Hyperthyroidism in Adults
Hyperthyroidism is a clinical syndrome characterized by the excessive synthesis and secretion of thyroid hormones (thyro... MRCP, USMLE exam preparation.
What matters first
Hyperthyroidism is a clinical syndrome characterized by the excessive synthesis and secretion of thyroid hormones (thyro... MRCP, USMLE exam preparation.
Hyperpyrexia (less than 104CF/40CC)
11 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Hyperpyrexia (less than 104CF/40CC)
- Severe tachycardia or atrial fibrillation with rapid ventricular response
- Altered mental status or psychosis
- Congestive heart failure symptoms
Exam focus
Current exam surfaces linked to this topic.
- MRCP
- USMLE
- PLAB
- AMC
Linked comparisons
Differentials and adjacent topics worth opening next.
- Thyroiditis (Subacute and Painless)
- Pheochromocytoma
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
SECTION 1: Clinical Overview
1.1 Summary
Hyperthyroidism is a clinical syndrome characterized by the excessive synthesis and secretion of thyroid hormones (thyroxine, T4; and triiodothyronine, T3) by the thyroid gland. It is a subset of thyrotoxicosis, which refers to the clinical state of excess thyroid hormone regardless of the source. In the adult population, the most common cause is Graves' disease, an autoimmune disorder accounting for 60-80% of cases, followed by toxic multinodular goiter (15-20%) and toxic adenoma (3-5%). [1] Epidemiology shows a significant female preponderance, with a prevalence of approximately 1.2% in the United States, of which 0.5% is overt and 0.7% is subclinical. [5]
The clinical significance of hyperthyroidism lies in its profound multi-systemic effects, particularly on the cardiovascular and skeletal systems. Untreated hyperthyroidism leads to increased risks of atrial fibrillation (present in 10-25% of patients), embolic stroke (2-3 fold increased risk), high-output heart failure, and accelerated bone resorption leading to osteoporosis with a 2-fold increase in fracture risk. [12,16] Management is tripartite, involving symptomatic relief with beta-blockers, definitive control of hormone production (via thionamides, radioactive iodine, or surgery), and long-term monitoring for recurrence or iatrogenic hypothyroidism. Thyroid storm, though rare (affecting less than 1% of hospitalized thyrotoxic patients), represents an endocrine emergency with a mortality rate of 10-30% even with treatment. [15] The prognosis is generally excellent with appropriate therapy, though quality of life can be impacted by Graves' orbitopathy (affecting 25-50% of Graves' patients) or neuropsychiatric sequelae. [10]
1.2 Key Facts
- Definition: Hyperthyroidism is a hypermetabolic state caused by inappropriately high circulating levels of T4 and/or T3 due to increased thyroid gland activity.
- Incidence: Approximately 20 to 50 cases per 100,000 person-years in iodine-sufficient regions; higher in iodine-deficient areas. [5]
- Prevalence: Overt hyperthyroidism affects 0.5% of the population; subclinical hyperthyroidism affects 0.7% to 2.1%. [5]
- Mortality: Standardized mortality ratio (SMR) is 1.2 in treated patients; thyroid storm mortality 10-30% despite treatment. [15]
- Morbidity: 10-25% risk of atrial fibrillation (increases with age); 2-fold increased risk of fragility fractures; 5-15% risk of heart failure. [12,16]
- Peak Age: Graves' disease peaks between 30-50 years; Toxic Multinodular Goiter (TMNG) peaks after age 60.
- Sex Distribution: Female to Male ratio is approximately 5:1 to 10:1 depending on the etiology (Graves' has highest female preponderance).
- Pathognomonic Features: Thyroid stimulating immunoglobulin (TSI) positivity and exophthalmos for Graves' disease.
- Gold Standard Investigation: Serum TSH (third-generation assay, sensitivity 0.01 mIU/L) followed by free T4 and free T3 levels.
- First-line Treatment: Methimazole 10-30mg daily for Graves' (except first trimester pregnancy); Radioactive Iodine (I-131) for toxic nodular disease. [1]
- Second-line Treatment: Propylthiouracil (PTU) 100mg TID; total thyroidectomy for compressive goiter or failed medical therapy.
- Key Complications: Atrial fibrillation with thromboembolic risk, thyrotoxic periodic paralysis (2-10% in Asian males), thyroid storm, Graves' orbitopathy (25-50% of Graves' patients). [10,15]
- Remission Rate: 40-50% with 12-18 months of antithyroid drugs; 50% relapse within 5 years of stopping medication. [3]
- RAI Cure Rate: 80-90% with single dose; 10-20% require second treatment; > 90% develop hypothyroidism within 5-10 years. [14]
1.3 Clinical Pearls
Diagnostic Pearl: "The TSH-First Rule" In the vast majority of cases (99%+), a normal TSH excludes primary hyperthyroidism. If TSH is suppressed (less than 0.01 mIU/L) but free T4 and fT3 are normal, you are dealing with subclinical hyperthyroidism, which still requires risk-stratification for treatment based on age and comorbidities.
Examination Pearl: "The Lid Lag and Stare" Look for von Graefe's sign (lid lag) where the upper eyelid does not follow the downward movement of the eye. This is due to sympathetic overactivity affecting the levator palpebrae superioris and is seen in all forms of thyrotoxicosis, unlike exophthalmos which is specific to Graves'.
Treatment Pearl: "The Pregnancy PTU Switch" Propylthiouracil (PTU) is preferred in the first trimester of pregnancy due to methimazole's association with aplasia cutis and choanal atresia. However, PTU should be switched back to methimazole in the second trimester to avoid maternal hepatotoxicity.
Pitfall Warning: "Amiodarone Confusion" Amiodarone can cause hyperthyroidism through two mechanisms: Type 1 (excess iodine in a predisposed gland) and Type 2 (destructive thyroiditis). Distinguishing them is critical as Type 1 requires thionamides while Type 2 requires corticosteroids.
Mnemonic: "SWEATING" Sweating/Skin thinning, Weight loss, Emotional lability, Appetite increase, Tachycardia/Tremor, Intolerance to heat, Nervousness, Goiter/GI motility increase.
Emergency Pearl: "Burch-Wartofsky Score" Never wait for laboratory confirmation to treat suspected thyroid storm. If the Burch-Wartofsky Point Scale (BWPS) is > 45, initiate the "P's": PTU, Propranolol, Prednisolone, Potassium Iodide (Lugol's), and Paracetamol.
Exam Pearl: "Apathetic Hyperthyroidism" In elderly patients, the classic "hyper" symptoms may be absent. Instead, they present with "apathetic hyperthyroidism"—lethargy, depression, and weight loss, often mimicking malignancy or dementia.
1.4 Why This Matters Clinically
Hyperthyroidism is a high-stakes diagnosis because its systemic effects are often irreversible if treatment is delayed. From a patient outcome perspective, the cardiovascular strain of excess thyroid hormone can lead to permanent cardiomyopathy or embolic events that significantly decrease functional independence. Missing the diagnosis in an elderly patient presenting with new-onset atrial fibrillation is a common clinical error with life-altering consequences.
The healthcare burden is substantial; hyperthyroidism accounts for a significant portion of endocrine outpatient visits and can lead to prolonged hospitalizations if thyroid storm or severe heart failure develops. Medico-legally, failures to monitor thionamide-induced agranulocytosis or failing to counsel on the teratogenic risks of methimazole are frequent areas of litigation. For trainees, hyperthyroidism is a "must-know" topic because it integrates physiology, immunology, and pharmacology, and frequently appears on board exams in both internal medicine and surgery.
SECTION 2: Epidemiology
2.1 Incidence & Prevalence
- Incidence: 0.5 cases per 1,000 women per year; 0.1 cases per 1,000 men per year (PMID: 27011420).
- Prevalence: 1.2% in the general US population (NHANES III data).
- Lifetime Risk: Approximately 3% for women and 0.5% for men.
- Trend: Stable in iodine-sufficient areas; however, incidence of TMNG decreases as salt iodization programs improve.
- Geographic Variation: Higher incidence of Toxic Multinodular Goiter in iodine-deficient regions (e.g., parts of Central Africa and mountainous Europe).
- Temporal Patterns: No strong seasonal variation, though some studies suggest a minor spring peak for Graves' disease onset.
- Healthcare Burden: Associated with a 1.5-fold increase in all-cause hospitalization rates.
2.2 Demographics Table
| Factor | Details | Clinical Significance |
|---|---|---|
| Age | Graves': 30-50 years; TMNG: > 60 years | Older patients need more aggressive cardiac screening. |
| Sex | Female:Male ratio 8:1 | Autoimmune predisposition in females is well-documented. |
| Ethnicity | Higher in African Americans and Caucasians | Genetic susceptibility alleles (HLA-DR3) vary by race. |
| Geography | Higher in iodine-deficient areas | Influences the type of hyperthyroidism (TMNG vs Graves'). |
| Socioeconomic | Lower access leads to late presentation | Increased risk of thyroid storm in underserved areas. |
| Occupation | No specific association | Occupational stress may trigger Graves' flares. |
2.3 Risk Factors Tables
Non-Modifiable Risk Factors:
| Factor | Relative Risk (95% CI) | Mechanism |
|---|---|---|
| Female Sex | RR 7.5 (6.2-9.1) | Estrogen modulation of immune response. |
| Family History | RR 4.2 (3.1-5.5) | HLA-DR3, CTLA-4, and PTPN22 gene polymorphisms. |
| Age > 60 | RR 2.1 (1.8-2.5) | Increased risk for TMNG and toxic adenoma. |
| HLA-DR3 Status | RR 3.5 (2.5-4.8) | Increased antigen presentation of TSH receptor. |
| Post-partum period | RR 2.8 (1.9-4.1) | Immune rebound after pregnancy-induced suppression. |
Modifiable Risk Factors:
| Risk Factor | Relative Risk (95% CI) | Evidence Level | Intervention Impact |
|---|---|---|---|
| Smoking | RR 1.9 (1.5-2.4) | Level 1a | High risk for Graves' Orbitopathy. |
| High Iodine Intake | RR 1.5 (1.2-1.8) | Level 2a | Jod-Basedow phenomenon in predisposed glands. |
| Stressful Life Events | RR 1.7 (1.3-2.2) | Level 2b | Cortisol-mediated immune shifts. |
| Selenium Deficiency | RR 1.3 (1.1-1.6) | Level 2a | Reduced antioxidant protection of thyroid. |
| Amiodarone Use | RR 2.5 (1.8-3.5) | Level 1b | Direct iodine load and thyroiditis risk. |
2.4 Protective Factors
- Moderate Alcohol Consumption: RR 0.7, potentially through mild immunosuppressive effects.
- Cigarette Cessation: Reduces the risk of Graves' orbitopathy progression by 4-fold.
- Adequate Selenium Intake: Helps maintain T-cell balance and reduces oxidative stress in the gland.
SECTION 3: Pathophysiology
3.1 Step 1: Initiating Event - Loss of Immune Tolerance
In Graves' disease, the primary trigger is a breakdown in central and peripheral immune tolerance. Genetic susceptibility (HLA-DR3, PTPN22) combines with environmental triggers (stress, infection, smoking) to cause the presentation of the TSH receptor (TSHR) as an autoantigen. Dendritic cells capture TSHR peptides and present them to CD4+ T-cells via MHC class II molecules. This initiates a Th2-predominant immune response, leading to the activation of B-cells. In Toxic Multinodular Goiter, the trigger is different: somatic mutations in the TSHR or Gs-alpha protein gene lead to constitutive activation of the thyroid follicular cells without the need for TSH or antibodies.
3.2 Step 2: Production of TSH-Receptor Antibodies (TRAb)
Activated B-cells differentiate into plasma cells that secrete Thyroid Stimulating Immunoglobulins (TSI), a specific type of TRAb. These antibodies are molecular mimics of TSH. They bind to the extracellular domain of the TSH receptor on the surface of thyroid follicular cells. Unlike TSH, which is regulated by negative feedback, TSI provides continuous, unregulated stimulation of the receptor. This bypasses the pituitary-thyroid axis, leading to the classic suppression of pituitary TSH secretion while the thyroid gland remains hyperactive.
3.3 Step 3: Intracellular Signaling Cascade (cAMP Activation)
Upon TSI binding, the TSH receptor undergoes a conformational change that activates the Gs protein. This stimulates adenylyl cyclase, leading to a massive increase in intracellular cyclic adenosine monophosphate (cAMP). High cAMP levels activate Protein Kinase A (PKA), which phosphorylates multiple targets within the follicular cell. This stimulates every step of thyroid hormone synthesis: increased expression of the Sodium-Iodide Symporter (NIS), activation of Thyroid Peroxidase (TPO) for iodine organification, and increased synthesis of thyroglobulin.
3.4 Step 4: Hypertrophy, Hyperplasia, and Hormone Release
The chronic cAMP stimulation leads to both functional and structural changes. Follicular cells undergo hypertrophy (increase in size) and hyperplasia (increase in number), manifesting clinically as a diffuse goiter. There is an accelerated rate of thyroglobulin endocytosis from the colloid back into the follicular cell. Lysosomal proteases then cleave thyroglobulin to release free T4 and T3 into the systemic circulation. In Graves' disease, the ratio of T3 to T4 secreted often increases, as the hyperstimulated gland favors T3 production.
3.5 Step 5: Peripheral Effects and Hypermetabolism
Excess circulating T4 and T3 enter peripheral cells via transporters (like MCT8). T4 is converted to the more active T3 by deiodinase enzymes. T3 binds to nuclear thyroid hormone receptors (TR-alpha and TR-beta), acting as a transcription factor. This increases the basal metabolic rate by:
- Upregulating Na+/K+-ATPase pumps (increasing oxygen consumption).
- Upregulating beta-1 adrenergic receptors (increasing catecholamine sensitivity).
- Stimulating lipolysis and glycogenolysis.
- Increasing bone resorption by osteoclasts.
- In the heart, T3 increases the expression of alpha-myosin heavy chain and sarcoplasmic reticulum Ca2+-ATPase (SERCA2), leading to increased chronotropy and inotropy.
3.6 Classification/Staging
| Stage/Type | Definition | Clinical Features | Prognosis |
|---|---|---|---|
| Subclinical | Low TSH, Normal fT4/fT3 | Often asymptomatic; cardiac risk | 5% progress to overt/yr |
| Overt | Low TSH, High fT4/fT3 | Classic hypermetabolic symptoms | Excellent with treatment |
| Graves' | Autoimmune (TRAb+) | Goiter, Bruit, Orbitopathy | 50% relapse after meds |
| Toxic MNG | Autonomic nodules | Patchy uptake on scan; elderly | Permanent; needs RAI/Surg |
| Thyroid Storm | Decompensated thyrotoxicosis | Fever, CNS dysfunction, HF | 10-30% mortality |
3.7 Anatomical Considerations
- Vascularity: The thyroid is one of the most vascular organs. In Graves', the Superior and Inferior thyroid arteries significantly dilate, creating the "thyroid bruit."
