Neutrophils release NETs containing DNA, histones, and antimicrobial proteins; NETs are immunogenic in SLE and source of autoantigens
- Macrophage activation: Phagocytosis of immune complexes via Fc receptors, release of pro-inflammatory cytokines (TNF-α, IL-1, IL-6)
- Tissue inflammation: Recruitment of T cells and B cells to inflamed tissues, formation of tertiary lymphoid structures
Step 7: Organ Damage
- Direct inflammation: Glomerulonephritis, vasculitis, serositis
- Vascular injury: Endothelial dysfunction, accelerated atherosclerosis (type I interferons, inflammatory cytokines, antiphospholipid antibodies, oxidised LDL)
- Chronic damage: Fibrosis, scarring, organ failure (SLICC/ACR Damage Index quantifies irreversible damage)
Autoantibody Profiles and Clinical Associations
| Antibody | Prevalence in SLE | Specificity | Clinical Association | Test Method |
|---|---|---|---|---|
| ANA | 95-99% | Low (also in other CTD, healthy) | Screening; negative ANA questions diagnosis | IIF on HEp-2 cells |
| Anti-dsDNA | 60-70% | High (95-98%) | Disease activity marker, lupus nephritis predictor | Farr assay, ELISA, Crithidia |
| Anti-Sm | 20-30% | Very high (>98%) | Pathognomonic for SLE; no activity correlation | ELISA, immunoprecipitation |
| Anti-Ro/SSA | 30-40% | Moderate | Photosensitivity, SCLE, neonatal lupus, CHB, Sjögren's overlap | ELISA |
| Anti-La/SSB | 10-15% | Moderate | Usually with anti-Ro; Sjögren's overlap | ELISA |
| Anti-RNP | 25-30% | Low-moderate | MCTD overlap; may protect against nephritis | ELISA |
| Anti-ribosomal P | 10-20% | High for SLE | Neuropsychiatric lupus (psychosis, depression), hepatitis | ELISA |
| Anti-histone | 50-70% (95% in DIL) | Low | Drug-induced lupus when isolated | ELISA |
| Antiphospholipid | 30-40% | Moderate | Thrombosis, pregnancy loss, livedo reticularis | LAC, aCL, anti-β2GPI |
| Anti-C1q | 30-40% | Moderate | Lupus nephritis, correlates with activity | ELISA |
Key Immunological Concepts for Exams:
- Type I interferons drive disease in the majority of patients and are therapeutic target (anifrolumab)
- BAFF/BLyS (B cell activating factor) is elevated in SLE, promotes B cell survival, and is therapeutic target (belimumab)
- Anti-dsDNA pathogenicity: Cross-reacts with glomerular basement membrane α-actinin and other renal antigens; forms immune complexes
- Complement serves dual role: deficiency predisposes to SLE (impaired clearance), but consumption indicates active disease
4. Clinical Presentation
SLE is the "great imitator" with protean manifestations. [4] The 2019 ACR/EULAR criteria capture the spectrum of clinical features, but many patients present with non-specific constitutional symptoms initially.
Constitutional Symptoms
- Fatigue: 90-95% of patients; often most debilitating symptom; multifactorial (disease activity, medications, fibromyalgia, depression, anaemia, hypothyroidism)
- Fever: 50-80% during flares; typically low-grade but can be high-grade mimicking infection; always exclude infection before attributing fever to SLE
- Weight loss: Common during active disease (10-15% of patients)
- Lymphadenopathy: 50% of patients; generalised or localised; rarely massive
- Splenomegaly: 10-20%; usually mild
Mucocutaneous Manifestations (80-90%)
Acute Cutaneous Lupus Erythematosus (ACLE):
- Malar (butterfly) rash: Erythematous rash over malar eminences and nasal bridge, characteristically sparing the nasolabial folds; photosensitive; may be flat or raised; may be transient or persistent
- Generalised ACLE: Maculopapular eruption resembling morbilliform rash, photodistributed
Subacute Cutaneous Lupus Erythematosus (SCLE):
- Papulosquamous/psoriasiform: Scaly erythematous plaques
- Annular-polycyclic: Ring-shaped lesions with central clearing
- Photodistributed (shoulders, extensor arms, upper back, V-neck)
- Strongly associated with anti-Ro/SSA antibodies
- Usually no scarring
Chronic Cutaneous Lupus Erythematosus (CCLE):
- Discoid lupus erythematosus (DLE): Scarring, atrophic plaques with erythema, follicular plugging, dyspigmentation; commonly on face, scalp (scarring alopecia), ears; 5-10% of DLE patients develop SLE
- Lupus profundus: Subcutaneous nodules
- Chilblain lupus: Violaceous lesions on acral sites
Other Cutaneous Features:
- Photosensitivity: 40-60%; rash provoked/exacerbated by UV light exposure
- Oral ulcers: 40-50%; usually painless (hard palate, buccal mucosa); nasal ulcers also occur
- Non-scarring alopecia: Diffuse hair loss or frontal alopecia; "lupus hair" (short, broken frontal hairs)
- Raynaud's phenomenon: 30-40%; colour changes (white-blue-red) with cold/stress
- Livedo reticularis: Net-like vascular pattern; associated with antiphospholipid antibodies
- Vasculitic lesions: Palpable purpura, digital infarcts, ulcers
Musculoskeletal Manifestations (90%)
- Arthralgia: Very common (>90%); may be presenting symptom
- Arthritis: 70-80%; inflammatory, symmetric, polyarticular; small joints of hands (MCP, PIP), wrists, knees commonly affected
- Non-erosive: Classically non-erosive on X-ray (unlike RA); occasionally mild erosions seen
- Jaccoud's arthropathy: Reducible joint deformities (swan-neck, ulnar deviation) due to ligamentous laxity without erosions; 5-10% of patients
- Myalgia: 50%; muscle tenderness and weakness
- Myositis: 5-10%; inflammatory myositis with elevated CK (overlap with polymyositis)
- Avascular necrosis (AVN): 5-15%; femoral head most common, also humeral head, knees; multifactorial (steroids, antiphospholipid antibodies, vasculopathy); presents with pain, limited range of motion; MRI diagnostic
Renal Manifestations (40-60%)
Lupus Nephritis:
Lupus nephritis is a major determinant of prognosis and occurs in 40-60% of SLE patients, more commonly in African, Asian, and Hispanic patients. [17] It may be present at diagnosis or develop during disease course.
Presentation:
- Asymptomatic: Abnormal urinalysis (proteinuria, haematuria) on routine screening
- Overt: Oedema (periorbital, peripheral), hypertension, oliguria, uraemic symptoms
- Nephrotic syndrome: Proteinuria >3.5g/24h, hypoalbuminaemia, oedema, hyperlipidaemia
- Nephritic syndrome: Haematuria, RBC casts, hypertension, oliguria, rising creatinine
- Rapidly progressive glomerulonephritis (RPGN): Rapid deterioration in renal function
ISN/RPS (International Society of Nephrology/Renal Pathology Society) Classification of Lupus Nephritis:
| Class | Description | Frequency | Clinical Features | Prognosis |
|---|---|---|---|---|
| I | Minimal mesangial | 5-10% | Normal urinalysis, normal renal function | Excellent; no treatment needed |
| II | Mesangial proliferative | 10-20% | Mild proteinuria, haematuria; normal function | Good; HCQ ± low-dose steroids |
| III | Focal proliferative (less than 50% glomeruli) | 15-25% | Proteinuria, haematuria, ± impaired function | Moderate; requires immunosuppression |
| IV | Diffuse proliferative (≥50% glomeruli) | 30-40% | Heavy proteinuria, haematuria, casts, hypertension, impaired function | Requires aggressive immunosuppression |
| V | Membranous | 10-20% | Nephrotic syndrome; ± impaired function | Variable; often requires immunosuppression |
| VI | Advanced sclerosing (>90% sclerosis) | 5-10% | Chronic kidney disease, hypertension | Poor; often ESRD; limited response to IS |
- Classes III and IV may be segmental (S) or global (G)
- Active (A) and chronic (C) lesions are scored separately
- Class III/IV + V = mixed proliferative and membranous
- Renal biopsy is essential for classification and guiding treatment
Biomarkers of Lupus Nephritis:
- Rising anti-dsDNA, falling C3/C4
- Proteinuria: spot urine protein:creatinine ratio (PCR) >50mg/mmol
- Active sediment: dysmorphic RBCs, RBC casts, WBC casts
- Rising serum creatinine, falling eGFR
Neuropsychiatric Manifestations (30-40%)
The ACR defines 19 neuropsychiatric SLE (NPSLE) syndromes divided into central and peripheral nervous system manifestations. [26] Attribution to SLE vs other causes (infection, medication, metabolic, psychiatric) is often challenging.
Central Nervous System:
- Headache: 25-50%; may be migraine-like or tension-type; severe refractory headache may indicate CNS vasculitis
- Cognitive dysfunction: 20-40%; impaired memory, attention, executive function; can be subtle
- Mood disorders: Depression (40-50%), anxiety; multifactorial (disease burden, steroids, chronic illness)
- Psychosis: 5-10%; hallucinations, delusions, paranoia; associated with anti-ribosomal P antibodies
- Seizures: 10-20%; focal or generalised; may be presenting feature
- Acute confusional state: 5-10%; delirium; exclude infection, metabolic causes
- Cerebrovascular disease: 5-15%; stroke (thrombotic or haemorrhagic), TIA; antiphospholipid antibodies major risk factor
- Aseptic meningitis: Rare; CSF pleocytosis, elevated protein, normal glucose; exclude infection
- Myelopathy: Rare; transverse myelitis with paraparesis, sensory level, sphincter dysfunction
- Movement disorders: Chorea (especially with antiphospholipid antibodies), parkinsonism, dystonia
- Demyelinating syndrome: Rare; mimics multiple sclerosis
Peripheral Nervous System:
- Peripheral neuropathy: 5-20%; sensory, motor, or mixed; mononeuritis multiplex (vasculitis)
- Cranial neuropathy: Optic neuritis, oculomotor palsies
- Guillain-Barré syndrome: Rare
- Autonomic neuropathy: Rare
Investigation of NPSLE:
- MRI brain: Focal lesions, white matter hyperintensities, atrophy; normal in many cases
- CSF analysis: Exclude infection; may show pleocytosis, elevated protein, oligoclonal bands
- EEG: Seizures, encephalopathy
- Neuropsychological testing: Quantify cognitive deficits
- Exclude alternative causes: Infection (HSV, cryptococcus), thrombosis (antiphospholipid syndrome), metabolic, medication effects (steroids), uraemia
Haematological Manifestations (50-80%)
Anaemia:
- Anaemia of chronic disease: Most common; normocytic, normochromic
- Autoimmune haemolytic anaemia (AIHA): 10-15%; Coombs-positive; reticulocytosis, elevated LDH, low haptoglobin, spherocytes on film
- Iron deficiency: GI bleeding (NSAIDs, steroids), menorrhagia
Leucopenia:
- Leucopenia (WBC less than 4.0 x 10⁹/L): 50-60%; usually mild; rarely problematic unless less than 2.0
- Lymphopenia (lymphocytes less than 1.0 x 10⁹/L): 70-80%; more specific for SLE than leucopenia; correlates with disease activity
Thrombocytopenia:
- Mild thrombocytopenia (platelets 100-150 x 10⁹/L): Common
- Moderate-severe (platelets less than 100 x 10⁹/L): 10-30%; immune-mediated (antiplatelet antibodies)
- Severe (platelets less than 20 x 10⁹/L): Requires urgent treatment (steroids ± IVIG)
- Distinguish from thrombotic thrombocytopenic purpura (TTP) if microangiopathic haemolytic anaemia present
Coagulation Abnormalities:
- Lupus anticoagulant: 20-40%; paradoxically increases thrombosis risk despite prolonging aPTT
- Acquired factor deficiencies: Rare; factor VIII inhibitor, prothrombin deficiency
Lymphoproliferative:
- Slightly increased risk of lymphoma (2-3x general population)
Cardiopulmonary Manifestations (30-50%)
Cardiac:
- Pericarditis: 25-50%; chest pain (positional, pleuritic), pericardial rub, ECG changes (widespread ST elevation, PR depression), effusion on echo (usually small); may be presenting feature
- Myocarditis: 5-10%; heart failure, arrhythmias, troponin elevation; may be subclinical
- Libman-Sacks endocarditis: 10-20%; sterile verrucous vegetations on mitral or aortic valves; usually asymptomatic; rarely causes regurgitation or emboli; associated with antiphospholipid antibodies
- Coronary artery disease: 2-10x increased risk vs age-matched controls; premature atherosclerosis (inflammation, type I interferons, dyslipidaemia, hypertension, steroids); now leading cause of late mortality [14]
- Conduction abnormalities: Heart block (especially in neonates exposed to maternal anti-Ro/SSA)
- Pulmonary hypertension: 5-10%; may be primary (vasculopathy) or secondary (ILD, chronic thromboembolism)
Pulmonary:
- Pleuritis/Pleurisy: 30-50%; pleuritic chest pain, pleural rub, pleural effusion (exudative, lymphocytic)
- Acute lupus pneumonitis: 5-10%; fever, dyspnoea, cough, hypoxia, bilateral infiltrates on CXR/CT; exclude infection; high mortality; requires aggressive immunosuppression
- Interstitial lung disease (ILD): 5-15%; progressive dyspnoea, bibasal crackles, restrictive pattern on PFTs, reticular changes on HRCT
- Pulmonary haemorrhage: Rare but life-threatening; haemoptysis, diffuse alveolar haemorrhage on imaging, falling haemoglobin; requires urgent immunosuppression
- Shrinking lung syndrome: Rare; progressive dyspnoea, restrictive defect, small lung volumes, elevated diaphragms on CXR; due to diaphragmatic myopathy
- Pulmonary embolism: Increased risk (antiphospholipid antibodies)
Gastrointestinal Manifestations (20-40%)
- Non-specific: Nausea, vomiting, diarrhoea, abdominal pain
- Serositis: Peritonitis (ascites, abdominal pain)
- Mesenteric vasculitis: Rare but serious; abdominal pain, bowel ischaemia, perforation; high mortality
- Lupus hepatitis: 5-10%; elevated transaminases; autoimmune hepatitis overlap
- Pancreatitis: 5%; may be drug-induced (azathioprine, steroids) or disease-related
- Protein-losing enteropathy: Rare
- Dysphagia/oesophageal dysmotility: Overlap with systemic sclerosis features
Other Manifestations
Ophthalmological:
- Keratoconjunctivitis sicca (dry eyes): 10-30%; Sjögren's overlap
- Retinal vasculitis: Cotton-wool spots, retinal haemorrhages, vascular occlusion
- Optic neuritis: Rare
Haematological - Antiphospholipid Syndrome (APS):
- 30-40% of SLE patients have antiphospholipid antibodies
- Arterial thrombosis: Stroke, MI, peripheral arterial thrombosis
- Venous thrombosis: DVT, PE, hepatic/portal vein thrombosis
- Obstetric: Recurrent pregnancy loss, pre-eclampsia, placental insufficiency, IUGR
- Catastrophic APS: Rare; multi-organ thrombosis; high mortality
Fever of Unknown Origin (FUO):
- SLE is a cause of FUO; always exclude infection
Red Flags - Life-Threatening Manifestations
[!CAUTION] Immediate Escalation Required:
- Lupus nephritis Class III-IV: Nephrotic/nephritic syndrome, rapidly rising creatinine → urgent nephrology, renal biopsy, aggressive immunosuppression
- Neuropsychiatric lupus: New seizures, psychosis, acute confusional state, myelopathy → exclude infection, brain imaging, consider pulse steroids
- Pulmonary haemorrhage: Haemoptysis, hypoxia, diffuse infiltrates → urgent HDU/ITU, pulse steroids, cyclophosphamide, plasmapheresis
- Macrophage activation syndrome (MAS): Fever, cytopenias, hepatosplenomegaly, high ferritin, low fibrinogen → high-dose steroids, consider anakinra/etoposide
- Catastrophic antiphospholipid syndrome: Multi-organ thrombosis → anticoagulation, steroids, plasmapheresis, IVIG
- Severe thrombocytopenia (less than 20 x 10⁹/L): Bleeding risk → steroids, IVIG, consider platelet transfusion if active bleeding
- Autoimmune haemolytic anaemia: Severe anaemia, haemodynamic compromise → steroids, transfusion (cross-match difficult)
- Mesenteric vasculitis: Acute abdomen, bowel ischaemia → surgical review, imaging, immunosuppression
5. Clinical Examination
Structured Examination Approach
General Inspection:
- Appearance: Cushingoid facies (moon face, buffalo hump, central obesity, striae) from chronic steroid use
- Mobility: Observe gait (arthritis, myopathy, AVN)
- Emotional state: Depression, anxiety (common comorbidities)
Hands:
- Skin: Raynaud's phenomenon (colour changes), digital infarcts (vasculitis, antiphospholipid), nail fold capillary changes
- Joints: Swelling (MCP, PIP, wrists), deformities (Jaccoud's arthropathy - swan-neck, ulnar deviation, but reducible), tenderness
- Nails: Onycholysis, splinter haemorrhages (vasculitis)