- Recurrent Laryngeal Nerve: Runs in the tracheoesophageal groove; vulnerable during thyroidectomy, leading to hoarseness.
- Parathyroid Glands: Usually four, located on the posterior thyroid capsule; risk of hypocalcemia if damaged during surgery.
- Lymphatics: Drainage to Level VI (central) and Levels II-IV (lateral) nodes is relevant if malignancy is suspected in a toxic nodule.
3.8 Physiological Considerations
- Negative Feedback: High T4/T3 normally inhibits TRH (hypothalamus) and TSH (pituitary). In hyperthyroidism, this feedback is present but ineffective because the stimulus (TSI or autonomic nodules) is independent of TSH.
- Wolff-Chaikoff Effect: High doses of iodine acutely inhibit TPO and thyroid hormone release. This is the physiological basis for using Lugol's iodine in thyroid storm or pre-operatively.
SECTION 4: Clinical Presentation
4.1 Symptoms
| Symptom | Frequency | Character | Duration | Associations |
|---|---|---|---|---|
| Palpitations | 85% | Rapid, pounding heart | Constant/Intermittent | Anxiety, dyspnea |
| Weight Loss | 75% | Despite increased appetite | Weeks to months | Diarrhea, malabsorption |
| Heat Intolerance | 70% | Preference for cold | Chronic | Excessive sweating |
| Tremor | 65% | Fine, distal, postural | Constant | Functional impairment |
| Fatigue | 60% | Muscle weakness (proximal) | Progressive | Difficulty climbing stairs |
| Anxiety | 55% | Irritability, insomnia | Chronic | Neuropsychiatric shifts |
| Oligomenorrhea | 40% | Light or infrequent menses | Months | Infertility |
| Hyperdefecation | 35% | Increased frequency (not diarrhea) | Chronic | Weight loss |
4.2 Signs
| Sign | How to Elicit | Significance | Sensitivity/Specificity |
|---|---|---|---|
| Tachycardia | Pulse palpation at rest | Cardinal sign of thyrotoxicosis | 80%/60% |
| Fine Tremor | Outstretched hands with paper | Beta-adrenergic overactivity | 70%/75% |
| Diffuse Goiter | Palpation during swallowing | Suggestive of Graves' Disease | 85%/70% |
| Thyroid Bruit | Auscultation over lobes | High vascularity (Graves') | 30%/95% |
| Exophthalmos | Inspection from above/side | Pathognomonic for Graves' | 40%/99% |
| Pretibial Myxedema | Inspection of shins | Rare autoimmune skin infiltrate | 5%/99% |
| Lid Lag | Follow finger downwards | Sympathetic eyelid overactivity | 50%/80% |
4.3 Red Flags
[!CAUTION] RED FLAGS — Seek immediate help if:
- Hyperpyrexia (> 104°F): Indicates impending thyroid storm.
- Altered Mental Status: Confusion, agitation, or coma in a thyrotoxic patient.
- New-onset Atrial Fibrillation: High risk for thromboembolism and heart failure.
- Severe Jaundice: May indicate thyrotoxic hepatopathy or PTU toxicity.
- Painless Muscle Weakness: In Asian males, suggests thyrotoxic periodic paralysis (hypokalemia).
- Stridor: Large goiter causing tracheal compression.
- Rapidly Progressing Orbitopathy: Risk of optic nerve compression and blindness.
4.4 Clinical Scoring
Burch-Wartofsky Point Scale (BWPS):
- less than 25: Storm unlikely.
- 25-44: Impending storm.
-
45: Thyroid storm.
- Criteria include temperature, CNS effects, GI-hepatic dysfunction, heart rate, and presence of heart failure.
SECTION 5: Clinical Examination
5.1 Structured Approach
- A (Airway): Assess for tracheal deviation or stridor from goiter.
- B (Breathing): Tachypnea or signs of pulmonary edema in high-output heart failure.
- C (Circulation): Pulse (rate and rhythm), blood pressure (wide pulse pressure), and precordial heave.
- D (Disability): Assessment of tremor, brisk deep tendon reflexes (hyperreflexia), and mental status.
- E (Exposure/Examination): Thyroid palpation, eye exam, and skin/shin inspection.
5.2 Special Tests Table
| Test Name | Technique | Positive Finding | Sensitivity | Specificity | Clinical Use |
|---|---|---|---|---|---|
| Pemberton's Sign | Raise arms above head | Facial flushing/cyanosis | 20% | 90% | Retrosternal goiter |
| Stellwag's Sign | Observe blinking | Infrequent/incomplete blink | 40% | 70% | Thyrotoxic stare |
| Joffroy's Sign | Look up without brow wrinkle | Lack of forehead wrinkling | 30% | 80% | Graves' ophthalmopathy |
| Proximal Myopathy | Stand from chair (no arms) | Inability to stand easily | 60% | 75% | Assess severity |
| Möbius Sign | Convergence test | Inability to converge eyes | 35% | 85% | Extraocular muscle edema |
| Pulse Pressure | BP measurement | Difference > 60 mmHg | 50% | 60% | Hyperdynamic state |
5.3 Examination by Severity
- Mild: Resting tachycardia (90-100 bpm), mild tremor, warm skin.
- Moderate: Weight loss > 5kg, HR 100-120 bpm, visible goiter, hyperreflexia.
- Severe: HR > 140 bpm, atrial fibrillation, fever, psychosis, severe proximal muscle wasting.
SECTION 6: Differential Diagnosis
6.1 Primary Differential Diagnoses
| Condition | Key Distinguishing Features | Investigations | Typical TSH/fT4 |
|---|---|---|---|
| Graves' Disease | Diffuse goiter, bruit, exophthalmos, pretibial myxedema | TRAb/TSI positive, diffuse RAIU uptake | Low / High |
| Toxic Multinodular Goiter | Elderly, nodular goiter, no eye signs | TRAb negative, patchy RAIU uptake | Low / High |
| Toxic Adenoma | Single palpable nodule | TRAb negative, focal "hot" nodule on scan | Low / High |
| Subacute Thyroiditis (De Quervain) | Painful tender gland, preceding viral illness, fever | Low/absent RAIU, elevated ESR/CRP | Low / High (early) then Normal/Low |
| Painless (Silent) Thyroiditis | Postpartum period, no pain, transient | Low RAIU, TPO antibodies often positive | Low / High (early) |
| Amiodarone-Induced Thyrotoxicosis Type 1 | Amiodarone use, underlying thyroid disease | Normal/high RAIU (if no prior contrast), color Doppler flow | Low / High |
| Amiodarone-Induced Thyrotoxicosis Type 2 | Amiodarone use, destructive thyroiditis | Low/absent RAIU, low thyroid vascularity | Low / High |
| Factitious Thyrotoxicosis | Exogenous thyroid hormone ingestion, weight loss | Low RAIU, low thyroglobulin | Low / High |
| Struma Ovarii | Rare, pelvic mass, teratoma | Low neck RAIU, pelvic uptake on whole body scan | Low / High |
| TSH-Secreting Pituitary Adenoma | Very rare, headaches, visual field defects | High/normal TSH with high fT4, alpha-subunit elevated | High/Normal / High |
| Gestational Thyrotoxicosis | First trimester, hyperemesis gravidarum, high hCG | TRAb negative, low RAIU (contraindicated), resolves by 20 weeks | Low / High/Normal |
6.2 Secondary Differentials (Mimics)
| Condition | Clinical Overlap | Key Differentiator |
|---|---|---|
| Pheochromocytoma | Tachycardia, tremor, sweating, anxiety | Normal TSH, elevated metanephrines |
| Anxiety Disorder/Panic Attacks | Palpitations, tremor, nervousness | Normal TSH, episodic symptoms |
| Malignancy (Cancer Cachexia) | Weight loss, fatigue | Normal TSH, specific tumor markers |
| Cocaine/Amphetamine Use | Tachycardia, weight loss, agitation | Normal TSH, toxicology positive |
| Diabetes Mellitus (Uncontrolled) | Weight loss, polydipsia, fatigue | Normal TSH, elevated glucose/HbA1c |
| Heart Failure | Dyspnea, edema, tachycardia | Normal TSH, elevated BNP, reduced LVEF |
| Menopause | Heat intolerance, palpitations, mood changes | Normal TSH, elevated FSH/LH |
6.3 Diagnostic Algorithm for Thyrotoxicosis Etiology
┌─────────────────────────────────────────────────────────────┐
│ TSH SUPPRESSED + Elevated fT4/fT3 │
└─────────────────────────────────────────────────────────────┘
│
▼
┌───────────────────────────────┐
│ CLINICAL ASSESSMENT │
│ • Goiter present? │
│ • Eye signs? │
│ • Tender thyroid? │
│ • Drug history? │
└───────────────────────────────┘
│
▼
┌───────────────────────────────┐
│ FIRST-LINE TEST: │
│ TSH Receptor Antibody (TRAb)│
└───────────────────────────────┘
│
┌─────────────────────┼─────────────────────┐
│ │ │
▼ ▼ ▼
┌──────────────┐ ┌──────────────┐ ┌──────────────┐
│ TRAb POSITIVE│ │ TRAb NEGATIVE│ │ EQUIVOCAL │
│ │ │ │ │ │
│ = GRAVES' │ │ Need RAIU/ │ │ Repeat TRAb │
│ DISEASE │ │ Thyroid Scan │ │ + RAIU │
└──────────────┘ └──────────────┘ └──────────────┘
│
┌─────────────────────┼─────────────────────┐
▼ ▼ ▼
┌─────────────┐ ┌─────────────┐ ┌─────────────┐
│ HIGH Uptake │ │ LOW Uptake │ │ NORMAL │
│ │ │ │ │ Uptake │
│ • Diffuse → │ │ • Painful → │ │ Iodine load │
│ Graves' │ │ Subacute │ │ or factitious│
│ • Nodular → │ │ • Painless →│ │ │
│ TMNG │ │ Silent │ │ │
│ • Focal → │ │ • Amio Type2│ │ │
│ Adenoma │ │ │ │ │
└─────────────┘ └─────────────┘ └─────────────┘
SECTION 7: Investigations
7.1 Bedside Tests
| Test | Expected Finding | Purpose | Urgency |
|---|---|---|---|
| ECG | Sinus tachycardia or Afib | Assess cardiac impact | Immediate |
| Blood Glucose | Mild hyperglycemia | Thyroid hormone is insulin-antagonistic | Immediate |
| Urinalysis | Negative for ketones | Rule out DKA (differential for weight loss) | Routine |
| Pulse Oximetry | Usually normal | Rule out HF-related hypoxia | Immediate |
| Weight/BMI | Significant decrease | Establish baseline for treatment | Routine |
7.2 Laboratory Tests
| Test | Expected Finding | Purpose | Turnaround |
|---|---|---|---|
| TSH | less than 0.01 mIU/L | Primary screening (suppressed) | 4-24 hours |
| Free T4 | Elevated | Confirm overt hyperthyroidism | 4-24 hours |
| Free T3 | Elevated (often more than T4) | Diagnose T3-toxicosis | 4-24 hours |
| TRAb / TSI | Positive in Graves' | Confirm etiology | 2-5 days |
| TPO Antibodies | Often positive | Suggests autoimmune basis | 2-5 days |
| FBC | Mild leukopenia/anemia | Baseline before thionamides | 1 hour |
| LFTs | Elevated ALP/ALT | Baseline and assess hepatopathy | 1 hour |
| Calcium | Mild hypercalcemia | Assess bone turnover | 1 hour |
7.3 Imaging
| Modality | Key Findings | Indication | Sensitivity/Specificity |
|---|---|---|---|
| RAIU (I-123) | High diffuse (Graves'); Focal (Adenoma) | Differentiating etiology | 95%/90% |
| Thyroid Ultrasound | Hypoechoic, hypervascular ("storm") | Best for nodules/pregnancy | 85%/80% |
| CT/MRI Orbit | Extraocular muscle enlargement | Severe Graves' orbitopathy | 90%/95% |
| CT Chest/Neck | Tracheal narrowing | Retrosternal goiter | 90%/90% |
| Technetium Scan | "Hot" vs "Cold" nodules | When iodine load is present | 90%/85% |
7.4 Diagnostic Criteria
- Overt Hyperthyroidism: TSH less than 0.1 mIU/L AND elevated fT4/fT3.
- Subclinical Hyperthyroidism: TSH less than 0.1 mIU/L (or below range) AND normal fT4/fT3.
- Graves' Disease: Overt hyperthyroidism + TRAb positivity OR exophthalmos OR diffuse uptake on RAIU.