Face and Head:
- Skin:
- "Malar (butterfly) rash: Erythema over malar eminences and nasal bridge, sparing nasolabial folds"
- "Discoid lesions: Scarring, atrophic plaques, follicular plugging"
- "Photosensitivity: Sun-exposed areas"
- Hair: Non-scarring alopecia (diffuse, frontal), "lupus hair" (short broken hairs at hairline), scarring alopecia (discoid lesions)
- Eyes: Conjunctival pallor (anaemia), jaundice (haemolysis, hepatitis), scleritis/episcleritis, fundoscopy (cotton-wool spots from retinal vasculitis)
- Mouth: Oral ulcers (hard palate, buccal mucosa - usually painless), nasal ulcers
Neck:
- Lymphadenopathy (generalised or localised)
- Thyroid (hypothyroidism common)
Chest:
- Cardiovascular: Pericardial rub (pericarditis), murmurs (Libman-Sacks endocarditis, valvular disease), signs of heart failure
- Respiratory: Pleural rub, reduced air entry (pleural effusion), bibasal crackles (ILD, pulmonary oedema)
Abdomen:
- Hepatomegaly, splenomegaly (10-20%)
- Ascites (serositis, nephrotic syndrome, cirrhosis)
- Tenderness (peritonitis, mesenteric vasculitis)
- Renal masses (rarely)
Legs:
- Oedema: Periorbital (nephrotic), peripheral/pitting (nephrotic, cardiac)
- Skin: Livedo reticularis (net-like vascular pattern - antiphospholipid), vasculitic lesions, ulcers
- Joints: Knee swelling, Jaccoud's deformities
Neurological Examination:
- Cognitive assessment (MMSE, MoCA)
- Cranial nerves (palsies, optic neuritis)
- Motor: Tone, power (myositis, neuropathy), reflexes
- Sensory: Glove-and-stocking (peripheral neuropathy), sensory level (myelopathy)
- Coordination: Cerebellar signs
- Gait: Hemiparesis (CVA), ataxia
Blood Pressure:
- Hypertension (renal disease, steroids)
Special Tests:
- Romberg's test (peripheral neuropathy, posterior column)
- Straight leg raise (radiculopathy rare)
Examination Findings Summary Table
| System | Key Findings | Frequency | Clinical Significance |
|---|---|---|---|
| Skin | Malar rash | 30-50% | Pathognomonic when classic |
| Discoid lesions | 15-25% | Scarring, need sun protection | |
| Photosensitivity | 40-60% | Triggers flares | |
| MSK | Non-erosive arthritis | 70-80% | Differentiates from RA |
| Jaccoud's | 5-10% | Deformity without erosions | |
| Renal | Hypertension | 30-50% with nephritis | Indicates renal involvement |
| Oedema | 20-40% with nephritis | Nephrotic syndrome | |
| Cardiac | Pericardial rub | 10-25% | Pericarditis common |
| Murmur | 10-20% | Libman-Sacks, valve disease | |
| Resp | Pleural rub/effusion | 30-50% | Serositis |
| Crackles | 5-15% | ILD | |
| Neuro | Cognitive impairment | 20-40% | Subtle, needs testing |
| Focal signs | Variable | CVA, vasculitis |
6. Diagnosis and Classification
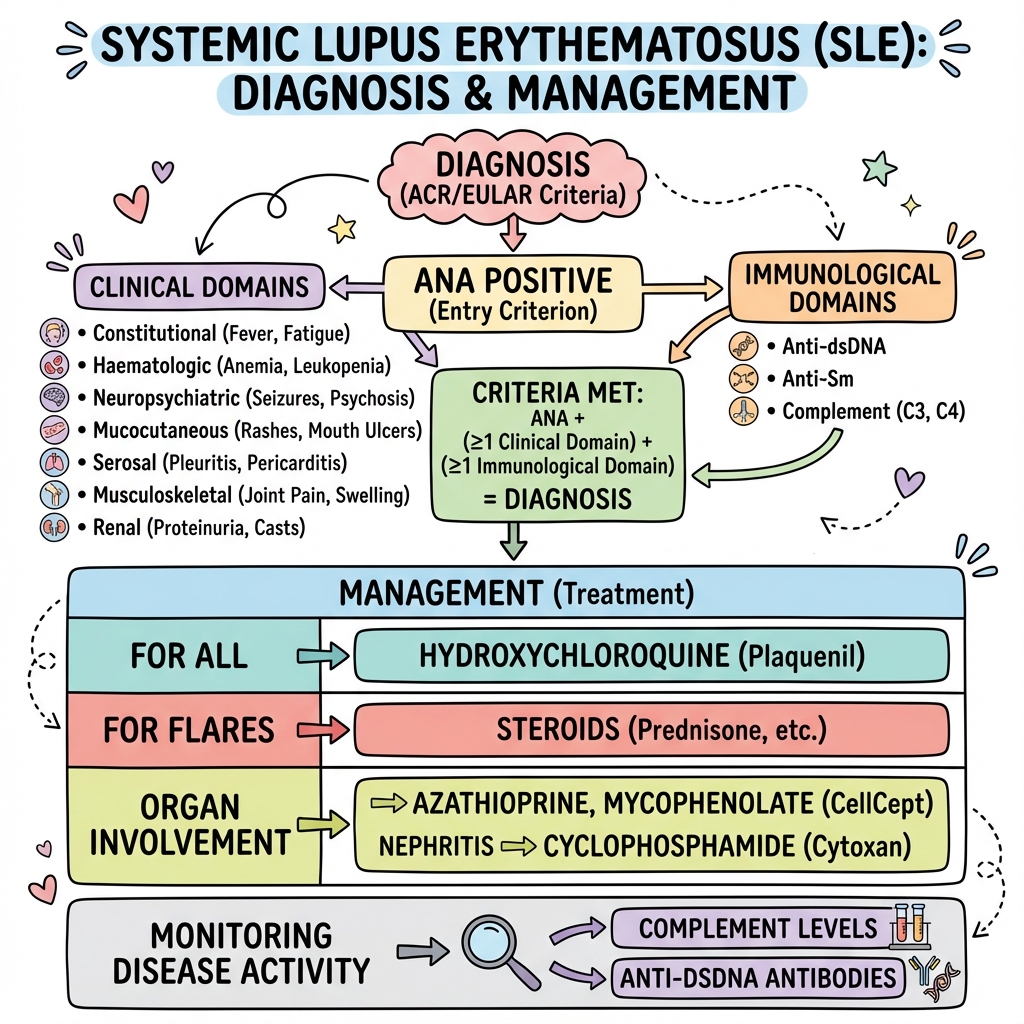
2019 ACR/EULAR Classification Criteria for SLE
The 2019 ACR/EULAR criteria represent the current gold standard for SLE classification with 96% sensitivity and 93% specificity. [5] These are classification criteria for research and clinical trials, not diagnostic criteria, but are widely used to guide diagnosis.
Entry Criterion (Mandatory):
- ANA titre ≥1:80 on HEp-2 cells or equivalent positive test
If ANA negative, patient is NOT classified as SLE (though rare ANA-negative SLE exists).
Additive Criteria (need ≥10 points for SLE classification):
Each domain scores only the highest weighted criterion within that domain:
| Domain | Criterion | Points |
|---|---|---|
| Constitutional | Fever (>38.3°C) | 2 |
| Haematological | Leucopenia (less than 4 x 10⁹/L) | 3 |
| Thrombocytopenia (less than 100 x 10⁹/L) | 4 | |
| Autoimmune haemolysis | 4 | |
| Neuropsychiatric | Delirium | 2 |
| Psychosis | 3 | |
| Seizure | 5 | |
| Mucocutaneous | Non-scarring alopecia | 2 |
| Oral ulcers | 2 | |
| Subacute cutaneous OR discoid lupus | 4 | |
| Acute cutaneous lupus (malar rash) | 6 | |
| Serosal | Pleural or pericardial effusion | 5 |
| Acute pericarditis | 6 | |
| Musculoskeletal | Joint involvement (≥2 joints with synovitis OR tenderness + ≥30min morning stiffness) | 6 |
| Renal | Proteinuria >0.5g/24h (or PCR ≥500mg/g or ≥50mg/mmol) | 4 |
| Renal biopsy Class II or V lupus nephritis | 8 | |
| Renal biopsy Class III or IV lupus nephritis | 10 | |
| Immunological | Anti-dsDNA OR Anti-Sm antibody | 6 |
| Antiphospholipid antibodies (any of: LAC, aCL IgG/IgM ≥40 units, anti-β2GPI IgG/IgM ≥40 units) | 2 | |
| Low complement (C3 OR C4 below lower limit of normal) | 3 | |
| Low complement (C3 AND C4 below lower limit of normal) | 4 |
Scoring Rules:
- Count highest criterion within each domain only
- Criteria need not be present simultaneously
- Criterion must occur at least once
- Exclude other explanations
Example Case:
- ANA 1:320 (✓ entry criterion)
- Malar rash (6 points)
- Oral ulcers (2 points, but malar rash already scored mucocutaneous domain = 6)
- Arthritis (6 points)
- Leucopenia (3 points)
- Low C3 and C4 (4 points)
- Total: 6+6+3+4 = 19 points → Classified as SLE
SLICC 2012 Classification Criteria (Alternative)
The SLICC criteria are an alternative validated system with higher sensitivity (97%) but lower specificity (84%). [6] Require either:
- ≥4 criteria (including at least 1 clinical and 1 immunological) OR
- Biopsy-proven lupus nephritis + ANA or anti-dsDNA
SLICC criteria are broader and may capture earlier/milder disease.
Differential Diagnosis
SLE mimics many conditions and must be differentiated from:
| Condition | Key Distinguishing Features | How to Differentiate |
|---|---|---|
| Drug-induced lupus | Anti-histone antibodies positive; Anti-dsDNA usually negative; Resolves with drug cessation; Common culprits: hydralazine, procainamide, isoniazid, minocycline | Medication history; Anti-histone +; Improvement after stopping drug |
| Mixed connective tissue disease (MCTD) | High titre anti-RNP antibodies; Features of SLE + SSc + PM/DM; Puffy hands, Raynaud's, myositis common | Anti-RNP (very high titre); Overlap features |
| Rheumatoid arthritis | Erosive arthritis on X-ray; RF and anti-CCP positive; Symmetric polyarthritis of small joints | Erosions on imaging; RF/anti-CCP; Less multi-system |
| Sjögren's syndrome | Dry eyes (keratoconjunctivitis sicca), dry mouth (xerostomia); Anti-Ro/La antibodies; Salivary gland biopsy | Schirmer's test; Lip biopsy; Anti-Ro/La without other features |
| Systemic sclerosis | Skin thickening (sclerodactyly), Raynaud's, ILD, PAH; Anti-Scl-70, anticentromere antibodies | Skin biopsy; Specific antibodies |
| Polymyositis/Dermatomyositis | Proximal muscle weakness, elevated CK; Heliotrope rash, Gottron's papules; Anti-Jo-1, anti-Mi-2 | Muscle biopsy; EMG; Specific antibodies |
| Viral infections | Acute onset; EBV, CMV, parvovirus B19, HIV can mimic SLE; Usually self-limited | Viral serology; Temporal course |
| Antiphospholipid syndrome (primary) | Thrombosis, pregnancy morbidity; aPL antibodies; Without other SLE features | Isolated aPL without SLE criteria |
| Vasculitis | ANCA-associated (GPA, MPA, EGPA); Henoch-Schönlein purpura; Behçet's disease | ANCA positive; Specific organ involvement patterns |
| Lymphoma | Fever, lymphadenopathy, weight loss; Lymph node biopsy diagnostic | Lymph node biopsy; Imaging |
| Sarcoidosis | Granulomas on biopsy; Hilar lymphadenopathy; Elevated ACE | Tissue biopsy; CXR pattern |
Drug-Induced Lupus Key Points:
- More common in elderly; equal sex distribution
- Develops months-years after drug exposure
- Constitutional and musculoskeletal features prominent
- Renal and CNS involvement rare
- Anti-histone antibodies in 95%; anti-dsDNA usually negative
- Resolves weeks-months after drug cessation
- Common culprits: hydralazine, procainamide, isoniazid, minocycline, TNF inhibitors (paradoxical)
Investigations
First-Line (Screening and Diagnosis):
| Investigation | Purpose | Expected Finding in SLE | Notes |
|---|---|---|---|
| ANA | Screening | Positive in 95-99% | Titre ≥1:80 on HEp-2 cells; Negative ANA makes SLE unlikely |
| ENA panel | Specificity | Anti-Sm (20-30%, highly specific); Anti-Ro/La (30-40%); Anti-RNP (25-30%) | Order if ANA positive |
| Anti-dsDNA | Specificity, activity | Positive in 60-70%; Correlates with activity and nephritis | Farr assay most specific |
| Complement C3/C4 | Disease activity | Low in active disease (consumption) | Serial monitoring useful |
| FBC | Cytopenias | Leucopenia, lymphopenia, thrombocytopenia, anaemia | Lymphopenia most specific |
| ESR | Inflammation | Elevated (often markedly) | Non-specific |
| CRP | Inflammation | Normal or mildly elevated | Discrepancy (high ESR, low CRP) typical of SLE; High CRP suggests infection |
| Urinalysis | Nephritis screening | Proteinuria, haematuria, casts | Essential at every visit |
| Urine PCR | Quantify proteinuria | Elevated (>50mg/mmol significant) | Monitors nephritis |
| Creatinine/eGFR | Renal function | May be elevated/reduced | Baseline and monitoring |
Second-Line (Specific Situations):
| Investigation | Indication | Findings | Interpretation |
|---|---|---|---|
| Antiphospholipid antibodies | Thrombosis, pregnancy loss, prolonged aPTT | LAC, aCL, anti-β2GPI positive | APS diagnosis requires positive tests on 2 occasions ≥12 weeks apart |
| Direct Coombs test | Suspected haemolysis | Positive | AIHA |
| Reticulocyte count | Anaemia | Elevated in haemolysis | Differentiates AIHA from ACD |
| LDH, haptoglobin, bilirubin | Suspected haemolysis | LDH ↑, haptoglobin ↓, indirect bilirubin ↑ | AIHA |
| Anti-ribosomal P | Neuropsychiatric symptoms | Positive in 10-20% (especially psychosis, depression) | Not routinely ordered |
| Anti-C1q | Suspected nephritis | Positive in 30-40% with active nephritis | Correlates with renal activity |
| Renal biopsy | Suspected lupus nephritis | ISN/RPS classification; Active vs chronic changes | Essential for nephritis classification and treatment planning |
| MRI brain | Neuropsychiatric symptoms | White matter lesions, infarcts, atrophy | May be normal |
| CSF analysis | CNS involvement | Pleocytosis, elevated protein | Exclude infection |
| CT chest | Pulmonary symptoms | ILD, pleuritis, pneumonitis | HRCT for ILD |
| Echocardiography | Cardiac symptoms | Pericardial effusion, valvular disease, pulmonary hypertension | Baseline recommended |
Monitoring Investigations:
- Regular (every 3-6 months in stable disease): FBC, U&E, urinalysis, anti-dsDNA, C3/C4
- Flare assessment: Repeat baseline bloods + organ-specific investigations
- Medication monitoring: LFTs (azathioprine, methotrexate, mycophenolate), TPMT (azathioprine), ophthalmology (hydroxychloroquine - annual after 5 years)
7. Management
Management Principles
SLE management aims to: [27]
- Prevent flares and maintain remission or low disease activity
- Prevent irreversible organ damage (SLICC/ACR Damage Index)
- Minimize glucocorticoid exposure (target less than 7.5mg/day prednisolone or discontinuation)
- Treat comorbidities (cardiovascular risk, osteoporosis, infection risk)
- Optimize quality of life (fatigue, pain, psychological support)
- Plan for pregnancy in women of childbearing age
Treat-to-Target Strategy:
- Define treatment target: remission or low disease activity (LLDAS)
- Use validated disease activity measures: SLEDAI-2K, BILAG
- Adjust treatment every 3 months until target reached
- Maintain target with minimal glucocorticoids
Management Algorithm
CONFIRMED SLE DIAGNOSIS
↓
┌───────────────────────────────────────────────┐
│ ALL PATIENTS: UNIVERSAL MEASURES │
│ • Hydroxychloroquine 5mg/kg/day (≤400mg) │
│ • Sun protection (SPF 50+, protective wear) │
│ • Smoking cessation │
│ • Vaccinations (pneumococcal, influenza) │
│ • CV risk management (BP, lipids, diabetes) │
│ • Bone protection (Ca/VitD, ± bisphosphonate)│
│ • Regular monitoring (bloods, urine) │
└───────────────────────────────────────────────┘
↓
┌───────────────────────────────────────────────┐
│ ASSESS DISEASE SEVERITY │
└───────────────────────────────────────────────┘
↓ ↓ ↓
┌─────────────┐ ┌────────────────┐ ┌─────────────────┐
│ MILD │ │ MODERATE │ │ SEVERE │
│ (skin, │ │ (serositis, │ │ (nephritis, │
│ joints) │ │ arthritis) │ │ CNS, severe │
│ │ │ │ │ haematologic) │
└─────────────┘ └────────────────┘ └─────────────────┘
↓ ↓ ↓
• HCQ • HCQ • HCQ
• NSAIDs • Pred 0.5mg/kg • IV methylpred
• Topical • Taper to less than 7.5mg 500-1000mg x3d
steroids • Add DMARD: • Then oral pred
• Low-dose - Azathioprine 0.5-1mg/kg
pred - Methotrexate • Immunosuppression:
(5-15mg) - Mycophenolate - MMF 2-3g/day
- Cyclophosphamide
• Consider biologics:
- Belimumab
- Anifrolumab
- Voclosporin (LN)
↓
┌─────────────────────────┐
│ REFRACTORY DISEASE │
│ • Rituximab │
│ • Belimumab │
│ • Anifrolumab │
│ • Voclosporin │
│ • Clinical trials │
│ • Combination therapy │
└─────────────────────────┘
Universal Measures (ALL Patients)
| Intervention | Details | Evidence/Rationale |
|---|---|---|
| Hydroxychloroquine | 5mg/kg actual body weight/day (maximum 400mg/day, typically 200-400mg) | Reduces flares 50%, prevents damage, reduces mortality 70%, safe in pregnancy [7,8] |
| Sun protection | SPF 50+ broad-spectrum sunscreen; Protective clothing; Avoid midday sun (10am-4pm) | UV light triggers flares; Photosensitivity in 40-60% |
| Smoking cessation | Complete cessation; Nicotine replacement if needed | Smoking worsens disease activity, cardiovascular risk, reduces HCQ efficacy |
| Vaccinations | Pneumococcal (PCV13 + PPSV23); Annual influenza; Avoid live vaccines if on immunosuppression | Infection major cause of morbidity/mortality |
| CV risk management | Aggressive BP control (less than 130/80); Statin for dyslipidaemia; Diabetes management | CV disease is leading cause of late mortality; 2-10x increased risk [14] |
| Bone protection | Calcium 1000-1500mg/day + Vitamin D 800-1000 IU/day; Bisphosphonate if on steroids ≥7.5mg >3 months or T-score < -1.5 | Steroids cause osteoporosis |
| Education | Disease education; Recognize flare symptoms; Medication adherence | Empowers patients; Improves outcomes |
| Fertility counseling | Plan pregnancies during remission; Discuss teratogenic medications | Optimize maternal and fetal outcomes [18] |
Hydroxychloroquine - The Foundation
Dosing:
- 5mg/kg actual body weight per day (NOT ideal body weight)
- Maximum 400mg/day (typically 200mg daily or 200mg BD)
- Continue lifelong even in remission
Benefits: [7,8]
- Reduces disease flares by 50%
- Prevents irreversible organ damage (SLICC Damage Index)
- Reduces mortality by up to 70%
- Reduces thrombosis risk (antithrombotic effect)
- Improves lipid profile (lowers cholesterol)
- Improves glycaemic control
- Safe in pregnancy and breastfeeding
Monitoring:
- Baseline ophthalmology examination (fundoscopy, visual fields, OCT)
- Annual screening after 5 years of use (or earlier if risk factors: renal/hepatic impairment, age >60, dose >5mg/kg, pre-existing retinopathy)
- Risk of retinopathy less than 1% with proper dosing; increases with cumulative dose >1000g
Toxicity:
- Retinopathy: Bull's eye maculopathy; irreversible if severe; detected on OCT
- QTc prolongation: Check baseline ECG if cardiac risk factors
- Haematological: Rare
- GI upset: Take with food
Contraindications:
- Known hypersensitivity
- Pre-existing maculopathy
- Relative: G6PD deficiency (haemolysis risk, but usually tolerated)
Glucocorticoids
Glucocorticoids are highly effective but cause cumulative organ damage. [19] Minimize exposure.