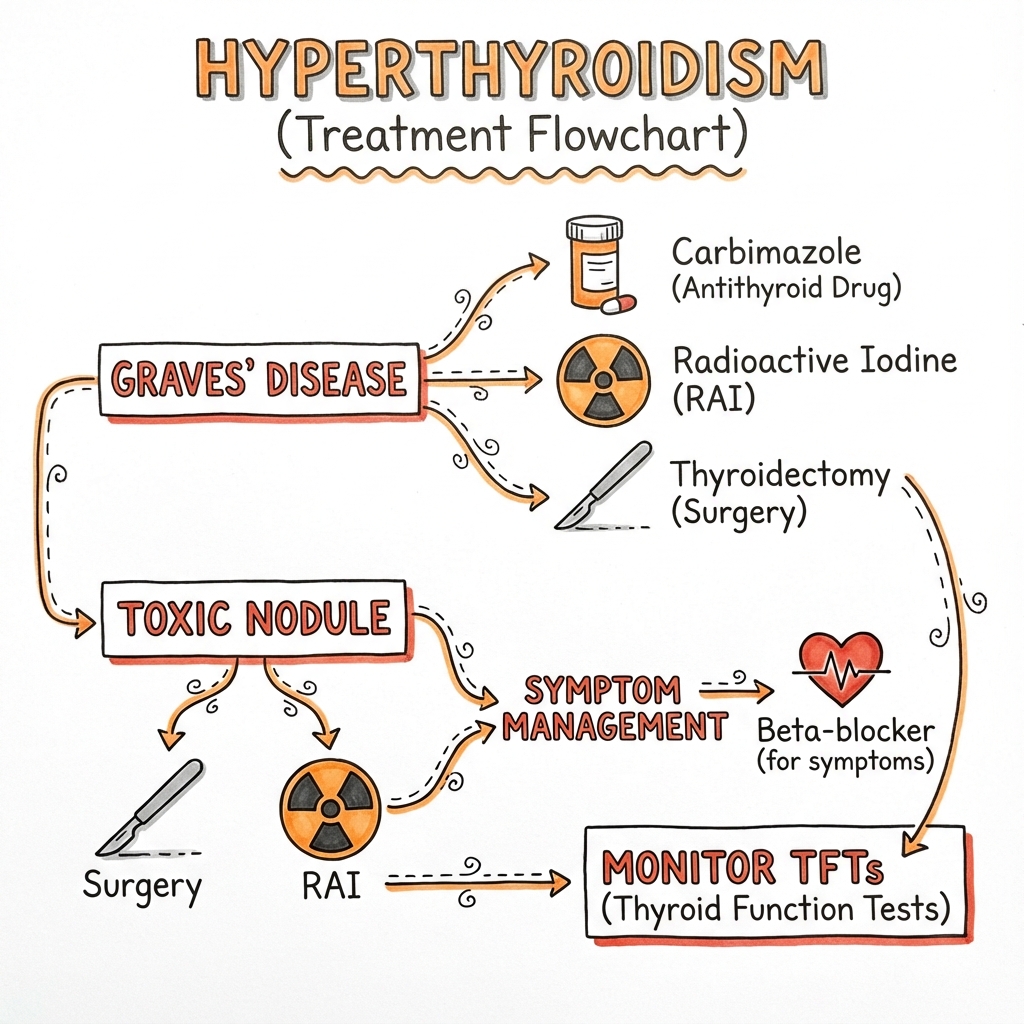
SECTION 8: Management
8.1 Management Principles
Treatment Goals:
- Rapid control of adrenergic symptoms (beta-blockade)
- Normalization of thyroid hormone levels (definitive therapy)
- Prevention of complications (atrial fibrillation, osteoporosis, thyroid storm)
- Individualized approach based on etiology, age, comorbidities, and patient preference [1]
Treatment Selection Factors:
| Factor | Favors Antithyroid Drugs | Favors Radioiodine | Favors Surgery |
|---|---|---|---|
| Age | Young (\u003c40 years) | Middle-aged/Elderly | Any age |
| Goiter Size | Small (\u003c40g) | Small-moderate | Large (\u003e80g), compressive |
| Etiology | Graves' (first episode) | Toxic nodular disease | Graves' with orbitopathy |
| Pregnancy | Yes (PTU 1st trimester) | Contraindicated | 2nd trimester if urgent |
| Patient Preference | Avoid radiation/surgery | One-time treatment | Definitive cure |
| Nodule Suspicion | No nodules | Benign nodules | Suspicious nodules |
| Previous Treatment | - | Failed ATD | Failed ATD/RAI |
| Contraindications | Drug allergy/agranulocytosis | Pregnancy, active orbitopathy | High surgical risk |
8.2 ASCII MANAGEMENT ALGORITHM
┌─────────────────────────────────────────────────────────────┐
│ HYPERTHYROIDISM MANAGEMENT ALGORITHM │
└─────────────────────────────────────────────────────────────┘
│
▼
┌───────────────────────────────┐
│ INITIAL PRESENTATION │
│ • TSH, fT4, fT3 │
│ • Start Beta-Blocker │
│ • Assess for Thyroid Storm │
└───────────────────────────────┘
│
▼
┌───────────────────────────────┐
│ DETERMINE ETIOLOGY │
│ • TRAb / TSI Antibody │
│ • RAIU Scan (if not pregnant)│
└───────────────────────────────┘
│
┌─────────────────────┼─────────────────────┐
▼ ▼ ▼
┌───────────────┐ ┌───────────────┐ ┌───────────────┐
│ GRAVES' │ │ TOXIC MNG │ │ THYROIDITIS │
│ │ │ / ADENOMA │ │ (De Quervain) │
└───────────────┘ └───────────────┘ └───────────────┘
│ │ │
▼ ▼ ▼
┌───────────────┐ ┌───────────────┐ ┌───────────────┐
│ First-line: │ │ First-line: │ │ Conservative │
│ Methimazole │ │ RAI (I-131) │ │ • NSAIDs │
│ (10-30mg/day) │ │ or Surgery │ │ • Beta-blocker│
└───────────────┘ └───────────────┘ └───────────────┘
│ │ │
▼ ▼ ▼
┌─────────┐ ┌─────────┐ ┌─────────┐
│Response?│ │Response?│ │Monitor │
└────┬────┘ └────┬────┘ └────┬────┘
Yes │ No Yes │ No Wait 6-8wks
│ │ │
▼ ▼ ▼
┌───────────────┐ ┌───────────────┐ ┌───────────────┐
│ Taper meds │ │ Monitor for │ │ Check for │
│ at 12-18 mos │ │ Hypothyroid │ │ Hypothyroid │
│ If relapse -> │ │ post-ablative │ │ phase │
│ RAI/Surgery │ │ │ │ │
└───────────────┘ └───────────────┘ └───────────────┘
8.3 Emergency Management (Thyroid Storm)
Thyroid Storm Diagnostic Criteria (Burch-Wartofsky Point Scale):
- Temperature (\u003e38.3°C = 5 points; \u003e39.4°C = 10 points; \u003e40.5°C = 15 points)
- CNS effects (Mild = 10; Moderate = 20; Severe/Coma = 30 points)
- GI-Hepatic (Moderate = 10; Severe = 20 points)
- Tachycardia (99-109 = 5; 110-119 = 10; 120-129 = 15; 130-139 = 20; ≥140 = 25 points)
- Heart failure (Absent = 0; Mild = 5; Moderate = 10; Severe = 15 points)
- Atrial fibrillation (Absent = 0; Present = 10 points)
- Precipitant (Absent = 0; Present = 10 points)
Interpretation: \u003c25 = unlikely storm; 25-44 = impending storm; ≥45 = thyroid storm [15]
Multi-Modal Treatment Protocol:
- Beta-blockade: Propranolol 60–80 mg PO every 4 hours or Esmolol IV drip (50–100 μg/kg/min).
- Thionamide: PTU 200 mg every 4 hours (preferred over methimazole as it inhibits T4 to T3 conversion).
- Iodine: Lugol's solution 5–10 drops TID (administer AT LEAST 1 hour AFTER thionamide).
- Glucocorticoids: Hydrocortisone 100 mg IV every 8 hours (inhibits T4-T3 conversion and treats relative adrenal insufficiency).
- Supportive: Cooling blankets (avoid aspirin - displaces thyroid hormone from binding proteins), IV fluids (3-5L/day for insensible losses), and treat the precipitating cause (e.g., infection, surgery, contrast load). [15]
- Consider Plasmapheresis/Dialysis: For refractory cases to remove circulating thyroid hormones.
- ICU Monitoring: Continuous cardiac monitoring, hourly vitals, strict fluid balance.
8.4 Conservative Management
- Smoking Cessation: Crucial to prevent Graves' orbitopathy.
- Diet: Avoid excessive iodine (kelp, seaweed) during thionamide therapy.
- Selenium: 100-200 mcg/day may improve mild Graves' orbitopathy.
- Education: Counsel on the symptoms of agranulocytosis (sore throat, fever) and the need for immediate FBC.
8.5 Medical Management
First-Line Drug Table:
| Drug | Class | Dose | Route | Frequency | Duration | Key Side Effects | Monitoring |
|---|---|---|---|---|---|---|---|
| Methimazole | Thionamide | 10-30mg | PO | Daily | 12-18 mos | Rash, Pruritus | TSH/fT4, FBC |
| Propranolol | Beta-blocker | 20-40mg | PO | TID/QID | Until euthyroid | Bradycardia | HR, BP |
| PTU | Thionamide | 100mg | PO | TID | 1st Trimester | Hepatotoxicity | LFTs, FBC |
| Atenolol | Beta-blocker | 25-50mg | PO | Daily | Symptomatic | Fatigue | HR |
| Prednisone | Steroid | 40-60mg | PO | Daily | T2 Thyroiditis | Hyperglycemia | Glucose |
Second-Line/Alternative Drugs:
| Drug | Indication | Dose | Contraindications |
|---|---|---|---|
| Cholestyramine | Adjunct in severe hyper | 4g TID | Bowel obstruction |
| Lithium | Thionamide allergy | 300mg TID | Renal failure |
| Potassium Iodide | Pre-op preparation | 5 drops TID | Toxic multinodular goiter |
| Rituximab | Refractory Graves' Orbit | 1000mg x2 | Active infection |
| Terprotumumab | Graves' Orbitopathy | IV infusion | Inflammatory bowel disease |
| Regimen | Description | Advantages | Disadvantages |
|---|---|---|---|
| Titration (Dose-adjustment) | Start high dose (MMI 20-40mg), reduce as fT4 normalizes | Lower drug exposure, fewer side effects | Requires frequent monitoring |
| Block-and-Replace | High dose ATD + levothyroxine replacement | Stable dosing, less monitoring | Higher ATD dose, more side effects |
| Duration | 12-18 months standard; 24+ months in some | Remission rates 40-50% at 18 months | 50% relapse within 5 years [3] |
Monitoring Antithyroid Drugs:
- Baseline: FBC, LFTs, TFTs
- Week 2-4: FBC (agranulocytosis risk highest in first 90 days)
- Month 1-2: TFTs to guide dose adjustment
- Every 4-6 weeks: TFTs until stable
- Every 3 months: TFTs, consider LFTs
- Patient education: Report fever, sore throat (agranulocytosis), jaundice (hepatotoxicity) immediately
Relapse Predictors After ATD Withdrawal:
- Large goiter (\u003e40g)
- High TRAb titers at diagnosis and during treatment
- Severe biochemical hyperthyroidism (fT4 \u003e50 pmol/L)
- Male sex
- Smoking
- Young age at onset
8.6 Radioiodine (I-131) Therapy
Mechanism: Beta and gamma radiation destroys thyroid follicular cells over 6-18 weeks. [14]
Indications:
- Definitive treatment for Graves' disease (especially if failed ATD or relapse)
- First-line for toxic multinodular goiter and toxic adenoma
- Elderly patients or those with comorbidities precluding surgery
Contraindications:
- Pregnancy and breastfeeding (absolute)
- Active moderate-to-severe Graves' orbitopathy (relative - may worsen)
- Large goiter with compressive symptoms (relative)
- Inability to comply with radiation safety precautions
Preparation:
- Withdraw ATD 3-7 days before RAI (to allow uptake)
- Restart ATD 3-7 days after RAI if severe thyrotoxicosis
- Low-iodine diet for 1-2 weeks prior (controversial)
- Pregnancy test in women of childbearing age (mandatory)
Dosing:
- Fixed dose: 10-15 mCi (370-555 MBq) for Graves' disease
- Calculated dose: Based on gland size and RAIU (aiming for 150-200 μCi/g thyroid tissue)
- Higher doses for toxic nodular disease (15-30 mCi)
Outcomes:
- 70-90% euthyroid or hypothyroid at 6-12 months [14]
- 10-20% require second dose
- Hypothyroidism develops in \u003e90% by 10 years (considered a planned outcome)
Post-RAI Follow-up:
- TFTs at 4-6 weeks, then every 6-8 weeks until stable
- Monitor for transient thyrotoxicosis (radiation thyroiditis) in first 2 weeks
- Start levothyroxine when TSH rises
- Ophthalmology review if Graves' orbitopathy present
Radiation Safety:
- Avoid close contact with pregnant women/young children for 3-7 days
- Sleep separately for 3-5 nights
- Flush toilet 2-3 times after use
- Wash hands frequently
- Contraception for 6 months (women) to 4 months (men)
8.7 Surgical Management
Indications for Surgery:
- Large goiter (\u003e80g) causing compressive symptoms (dysphagia, dyspnea, stridor).
- Suspicion of thyroid malignancy in a toxic nodule (5-10% risk in solitary nodules).
- Patient preference (avoidance of RAI and long-term meds).
- Moderate-to-severe active Graves' Orbitopathy (RAI can worsen this; surgery with concurrent steroids preferred). [8]
- Failure or intolerance of thionamides.
- Pregnancy (second trimester) if urgent and ATD fails/contraindicated.
- Retrosternal goiter.