| Situation | Dose | Duration | Notes |
|---|---|---|---|
| Mild flare | Prednisolone 5-15mg/day PO | Taper rapidly over weeks | Add/optimize DMARD |
| Moderate flare | Prednisolone 0.5mg/kg/day PO | Taper to less than 7.5mg within 3 months | Add steroid-sparing agent |
| Severe flare/organ-threatening | IV methylprednisolone 500-1000mg daily x 3 days → Prednisolone 0.5-1mg/kg PO | Taper to less than 7.5mg by 6 months | Always add potent immunosuppressive (MMF, CYC) |
| Maintenance | Prednisolone less than 7.5mg/day OR discontinue | Long-term | Use steroid-sparing agents to enable taper |
Adverse Effects of Prolonged Glucocorticoids:
- Infection risk (dose-dependent)
- Avascular necrosis
- Osteoporosis and fractures
- Cataracts, glaucoma
- Hyperglycaemia/diabetes
- Hypertension
- Dyslipidaemia, cardiovascular disease
- Weight gain, cushingoid features
- Mood disturbance (depression, mania, psychosis)
- Myopathy
- Skin thinning, bruising, striae
Bone Protection:
- All patients on prednisolone ≥7.5mg/day for >3 months require:
- Calcium 1000-1500mg/day + Vitamin D 800-1000 IU/day
- Bisphosphonate (alendronate 70mg weekly or risedronate 35mg weekly)
- DEXA scan baseline and every 1-2 years
Immunosuppressive Agents (DMARDs)
| Drug | Indication | Dose | Mechanism | Monitoring | Key Points |
|---|---|---|---|---|---|
| Azathioprine | Steroid-sparing; Maintenance therapy for nephritis | 2-3mg/kg/day PO (100-200mg) | Purine synthesis inhibitor | FBC (leucopenia), LFTs; TPMT testing before initiation | Safe in pregnancy; Risk of malignancy long-term |
| Mycophenolate mofetil (MMF) | Lupus nephritis (induction and maintenance); Severe SLE | 2-3g/day PO (divided BD) | Inosine monophosphate dehydrogenase inhibitor; Blocks lymphocyte proliferation | FBC, LFTs | Teratogenic (avoid pregnancy); More GI side effects than azathioprine |
| Methotrexate | Skin and joint disease; Steroid-sparing | 10-25mg once weekly PO/SC + folic acid 5mg 24h later | Folate antagonist; Anti-inflammatory | FBC, LFTs, creatinine; CXR baseline (pneumonitis risk) | Teratogenic; Avoid alcohol; Risk of hepatotoxicity, pneumonitis |
| Cyclophosphamide | Severe nephritis (Class III/IV); CNS lupus; Severe haematological | Euro-Lupus: 500mg IV every 2 weeks x 6 doses (total 3g) OR NIH: 500-1000mg/m² IV monthly x 6 months | Alkylating agent; Lymphocyte depletion | FBC (nadir 10-14 days); Urinalysis (haemorrhagic cystitis) | Teratogenic; Gonadotoxic (ovarian failure, infertility); Mesna co-administration; Risk of malignancy; Consider fertility preservation |
| Tacrolimus/Ciclosporin | Lupus nephritis (especially Class V); Refractory | Tacrolimus 0.05mg/kg/day; Ciclosporin 2-3mg/kg/day | Calcineurin inhibitor; T cell suppression | Drug levels, creatinine, BP | Nephrotoxic; Hypertension |
| Voclosporin | Lupus nephritis (with MMF) | 23.7mg BD PO | Calcineurin inhibitor | Creatinine, BP | Approved 2021; AURORA trial [11] |
Choice of Immunosuppressive Agent:
- Lupus nephritis (Class III/IV): MMF or cyclophosphamide for induction; MMF or azathioprine for maintenance
- CNS lupus: Cyclophosphamide + high-dose steroids
- Skin/joint predominant: Methotrexate
- General steroid-sparing: Azathioprine (safe in pregnancy), methotrexate (if not childbearing age)
Lupus Nephritis Management
Lupus nephritis (LN) requires aggressive treatment to prevent progression to ESRD. [17]
Induction Therapy (Class III or IV Nephritis) - Duration 6 months:
Option 1: Mycophenolate Mofetil (PREFERRED)
- MMF 2-3g/day PO (divided BD)
- PLUS glucocorticoids: IV methylprednisolone 500-1000mg x 3 days → prednisolone 0.5-1mg/kg/day → taper to ≤7.5mg by 6 months
Option 2: Cyclophosphamide
- Euro-Lupus regimen (PREFERRED for Caucasians, less gonadotoxic): 500mg IV every 2 weeks x 6 doses (total 3g over 12 weeks)
- NIH regimen (for African/Hispanic descent, severe disease): 500-1000mg/m² IV monthly x 6 months
- PLUS glucocorticoids as above
Adjunctive Therapy:
- Belimumab: Consider adding to standard therapy; BLISS-LN trial showed benefit [9]
- Voclosporin: AURORA trial demonstrated superior renal response (40.8% vs 22.5%) when added to MMF [11]
Maintenance Therapy (Following induction) - Duration ≥3 years:
- MMF 1-2g/day OR Azathioprine 2mg/kg/day
- Prednisolone ≤7.5mg/day (aim to discontinue if possible)
- Continue hydroxychloroquine
- Monitor: Urine PCR, creatinine, C3/C4, anti-dsDNA every 3 months
Class V (Membranous) Nephritis:
- If nephrotic-range proteinuria or declining renal function: Treat as Class III/IV
- If non-nephrotic: Conservative management + HCQ + ACE inhibitor/ARB
Refractory Nephritis:
- Switch from MMF to cyclophosphamide (or vice versa)
- Rituximab (anti-CD20): 1000mg IV x 2 doses 2 weeks apart
- Consider voclosporin, belimumab
- Plasmapheresis (limited evidence)
Renal Response Definitions:
- Complete renal response: Urine PCR less than 50mg/mmol + normal/near-normal renal function + inactive sediment
- Partial renal response: ≥50% reduction in proteinuria + stable renal function
Target: Complete renal response by 12 months
Biologic Therapies
| Drug | Mechanism | Indication | Dose | Evidence | Key Points |
|---|---|---|---|---|---|
| Belimumab | Anti-BAFF/BLyS monoclonal antibody; Reduces B cell survival | Active SLE despite standard therapy; Lupus nephritis (adjunct) | 10mg/kg IV every 2 weeks x 3, then every 4 weeks OR 200mg SC weekly | BLISS-52, BLISS-76, BLISS-LN [9] | First biologic approved for SLE (2011); Reduces flares, steroid use; Takes 6 months for full effect |
| Anifrolumab | Anti-type I interferon receptor antibody | Moderate-severe SLE (skin, joints, serositis) | 300mg IV every 4 weeks | TULIP-2 [10] | Approved 2021; Efficacy in high IFN signature patients; Reduces BILAG A/B flares, skin disease; Herpes zoster risk (vaccinate) |
| Rituximab | Anti-CD20 monoclonal antibody; B cell depletion | Refractory SLE (off-label); Lupus nephritis; Severe haematological | 1000mg IV x 2 doses 2 weeks apart OR 375mg/m² weekly x 4 | EXPLORER, LUNAR (neutral), but clinical experience positive | Not FDA-approved for SLE but widely used; Effective in refractory disease; Monitor for infusion reactions, PML (rare) |
| Voclosporin | Calcineurin inhibitor | Lupus nephritis (Class III-V) with MMF | 23.7mg BD PO | AURORA-1 [11] | Approved 2021; Superior complete renal response vs MMF alone (40.8% vs 22.5%); Monitor creatinine, BP |
When to Consider Biologics:
- Refractory disease despite standard therapy (HCQ + steroid + DMARD)
- Inability to taper steroids below 7.5mg/day
- Frequent relapses
- Severe organ involvement (nephritis, CNS)
- Contraindications/intolerance to conventional DMARDs
Specific Manifestations Management
| Manifestation | Treatment | Notes |
|---|---|---|
| Cutaneous lupus | Topical corticosteroids; HCQ; Sun protection; Methotrexate if refractory | Discoid lupus may need intralesional steroids |
| Arthritis | NSAIDs; HCQ; Low-dose prednisolone; Methotrexate if refractory | Usually non-erosive |
| Serositis | NSAIDs; Prednisolone 0.5mg/kg; Colchicine (pericarditis) | Large effusions may need drainage |
| Haematological (AIHA, ITP) | Prednisolone 1mg/kg; IVIG (ITP); Rituximab if refractory | Severe thrombocytopenia may need platelet transfusion |
| Neuropsychiatric | High-dose steroids; Cyclophosphamide; Exclude infection/metabolic | Anticonvulsants for seizures; Antipsychotics for psychosis |
| APS (thrombosis) | Anticoagulation: Warfarin (INR 2-3 for venous, 3-4 for arterial) or DOAC | Aspirin if aPL+ without thrombosis |
| Pulmonary haemorrhage | IV methylprednisolone 1g x 3d; Cyclophosphamide; Plasmapheresis; ITU | Life-threatening; high mortality |
Pregnancy in SLE
Pre-Conception Counseling: [18]
- Plan pregnancy during disease quiescence (≥6 months remission)
- Screen for anti-Ro/SSA (congenital heart block risk), antiphospholipid antibodies (pregnancy morbidity)
- Discuss medication safety
Safe Medications in Pregnancy:
- ✅ Hydroxychloroquine (continue throughout)
- ✅ Azathioprine
- ✅ Prednisolone (avoid high-dose first trimester if possible)
- ✅ Tacrolimus
- ✅ Aspirin 75-150mg (if aPL+)
- ❌ Mycophenolate (teratogenic - stop 6 months before conception)
- ❌ Cyclophosphamide (teratogenic, gonadotoxic)
- ❌ Methotrexate (teratogenic - stop 3 months before conception)
- ⚠️ Warfarin (teratogenic first trimester - switch to LMWH)
Monitoring During Pregnancy:
- Joint rheumatology-obstetrics care
- Monthly clinical assessment
- Urine PCR, FBC, C3/C4, anti-dsDNA each trimester
- Fetal echocardiography 18-24 weeks if anti-Ro/SSA positive (congenital heart block)
- Low-dose aspirin if antiphospholipid antibodies
Flare Management:
- 30% of patients flare during pregnancy
- Prednisolone preferred (does not cross placenta significantly)
- Azathioprine if needed
Post-Partum:
- High flare risk in first 3 months post-partum
- Resume pre-pregnancy medications if not breastfeeding
- Hydroxychloroquine, azathioprine, prednisolone compatible with breastfeeding
Monitoring and Follow-Up
Stable Disease (Remission/Low Activity):
- Clinical review every 3-6 months
- Bloods: FBC, U&E, LFTs, anti-dsDNA, C3/C4, urinalysis every 3-6 months
- Annual ophthalmology after 5 years HCQ
- DEXA scan if on steroids
Active Disease/Flare:
- Clinical review monthly (or more frequent)
- Bloods: FBC, U&E, anti-dsDNA, C3/C4, urinalysis monthly
- Organ-specific investigations as needed
Disease Activity Measures:
- SLEDAI-2K (SLE Disease Activity Index): 0-105 score; ≥6 = active disease
- BILAG (British Isles Lupus Assessment Group): Grades activity by organ system (A-E)
- LLDAS (Lupus Low Disease Activity State): Composite measure; target for treat-to-target
Damage Assessment:
- SLICC/ACR Damage Index: Quantifies irreversible organ damage; Predicts mortality [21]
8. Complications
| Complication | Incidence | Mechanism | Prevention | Management |
|---|---|---|---|---|
| End-stage renal disease | 10-20% of LN patients | Untreated/refractory nephritis | Early aggressive treatment of nephritis | Dialysis, transplant (good outcomes) |
| Cerebrovascular disease | 10-15% | Accelerated atherosclerosis, antiphospholipid antibodies, vasculitis | CV risk management; Anticoagulation if aPL+ | Aspirin/anticoagulation; Immunosuppression if vasculitis |
| Coronary artery disease | 2-10x general population | Accelerated atherosclerosis (inflammation, type I IFN, dyslipidaemia, hypertension, steroids) | Aggressive CV risk control; Minimize steroids | Statins, antihypertensives, revascularization if needed |
| Avascular necrosis | 5-15% | Steroids, antiphospholipid antibodies, vasculopathy | Minimize steroids; Avoid high-dose pulses if possible | Analgesia; Joint replacement |
| Infections | Leading cause early mortality | Immunosuppression, complement deficiency, neutropenia | Vaccinations; Prophylaxis (PCP if on CYC); Early recognition | Aggressive antibiotic therapy; Low threshold for admission |
| Malignancy | 1.5-2x general population | Immunosuppression (especially cyclophosphamide), chronic inflammation | Minimize cyclophosphamide exposure; Cancer screening | Standard oncological management |
| Osteoporosis/fractures | 25% (steroid-induced) | Glucocorticoids | Bone protection (Ca/VitD, bisphosphonate) | Bisphosphonates, denosumab, teriparatide |
| Pregnancy complications | 20-30% | Disease activity, antiphospholipid antibodies, anti-Ro | Plan pregnancy during remission; Aspirin if aPL+ | Joint rheumatology-obstetrics care |
| Antiphospholipid syndrome | 30-40% have aPL; ~50% of those develop thrombosis | Antiphospholipid antibodies | Aspirin if aPL+ without thrombosis | Anticoagulation (warfarin INR 2-3) |
| Pulmonary hypertension | 5-10% | Vasculopathy, chronic thromboembolism, ILD | Early detection (echocardiography) | Pulmonary vasodilators, anticoagulation |
| Accelerated atherosclerosis | 50% subclinical disease | Chronic inflammation, type I interferons, dyslipidaemia, hypertension | Aggressive CV risk management | Statins, antihypertensives, aspirin |
9. Prognosis and Outcomes
Survival
Modern treatment has dramatically improved SLE prognosis: [12]
- 5-year survival: >95% (vs less than 50% in 1950s)
- 10-year survival: >90% (vs less than 70% in 1970s)
- 20-year survival: 75-85%
Causes of Death
Early (less than 5 years):
- Active SLE (multi-organ involvement)
- Infection (most common early cause)
- Lupus nephritis
- Neuropsychiatric disease
Late (>5 years):
- Cardiovascular disease (myocardial infarction, stroke) - NOW LEADING CAUSE [14]
- Infection
- Malignancy
- End-stage renal disease
Prognostic Factors
Poor Prognosis:
- Lupus nephritis (especially Class IV diffuse proliferative)
- Neuropsychiatric involvement
- Antiphospholipid syndrome with thrombosis
- African, Asian, or Hispanic ethnicity
- Low socioeconomic status
- Male sex
- Childhood-onset (less than 18 years)
- High disease activity at presentation
- Delayed diagnosis
- Poor medication adherence
Good Prognosis:
- Limited disease (skin, joints only)
- Good response to initial therapy
- Adherence to hydroxychloroquine
- European ethnicity
- Female sex
- Adult-onset
- Low cumulative organ damage
Disease Course
- Relapsing-remitting: Most common pattern; flares interspersed with remissions
- Chronic active: Persistent disease activity despite treatment
- Long quiescent: Prolonged remission (rare)
Organ Damage
SLICC/ACR Damage Index: [21]
- Quantifies irreversible damage across 12 organ systems
- Each item scored once after being present for ≥6 months
- Examples: Cognitive impairment, seizures, stroke, myocardial infarction, heart failure, pulmonary hypertension, eGFR less than 50%, proteinuria >3.5g/24h, ESRD, AVN, osteoporotic fracture, cataracts, retinopathy
- Higher damage scores predict mortality and morbidity
- Damage accrual is partly preventable with tight disease control and steroid minimization
Key Concept for Exams:
- Disease activity (SLEDAI, BILAG) is reversible with treatment
- Organ damage (SLICC/ACR Damage Index) is irreversible
- Goal: Control activity to prevent damage
10. Evidence and Guidelines
Key Guidelines
-
2019 EULAR Recommendations for SLE Management — Fanouriakis A et al. Ann Rheum Dis. 2019;78(6):736-745. PMID: 30926722 [1]
- Comprehensive European guidelines covering all aspects of SLE management
- Emphasizes hydroxychloroquine for all, minimizing glucocorticoids, treat-to-target
-
2019 ACR/EULAR Classification Criteria for SLE — Aringer M et al. Ann Rheum Dis. 2019;78(9):1151-1159. PMID: 31383717 [5]
- Current gold standard classification criteria with 96% sensitivity, 93% specificity
- ANA ≥1:80 entry criterion + weighted domains ≥10 points
-
SLICC 2012 Classification Criteria for SLE — Petri M et al. Arthritis Rheum. 2012;64(8):2677-2686. PMID: 22553077 [6]
- Alternative validated criteria; higher sensitivity (97%), lower specificity (84%)
-
2020 ACR Guideline for Lupus Nephritis — Hahn BH et al. Arthritis Care Res. 2012;64(6):797-808. PMID: 22556106 [17]
- Detailed recommendations for lupus nephritis management
-
EULAR/ACR Classification Criteria for Antiphospholipid Syndrome — Barbhaiya M et al. Arthritis Rheumatol. 2023;75(10):1687-1702. PMID: 37596173
- Updated APS criteria
Landmark Trials and Evidence
BLISS-52 and BLISS-76 (Belimumab) [9]
- Population: Active SLE (SELENA-SLEDAI ≥6) despite standard therapy
- Intervention: Belimumab 10mg/kg IV vs placebo (+ standard therapy)
- Primary outcome: SRI-4 response at 52/76 weeks
- Results: BLISS-52: 58% vs 44% (p=0.0006); BLISS-76: 43% vs 34% (p=0.017)
- Impact: First biologic approved for SLE (2011); Established BAFF/BLyS as therapeutic target
- PMID: 21296403
BLISS-LN (Belimumab for Lupus Nephritis)
- Population: Active lupus nephritis (Class III, IV, or V)
- Intervention: Belimumab + standard therapy (MMF/CYC) vs placebo + standard therapy
- Results: Higher complete renal response with belimumab (43% vs 32% at 104 weeks)
- Impact: Belimumab approved as adjunctive therapy for lupus nephritis
- PMID: 33631841
TULIP-2 (Anifrolumab) [10]
- Population: Moderate-severe SLE
- Intervention: Anifrolumab 300mg IV every 4 weeks vs placebo
- Primary outcome: BICLA response at 52 weeks
- Results: 47.8% vs 31.5% (pless than 0.001); Particular efficacy in skin disease
- Impact: Validated type I interferon inhibition; Approved 2021
- PMID: 31851795
AURORA-1 (Voclosporin for Lupus Nephritis) [11]
- Population: Active lupus nephritis (Class III, IV, V, or III+V, IV+V)
- Intervention: Voclosporin 23.7mg BD + MMF vs placebo + MMF
- Primary outcome: Complete renal response at 52 weeks
- Results: 40.8% vs 22.5% (pless than 0.001)
- Impact: Voclosporin approved for lupus nephritis 2021
- PMID: 33497604
ALMS (Mycophenolate vs Cyclophosphamide for Lupus Nephritis)
- Population: Lupus nephritis (Class III, IV, V, or III+V, IV+V)
- Intervention: MMF vs IV cyclophosphamide (NIH protocol) for induction
- Results: MMF non-inferior to CYC for inducing renal response
- Impact: MMF became preferred first-line due to better side effect profile (less gonadotoxicity)
- PMID: 19369404
Canadian Hydroxychloroquine Study Group [7]
- Findings: HCQ reduces SLE flares by 50%; Discontinuation increases flare risk 3-fold
- Impact: Established HCQ as cornerstone therapy for all SLE patients
- PMID: 1991513
Hydroxychloroquine and Survival in SLE [8]
- Findings: HCQ use associated with 70% reduction in mortality
- Impact: Reinforced universal HCQ recommendation
- PMID: 19103632
11. Patient Explanation (Layperson Level)
What is Systemic Lupus Erythematosus?
Lupus is a condition where your immune system, which normally protects you from infections, becomes overactive and mistakenly attacks your own body's tissues. This is called an "autoimmune" disease. Lupus can affect many parts of your body including your skin, joints, kidneys, heart, lungs, brain, and blood cells. The disease comes and goes in episodes called "flares."
What Causes Lupus?
We don't know exactly what causes lupus, but it involves a combination of:
- Your genes (it can run in families)
- Hormones (which is why it's more common in women)
- Environmental triggers like sunlight, infections, or certain medications
Lupus is not contagious - you cannot catch it from someone else.
What Are the Symptoms?
Lupus affects everyone differently, but common symptoms include:
- Extreme tiredness (fatigue)
- Joint pain and swelling
- Skin rashes, especially a "butterfly" rash across the cheeks and nose
- Sensitivity to sunlight
- Fever
- Hair loss
- Mouth sores
Some people have serious problems with their kidneys, heart, lungs, or brain that need urgent treatment.
How is Lupus Diagnosed?
Your doctor will:
- Ask about your symptoms
- Examine you
- Order blood tests (especially an "ANA" test and other antibody tests)
- Check your urine for signs of kidney problems
There's no single test for lupus - diagnosis is based on a combination of symptoms and test results.
How is Lupus Treated?
Medication You'll Take Every Day:
- Hydroxychloroquine (also called Plaquenil): Nearly everyone with lupus takes this medication. It prevents flares, protects your organs, and is safe even during pregnancy. You'll need eye checks every year after 5 years of use.
Medications for Flares:
- Steroids (like prednisolone): Very effective for controlling active lupus, but we try to use the lowest dose possible because long-term use can cause side effects like weak bones, weight gain, and increased infection risk.
- Other immunosuppressive medications: These help control your immune system and allow us to reduce your steroid dose. Examples include azathioprine, mycophenolate, and methotrexate.
Newer Treatments:
- Some people need specialized medications called "biologics" (like belimumab or anifrolumab) if standard treatments aren't working well enough.
Living with Lupus
Things You Can Do:
- Protect yourself from the sun: Use SPF 50+ sunscreen every day, wear hats and long sleeves, and avoid the midday sun. Sunlight can trigger lupus flares.