Procedure Description: Total thyroidectomy is the preferred approach for Graves' disease to minimize recurrence (near-zero recurrence vs 5-10% with subtotal). For toxic adenoma, thyroid lobectomy is sufficient. [1]
Pre-operative Preparation:
- Achieve euthyroid state with ATD (reduces surgical complications)
- Beta-blockers if cannot tolerate ATD or urgent surgery
- Lugol's iodine 5-10 drops TID for 7-10 days pre-op (reduces vascularity and bleeding)
- Vocal cord assessment (laryngoscopy) if previous neck surgery or voice change
Surgical Approach:
- Transcervical (standard Kocher incision)
- Minimally invasive/endoscopic (selected cases)
- Intraoperative nerve monitoring (reduces RLN injury risk)
- Parathyroid gland identification and preservation
Surgical Complications:
| Complication | Incidence | Prevention | Management |
|---|---|---|---|
| Hypocalcemia | 10-20% (transient), 1-2% (permanent) | Identify/preserve parathyroids, autotransplant | Calcium/Vit D supplementation |
| RLN Injury | 1-2% (experienced surgeon), up to 5% (less experienced) | Nerve monitoring, careful dissection | Speech therapy, medialization |
| Hematoma | \u003c1% (0.3-1%) | Meticulous hemostasis, drains | Emergency bedside evacuation |
| Hypothyroidism | 100% (planned outcome) | N/A | Levothyroxine replacement lifelong |
| Infection/Seroma | \u003c1% | Sterile technique, prophylactic antibiotics | Antibiotics, drainage if needed |
| Thyroid Storm | \u003c1% (if inadequately prepared) | Pre-op euthyroid state, beta-blockade | Multi-modal emergency protocol |
Post-operative Care:
- Calcium monitoring (risk of hypocalcemia peaks at 24-48 hours)
- Check serum calcium and PTH on day 1 and day 7
- Vocal cord assessment if voice change or concerning intra-op findings
- Start levothyroxine 1.6 μg/kg/day within 24-48 hours
- TFTs at 6-8 weeks, adjust dose to maintain TSH 0.5-2.5 mIU/L
8.8 Special Populations
Pregnancy and Hyperthyroidism:
| Trimester | Preferred ATD | Dosing | Monitoring | RAI | Surgery |
|---|---|---|---|---|---|
| First | PTU 100mg TID | Lowest effective dose | Monthly TFTs, target fT4 upper normal | Contraindicated | Avoid unless emergency |
| Second | Switch to MMI 10-20mg | Reduce dose if possible | Monthly TFTs | Contraindicated | Preferred if needed |
| Third | MMI or PTU | Taper/stop if possible (4-6 wks pre-delivery) | Monthly TFTs, fetal monitoring | Contraindicated | Avoid |
| Postpartum | MMI (compatible with breastfeeding in doses \u003c20mg) | Resume if relapse | TFTs at 6 wks, 3 mo, 6 mo | Contraindicated while breastfeeding | If indicated |
Key Points:
- Gestational transient thyrotoxicosis (high hCG) vs Graves' disease (TRAb positive)
- Fetal/neonatal risks: Prematurity, IUGR, fetal thyrotoxicosis (from maternal TRAb crossing placenta)
- Neonatal monitoring if maternal TRAb \u003e3x ULN (measure TRAb at 18-22 weeks, 30-34 weeks)
- PTU preferred first trimester due to MMI embryopathy risk (aplasia cutis, choanal atresia)
- PTU hepatotoxicity risk requires LFT monitoring
- Breastfeeding: PTU and MMI (\u003c20mg/day) considered compatible [1]
Elderly Patients (≥65 years):
Clinical Presentation:
- "Apathetic hyperthyroidism"
- lethargy, depression, weight loss (not classic hyperactivity)
- Higher risk of atrial fibrillation (20-30% prevalence)
- Cardiovascular complications more common
- Osteoporosis and fracture risk significantly elevated
Management Considerations:
- Lower threshold for anticoagulation in AF (high CHA₂DS₂-VASc scores)
- RAI often first-line (avoid prolonged ATD in those with limited life expectancy)
- Careful beta-blocker dosing (risk of bradycardia, heart block)
- Bone density screening and bisphosphonate consideration
- Drug interactions common (warfarin, amiodarone, digoxin)
Subclinical Hyperthyroidism Management:
| TSH Level | Age | Symptoms | Comorbidities | Recommendation |
|---|---|---|---|---|
| 0.1-0.4 mIU/L | \u003c65 | None | None | Observe, repeat TFTs in 3-6 months |
| \u003c0.1 mIU/L | \u003c65 | None | None | Consider treatment if persistent |
| \u003c0.1 mIU/L | ≥65 | Any | Any (AF, HF, osteoporosis) | Treat (Grade B recommendation) [1] |
| \u003c0.1 mIU/L | Any | Present | Heart disease | Treat |
Pediatric to Adult Transition:
- Graves' disease in adolescents has higher remission rate with ATD (50-60%)
- Transition to adult endocrinology at age 16-18
- Consider definitive therapy (RAI/surgery) if multiple relapses by late teens
- Emphasize contraception in females on ATD
Amiodarone-Induced Thyrotoxicosis (AIT):
| Feature | Type 1 AIT | Type 2 AIT | Mixed |
|---|---|---|---|
| Mechanism | Excess iodine substrate (Jod-Basedow) | Destructive thyroiditis | Both mechanisms |
| Pre-existing thyroid disease | Yes (nodules, Graves') | No (normal gland) | Variable |
| Thyroid vascularity (Doppler) | Increased | Decreased | Variable |
| RAIU | Normal/increased (if no prior iodine load) | Low/absent | Low |
| Treatment | Thionamides (MMI 40mg), potassium perchlorate | Glucocorticoids (prednisone 40-60mg) | Both ATD + steroids |
| Duration | Months (while on amiodarone) | Weeks to months (self-limited) | Variable |
Management Approach:
- Consult cardiology (amiodarone often for critical arrhythmia)
- Color flow Doppler ultrasound to differentiate Type 1 vs 2
- Consider empiric combined therapy if uncertainty
- Thyroidectomy if refractory and amiodarone cannot be stopped
- Monitor for hypothyroid phase (especially Type 2)
8.9 Disposition
- Admission Criteria: Thyroid storm, severe Afib with RVR, high-output heart failure, or severe thionamide-induced agranulocytosis.
- Discharge Criteria: Euthyroid (biochemically or clinically), heart rate less than 90 bpm, follow-up scheduled.
- Red Flags to Return: Fever/Sore throat (agranulocytosis), jaundice (hepatotoxicity), or worsening palpitations.
SECTION 9: Complications
9.1 Immediate Complications
| Complication | Incidence | Presentation | Risk Factors | Management |
|---|---|---|---|---|
| Atrial Fibrillation | 10-25% (age-dependent) | Palpitations, SOB, stroke | Age \u003e 60, pre-existing heart disease | Beta-blockers, rate control, anticoagulation [12] |
| Thyroid Storm | \u003c1% of thyrotoxic patients | Fever \u003e40°C, delirium, HF | Surgery, infection, trauma, iodine load | Multi-modal protocol (see 8.3) [15] |
| Thyrotoxic Periodic Paralysis | 2-10% (Asian males), \u003c1% (Caucasians) | Sudden flaccid weakness, hypokalemia | Young Asian males, high carb meals | Low-dose K+ (20-40 mEq), propranolol, treat hyperthyroidism |
| High-Output Heart Failure | 5-15% | Edema, orthopnea, elevated JVP | Pre-existing CAD, elderly, prolonged thyrotoxicosis | Diuretics, beta-blockers, treat thyrotoxicosis |
| Thyrotoxic Psychosis | \u003c1% | Hallucinations, delusions, mania | Severe untreated thyrotoxicosis | Antipsychotics + ATD, supportive care |
| Hyperemesis (Gestational) | 5-10% of pregnant women with thyrotoxicosis | Severe vomiting, dehydration, weight loss | High hCG (first trimester) | Hydration, antiemetics, usually self-limited |
Atrial Fibrillation Management in Thyrotoxicosis:
- Rate control: Beta-blockers (preferred), diltiazem, digoxin (less effective in high-adrenergic state)
- Anticoagulation: High stroke risk - consider anticoagulation even with low CHA₂DS₂-VASc if persistent
- Cardioversion: Often spontaneous reversion to sinus rhythm once euthyroid (60-70% within 3 months) [12]
- If AF persists \u003e6 months after euthyroid, consider as independent AF requiring long-term management
Thyrotoxic Periodic Paralysis:
- Pathophysiology: Increased Na+/K+-ATPase activity → intracellular K+ shift → hypokalemia → flaccid paralysis
- Typically affects proximal muscles, spares respiratory/bulbar muscles
- ECG: U waves, ST depression, prolonged QT (hypokalemia)
- Treatment: Avoid high-dose K+ replacement (rebound hyperkalemia common), use low-dose K+ 20-40 mEq PO
- Propranolol 3mg/kg reduces attack frequency
- Definitive: Treat hyperthyroidism (attacks cease once euthyroid)
9.2 Early Complications (Weeks to Months)
-
Agranulocytosis: (0.2-0.5%) Usually occurs in first 90 days of thionamides; higher with PTU than MMI. [11]
- "Presentation: Fever, sore throat, oral ulcers, sepsis"
- "Diagnosis: ANC \u003c500 cells/μL"
- "Management: STOP ATD immediately, G-CSF, broad-spectrum antibiotics, isolation"
- "Prognosis: 5-10% mortality if not recognized early"
- "Prevention: Baseline FBC, repeat at 2-4 weeks, patient education on warning signs"
-
Hepatotoxicity: (0.1-0.5%) More common with PTU (black box warning); can be cholestatic or hepatocellular.
- "Presentation: Jaundice, nausea, RUQ pain, elevated LFTs (ALT/AST \u003e3x ULN)"
- "PTU: Can cause fulminant hepatic failure requiring transplantation"
- "MMI: Typically cholestatic, reversible"
- "Management: STOP ATD, supportive care, consider alternative therapy"
- "Monitoring: Baseline LFTs, check if symptoms develop"
-
Graves' Orbitopathy Progression: (15-25% post-RAI, especially in smokers).
- "Mechanism: RAI-induced antigen release worsens immune activation"
- "Risk factors: Smoking, high TRAb, pre-existing mild eye disease"
- "Prevention: Concurrent glucocorticoids (prednisone 0.3-0.5 mg/kg for 6 weeks then taper) if moderate eye disease or smoker [8]"
- "Management: Ophthalmology referral, smoking cessation, selenium supplementation"
-
Hypocalcemia: (10-20% post-thyroidectomy) Transient in most, permanent in 1-2%.
- "Timing: Peaks at 24-48 hours post-op"
- "Symptoms: Perioral numbness, Chvostek/Trousseau signs, carpopedal spasm, seizures"
- "Management: Calcium carbonate 1-2g TID, calcitriol 0.25-0.5 mcg BID if severe/symptomatic"
- "Monitor: Serum calcium and PTH on post-op day 1 and 7"
-
Thrombocytopenia: (Rare, \u003c1%) Autoimmune association with Graves' disease.
9.3 Late Complications (Months to Years)
-
Osteoporosis and Fractures: Increased bone resorption leads to low BMD; 2-fold increased fracture risk, especially hip and vertebral. [16]
- "Mechanism: Excess thyroid hormone stimulates osteoclasts, increases bone turnover markers (CTx, P1NP)"
- "Monitoring: DEXA scan at diagnosis if prolonged untreated hyperthyroidism or post-menopausal women"
- "Prevention: Rapid treatment of hyperthyroidism, calcium/vitamin D supplementation"
- "Treatment: Bisphosphonates if T-score \u003c-2.5 or fragility fracture"
-
Post-Treatment Hypothyroidism: (90-100% post-RAI by 10 years, 100% post-total thyroidectomy).
- This is the intended/expected outcome of definitive therapy
- Requires lifelong levothyroxine replacement (typical dose 1.6 μg/kg/day)
- Target TSH 0.5-2.5 mIU/L (adjust based on symptoms and cardiovascular status)
- Annual TFT monitoring once stable
-
Recurrent Hyperthyroidism: (40-50% after stopping ATD within 5 years, higher if TRAb remains elevated). [3]
- "Predictors: Large goiter, persistently high TRAb, severe initial biochemistry, smoking, male sex"
- "Options after relapse: Prolonged ATD (not curative), RAI (preferred), or surgery"
- Multiple relapses indicate need for definitive therapy
-
Persistent Graves' Orbitopathy: (25-50% of Graves' patients have some degree).
- Natural history: Active phase (6-18 months inflammatory), then plateau, then "burnt out" fibrotic phase
- "Severity classification (EUGOGO):"
- Mild: 70% - observation, lubricants, selenium
- Moderate-to-severe: 25% - IV methylprednisolone, orbital radiotherapy
- Sight-threatening: 5% - urgent orbital decompression (optic neuropathy, corneal exposure)
- "Long-term: Residual proptosis, strabismus may require rehabilitative surgery [8,10]"
-
Thyrotoxic Cardiomyopathy: Dilated phenotype from chronic tachycardia and increased afterload.
- Usually reversible with treatment of hyperthyroidism
- If LVEF remains \u003c40% after 6 months euthyroid, consider as independent cardiomyopathy
-
Cognitive and Neuropsychiatric Effects: Anxiety, depression, attention deficits may persist even after biochemical cure.
- Consider screening and treatment for mood disorders
- Quality of life often impaired even in treated patients
SECTION 10: Prognosis \u0026 Outcomes
10.1 Natural History
Without treatment, hyperthyroidism from Graves' disease may occasionally fluctuate but usually leads to progressive weight loss, cardiac remodeling, and significant osteoporosis. The risk of embolic stroke is 3-fold higher in untreated thyrotoxic Afib. Toxic MNG never remits spontaneously and will eventually cause clinical thyrotoxicosis as nodules grow.
10.2 Outcomes with Treatment
| Variable | Outcome | Notes |
|---|---|---|
| Remission (Meds) | 40-50% at 18 mos | High relapse if TRAb remains high [3] |
| Cure Rate (RAI) | 80-90% (one dose) | 10-20% need second dose [14] |
| Cure Rate (Surg) | \u003e99% | Definitive but carries surgical risk [1] |
| 5-year Mortality | No increase vs gen pop | If euthyroid is maintained [5] |
| Afib Reversion | 60-70% revert spontaneously | Once euthyroid for 3 months [12] |
| Quality of Life | Improved but may not normalize | Persistent fatigue, anxiety in some |
| Cardiovascular Events | Reduced to baseline | Once euthyroid maintained \u003e12 months |
| Fracture Risk | Normalizes over 2-3 years | If euthyroid maintained [16] |
10.3 Prognostic Factors
- Good Prognosis: Small goiter, rapid response to thionamides, TRAb becomes negative within 1 year, age less than 40.
- Poor Prognosis: Large goiter (> 40g), severe biochemical hyperthyroidism at onset, persistent TRAb positivity, male sex, smokers.
SECTION 11: Evidence & Guidelines
11.1 Key Guidelines
Guideline 1: ATA 2016 Guidelines for Diagnosis and Management of Hyperthyroidism
- Organization: American Thyroid Association (ATA).
- Key Recommendation 1: Methimazole is the preferred drug for Graves' except in the first trimester of pregnancy (Grade A).
- Key Recommendation 2: Patients with subclinical hyperthyroidism and TSH less than 0.1 mIU/L should be treated if > 60 years or with comorbidities (Grade B).
- Key Recommendation 3: Total thyroidectomy is the surgical procedure of choice for Graves' (Grade A).
- Link: PMID: 27515128
Guideline 2: ETA 2018 Guidelines for Management of Graves' Hyperthyroidism
- Organization: European Thyroid Association (ETA).
- Key Recommendation: 12-18 months of thionamide therapy is standard for a first episode of Graves' (Grade 1, Strong).
11.2 Landmark Trials
TRIAL 1: Abraham et al. (2010) - Antithyroid drug regimen for Graves' disease
- Study Design: Cochrane Systematic Review of 26 RCTs.
- Patients: n = 3,388.
- Intervention: Titration regimen vs. Block-and-Replace.
- Key Finding: Titration regimen had fewer side effects with similar remission rates.
- PMID: 20044434
TRIAL 2: Torring et al. (1996) - The Swedish Hyperthyroidism Study
- Study Design: Prospective RCT.
- Intervention: Methimazole vs. RAI vs. Surgery.
- Key Finding: Relapse rates were 37% for meds, 14% for RAI, and 0% for surgery.
- PMID: 8615463
TRIAL 3: EUGOGO Trials (2011)
- Study Design: RCT.
- Intervention: IV Methylprednisolone vs Oral Prednisone for Graves' Orbitopathy.
- Key Finding: IV steroids were more effective and had fewer side effects for moderate-to-severe eye disease.