- Don't smoke: Smoking makes lupus worse and reduces the effectiveness of your medications.
- Take your medications every day: Especially hydroxychloroquine - don't stop even when you feel well.
- Get vaccinated: Flu and pneumonia vaccines are important because you're at higher risk of infections.
- Eat a healthy diet and stay as active as you can.
- Get enough rest: Fatigue is common, so pace yourself.
Planning for Pregnancy:
- If you're thinking about having a baby, talk to your doctor first
- We need to make sure your lupus is well-controlled and switch any medications that aren't safe in pregnancy
- Most women with lupus can have healthy pregnancies with careful planning and monitoring
Warning Signs to Report Immediately
Contact your doctor urgently if you develop:
- New or worsening rash
- Swelling in your legs, feet, or face
- Blood in your urine or foamy urine
- Chest pain or difficulty breathing
- Severe headaches
- Confusion, seizures, or severe mood changes
- High fever
What's the Outlook?
With modern treatment, most people with lupus live long, full lives. The key is:
- Regular monitoring and blood tests
- Taking your medications as prescribed
- Recognizing and reporting flare symptoms early
- Looking after your overall health
We'll work together as a team to keep your lupus under control and prevent complications.
12. Examination Focus (Viva and Clinical Exam Preparation)
Opening Statement (Viva)
"Systemic lupus erythematosus is a chronic, multisystem autoimmune disease characterised by loss of immune tolerance to nuclear antigens, production of pathogenic autoantibodies - particularly anti-dsDNA and anti-Sm - immune complex deposition, and complement-mediated inflammation affecting virtually any organ system. It predominantly affects women of childbearing age with a 9:1 female-to-male ratio and shows marked ethnic variation with higher incidence in African, Asian, and Hispanic populations. Classification is based on the 2019 ACR/EULAR criteria requiring ANA positivity as entry criterion followed by weighted clinical and immunological domains totalling 10 or more points. Hydroxychloroquine is the cornerstone of therapy for all patients, and lupus nephritis is treated with mycophenolate or cyclophosphamide. Modern biologics including belimumab, anifrolumab, and voclosporin provide additional therapeutic options. With current treatment, 10-year survival exceeds 90%."
Key Viva Points to Memorize
Classification (2019 ACR/EULAR):
- Entry: ANA ≥1:80 (mandatory)
- Need ≥10 points across weighted domains
- Highest scorers: Acute cutaneous lupus (6), joint involvement (6), pericarditis (6), anti-dsDNA/anti-Sm (6), Class III/IV nephritis (10)
Autoantibodies:
- ANA: 95-99% sensitive, low specificity
- Anti-dsDNA: 60-70%, highly specific, activity marker, nephritis
- Anti-Sm: 20-30%, pathognomonic (>98% specific), no activity correlation
- Anti-Ro/SSA: 30-40%, photosensitivity, neonatal lupus, congenital heart block
ISN/RPS Lupus Nephritis Classification:
- Class I: Minimal mesangial (no treatment)
- Class II: Mesangial proliferative (HCQ ± low-dose steroids)
- Class III: Focal proliferative less than 50% glomeruli (immunosuppression required)
- Class IV: Diffuse proliferative ≥50% glomeruli (aggressive immunosuppression)
- Class V: Membranous (treat if nephrotic or declining function)
- Class VI: Advanced sclerosing (ESRD, limited response)
Management Essentials:
- ALL patients: HCQ 5mg/kg/day (≤400mg)
- Lupus nephritis induction: MMF 2-3g/day OR cyclophosphamide (Euro-Lupus 500mg q2wk x6)
- Maintenance: MMF or azathioprine for ≥3 years
- Target: Prednisolone less than 7.5mg/day or discontinue
- Biologics: Belimumab (anti-BAFF), anifrolumab (anti-IFN receptor), voclosporin (calcineurin inhibitor for LN)
Prognostic Factors:
- Poor: Nephritis (especially Class IV), NPSLE, APS, African/Asian/Hispanic ethnicity, male sex, childhood onset
- Good: Limited disease, good initial response, HCQ adherence
Common Viva Questions and Model Answers
Q1: "How would you diagnose SLE in a 28-year-old woman presenting with arthralgia, malar rash, and fatigue?"
A: "I would approach this systematically. First, I would take a detailed history including onset, pattern of symptoms, photosensitivity, systemic features, and medication history to exclude drug-induced lupus. Examination would include looking for malar rash distribution - specifically whether it spares the nasolabial folds - joint swelling, oral ulcers, and signs of organ involvement.
My investigations would start with ANA as a screening test - the 2019 ACR/EULAR criteria require ANA positivity at titre 1:80 or higher as an entry criterion. If ANA is positive, I would send an ENA panel including anti-dsDNA and anti-Sm for specificity, complement levels C3 and C4, full blood count looking for cytopenias particularly lymphopenia, ESR and CRP - noting that discordance with high ESR but low CRP is typical of SLE - and urinalysis with protein:creatinine ratio to screen for nephritis.
For classification, I would apply the 2019 ACR/EULAR criteria: the malar rash scores 6 points, if she has inflammatory arthritis that would score 6 points, and depending on other features like cytopenias, low complement, or positive anti-dsDNA she could easily exceed the threshold of 10 points for SLE classification. However, I would also exclude important differentials including rheumatoid arthritis, mixed connective tissue disease, and drug-induced lupus."
Q2: "A patient with known SLE presents with new-onset proteinuria. How would you manage this?"
A: "New proteinuria in an SLE patient is lupus nephritis until proven otherwise and requires urgent assessment. I would quantify the proteinuria using a spot urine protein:creatinine ratio, check renal function with U&Es, examine the urinary sediment for dysmorphic red cells and casts which indicate active nephritis, and check disease activity markers including anti-dsDNA and complement C3/C4.
I would refer urgently to nephrology for renal biopsy, which is essential for ISN/RPS classification to guide treatment intensity. The classification ranges from Class I minimal mesangial requiring no treatment, through Class III focal proliferative and Class IV diffuse proliferative which require aggressive immunosuppression, to Class V membranous and Class VI advanced sclerosing.
If biopsy confirms Class III or IV nephritis, I would initiate induction therapy with either mycophenolate mofetil 2-3 grams daily or cyclophosphamide using the Euro-Lupus regimen of 500mg every 2 weeks for 6 doses, combined with glucocorticoids - typically IV methylprednisolone pulses followed by oral prednisolone with a taper to under 7.5mg daily by 6 months. I would also consider adding belimumab based on the BLISS-LN trial data, or voclosporin which showed superior complete renal response in the AURORA trial.
After 6 months induction, I would switch to maintenance therapy with mycophenolate or azathioprine for at least 3 years, ensuring the patient remains on hydroxychloroquine. I would monitor closely with urine protein:creatinine ratio, renal function, and serology every 3 months, aiming for complete renal response - that is PCR under 50mg/mmol with normal renal function - by 12 months."
Q3: "What is the mechanism of action of belimumab and what is the evidence for its use in SLE?"
A: "Belimumab is a fully human monoclonal antibody that targets B-cell activating factor, also called BAFF or BLyS. BAFF is a cytokine elevated in SLE patients that promotes B cell survival, maturation, and differentiation into antibody-producing plasma cells. By blocking BAFF, belimumab reduces autoreactive B cell survival and autoantibody production.
The evidence comes from the BLISS-52 and BLISS-76 trials published in 2011, which demonstrated that belimumab 10mg/kg intravenous every 4 weeks, added to standard therapy, significantly improved the SRI-4 response rate compared to placebo - 58% versus 44% in BLISS-52. This led to belimumab becoming the first biologic approved specifically for SLE.
More recently, the BLISS-LN trial demonstrated benefit when belimumab is added to standard therapy in lupus nephritis, showing higher complete renal response rates at 104 weeks - 43% versus 32%. Belimumab is now indicated for active SLE despite standard therapy, and as adjunctive treatment for lupus nephritis. The main limitation is that it takes approximately 6 months to see full clinical effect, so it's not suitable for acute severe disease. The evidence supports its use particularly in patients with serologically active disease - high anti-dsDNA and low complement - and frequent relapses requiring steroids."
Q4: "Why is hydroxychloroquine considered essential for all SLE patients?"
A: "Hydroxychloroquine has multiple beneficial mechanisms and is supported by robust long-term data. The Canadian Hydroxychloroquine Study Group demonstrated that hydroxychloroquine reduces SLE flares by 50%, and discontinuation leads to a 3-fold increased flare risk. Subsequent studies have shown that hydroxychloroquine prevents irreversible organ damage accumulation as measured by the SLICC Damage Index and reduces mortality by up to 70%.
Beyond its immunomodulatory effects, hydroxychloroquine has important ancillary benefits: it has antithrombotic properties reducing thrombosis risk particularly important in patients with antiphospholipid antibodies, improves lipid profiles by lowering cholesterol, improves glycaemic control reducing diabetes risk, and most importantly is safe in pregnancy - in fact we actively continue it throughout pregnancy.
The dosing is weight-based at 5mg/kg actual body weight per day with a maximum of 400mg daily. The main toxicity concern is retinopathy, but this is rare - less than 1% - with proper dosing. We perform baseline ophthalmology and then annual screening after 5 years of use, or earlier if risk factors such as renal impairment, age over 60, or cumulative dose exceeding 1000 grams.
Given this favorable benefit-to-risk ratio and robust evidence base, current EULAR guidelines recommend hydroxychloroquine for all SLE patients unless contraindicated, even those in remission."
Q5: "How would you differentiate drug-induced lupus from idiopathic SLE?"
A: "Drug-induced lupus shares some features with idiopathic SLE but has important distinguishing characteristics. First, there must be temporal association with medication exposure - common culprits include hydralazine, procainamide, isoniazid, and minocycline, with TNF inhibitors causing paradoxical drug-induced lupus. Drug-induced lupus typically develops months to years after drug initiation.
Clinically, drug-induced lupus presents predominantly with constitutional symptoms, arthralgia, myalgia, and serositis - particularly pleuritis and pericarditis. Crucially, severe manifestations like lupus nephritis and neuropsychiatric involvement are extremely rare in drug-induced lupus, which helps differentiate it.
Serologically, ANA is positive in both, but the antibody profile differs significantly. Drug-induced lupus characteristically shows anti-histone antibodies positive in over 95% of cases, whereas anti-dsDNA antibodies - which are hallmark of idiopathic SLE and correlate with nephritis - are usually negative in drug-induced lupus. Anti-Sm antibodies, which are pathognomonic for SLE, are also typically absent.
Perhaps most importantly, drug-induced lupus resolves with cessation of the offending medication, usually within weeks to months, whereas idiopathic SLE persists. So the key differentiating factors are: medication history, absence of major organ involvement especially renal and CNS, presence of anti-histone antibodies, absence of anti-dsDNA, and resolution after drug withdrawal."
Clinical Examination Findings to Demonstrate
Malar (butterfly) rash:
- Erythema over malar eminences and nasal bridge
- Characteristically spares nasolabial folds ← Pathognomonic feature
- Photosensitive
Discoid lupus:
- Scarring, atrophic plaques
- Follicular plugging
- Hyper/hypopigmentation
- Commonly on face, scalp (causing scarring alopecia), ears
Jaccoud's arthropathy:
- Reducible joint deformities (swan-neck deformity, ulnar deviation)
- Soft tissue laxity, not bony erosions
- Differentiates from rheumatoid arthritis
Oral ulcers:
- Usually painless (unlike Behçet's which are painful)
- Hard palate, buccal mucosa
Oedema:
- Periorbital (nephrotic syndrome)
- Peripheral pitting oedema (nephrotic syndrome, cardiac failure)
Pericardial/pleural rubs:
- Serositis
Livedo reticularis:
- Net-like vascular pattern
- Associated with antiphospholipid antibodies
Common Mistakes to Avoid
❌ Not starting hydroxychloroquine in all patients - it is foundational therapy ❌ Missing lupus nephritis by not checking urinalysis at every visit ❌ Prolonged high-dose glucocorticoids without steroid-sparing agents - causes irreversible damage ❌ Confusing drug-induced lupus with SLE - check medication history, anti-histone antibodies ❌ Not screening for antiphospholipid antibodies - 30-40% have aPL; significant thrombosis risk ❌ Forgetting ANA is entry criterion in 2019 ACR/EULAR criteria - without ANA ≥1:80 cannot classify as SLE ❌ Using ideal body weight for HCQ dosing - should use actual body weight at 5mg/kg ❌ Not counseling about pregnancy planning in women of childbearing age - need to switch teratogenic medications ❌ Attributing all symptoms to SLE without excluding infection, especially in immunosuppressed patients ❌ Not monitoring for cardiovascular risk - leading cause of late mortality
Differential Diagnosis Mnemonics
ANA-positive conditions (SOAP BRAIN MD):
- Scleroderma
- Other CTD
- Autoimmune hepatitis
- Polymyositis/dermatomyositis
- Berg (Sjögren's)
- RA (rheumatoid arthritis)
- Autoinflammatory
- Infections (EBV, HIV)
- Neoplasm (lymphoma)
- MCTD
- Drug-induced
13. References
-
Fanouriakis A, Kostopoulou M, Alunno A, et al. 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann Rheum Dis. 2019;78(6):736-745. doi:10.1136/annrheumdis-2019-215089
-
Tsokos GC. Systemic lupus erythematosus. N Engl J Med. 2011;365(22):2110-2121. doi:10.1056/NEJMra1100359
-
Pons-Estel GJ, Alarcón GS, Scofield L, Reinlib L, Cooper GS. Understanding the epidemiology and progression of systemic lupus erythematosus. Semin Arthritis Rheum. 2010;39(4):257-268. doi:10.1016/j.semarthrit.2008.10.007
-
Lisnevskaia L, Murphy G, Isenberg D. Systemic lupus erythematosus. Lancet. 2014;384(9957):1878-1888. doi:10.1016/S0140-6736(14)60128-8
-
Aringer M, Costenbader K, Daikh D, et al. 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus. Ann Rheum Dis. 2019;78(9):1151-1159. doi:10.1136/annrheumdis-2018-214819
-
Petri M, Orbai AM, Alarcón GS, et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 2012;64(8):2677-2686. doi:10.1002/art.34473
-
Ruiz-Irastorza G, Ramos-Casals M, Brito-Zeron P, Khamashta MA. Clinical efficacy and side effects of antimalarials in systemic lupus erythematosus: a systematic review. Ann Rheum Dis. 2010;69(1):20-28. doi:10.1136/ard.2008.101766
-
Alarcón GS, McGwin G, Bertoli AM, et al. Effect of hydroxychloroquine on the survival of patients with systemic lupus erythematosus: data from LUMINA, a multiethnic US cohort (LUMINA L). Ann Rheum Dis. 2007;66(9):1168-1172. doi:10.1136/ard.2006.068676
-
Navarra SV, Guzmán RM, Gallacher AE, et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial. Lancet. 2011;377(9767):721-731. doi:10.1016/S0140-6736(10)61354-2
-
Morand EF, Furie R, Tanaka Y, et al. Trial of anifrolumab in active systemic lupus erythematosus. N Engl J Med. 2020;382(3):211-221. doi:10.1056/NEJMoa1912196
-
Rovin BH, Teng YKO, Ginzler EM, et al. Efficacy and safety of voclosporin versus placebo for lupus nephritis (AURORA 1): a double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2021;397(10289):2070-2080. doi:10.1016/S0140-6736(21)00578-X
-
Cervera R, Khamashta MA, Font J, et al. Morbidity and mortality in systemic lupus erythematosus during a 10-year period: a comparison of early and late manifestations in a cohort of 1,000 patients. Medicine (Baltimore). 2003;82(5):299-308. doi:10.1097/01.md.0000091181.93122.55
-
Tektonidou MG, Dasgupta A, Ward MM. Risk of end-stage renal disease in patients with lupus nephritis, 1971-2015: a systematic review and Bayesian meta-analysis. Arthritis Rheumatol. 2016;68(6):1432-1441. doi:10.1002/art.39594
-
Manzi S, Meilahn EN, Rairie JE, et al. Age-specific incidence rates of myocardial infarction and angina in women with systemic lupus erythematosus: comparison with the Framingham Study. Am J Epidemiol. 1997;145(5):408-415. doi:10.1093/oxfordjournals.aje.a009122
-
Pisetsky DS, Lipsky PE. New insights into the role of antinuclear antibodies in systemic lupus erythematosus. Nat Rev Rheumatol. 2020;16(10):565-579. doi:10.1038/s41584-020-0480-7
-
Birmingham DJ, Hebert LA, Song H, et al. Evidence that abnormally large seasonal declines in vitamin D status may trigger SLE flare in non-African Americans. Lupus. 2012;21(8):855-864. doi:10.1177/0961203312439640
-
Hahn BH, McMahon MA, Wilkinson A, et al. American College of Rheumatology guidelines for screening, treatment, and management of lupus nephritis. Arthritis Care Res (Hoboken). 2012;64(6):797-808. doi:10.1002/acr.21664
-
Clowse ME, Magder LS, Petri M. The clinical utility of measuring complement and anti-dsDNA antibodies during pregnancy in patients with systemic lupus erythematosus. J Rheumatol. 2011;38(6):1012-1016. doi:10.3899/jrheum.100746
-
Zonana-Nacach A, Barr SG, Magder LS, Petri M. Damage in systemic lupus erythematosus and its association with corticosteroids. Arthritis Rheum. 2000;43(8):1801-1808. doi:10.1002/1529-0131(200008)43:8less than 1801::AID-ANR17>3.0.CO;2-O
-
Banchereau R, Hong S, Cantarel B, et al. Personalized immunomonitoring uncovers molecular networks that stratify lupus patients. Cell. 2016;165(6):1548-1550. doi:10.1016/j.cell.2016.05.057
-
Gladman DD, Urowitz MB, Goldsmith CH, et al. The reliability of the Systemic Lupus International Collaborating Clinics/American College of Rheumatology Damage Index in patients with systemic lupus erythematosus. Arthritis Rheum. 1997;40(5):809-813. doi:10.1002/art.1780400506
-
Rees F, Doherty M, Grainge MJ, Lanyon P, Zhang W. The worldwide incidence and prevalence of systemic lupus erythematosus: a systematic review of epidemiological studies. Rheumatology (Oxford). 2017;56(11):1945-1961. doi:10.1093/rheumatology/kex260
-
Bentham J, Morris DL, Graham DSC, et al. Genetic association analyses implicate aberrant regulation of innate and adaptive immunity genes in the pathogenesis of systemic lupus erythematosus. Nat Genet. 2015;47(12):1457-1464. doi:10.1038/ng.3434
-
Tsokos GC, Lo MS, Costa Reis P, Sullivan KE. New insights into the immunopathogenesis of systemic lupus erythematosus. Nat Rev Rheumatol. 2016;12(12):716-730. doi:10.1038/nrrheum.2016.186
-
Rahman A, Isenberg DA. Systemic lupus erythematosus. N Engl J Med. 2008;358(9):929-939. doi:10.1056/NEJMra071297
-
Bertsias GK, Ioannidis JP, Aringer M, et al. EULAR recommendations for the management of systemic lupus erythematosus with neuropsychiatric manifestations: report of a task force of the EULAR standing committee for clinical affairs. Ann Rheum Dis. 2010;69(12):2074-2082. doi:10.1136/ard.2010.130476
-
van Vollenhoven RF, Mosca M, Bertsias G, et al. Treat-to-target in systemic lupus erythematosus: recommendations from an international task force. Ann Rheum Dis. 2014;73(6):958-967. doi:10.1136/annrheumdis-2013-205139
Last Updated: 2026-01-10
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Autoimmunity and Immune Tolerance
- Complement System
Differentials
Competing diagnoses and look-alikes to compare.
- Drug-Induced Lupus
- Mixed Connective Tissue Disease
- Rheumatoid Arthritis
- Sjögren's Syndrome
Consequences
Complications and downstream problems to keep in mind.
- Lupus Nephritis
- End-Stage Renal Disease
- Accelerated Atherosclerosis