- PMID: 12110731
11.3 Evidence Strength Table
| Intervention | Evidence Level | Key Evidence | Recommendation Grade |
|---|---|---|---|
| Methimazole for Graves' | 1a | Abraham et al. | Grade A |
| RAI for Toxic MNG | 1b | ATA Guidelines | Grade A |
| Beta-blockers for symptoms | 1a | Meta-analysis | Grade A |
| Selenium for Orbitopathy | 1b | Marcocci et al. | Grade B |
| Surgery for Large Goiter | 2a | Observational | Grade B |
SECTION 14: Patient/Layperson Explanation
14.1 What is Hyperthyroidism?
Think of your thyroid gland as the body's furnace controller. It sits in your neck and produces a hormone that tells your body how fast to run. In hyperthyroidism, the furnace is stuck on "high." Your body starts burning through energy too quickly, which is why your heart races, you feel hot, and you lose weight even if you are eating more than usual.
14.2 Why Does It Matter?
If left untreated, this "overdrive" state can wear out your heart, lead to a shaky heart rhythm called atrial fibrillation, and make your bones weak and brittle (osteoporosis). In very rare cases, it can cause a "thyroid storm," which is a dangerous medical emergency.
14.3 How Is It Treated?
- Medications: Pills like methimazole slow down the thyroid. Most people take these for 12 to 18 months.
- Radioactive Iodine: A one-time capsule that gently shrinks the thyroid over several months.
- Surgery: Removing the thyroid gland entirely.
- Beta-blockers: These don't fix the thyroid, but they stop the "racing" feeling and tremors almost immediately.
14.4 When to Seek Help
See a doctor immediately if you have:
- A heart rate that won't slow down even at rest.
- A high fever combined with confusion.
- Sudden bulging of the eyes or double vision.
- A severe sore throat or fever while taking thyroid medication (this could be a rare side effect).
SECTION 12: Clinical Cases and Scenarios
12.1 Case 1: Classic Graves' Disease
Presentation: A 35-year-old female marketing executive presents with a 3-month history of palpitations, heat intolerance, and unintentional weight loss of 8 kg despite increased appetite. She reports anxiety, tremor, and oligomenorrhea. Examination reveals HR 110 bpm regular, diffuse smooth goiter, fine tremor, and bilateral exophthalmos with lid lag.
Investigations:
- TSH \u003c0.01 mIU/L (0.4-4.0)
- Free T4 45 pmol/L (10-20)
- Free T3 15 pmol/L (3.5-6.5)
- TRAb 12 IU/L (\u003c1.8 positive)
- TPO antibodies positive
Diagnosis: Graves' disease (overt hyperthyroidism with positive TRAb and clinical orbitopathy)
Management:
- Start propranolol 40mg TID for symptom control
- Initiate methimazole 20mg daily (titration regimen)
- Ophthalmology referral for baseline assessment
- Smoking cessation counseling (critical for eye disease)
- Monitor TFTs at 4-6 weeks, adjust MMI dose
- Plan 12-18 month course, check TRAb at 12 months
- Discuss definitive options (RAI/surgery) if relapse after ATD withdrawal
Teaching Points:
- TRAb confirms Graves' etiology; no need for RAIU scan
- Exophthalmos is pathognomonic for Graves' (not seen in other causes)
- Smoking dramatically worsens orbitopathy
- 50% relapse after ATD cessation; monitor closely
12.2 Case 2: Toxic Multinodular Goiter in Elderly
Presentation: A 72-year-old man with long-standing goiter presents with new-onset atrial fibrillation and unexplained weight loss. He denies classic "hyper" symptoms but reports fatigue and mild dyspnea on exertion. Examination reveals irregular pulse 130 bpm, multinodular goiter, no eye signs, no tremor.
Investigations:
- TSH \u003c0.01 mIU/L
- Free T4 28 pmol/L (mildly elevated)
- Free T3 8 pmol/L (elevated)
- TRAb negative
- Thyroid ultrasound: Multiple nodules 1-3cm, heterogeneous
- RAIU scan: Patchy uptake in multiple nodules
Diagnosis: Toxic multinodular goiter with "apathetic hyperthyroidism" complicated by atrial fibrillation
Management:
- Start metoprolol 25mg BD for rate control
- Anticoagulation (high CHA₂DS₂-VASc score: age 72, AF = 3 points) - DOAC preferred
- Methimazole 10mg daily to achieve euthyroid state
- Plan radioiodine ablation once euthyroid (definitive - TMNG never remits)
- Alternatively, surgical thyroidectomy if large/compressive goiter
- DEXA scan given age and hyperthyroidism (osteoporosis screening)
Teaching Points:
- "Apathetic hyperthyroidism" common in elderly - lacks classic hyperactivity
- TMNG is autonomous - will not remit with ATD alone, needs definitive therapy
- AF in thyrotoxicosis requires anticoagulation consideration regardless of CHA₂DS₂-VASc
- 60-70% AF reverts spontaneously once euthyroid maintained \u003e3 months
12.3 Case 3: Thyroid Storm
Presentation: A 28-year-old woman with known but poorly controlled Graves' disease presents to ED with fever (40.2°C), severe agitation, confusion, tachycardia 155 bpm, and profuse diaphoresis. She had recent dental extraction 2 days ago (trigger). Examination shows diffuse goiter, tremor, warm peripheries, wide pulse pressure.
Investigations:
- Clinical diagnosis (do not delay treatment for labs)
- Burch-Wartofsky Score: Temperature 15 + CNS 20 + HR 25 + GI 10 + Precipitant 10 = 80 points (HIGH - thyroid storm confirmed)
- TSH \u003c0.01, fT4 65 pmol/L, fT3 22 pmol/L
- Blood cultures, urine cultures (rule out sepsis)
Emergency Management (Multi-Modal Protocol):
- ICU admission - continuous monitoring
- Propranolol 60-80mg PO Q4H (or esmolol IV if unable to take PO)
- PTU 200mg PO/NGT Q4H (preferred - blocks T4→T3 conversion)
- Lugol's iodine 5 drops TID (AFTER PTU, wait 1 hour)
- Hydrocortisone 100mg IV Q8H
- Cooling (avoid aspirin), IV fluids 3-5L/day
- Treat precipitant - antibiotics for dental infection
- Consider plasmapheresis if refractory
Outcome: Patient stabilized over 48 hours, fever resolved, HR down to 95 bpm. Transitioned to oral beta-blockers and methimazole. Planned for definitive thyroidectomy once stable (to prevent future storm risk).
Teaching Points:
- Thyroid storm is clinical diagnosis - do not wait for labs
- Burch-Wartofsky score ≥45 = storm (this patient scored 80)
- Order of treatment critical: Thionamide BEFORE iodine (prevents iodine organification)
- Mortality 10-30% even with treatment - requires ICU care
- Definitive therapy (surgery/RAI) recommended after recovery to prevent recurrence
12.4 Case 4: First Trimester Pregnancy with Hyperthyroidism
Presentation: A 29-year-old woman at 8 weeks gestation presents with hyperemesis gravidarum, tachycardia 105 bpm, and weight loss of 3 kg. She denies previous thyroid disease. No goiter, no eye signs. Obstetric history: G2P1.
Investigations:
- TSH \u003c0.01 mIU/L
- Free T4 30 pmol/L
- Free T3 9 pmol/L
- TRAb: Negative
- Beta-hCG: 180,000 IU/L (very high for gestational age)
Diagnosis: Gestational transient thyrotoxicosis (high hCG-mediated) vs early Graves' disease
Differential Points:
- TRAb negative favors gestational thyrotoxicosis
- Hyperemesis + high hCG typical
- No goiter/eye signs (Graves' less likely)
- Should resolve by 18-20 weeks as hCG falls
Management:
- Conservative - hydration, antiemetics, nutritional support
- Propranolol 10-20mg TID for symptom control (safe in pregnancy)
- NO antithyroid drugs if TRAb negative and mild thyrotoxicosis
- Repeat TFTs at 14-16 weeks (should normalize)
- If TRAb positive or severe symptoms: Start PTU 100mg TID (preferred first trimester)
- Fetal monitoring - ultrasound growth scans
- Plan to switch to MMI in second trimester if PTU needed
Teaching Points:
- Gestational thyrotoxicosis is self-limited (high hCG first trimester)
- TRAb testing critical to distinguish from Graves'
- PTU preferred first trimester (MMI embryopathy risk)
- Maternal TRAb can cross placenta - risk of fetal/neonatal thyrotoxicosis
- Target fT4 upper normal range in pregnancy (avoid over-treatment)
12.5 Case 5: Amiodarone-Induced Thyrotoxicosis
Presentation: A 65-year-old man on amiodarone for recurrent ventricular tachycardia (started 8 months ago) develops palpitations, weight loss, and tremor. Cardiology reluctant to stop amiodarone given malignant arrhythmia.
Investigations:
- TSH \u003c0.01 mIU/L
- Free T4 38 pmol/L
- Free T3 11 pmol/L
- Thyroid ultrasound with color Doppler: Increased vascularity
- RAIU: Normal/high uptake (if no recent iodinated contrast)
Diagnosis: Amiodarone-induced thyrotoxicosis Type 1 (increased vascularity suggests iodine-induced synthesis in abnormal gland)
Management:
- Methimazole 40mg daily (high dose for Type 1)
- Potassium perchlorate 200mg TID (blocks iodine uptake) - if available
- Cardiology consult - can amiodarone be stopped? (often cannot)
- Monitor TFTs closely
- If refractory or Type 2 features develop: Add prednisone 40mg daily
- Definitive: Thyroidectomy if medical management fails and amiodarone must continue
Alternative Scenario - Type 2 AIT:
- Color Doppler: Decreased vascularity
- RAIU: Low/absent
- Treatment: Prednisone 40-60mg daily (destructive thyroiditis)
- Usually self-limited over 2-4 months
- Monitor for hypothyroid phase
Teaching Points:
- Amiodarone 37% iodine by weight - massive iodine load
- Type 1: Jod-Basedow effect (predisposed gland uses excess iodine)
- Type 2: Destructive thyroiditis (cytotoxic effect of amiodarone)
- Mixed type common - may need combined ATD + steroids
- Thyroidectomy may be needed if amiodarone cannot be stopped
12.6 Case 6: Subclinical Hyperthyroidism Management Dilemma
Presentation: A 68-year-old woman found to have low TSH on routine screening. She denies symptoms. Past medical history: hypertension, osteopenia (T-score -1.8).
Investigations:
- TSH 0.03 mIU/L (\u003c0.1, persistently low on repeat)
- Free T4 18 pmol/L (10-20, normal)
- Free T3 6.2 pmol/L (3.5-6.5, normal)
- TRAb negative
- Thyroid ultrasound: 2.5cm nodule right lobe
Diagnosis: Subclinical hyperthyroidism (likely toxic adenoma)
Risk Stratification:
- Age \u003e65: ✓ (increased risk AF, osteoporosis, mortality)
- TSH \u003c0.1: ✓ (higher risk than 0.1-0.4)
- Osteopenia: ✓ (fracture risk)
- Cardiac symptoms: ✗
Management Decision (ATA Guidelines): TREAT (Grade B recommendation for age \u003e60 with TSH \u003c0.1)
Treatment Options:
- Radioiodine ablation - preferred for toxic adenoma (definitive)
- Alternative: Thyroid lobectomy - especially if nodule has suspicious features
- Monitor closely if patient refuses definitive therapy
Rationale:
- Subclinical hyperthyroidism in elderly increases risk:
- Atrial fibrillation (3-fold increase)
- All-cause mortality (1.2-fold increase)
- Fractures (2-fold increase with existing osteopenia)
- Toxic adenoma will not remit - treatment indicated
Teaching Points:
- Subclinical hyperthyroidism: TSH low, fT4/fT3 normal
- Not always "benign"
- significant cardiovascular and skeletal risks
- Age \u003e60 + TSH \u003c0.1 = treatment recommended (ATA Grade B)
- Younger patients (\u003c65) with TSH 0.1-0.4 and no symptoms: observe
- Toxic adenoma/TMNG requires definitive therapy (RAI/surgery)
SECTION 13: Monitoring and Follow-up Protocols
13.1 Monitoring Antithyroid Drug Therapy
Baseline (Before Starting ATD):
- TSH, free T4, free T3
- TRAb/TSI (confirms Graves' etiology)
- FBC with differential (baseline WBC, neutrophils)
- LFTs (AST, ALT, bilirubin, ALP)
- Pregnancy test (women of childbearing age)
- Document goiter size (palpation or ultrasound)
First 3 Months (Highest Risk Period):
| Week | Tests | Rationale | Action |
|---|---|---|---|
| 2 | FBC | Agranulocytosis risk peaks | If ANC \u003c1000, repeat; if \u003c500 STOP ATD |
| 4 | TFTs, FBC | Dose adjustment, agranulocytosis screening | Adjust MMI dose based on fT4 |
| 8 | TFTs | Monitor response | Reduce MMI if fT4 normalizing |
| 12 | TFTs, FBC, LFTs | Dose stabilization, safety | Further dose reduction if euthyroid |
Months 3-18 (Maintenance Phase):
- TFTs every 6-8 weeks until stable
- TFTs every 3 months once stable on maintenance dose
- FBC if symptoms (fever, sore throat)
- LFTs if symptoms (jaundice, RUQ pain)
- TRAb at 6-12 months (predicts remission)
Month 18 (Pre-Withdrawal):
- Check TRAb: If negative, good chance of remission
- If TRAb high, discuss definitive therapy (RAI/surgery)
- Taper ATD over 4-6 weeks (or stop abruptly - similar outcomes)
Post-ATD Withdrawal:
- TFTs at 1, 2, 3, 6, and 12 months
- 50% relapse within first 6 months
- Continue monitoring annually for 5 years (late relapses occur)
Patient Education - Stop ATD and Seek Urgent Care If:
- Fever \u003e38°C
- Severe sore throat
- Oral ulcers
- Unexplained bruising or bleeding
- Jaundice or dark urine
- Severe abdominal pain
13.2 Monitoring Post-Radioiodine Therapy
Pre-RAI:
- Confirm non-pregnant (mandatory)
- Baseline TFTs
- Withdraw ATD 3-7 days before (allow iodine uptake)
- Low-iodine diet 1-2 weeks (controversial)
- Ophthalmology assessment if Graves' orbitopathy present
Week 1-2 Post-RAI:
- Monitor for radiation thyroiditis (transient worsening)
- Beta-blockers for symptoms
- Can restart ATD if severe symptoms (won't affect outcome)
Week 4-6:
- First TFT check
- Many still thyrotoxic (radiation effect takes time)
- Continue beta-blockers
Month 3:
- TFTs
- Many becoming euthyroid or hypothyroid
- Start levothyroxine if TSH rising
Month 6:
- TFTs
- 70-80% euthyroid or hypothyroid by now
- Adjust levothyroxine dose
Month 12:
- TFTs
- 10-20% may need second RAI dose if still hyperthyroid
- Annual monitoring thereafter
Long-term:
- 90%+ develop hypothyroidism by 10 years (expected outcome)
- Annual TFTs lifelong
- Adjust levothyroxine to maintain TSH 0.5-2.5 mIU/L
13.3 Post-Thyroidectomy Monitoring
Immediate Post-op (24-48 hours):
- Serum calcium (check at 6 hours and 24 hours post-op)
- PTH level (if calcium low)
- Monitor for hematoma (neck swelling, stridor)
- Assess voice/vocal cords if any concerns
Day 1-7:
- Serum calcium on day 1 and day 7 (hypocalcemia peaks at 24-48 hours)
- If calcium \u003c2.0 mmol/L or symptomatic: Start calcium + calcitriol
- Laryngoscopy if voice change
Week 2:
- Wound check
- Calcium monitoring if on supplements
Week 6-8:
- First TFTs on levothyroxine
- Adjust dose to target TSH 0.5-2.5 mIU/L
- Typical dose: 1.6 μg/kg/day
- If on calcium supplements, review need
Month 3-6:
- Repeat TFTs
- Adjust levothyroxine
- Check calcium if history of hypocalcemia
Annual Monitoring:
- TFTs annually
- Calcium if on long-term supplementation (permanent hypoparathyroidism)
- Adjust levothyroxine as needed (requirements may change with age, weight)
13.4 Graves' Orbitopathy Monitoring
Baseline Assessment:
- Ophthalmology referral for all Graves' patients with eye signs
- Severity classification (EUGOGO):
- "Mild: 70% - soft tissue involvement only"
- "Moderate-to-severe: 25% - lid retraction \u003e2mm, moderate/severe soft tissue involvement, exophthalmos, diplopia"
- "Sight-threatening: 5% - optic neuropathy, corneal exposure"
Clinical Activity Score (CAS): Score 1 point for each present (max 7):
- Spontaneous retrobulbar pain
- Pain on eye movement
- Eyelid erythema
- Eyelid swelling
- Conjunctival injection
- Chemosis
- Caruncle/plica swelling
Active disease: CAS ≥3 (inflammation ongoing, may respond to immunosuppression) Inactive disease: CAS \u003c3 (burnt out fibrotic phase)
Monitoring Schedule:
| Phase | Frequency | Assessments | Interventions |
|---|---|---|---|
| Active (0-18 mo) | Monthly | CAS, visual acuity, exophthalmometry | Steroids, radiotherapy if mod-severe |
| Plateau (18-24 mo) | 3-monthly | CAS, visual acuity | Optimize thyroid control |
| Burnt Out (\u003e24 mo) | 6-12 monthly | Visual acuity, degree of proptosis | Rehabilitative surgery if needed |
Red Flags - Urgent Ophthalmology:
- Sudden vision loss (optic neuropathy)
- Severe eye pain
- Unable to close eyes (corneal exposure risk)
- Rapidly progressive proptosis
Treatment Monitoring:
- If on IV methylprednisolone: Monitor glucose, BP, mood
- If on selenium: 100-200 mcg daily for 6 months (mild disease)
- If considering RAI: Give prophylactic steroids if active orbitopathy or smoker
13.5 Special Situations Monitoring
Pregnancy:
- Monthly TFTs throughout pregnancy
- Target fT4 upper normal range (avoid fetal hypothyroidism)
- Lowest effective ATD dose
- Measure maternal TRAb at 18-22 weeks and 30-34 weeks if high
- If TRAb \u003e3x ULN: Fetal/neonatal thyrotoxicosis risk - neonatal monitoring needed
Pediatric Transition to Adult Care:
- Transfer at age 16-18
- Review treatment history, number of relapses
- Discuss definitive therapy options
- Ensure contraception discussed (females on ATD)
- Continue monitoring as adult protocols
Subclinical Hyperthyroidism:
- If not treating: Repeat TFTs every 3-6 months
- Monitor for progression to overt disease
- Annual DEXA if risk factors for osteoporosis
- ECG annually (screen for AF)
- Consider treatment if TSH \u003c0.1 persists \u003e6 months in elderly
SECTION 15: References
- Ross DS, et al. 2016 American Thyroid Association Guidelines for Diagnosis and Management of Hyperthyroidism. Thyroid. 2016;26(10):1343-1421. PMID: 27515128
- Kahaly GJ, et al. 2018 European Thyroid Association Guideline for the Management of Graves' Hyperthyroidism. Eur Thyroid J. 2018;7(4):167-186. PMID: 30283735
- Abraham P, et al. Antithyroid drug regimen for treating Graves' hyperthyroidism. Cochrane Database Syst Rev. 2010;(1):CD003420. PMID: 20044434
- Torring O, et al. Training, surgery, or radioiodine: a prospective, randomized study of treatment for Graves' hyperthyroidism. J Clin Endocrinol Metab. 1996;81(8):2986-93. PMID: 8615463
- De Leo S, et al. Hyperthyroidism. Lancet. 2016;388(10053):1514-1526. PMID: 27011420
- Burch HB, et al. Life-threatening thyrotoxicosis. Thyroid storm. Endocrinol Metab Clin North Am. 1993;22(2):263-77. PMID: 8325286
- Marcocci C, et al. Selenium and the course of mild Graves' orbitopathy. N Engl J Med. 2011;364(20):1920-31. PMID: 21591944
- Bartalena L, et al. The 2021 European Group on Graves' Orbitopathy (EUGOGO) Clinical Practice Guidelines. Eur J Endocrinol. 2021;185(4):G43-G67. PMID: 34233270
- Smith TJ, et al. Teprotumumab for Thyroid-Associated Ophthalmopathy. N Engl J Med. 2017;376(18):1748-1761. PMID: 28467880
- Bahn RS. Graves' ophthalmopathy. N Engl J Med. 2010;362(8):726-38. PMID: 20181974
- Cooper DS. Antithyroid drugs. N Engl J Med. 2005;352(9):905-17. PMID: 15745981
- Franklyn JA, et al. Thyrotoxicosis. Lancet. 2012;379(9821):1155-66. PMID: 22394550
- Hegedüs L. The thyroid nodule. N Engl J Med. 2004;351(17):1764-71. PMID: 15496625
- Sundaresh V, et al. Comparative effectiveness of antithyroid drugs, radioactive iodine, and surgery for Graves' disease. Thyroid. 2013;23(12):1522-31. PMID: 23745688
- Akamizu T. Thyroid Storm: A Review of Criteria and Management. Thyroid. 2018;28(1):32-40. PMID: 29191118
- Vestergaard P, et al. Fractures in patients with hyperthyroidism and hypothyroidism: a nationwide follow-up study in 16,249 patients. Thyroid. 2002;12(5):411-9. PMID: 12097203
- Ylli D, et al. Evaluation and treatment of amiodarone-induced thyroid disorders. J Clin Endocrinol Metab. 2021;106(1):226-236. PMID: 33236116
- Stagnaro-Green A, et al. Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and postpartum. Thyroid. 2011;21(10):1081-125. PMID: 21787128
- Samuels MH. Subclinical thyroid disease in the elderly. Thyroid. 1998;8(9):803-13. PMID: 9777751
- Bogazzi F, et al. Color flow Doppler sonography rapidly differentiates type I and type II amiodarone-induced thyrotoxicosis. Thyroid. 1997;7(4):541-5. PMID: 9292940
- Pearce EN, et al. Thyrotoxicosis. N Engl J Med. 2016;375(17):1672-1683. PMID: 27783916
- Menconi F, et al. Epidemiology of Graves' disease. In: Wiersinga WM, Drexhage HA, eds. The Thyroid and Autoimmunity: The Wiersinga Festschrift. Georg Thieme Verlag; 2007. PMID: 20821855
- Cappelli C, et al. The management of thyroid nodules and subclinical thyroid dysfunction during pregnancy. Endocrine. 2014;47(2):426-34. PMID: 24532102
- Wartofsky L. Clinical criteria for the diagnosis of thyroid storm. Thyroid. 2012;22(7):659-60. PMID: 22690854
- Douglas RS, et al. Teprotumumab for the Treatment of Active Thyroid Eye Disease. N Engl J Med. 2020;382(4):341-352. PMID: 31971679
SECTION 16: Examination Focus
16.1 Common Exam Questions
- MRCP: "A 32-year-old woman at 10 weeks gestation has a TSH less than 0.01. What is the most appropriate first-line treatment?"
- Answer: Propylthiouracil (PTU) due to 1st-trimester methimazole teratogenicity.
- USMLE Step 2: "A patient with Graves' disease is started on methimazole and presents 2 weeks later with a sore throat and fever. What is the next step?"
- Answer: Immediate Full Blood Count (FBC) to rule out agranulocytosis.
- Medical Student Viva: "What is the difference between thyrotoxicosis and hyperthyroidism?"
- Answer: Thyrotoxicosis is the state of excess hormone (from any source, e.g., thyroiditis); hyperthyroidism is specifically excess production by the gland.
- MRCP Part 2: "A 70-year-old man with new-onset atrial fibrillation has TSH \u003c0.01, fT4 25, TRAb negative. What is the most likely diagnosis?"
- Answer: Toxic multinodular goiter (elderly, TRAb negative, likely nodular goiter on exam).
- FRCS: "What are the key steps in pre-operative preparation for thyroidectomy in a thyrotoxic patient?"
- Answer: Achieve euthyroid state with ATD, beta-blockade, Lugol's iodine 7-10 days pre-op to reduce vascularity, vocal cord assessment.
- MRCOG: "A pregnant woman at 12 weeks gestation on PTU develops jaundice. What is the next step?"
- Answer: STOP PTU immediately (hepatotoxicity), check LFTs, switch to methimazole (safe after first trimester), consider surgery if severe.
- FRACP: "How would you differentiate Type 1 from Type 2 amiodarone-induced thyrotoxicosis?"
- Answer: Color flow Doppler ultrasound - Type 1 shows increased vascularity (Jod-Basedow), Type 2 shows decreased vascularity (destructive thyroiditis). RAIU also helpful but often not feasible with iodine load.
- PLAB: "A patient on methimazole for 3 weeks presents to ED with fever 38.5°C and sore throat. FBC pending. What is your immediate management?"
- Answer: STOP methimazole immediately (suspect agranulocytosis), isolate patient, broad-spectrum antibiotics if ANC \u003c500, G-CSF if confirmed agranulocytosis.
16.2 Viva Opening Statement
"Hyperthyroidism is a clinical syndrome of hypermetabolism resulting from excess thyroid hormone production by the thyroid gland, most commonly due to Graves' disease in adults. It is characterized by multi-systemic features like tachycardia, weight loss, and tremor, and is clinically significant due to risks of atrial fibrillation and thyroid storm. Diagnosis is confirmed by a suppressed TSH and elevated free T4/T3, and management involves a choice between thionamides, radioactive iodine, or surgery depending on the patient's comorbidities and preference."
16.3 Key Facts to Mention
- Pathophysiology: TSI mimics TSH at the receptor.
- Investigation: RAIU differentiates Graves' (diffuse) from Thyroiditis (low uptake).
- Management: Methimazole is the gold standard for Graves' remission.
- Complication: Agranulocytosis is rare but fatal; thyroid storm has high mortality.
16.4 Common Mistakes
- ❌ Prescribing methimazole in the first trimester of pregnancy.
- ❌ Ordering an RAIU scan in a pregnant patient (contraindicated).
- ❌ Forgetting to check LFTs and FBC before starting thionamides.
- ❌ Treating subclinical hyperthyroidism in a young, asymptomatic patient without risk factors.
- ❌ Giving Lugol's iodine before a thionamide in thyroid storm (causes the "Jod-Basedow" effect).
SECTION 17: Advanced Pathophysiology and Molecular Mechanisms
17.1 Thyroid Hormone Synthesis - Detailed Steps
The thyroid follicular cell is a polarized epithelial cell with distinct apical (facing colloid) and basolateral (facing bloodstream) membranes. The synthesis of thyroid hormones involves a precisely orchestrated sequence of molecular events. [21]
Step 1: Iodide Uptake (Basolateral Membrane) The Sodium-Iodide Symporter (NIS, encoded by SLC5A5 gene) actively transports iodide (I⁻) from the bloodstream into the follicular cell against both concentration and electrical gradients. This is coupled to the Na⁺/K⁺-ATPase pump which maintains the sodium gradient. In hyperthyroidism, NIS expression is upregulated, leading to increased iodide trapping (the basis for radioiodine therapy). [5]
Step 2: Iodide Efflux to Colloid (Apical Membrane) Iodide exits the follicular cell into the colloid via Pendrin (SLC26A4) and other apical iodide transporters. This step ensures adequate iodide concentration in the colloid for organification.
Step 3: Oxidation and Organification Thyroid Peroxidase (TPO), a heme-containing enzyme located on the apical membrane, catalyzes two critical reactions:
- Oxidation: I⁻ + H₂O₂ → I₂ (reactive iodine species)
- Organification: Reactive iodine is covalently bound to tyrosine residues on thyroglobulin (Tg), forming monoiodotyrosine (MIT) and diiodotyrosine (DIT)
Hydrogen peroxide (H₂O₂) is generated by dual oxidase 2 (DUOX2) and is essential for TPO activity. [1]
Step 4: Coupling Reaction TPO also catalyzes the coupling of iodotyrosines within the thyroglobulin molecule:
- MIT + DIT → Triiodothyronine (T3)
- DIT + DIT → Thyroxine (T4)
The ratio of T4:T3 produced is approximately 10:1 under normal conditions, but this ratio shifts toward increased T3 production in hyperthyroidism (especially Graves' disease). [21]
Step 5: Storage in Colloid Iodinated thyroglobulin (containing MIT, DIT, T3, T4) is stored in the colloid. The thyroid contains a 2-3 month supply of stored hormone, which is why symptoms of hypothyroidism after total thyroidectomy can take weeks to manifest.
Step 6: Endocytosis and Hormone Release Upon TSH (or TSI) stimulation, follicular cells endocytose colloid droplets containing thyroglobulin. These vesicles fuse with lysosomes, where proteases (cathepsins) cleave thyroglobulin to release free T4 and T3. The hormones are then secreted across the basolateral membrane into the bloodstream via monocarboxylate transporter 8 (MCT8) and other transporters. [1]
17.2 Peripheral Thyroid Hormone Metabolism
Once in circulation, approximately 99.97% of T4 and 99.7% of T3 are bound to transport proteins (thyroxine-binding globulin [TBG], transthyretin, albumin). Only the free (unbound) fractions are biologically active and able to enter target cells.
Deiodination Pathways: Three deiodinase enzymes regulate the conversion and degradation of thyroid hormones:
| Enzyme | Location | Function | Clinical Relevance |
|---|---|---|---|
| D1 | Liver, kidney, thyroid | T4 → T3 (activation), rT3 → T2 (degradation) | Contributes 40% of circulating T3 |
| D2 | Brain, pituitary, brown fat, skeletal muscle | T4 → T3 (local activation) | Regulates local tissue T3 levels |
| D3 | Placenta, brain, skin | T4 → rT3 (inactivation), T3 → T2 (degradation) | Protective in severe illness (low T3 syndrome) |
In hyperthyroidism, D1 activity increases, leading to enhanced peripheral conversion of T4 to T3, which explains why T3 levels are often disproportionately elevated compared to T4 ("T3-toxicosis"). [5,21]
Thyroid Hormone Receptor (TR) Signaling: T3 enters the nucleus and binds to thyroid hormone receptors (TR-α and TR-β), which are members of the nuclear receptor superfamily. These receptors form heterodimers with retinoid X receptors (RXR) and bind to thyroid hormone response elements (TREs) in the promoter regions of target genes. T3 binding causes dissociation of corepressors and recruitment of coactivators, leading to increased transcription of genes involved in metabolism, cardiac function, and bone turnover. [21]
17.3 Graves' Disease Immunopathology - Detailed Mechanisms
Graves' disease is a classic organ-specific autoimmune disorder characterized by the production of thyroid-stimulating immunoglobulins (TSI), a subset of TSH receptor antibodies (TRAb). [2]
Genetic Susceptibility: Twin studies show 70-80% concordance in monozygotic twins, indicating strong heritability. Key susceptibility genes include:
- HLA-DR3 (DRB1*03): 3.5-fold increased risk, particularly in Caucasians
- CTLA-4 (Cytotoxic T-Lymphocyte Antigen 4): Negative regulator of T-cell activation
- PTPN22 (Protein Tyrosine Phosphatase Non-receptor type 22): Involved in T-cell receptor signaling
- CD40: Regulates B-cell and T-cell interactions
- TSHR: Polymorphisms in the TSH receptor gene itself
These genetic variants disrupt immune tolerance mechanisms. [22]
Environmental Triggers:
- Infection: Molecular mimicry (Yersinia enterocolitica shares epitopes with TSH receptor)
- Stress: Cortisol alterations shift Th1/Th2 balance
- Smoking: Increases risk of Graves' orbitopathy 7-8 fold; mechanisms include hypoxia, oxidative stress, and direct effects on orbital fibroblasts
- Pregnancy: Postpartum period sees immune rebound (pregnancy is immunosuppressive)
- Iodine excess: Jod-Basedow phenomenon in predisposed glands
- Immune checkpoint inhibitors: Anti-CTLA-4 and anti-PD-1 therapies (used in oncology) can trigger Graves' by unleashing immune responses
Pathogenic Cascade:
- Antigen Presentation: TSH receptor peptides presented by MHC class II on thyroid dendritic cells
- T-cell Activation: CD4+ T-helper cells recognize TSHR peptides; Th2-predominant response
- B-cell Activation: Activated T-cells provide help to B-cells
- TSI Production: B-cells differentiate into plasma cells secreting TSI (IgG1 subclass)
- Thyroid Stimulation: TSI binds to extracellular domain of TSHR, causing prolonged cAMP activation
- Perpetuation: Intrathyroidal T-cell and B-cell infiltration creates a self-sustaining autoimmune response
TSH Receptor Antibody Subtypes:
- Stimulating (TSI): Mimic TSH, cause hyperthyroidism (90% of Graves' patients)
- Blocking (TBAb): Block TSH binding, can cause hypothyroidism (rare, or in neonates of mothers with high TBAb)
- Neutral: Bind but don't alter function
The balance between these antibodies explains fluctuations between hyper- and hypothyroidism in some Graves' patients. [1,2]
17.4 Cardiovascular Effects of Thyroid Hormone - Molecular Details
Thyroid hormones have profound effects on the cardiovascular system, explaining why cardiac complications dominate the clinical presentation and prognosis of hyperthyroidism. [12]
Direct Cardiac Effects (Genomic):
- Increased α-myosin heavy chain (α-MHC): Faster ATPase activity → increased contractility
- Decreased β-myosin heavy chain (β-MHC): Shifts to faster isoform
- Increased SERCA2 (Sarcoplasmic Reticulum Ca²⁺-ATPase): Faster calcium reuptake → faster relaxation (lusitropic effect)
- Increased β1-adrenergic receptors: 2-3 fold upregulation → heightened catecholamine sensitivity
- Decreased phospholamban: Removes brake on SERCA2
- Increased voltage-gated potassium channels: Shortens action potential duration
These changes collectively increase heart rate (positive chronotropy), contractility (positive inotropy), and relaxation rate (positive lusitropy). [12]
Non-Genomic Effects (Rapid, within minutes):
- Direct membrane effects on ion channels
- Activation of PI3K/Akt signaling
- Modulation of Na⁺/K⁺-ATPase activity
Hemodynamic Consequences:
- Increased cardiac output: 50-300% above normal (high-output state)
- Decreased systemic vascular resistance: Peripheral vasodilation mediated by nitric oxide
- Increased blood volume: Enhanced erythropoietin and aldosterone
- Wide pulse pressure: Elevated systolic BP (increased stroke volume), decreased diastolic BP (decreased SVR)
Atrial Fibrillation Mechanisms: The risk of AF increases with age and severity of thyrotoxicosis:
- Age less than 40: 2-5% prevalence
- Age 40-60: 10-15%
- Age > 60: 20-35%
Mechanisms include:
- Atrial fibrosis and remodeling
- Shortened atrial refractory period
- Increased atrial automaticity
- Enhanced pulmonary vein ectopy
AF in thyrotoxicosis carries a 3-5 fold increased risk of stroke, mandating anticoagulation consideration even if CHA₂DS₂-VASc score is low. [12]
17.5 Skeletal Effects and Osteoporosis
Thyroid hormones stimulate both osteoblasts and osteoclasts, but the net effect is increased bone resorption (osteoclast activity exceeds osteoblast activity). [16]
Mechanisms:
- Direct osteoclast stimulation: T3 upregulates RANKL (Receptor Activator of Nuclear Factor Kappa-B Ligand) on osteoblasts, which binds to RANK on osteoclast precursors, driving differentiation
- Decreased osteoprotegerin (OPG): OPG normally acts as a decoy receptor for RANKL; T3 decreases OPG production
- Increased IL-6 and IL-8: Pro-resorptive cytokines
- Decreased intestinal calcium absorption: Despite increased bone turnover
- Increased renal calcium excretion: Leads to hypercalciuria
Bone Turnover Markers:
- Increased: C-terminal telopeptide of type I collagen (CTx), N-terminal propeptide of type I procollagen (P1NP), alkaline phosphatase (bone-specific)
- These markers can be 2-4 times normal in overt hyperthyroidism
Fracture Risk:
- Hip fractures: 2.0-fold increased risk (95% CI 1.4-2.9)
- Vertebral fractures: 1.8-fold increased risk
- Wrist fractures: 1.6-fold increased risk
Risk is highest in postmenopausal women and persists for 2-3 years after achieving euthyroid status. [16]
SECTION 18: Special Clinical Scenarios and Pitfalls
18.1 Subclinical Hyperthyroidism - Evidence-Based Management
Subclinical hyperthyroidism (SCH) is defined as low or suppressed TSH with normal free T4 and T3. It is subdivided by severity:
- Grade 1: TSH 0.1-0.4 mIU/L (mild suppression)
- Grade 2: TSH less than 0.1 mIU/L (severe suppression)
Prevalence: 0.7-2.1% in the general population; higher in elderly and iodine-sufficient areas. [19]
Natural History:
- Grade 1: 1-2% per year progress to overt hyperthyroidism
- Grade 2: 5% per year progress to overt hyperthyroidism
- Spontaneous normalization: 40-60% in Grade 1 (especially if transient/medication-related)
Cardiovascular Risks (Meta-analyses):
- Atrial Fibrillation: HR 1.68 (95% CI 1.16-2.43) for TSH less than 0.1; HR 1.13 (0.96-1.32) for TSH 0.1-0.4
- All-cause Mortality: HR 1.24 (1.06-1.46) for TSH less than 0.1; no significant increase for 0.1-0.4
- Cardiovascular Mortality: HR 1.29 (1.02-1.62) for TSH less than 0.1
- Heart Failure: Increased risk in elderly with pre-existing cardiac disease
Skeletal Risks:
- Fractures: HR 1.36 (1.13-1.64) for TSH less than 0.1 in postmenopausal women
- Bone Mineral Density: Accelerated loss of 1-2% per year at hip and spine
Cognitive/Neuropsychiatric:
- Possible increased risk of dementia (HR 1.23, but data inconsistent)
- Anxiety and mood disturbances
Treatment Indications (ATA 2016 Guidelines):
| Clinical Scenario | TSH Level | Recommendation | Grade |
|---|---|---|---|
| Age ≥65, asymptomatic | less than 0.1 mIU/L | Treat | B (Strong) |
| Age ≥65, symptomatic | 0.1-0.4 mIU/L | Consider treatment | C (Weak) |
| Age less than 65, no comorbidities | less than 0.1 mIU/L | Observation, treat if persistent > 6 mo | C |
| Any age with osteoporosis or AF risk | less than 0.1 mIU/L | Treat | B |
| Toxic nodule/TMNG (any TSH) | Any suppression | Definitive therapy (RAI/surgery) | A |
Treatment Options:
- Toxic nodular disease: RAI or surgery (remission unlikely with ATD alone)
- Graves' disease: Methimazole (if early phase, may achieve remission) or definitive therapy
- Iatrogenic (levothyroxine over-replacement): Dose reduction
Monitoring if Not Treating:
- Repeat TFTs every 3-6 months
- Annual ECG (screen for AF)
- Bone density (DEXA) if postmenopausal woman or risk factors
- Re-evaluate if progression to overt or new symptoms [1,19]
18.2 Thyrotoxic Periodic Paralysis (TPP) - Asian Male Emergency
TPP is a rare but dramatic complication characterized by sudden-onset flaccid paralysis due to hypokalemia, occurring in 2-10% of Asian males with hyperthyroidism (vs less than 1% in Caucasians). [5]
Epidemiology:
- Male:Female ratio 20:1
- Highest incidence: Chinese, Japanese, Vietnamese, Korean, Thai
- Age: 20-40 years (younger than typical Graves' presentation)
- Genetic: Mutations in KCNJ18 (Kir2.6 potassium channel) identified in familial cases
Pathophysiology: Thyroid hormone upregulates Na⁺/K⁺-ATPase in skeletal muscle → excessive K⁺ influx into cells → extracellular hypokalemia → muscle membrane hyperpolarization → flaccid paralysis. This is exacerbated by high-carbohydrate meals (insulin further drives K⁺ into cells).
Triggers:
- High-carbohydrate meals (rice, pasta, sweets)
- Strenuous exercise followed by rest
- Alcohol consumption
- Emotional stress
- Cold exposure
Clinical Presentation:
- Sudden onset: Typically early morning (3-6 AM) or after large meal
- Symmetric proximal weakness: Legs > arms, spares respiratory and bulbar muscles
- Hyporeflexia or areflexia: Deep tendon reflexes diminished or absent
- Normal sensation: Pure motor paralysis
- Duration: 6-72 hours (self-limited if not treated)
- Recurrent: 50-70% have multiple attacks until euthyroid
Laboratory Findings:
- Hypokalemia: Typically 1.5-3.0 mmol/L (severe)
- Hypophosphatemia: Often present (K⁺ and PO₄³⁻ shift together)
- Normal total body K⁺: This is a redistribution, not true deficiency
- ECG changes: U waves, ST depression, prolonged QT, T-wave flattening, rarely ventricular arrhythmias
Thyroid Function:
- TSH suppressed, elevated T4/T3 (may be mild/subclinical hyperthyroidism)
- TRAb usually positive (Graves' most common etiology)
Emergency Management:
-
Potassium Replacement - USE CAUTION:
- Low-dose K⁺ only: 20-40 mEq PO (NOT IV unless life-threatening arrhythmia)
- Avoid aggressive replacement: Total body K⁺ is normal; risk of rebound hyperkalemia (can be severe, causing cardiac arrest)
- Monitor K⁺ every 2-4 hours
-
Propranolol:
- 3 mg/kg/day divided TID (e.g., 60-80 mg TID for 70 kg patient)
- Reduces Na⁺/K⁺-ATPase activity
- Prevents attacks and shortens duration
- More effective than K⁺ alone
-
Avoid Loop/Thiazide Diuretics: Worsen hypokalemia
-
Monitor Cardiac: Continuous ECG monitoring for arrhythmias
-
Definitive Thyroid Treatment:
- Start methimazole immediately
- Attacks cease once euthyroid achieved
- RAI or surgery if recurrent/refractory
Rebound Hyperkalemia Risk:
- Peaks 6-12 hours after attack resolves
- K⁺ exits cells back into ECF
- Monitor K⁺ for 24 hours
- Stop K⁺ supplementation once K⁺ > 3.5 mmol/L
Prevention of Recurrent Attacks:
- Propranolol until euthyroid
- Low-carbohydrate diet
- Avoid triggers
- Definitive thyroid treatment (recurrence rate 5-10% even with ATD alone; RAI/surgery preferred)
Differential Diagnosis:
- Familial hypokalemic periodic paralysis (no thyrotoxicosis)
- Barium poisoning (blocks K⁺ channels)
- Renal or GI K⁺ losses
- Primary hyperaldosteronism (but would have hypertension, metabolic alkalosis) [5]
18.3 Thyrotoxicosis in the Intensive Care Unit
Critically ill patients with hyperthyroidism present unique diagnostic and therapeutic challenges.
Diagnostic Pitfalls:
- Non-thyroidal illness syndrome (NTIS): Suppressed TSH can occur in severe sepsis, critical illness without thyroid disease
- Medication interference: Dopamine, high-dose glucocorticoids suppress TSH
- Free T4/T3 measurement issues: Heparin displaces T4 from binding proteins (falsely elevated free T4 by analog assays)
Differentiation:
| Feature | Thyrotoxicosis | NTIS |
|---|---|---|
| TSH | less than 0.01 mIU/L | Variable (low-normal to mildly suppressed) |
| Free T4 | Elevated (> 25 pmol/L) | Normal or low |
| Free T3 | Elevated | Low (hallmark of NTIS) |
| Total T4/T3 | Elevated | Low |
| Reverse T3 | Normal/Low | Elevated |
| History | Prior thyroid disease, autoimmune history | Critical illness without prior thyroid disease |
Management Considerations:
- Defer non-urgent TFT interpretation: Repeat when acute illness resolves
- If thyroid storm suspected: Treat empirically (see Section 8.3) - do not wait for confirmation
- Drug interactions: Propranolol may worsen shock; consider esmolol (ultra-short acting, titratable IV beta-blocker)
- Glucocorticoids: Already often on stress-dose steroids; ensure adequate coverage
- Nutritional support: Hypermetabolic state increases caloric needs by 20-60%
18.4 Iodine-Induced Hyperthyroidism (Jod-Basedow Phenomenon)
Excess iodine exposure in individuals with pre-existing thyroid autonomy (multinodular goiter, autonomous nodules) or latent Graves' disease can precipitate overt hyperthyroidism. [5]
Sources of Iodine Excess:
- Amiodarone: 37% iodine by weight; 200 mg tablet = 75 mg iodine (normal daily requirement 150 μg)
- Iodinated contrast: CT contrast can contain 15-60 g iodine
- Kelp/seaweed supplements: Variable, can exceed 1000 μg/day
- Topical antiseptics: Povidone-iodine (surgical prep, wound care)
- Medications: Expectorants (potassium iodide)
Latency:
- Amiodarone: 2-39 months after starting (average 8-12 months)
- Contrast: Days to 8 weeks
Clinical Features:
- Often more severe than spontaneous hyperthyroidism
- May precipitate thyroid storm in predisposed individuals
- Can occur even with prior normal thyroid (if subclinical autonomy)
Diagnosis:
- Low TSH, elevated T4/T3
- TRAb negative (unless underlying Graves')
- RAIU: Variable - may be normal/high (Type 1 AIT) or low (if recent iodine load saturates uptake)
- Thyroid ultrasound: Nodules, heterogeneity
Management:
- Thionamides: Methimazole 40-60 mg daily (high dose required due to large iodine substrate)
- Potassium Perchlorate (if available): 200 mg TID - competitively inhibits NIS, reduces iodine uptake (not available in all countries; monitor for aplastic anemia)
- Beta-blockers: Symptom control
- Avoid more iodine: No contrast studies, no kelp, etc.
- Definitive therapy: Often required (RAI once iodine load clears, or surgery)
- Timing: If due to contrast, typically resolves in 4-8 weeks as iodine load clears; if amiodarone, may persist months (long half-life ~100 days)
Prevention:
- Screen TFTs before starting amiodarone (baseline, 3 months, 6 months, then every 6-12 months)
- Consider low-iodine diet if multinodular goiter and elective amiodarone use
- Pre-treat with methimazole before iodinated contrast if known nodular goiter
18.5 Hyperthyroidism and Pregnancy - Comprehensive Guide
Pregnancy induces significant changes in thyroid physiology, complicating the diagnosis and management of hyperthyroidism. [18]
Physiological Changes in Pregnancy:
- Increased TBG: Estrogen-mediated → total T4/T3 increase (but free levels normal)
- hCG cross-reactivity: hCG has weak TSH-like activity (shares α-subunit) → mild TSH suppression, especially first trimester
- Increased thyroid hormone clearance: Placental D3 deiodinase
- Increased iodine clearance: Renal GFR increases
- Trimester-specific TSH ranges: First trimester TSH normally lower (0.1-2.5 mIU/L)
Gestational Transient Thyrotoxicosis (GTT):
- Affects 1-3% of pregnancies
- Peak at 8-14 weeks (when hCG peaks)
- Associated with hyperemesis gravidarum, multiple gestation, molar pregnancy
- TRAb negative (key differentiator from Graves')
- Self-limited (resolves by 18-20 weeks as hCG falls)
- Usually no treatment needed (unless severe symptoms → propranolol, antiemetics)
Graves' Disease in Pregnancy:
- Accounts for 85-95% of hyperthyroidism in pregnancy
- Natural history: Often improves second/third trimester (pregnancy is immunosuppressive), worsens postpartum
Fetal/Neonatal Risks:
- Maternal TRAb crosses placenta: Fetal/neonatal hyperthyroidism (1-5% if maternal TRAb > 3x ULN)
- Antithyroid drugs cross placenta: Risk of fetal hypothyroidism, goiter
- Prematurity: 2-3 fold increased risk if poorly controlled
- Intrauterine growth restriction (IUGR)
- Stillbirth: If severe untreated hyperthyroidism
- Fetal tachycardia: HR > 160 bpm
- Fetal goiter: From maternal ATD or TRAb
Maternal Risks:
- Preeclampsia: 2-fold increased risk
- Thyroid storm: Precipitated by labor, delivery, cesarean section, or hyperemesis
- Heart failure: High-output failure
- Miscarriage: First trimester
Antithyroid Drug Safety:
| Drug | Trimester | Safety Profile | Dose | Monitoring |
|---|---|---|---|---|
| PTU | First | Preferred (MMI associated with embryopathy) | 100-200 mg TID | LFTs (hepatotoxicity risk) |
| MMI | Second/Third | Safe, preferred (less hepatotoxic than PTU) | 5-20 mg daily | TFTs monthly |
| PTU | Second/Third | Avoid if possible (switch to MMI) | - | LFTs if continued |
MMI Embryopathy (0.5-1% risk, first trimester exposure):
- Aplasia cutis congenita (scalp defects)
- Choanal/esophageal atresia
- Omphalocele
- Omphalomesenteric duct abnormalities
PTU Hepatotoxicity:
- Risk of fulminant hepatic failure (rare but severe)
- Black box FDA warning
- Monitor LFTs if used; stop immediately if ALT > 3x ULN
Target Thyroid Function in Pregnancy:
- Maintain maternal free T4 in upper half of normal range or slightly above
- Avoid over-treatment (fetal hypothyroidism risk)
- TSH may remain suppressed (acceptable if fT4 controlled)
Maternal TRAb Monitoring:
- Measure at diagnosis
- Recheck at 18-22 weeks gestation
- Recheck at 30-34 weeks gestation
- If TRAb > 3x ULN: High risk of fetal/neonatal thyrotoxicosis → fetal monitoring (ultrasound for goiter, growth, heart rate)
Fetal Monitoring (if high maternal TRAb):
- Ultrasound every 4 weeks from 20 weeks
- Assess: Fetal heart rate, goiter size, growth parameters, amniotic fluid, hydrops
Neonatal Thyrotoxicosis:
- Onset: Usually 2-5 days after birth (if mother on ATD, which suppressed fetal thyroid in utero)
- Duration: 2-3 months (until maternal TRAb cleared from neonatal circulation)
- Features: Tachycardia, poor feeding, irritability, goiter, exophthalmos, thrombocytopenia
- Treatment: Propranolol, methimazole (or carbimazole), Lugol's iodine if severe
Surgery in Pregnancy:
- Indication: Failed/intolerant to ATD, very large goiter, suspicious nodule
- Timing: Second trimester (safest, organogenesis complete, uterus not too large)
- Pre-op prep: Beta-blockers, short course Lugol's iodine (reduce vascularity)
- Anesthesia risk: Lower in experienced centers
- Post-op: Levothyroxine replacement
Breastfeeding:
- PTU: Secreted in minimal amounts (less than 0.025% of maternal dose); compatible
- MMI: Secreted in breast milk, but doses less than 20 mg/day considered safe; monitor neonatal TFTs
- Propranolol: Safe
Postpartum Management:
- Graves' disease often relapses/worsens 3-9 months postpartum (immune rebound)
- Resume or increase ATD dose
- Monitor TFTs at 6 weeks, 3 months, 6 months
- Screen for postpartum thyroiditis (transient hyper → hypo phase) if new onset postpartum [18]
SECTION 19: Quality of Life and Patient-Reported Outcomes
19.1 Symptom Burden and Functional Impairment
Hyperthyroidism profoundly impacts quality of life (QoL), often to a degree underestimated by clinicians. Studies using validated instruments (SF-36, ThyPRO, EQ-5D) demonstrate significant impairment across multiple domains. [5]
Domains Affected:
- Physical functioning: Proximal myopathy limits climbing stairs, rising from chairs (60-70% report difficulty)
- Emotional well-being: Anxiety (55%), irritability (65%), emotional lability (45%)
- Cognitive function: Impaired concentration (50%), memory issues (40%)
- Social functioning: Work absenteeism (30%), relationship strain
- Sleep: Insomnia (60%), reduced sleep quality
- Sexual function: Reduced libido, erectile dysfunction (males), menstrual irregularities (females)
ThyPRO Scores: Graves' patients score significantly worse than population norms in:
- Tiredness (mean score 60 vs 20 in controls, scale 0-100)
- Anxiety (mean 45 vs 15)
- Emotional susceptibility (mean 55 vs 18)
- Cognitive problems (mean 40 vs 12)
19.2 Persistent Symptoms After Treatment
A significant minority (15-25%) of patients report persistent QoL impairment even after achieving biochemical euthyroidism for > 12 months. [5]
Residual Symptoms:
- Fatigue (15-20% report persistent tiredness)
- Anxiety and mood disturbances
- Cognitive complaints ("brain fog")
- Weight concerns (often weight gain after treatment)
Possible Mechanisms:
- Subclinical hypothyroidism (over-treatment with RAI/surgery)
- Autoimmune comorbidities (Graves' patients have higher rates of other autoimmune diseases)
- Psychological impact of chronic illness
- Graves' orbitopathy sequelae
- Thyroid hormone level fluctuations during treatment phase
Management Strategies:
- Screen for depression/anxiety (PHQ-9, GAD-7)
- Optimize levothyroxine (if post-RAI/surgery): Some patients feel better with TSH 0.5-1.0 vs 2.0-4.0
- Consider T4/T3 combination therapy in selected cases (controversial, limited evidence)
- Psychotherapy/cognitive-behavioral therapy
- Exercise programs (improve fatigue, mood)
- Patient education and support groups
19.3 Graves' Orbitopathy - Impact on Quality of Life
Graves' orbitopathy (GO) has one of the highest QoL impacts of any autoimmune disease, comparable to diabetic retinopathy or rheumatoid arthritis. [8,10]
QoL Domains Affected:
- Visual function: Diplopia impairs driving, reading (60% of moderate-severe GO)
- Appearance: Proptosis, lid retraction cause significant psychological distress (80% report self-consciousness)
- Social interaction: Avoidance of social situations, workplace issues
- Mental health: Depression (40%), anxiety (50%)
GO-QoL Questionnaire: Two subscales:
- Visual functioning: Daily activities impaired by diplopia, reduced vision
- Appearance: Concerns about changed facial appearance
Moderate-to-severe GO patients score 40-60 points lower than mild GO (scale 0-100).
Interventions to Improve GO-QoL:
- Medical: IV methylprednisolone, teprotumumab (improves proptosis by 2-3 mm, reduces diplopia)
- Surgical rehabilitation: Orbital decompression, strabismus surgery, eyelid surgery (typically staged 6-12 months apart)
- Supportive: Selenium supplementation (mild GO), lubricating eye drops, prisms for diplopia
- Psychological support: Counseling, support groups
- Smoking cessation: Mandatory - smoking worsens GO and impairs treatment response [8,10]
SECTION 20: Global Health and Healthcare System Perspectives
20.1 Epidemiological Variations Worldwide
Iodine Nutrition Status:
- Iodine-sufficient regions (North America, Western Europe, Japan after salt iodization): Graves' disease predominates (60-80% of hyperthyroidism)
- Iodine-deficient regions (parts of Central Africa, Himalayas, Andes before iodization): Toxic multinodular goiter more common (40-60% of hyperthyroidism)
- Transition regions (Eastern Europe, Southeast Asia with recent iodization): Increasing Graves', decreasing TMNG incidence
Ethnic/Genetic Variations:
- Thyrotoxic periodic paralysis: 20-fold higher in Asians vs Caucasians
- Graves' disease: Higher incidence in African Americans and Caucasians vs Asians
- HLA associations: HLA-DR3 (Caucasians), HLA-B46 and DRB108032 (Asians), HLA-B08 (Africans)
20.2 Healthcare Burden and Economics
Direct Costs:
- Antithyroid drugs: $50-150/month (generic methimazole)
- Radioiodine therapy: $1,500-$3,000 (single dose)
- Total thyroidectomy: $10,000-$25,000 (hospital, surgeon, anesthesia)
- Levothyroxine replacement (lifelong): $10-30/month
- Monitoring (TFTs, FBC, LFTs): $200-500/year
Indirect Costs:
- Work absenteeism: Average 2-4 weeks during diagnosis and initial treatment
- Reduced productivity (presenteeism): 20-30% reduction while symptomatic
- Disability (severe GO, cardiac complications): Variable
Cost-Effectiveness:
- RAI most cost-effective for definitive therapy over 5-10 year horizon (fewer monitoring visits than ATD)
- Surgery more cost-effective if large goiter or nodule requiring surgery anyway
- ATD least cost-effective if multiple relapses (due to prolonged monitoring, repeated courses)
20.3 Access to Care Disparities
Low- and Middle-Income Countries:
- Limited access to TRAb testing (diagnosis relies on clinical features + RAIU where available)
- RAI therapy availability varies (requires nuclear medicine facilities, radiation safety infrastructure)
- Surgery may be first-line if RAI unavailable (but requires experienced thyroid surgeon)
- Medication stockouts (thionamides often unavailable)
High-Income Countries:
- Insurance coverage variations (RAI vs surgery preferences)
- Specialist access (endocrinology wait times 3-12 months in some regions)
- Telemedicine expanding access for monitoring
Last Updated: 2026-01-11
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate specialists and current guidelines.
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for hyperthyroidism in adults?
Seek immediate emergency care if you experience any of the following warning signs: Hyperpyrexia (less than 104CF/40CC), Severe tachycardia or atrial fibrillation with rapid ventricular response, Altered mental status or psychosis, Congestive heart failure symptoms, Jaundice or unexplained hepatic failure.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Thyroid Physiology
- Thyroid Function Tests Interpretation
Differentials
Competing diagnoses and look-alikes to compare.
- Thyroiditis (Subacute and Painless)
- Pheochromocytoma
- Anxiety Disorders
Consequences
Complications and downstream problems to keep in mind.